Single and Multiplex Immunohistochemistry to Detect Platelets and Neutrophils in Rat and Porcine Tissues
Abstract
:1. Introduction
2. Materials and Methods
2.1. Background to In Vivo Experimentation and Tissue Collection
2.2. Tissue Collection
2.3. Preparation of Rat and Pig Tissue for Immunohistochemistry (IHC)
2.3.1. Frozen Tissue
2.3.2. FPPE Tissue
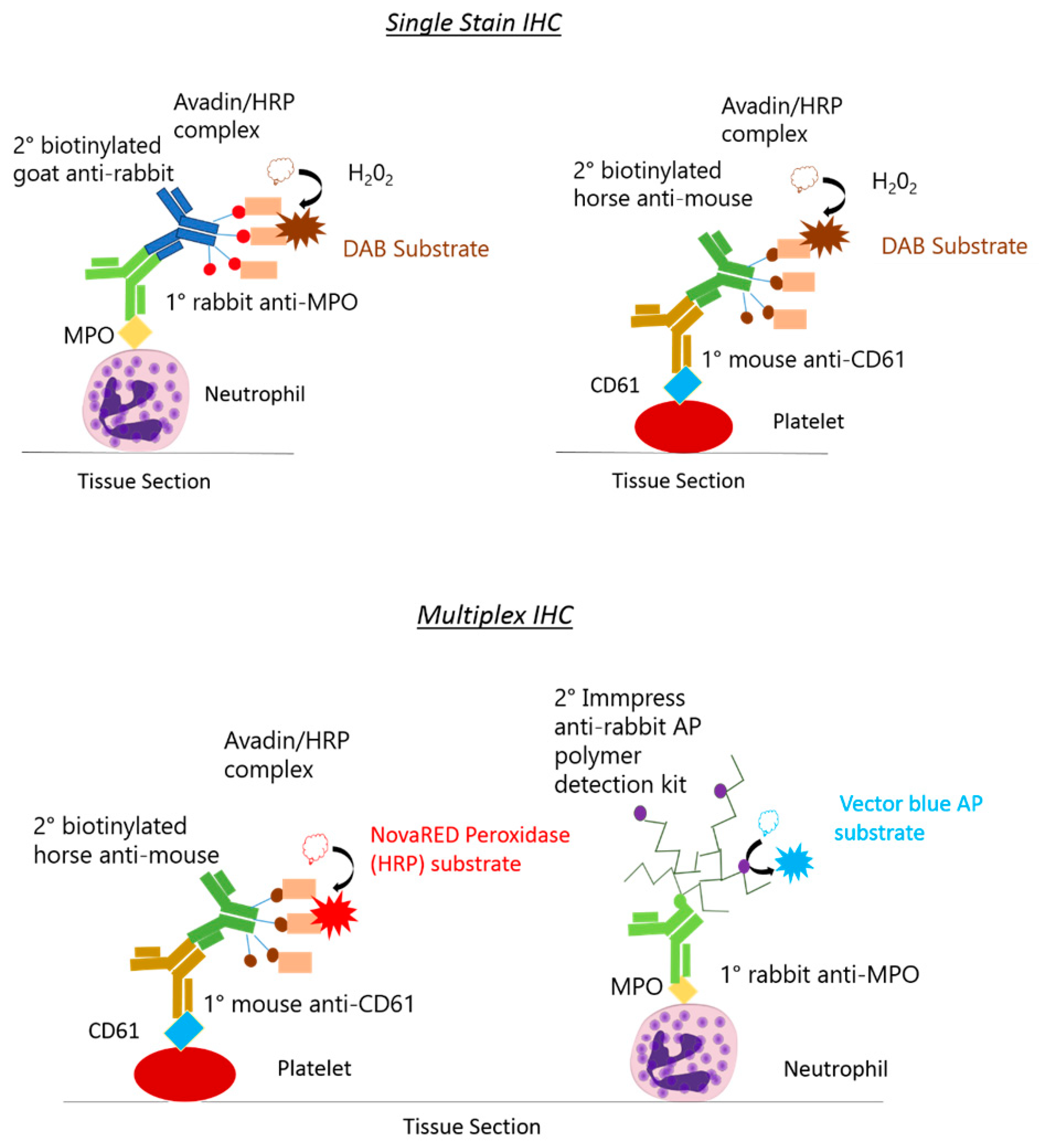
2.4. Rat Monoplex Horse Radish Peroxidase Detection and Colour Development
2.5. Rat Multiplex Alkaline Phosphatase and Horse Radish Peroxidase Detection and Colour Development
2.6. Counterstaining, Dehydration and Clearing
3. Results
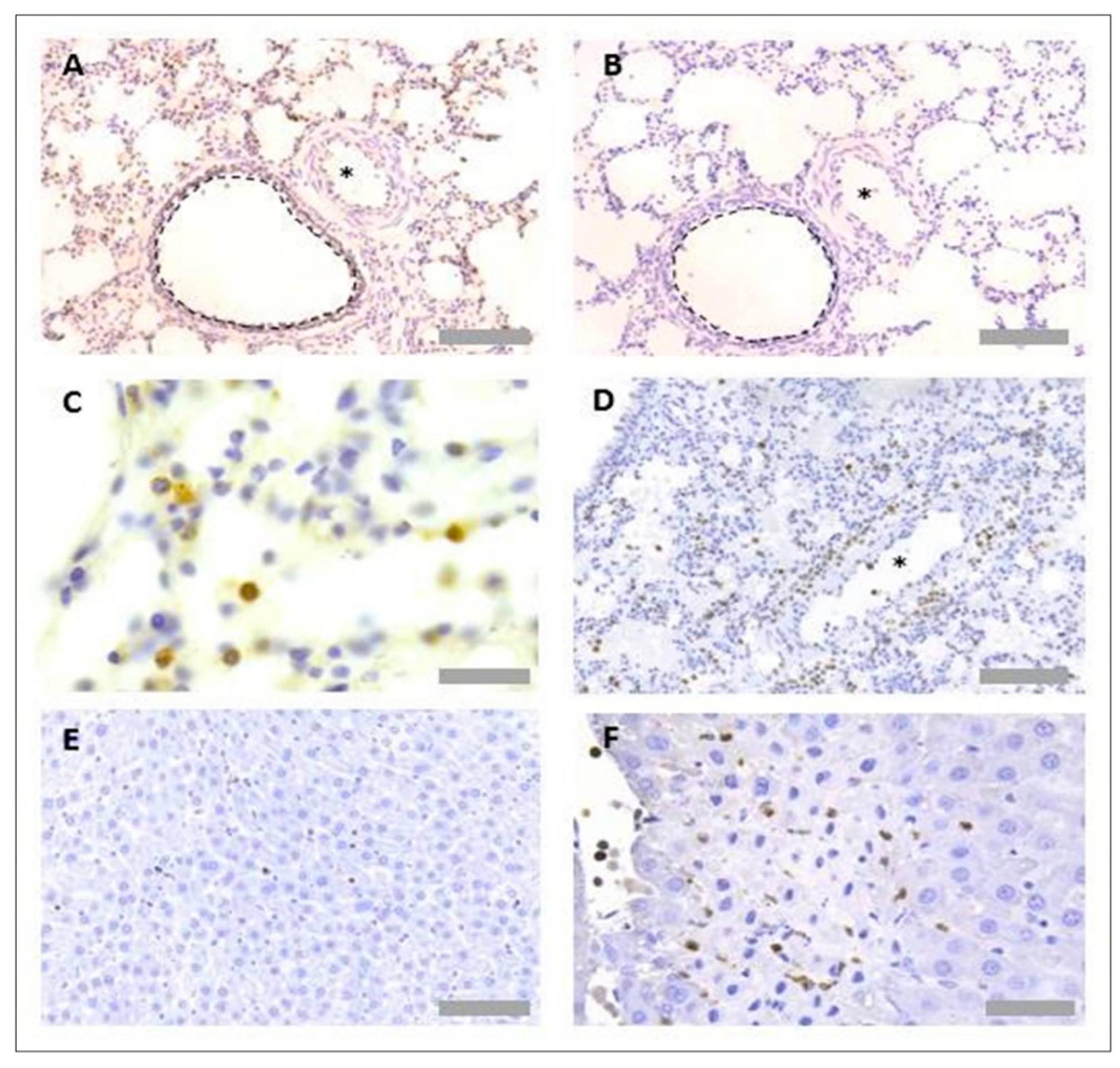
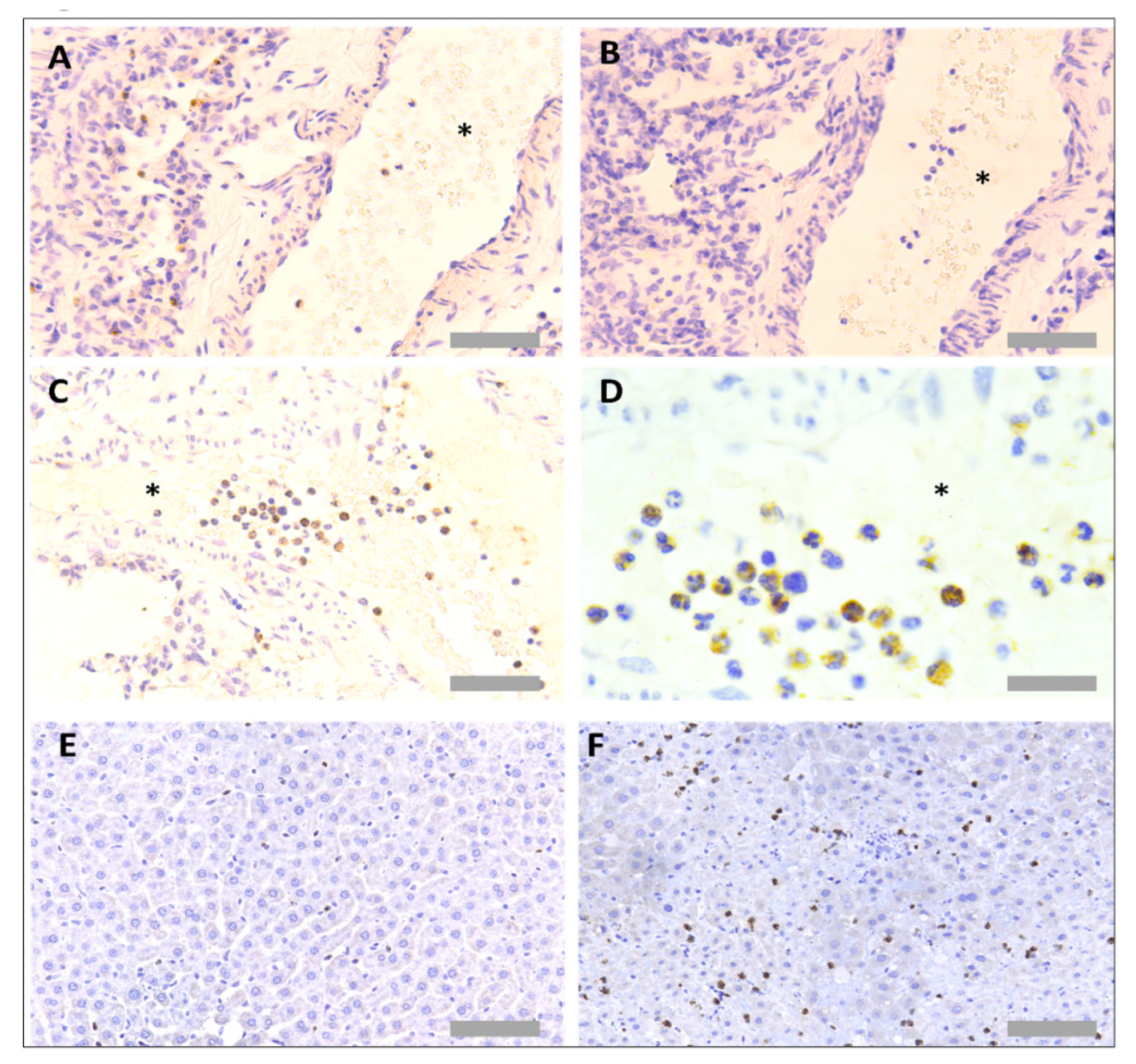
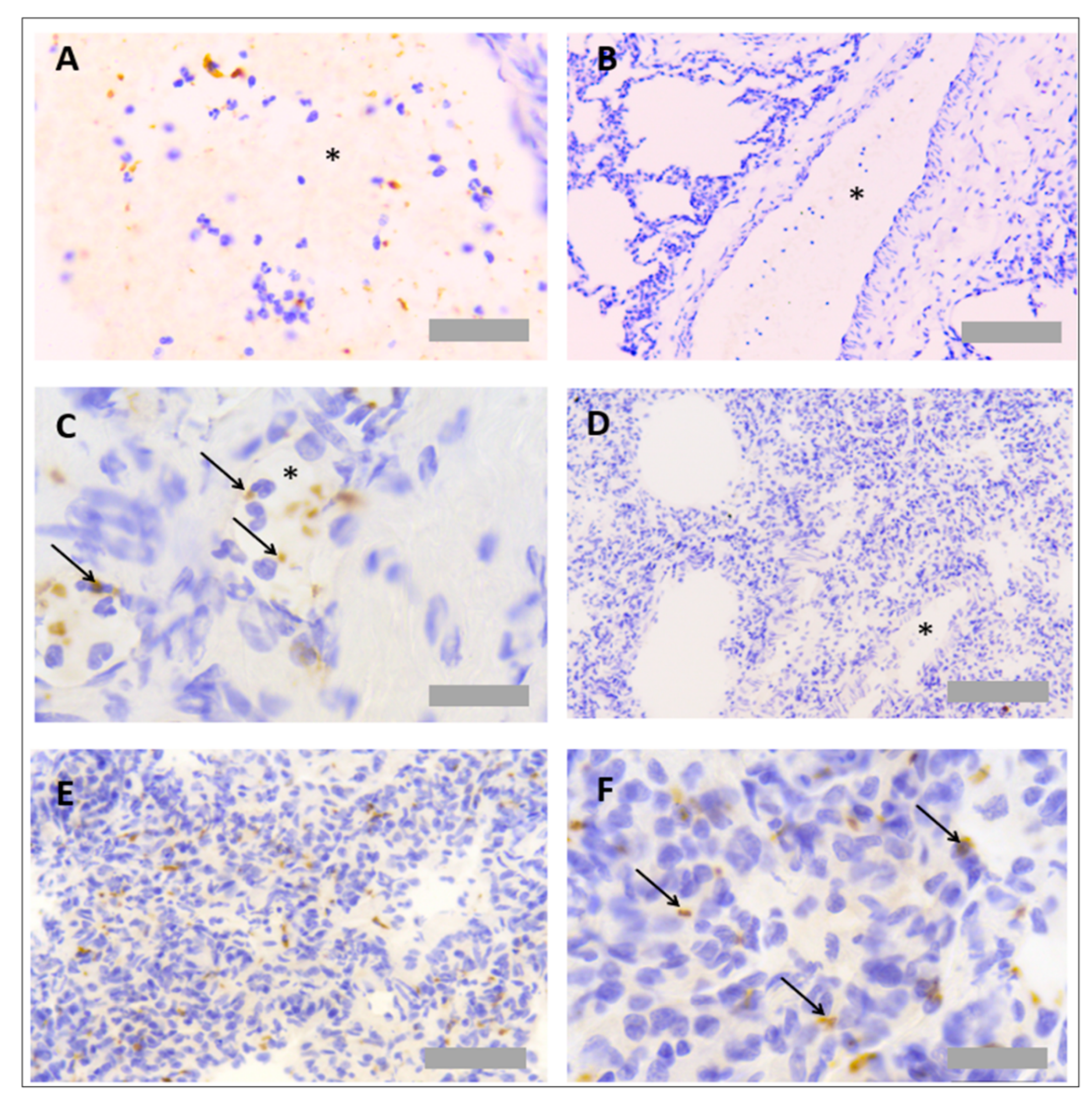
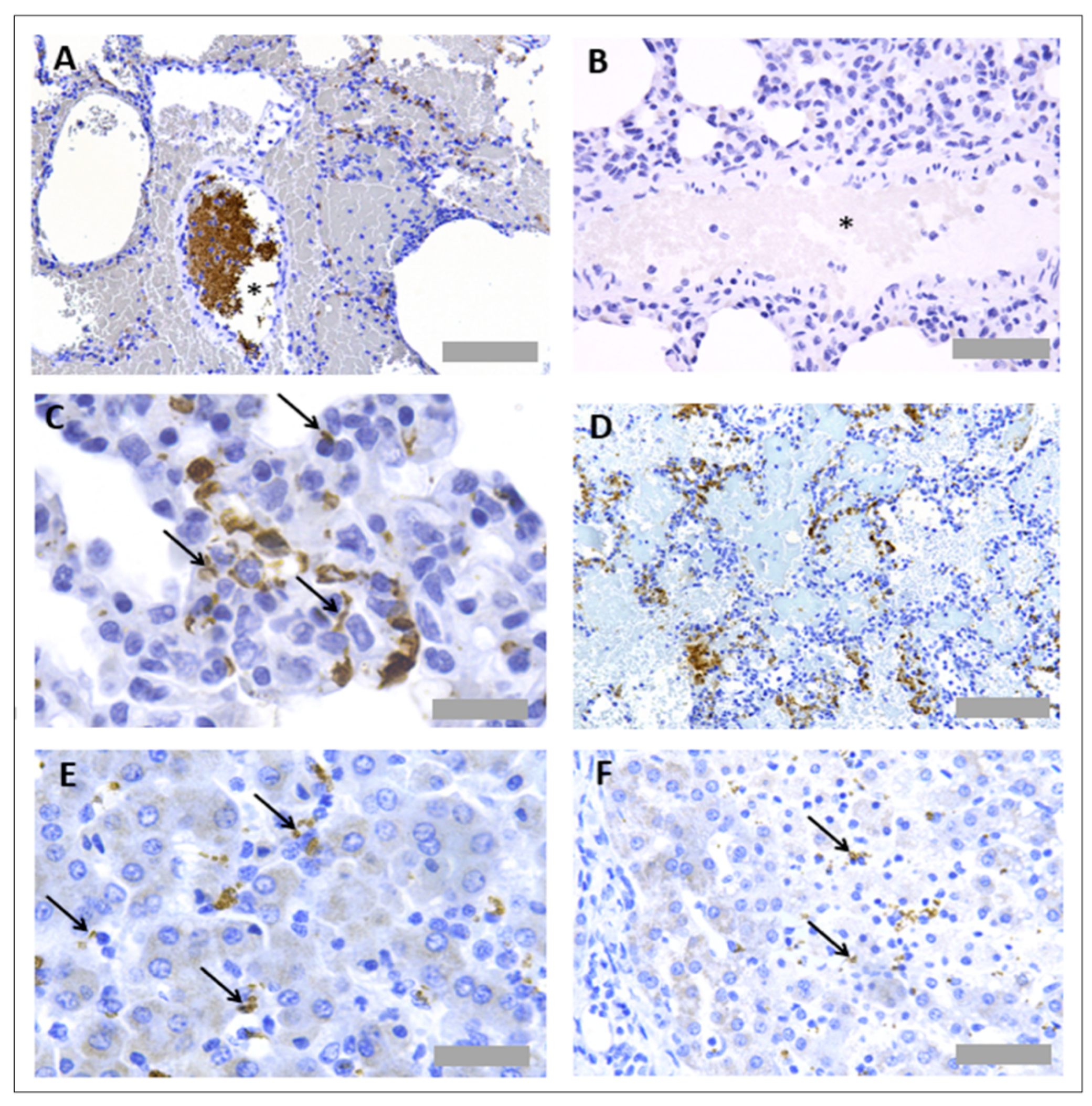
3.1. Protocol Optimisation for IHC in Rat Tissues to Identify Neutrophils and Platelets Using Singleplex Protocols
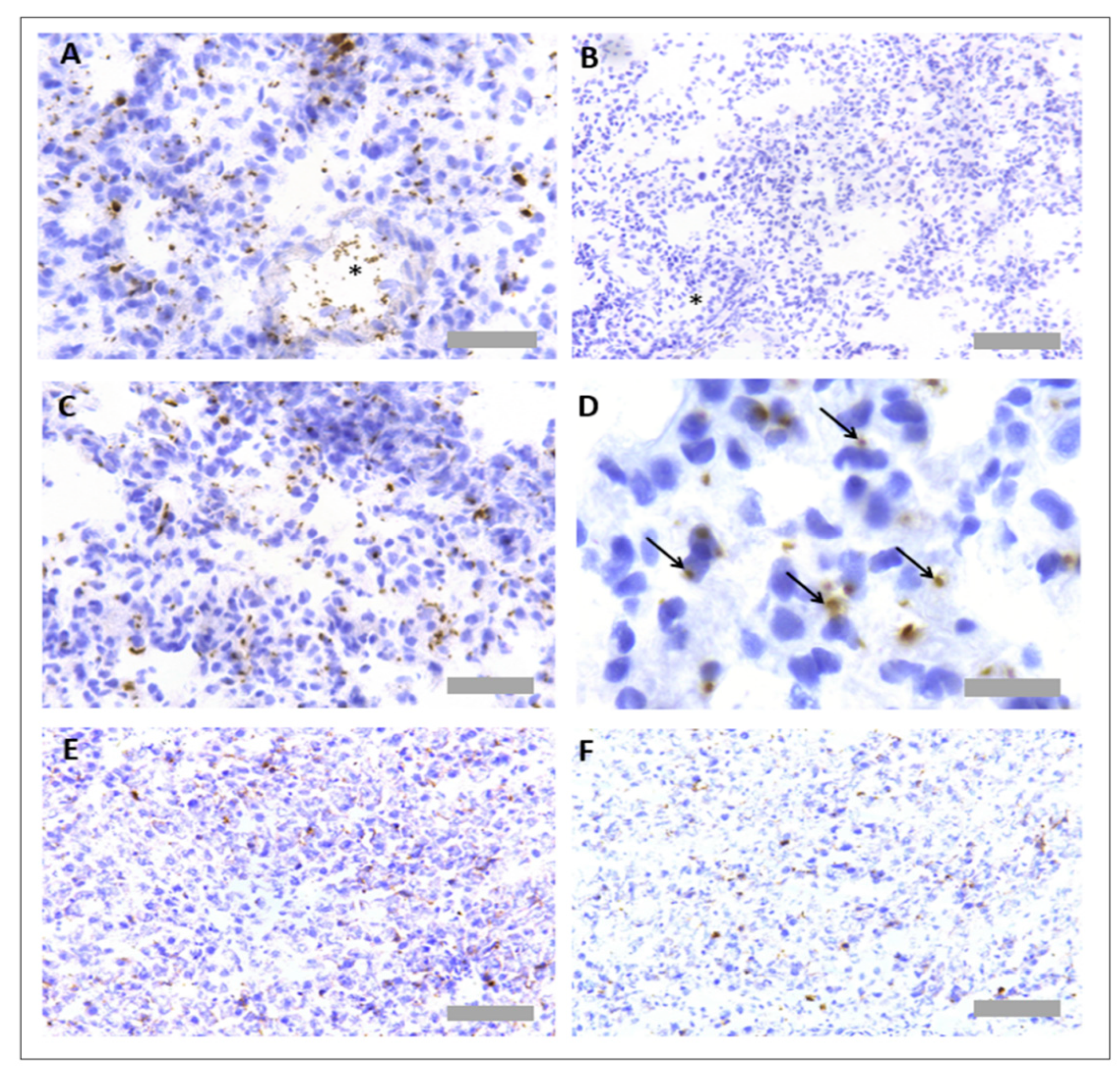
3.2. Protocol Optimisation for IHC to Identify Neutrophils and Platelets in Pig Tissues
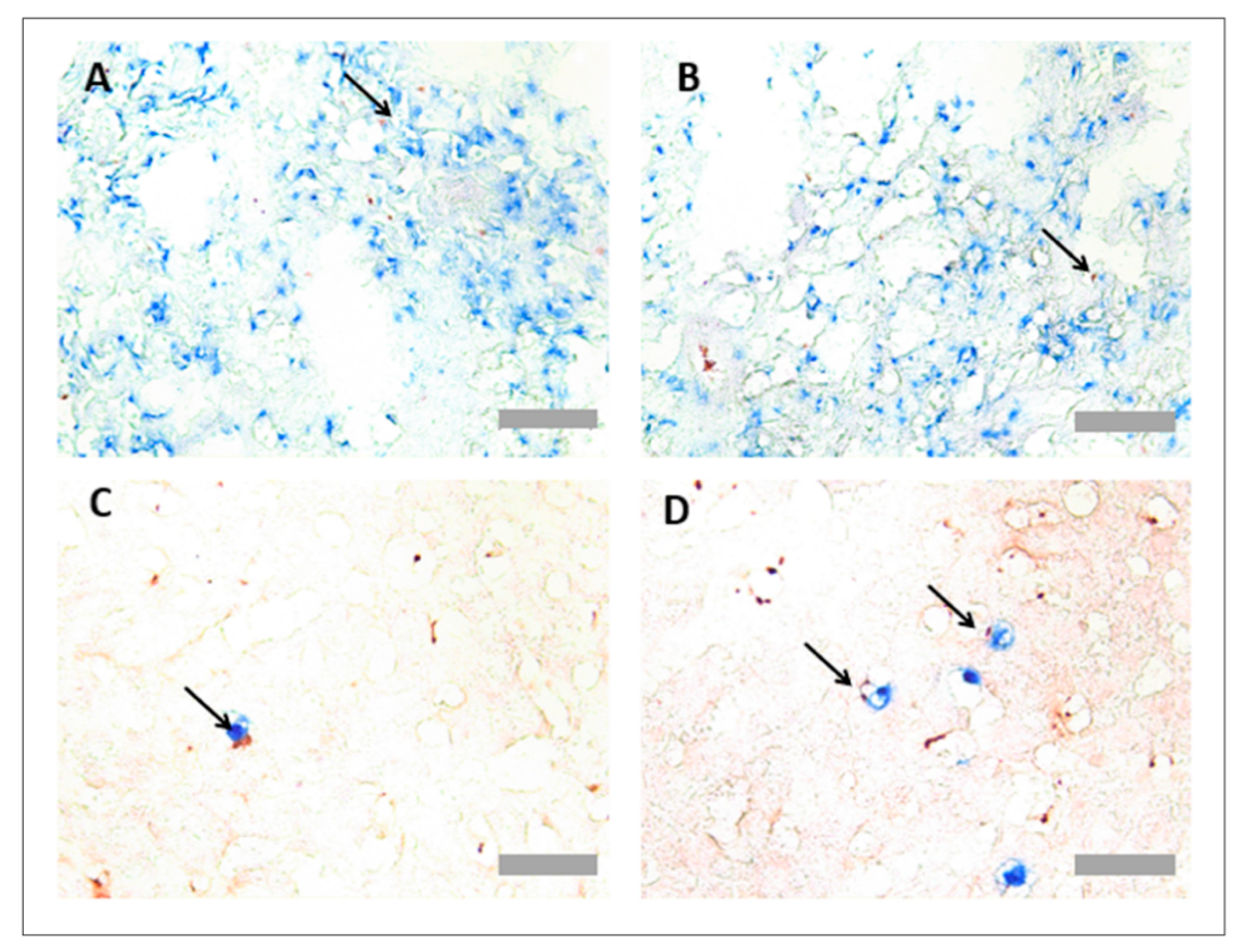
3.3. Protocol Optimisation for IHC in Porcine Tissues to Identify PNCs Using a Multiplex Protocol
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Middleton, E.A.; Weyrich, A.S.; Zimmerman, G.A. Platelets in Pulmonary Immune Responses and Inflammatory Lung Diseases. Physiol. Rev. 2016, 96, 1211–1259. [Google Scholar] [CrossRef]
- Cleary, S.J.; Hobbs, C.; Amison, R.T.; Arnold, S.; O’Shaughnessy, B.G.; Lefrançais, E.; Mallavia, B.; Looney, M.R.; Page, C.P.; Pitchford, S.C. LPS-induced Lung Platelet Recruitment Occurs Independently from Neutrophils, PSGL-1, and P-Selectin. Am. J. Respir. Cell Mol. Biol. 2019, 61, 232–243. [Google Scholar] [CrossRef]
- Looney, M.R.; Nguyen, J.X.; Hu, Y.; Van Ziffle, J.A.; Lowell, C.A.; Matthay, M.A. Platelet depletion and aspirin treatment protect mice in a two-event model of transfusion-related acute lung injury. J. Clin. Investig. 2009, 119, 3450–3461. [Google Scholar] [CrossRef]
- Chavko, M.; Prusaczyk, W.K.; McCarron, R.M. Lung Injury and Recovery After Exposure to Blast Overpressure. J. Trauma 2006, 61, 933–942. [Google Scholar] [CrossRef]
- Scott, T.; Kirkman, E.; Haque, M.; Gibb, I.E.; Mahoney, P.; Hardman, J. Primary blast lung injury—A review. Br. J. Anaesth. 2017, 118, 311–316. [Google Scholar] [CrossRef]
- Kirkman, E.; Watts, S. Characterization of the response to primary blast injury. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 286–290. [Google Scholar] [CrossRef]
- Garner, J.P.; Watts, S.; Parry, C.; Bird, J.; Kirkman, E. Development of a Large Animal Model for Investigating Resuscitation After Blast and Hemorrhage. World J. Surg. 2009, 33, 2194–2202. [Google Scholar] [CrossRef]
- Garner, J.; Watts, S.; Parry, C.; Bird, J.; Cooper, G.; Kirkman, E. Prolonged Permissive Hypotensive Resuscitation Is Associated with Poor Outcome in Primary Blast Injury With Controlled Hemorrhage. Ann. Surg. 2010, 251, 1131–1139. [Google Scholar] [CrossRef]
- Chavko, M.; Adeeb, S.; Ahlers, S.T.; McCarron, R.M. Attenuation of pulmonary inflammation after exposure to blast overpressure by N-acetylcysteine amide. Shock 2009, 32, 325–331. [Google Scholar] [CrossRef]
- Sawdon, M.; Ohnishi, M.; Watkins, P.E.; Kirkman, E. The effects of primary thoracic blast injury and morphine on the response to haemorrhage in the anaesthetised rat. Exp. Physiol. 2002, 87, 683–689. [Google Scholar] [CrossRef] [Green Version]
- Eftaxiopoulou, T.; Barnett-Vanes, A.; Arora, H.; Macdonald, W.; Nguyen, T.-T.N.; Itadani, M.; Sharrock, A.E.; Britzman, D.; Proud, W.G.; Bull, A.M.; et al. Prolonged but not short-duration blast waves elicit acute inflammation in a rodent model of primary blast limb trauma. Injury 2016, 47, 625–632. [Google Scholar] [CrossRef]
- Barnett-Vanes, A.; Sharrock, A.; Eftaxiopoulou, T.; Arora, H.; Macdonald, W.; Bull, A.M.; Rankin, S.M. CD43Lo classical monocytes participate in the cellular immune response to isolated primary blast lung injury. J. Trauma Acute Care Surg. 2016, 81, 500–511. [Google Scholar] [CrossRef]
- Skotak, M.; Wang, F.; Alai, A.; Holmberg, A.; Harris, S.; Switzer, R.C.; Chandra, N. Rat Injury Model under Controlled Field-Relevant Primary Blast Conditions: Acute Response to a Wide Range of Peak Overpressures. J. Neurotrauma 2013, 30, 1147–1160. [Google Scholar] [CrossRef]
- Chai, J.-K.; Liu, W.; Deng, H.-P.; Cai, J.-H.; Hu, Q.-G.; Zou, X.-F.; Shen, C.-A.; Yin, H.-N.; Han, Y.-F.; Zhang, X.-B.; et al. A Novel Model of Burn-Blast Combined Injury and Its Phasic Changes of Blood Coagulation in Rats. Shock 2013, 40, 297–302. [Google Scholar] [CrossRef]
- Shah, S.A.; Kanabar, V.; Riffo-Vasquez, Y.; Mohamed, Z.; Cleary, S.J.; Corrigan, C.; James, A.L.; Elliot, J.G.; Shute, J.K.; Page, C.P.; et al. Platelets Independently Recruit into Asthmatic Lungs and Models of Allergic Inflammation via CCR3. Am. J. Respir. Cell Mol. Biol. 2021, 64, 557–568. [Google Scholar] [CrossRef]
- Zarbock, A.; Singbartl, K.; Ley, K. Complete reversal of acid-induced acute lung injury by blocking of platelet-neutrophil aggregation. J. Clin. Investig. 2006, 116, 3211–3219. [Google Scholar] [CrossRef]
- Francis, W.R.; Ireland, R.E.; Spear, A.M.; Jenner, D.; Watts, S.A.; Kirkman, E.; Pallister, I. Flow Cytometric Analysis of Hematopoietic Populations in Rat Bone Marrow. Impact of Trauma and Hemorrhagic Shock. Cytom. Part A 2019, 95, 1167–1177. [Google Scholar] [CrossRef]
- Watts, S.; Nordmann, G.; Brohi, K.; Midwinter, M.; Woolley, T.; Gwyther, R.; Wilson, C.; Poon, H.; Kirkman, E. Evaluation of Prehospital Blood Products to Attenuate Acute Coagulopathy of Trauma in a Model of Severe Injury and Shock in Anesthetized Pigs. Shock 2015, 44, 138–148. [Google Scholar] [CrossRef]
- Sorrelle, N.; Ganguly, D.; Dominguez, A.T.A.; Zhang, Y.; Huang, H.; Dahal, L.N.; Burton, N.; Ziemys, A.; Brekken, R.A. Improved Multiplex Immunohistochemistry for Immune Microenvironment Evaluation of Mouse Formalin-Fixed, Paraffin-Embedded Tissues. J. Immunol. 2019, 202, 292–299. [Google Scholar] [CrossRef]
- Pirici, D.; Mogoanta, L.; Kumar-Singh, S.; Pirici, I.; Margaritescu, C.; Simionescu, C.; Stanescu, R. Antibody Elution Method for Multiple Immunohistochemistry on Primary Antibodies Raised in the Same Species and of the Same Subtype. J. Histochem. Cytochem. 2009, 57, 567–575. [Google Scholar] [CrossRef] [Green Version]
- Bochicchio, G.V.; Napolitano, L.M.; Joshi, M.; Knorr, K.; Tracy, J.K.; Ilahi, O.; Scalea, T.M. Persistent Systemic Inflammatory Response Syndrome Is Predictive of Nosocomial Infection in Trauma. J. Trauma 2002, 53, 245–251. [Google Scholar] [CrossRef]
- Malone, D.L.; Kuhls, D.; Napolitano, L.M.; McCarter, R.; Scalea, T. Back to basics: Validation of the admission systemic in-flammatory response syndrome score in predicting outcome in trauma. J. Trauma 2001, 51, 458–463. [Google Scholar] [CrossRef]
- Lord, J.M.; Midwinter, M.J.; Chen, Y.-F.; Belli, A.; Brohi, K.; Kovacs, E.J.; Koenderman, L.; Kubes, P.; Lilford, R.J. The systemic immune response to trauma: An overview of pathophysiology and treatment. Lancet 2014, 384, 1455–1465. [Google Scholar] [CrossRef]
- Pitchford, S.C.; Momi, S.; Baglioni, S.; Casali, L.; Giannini, S.; Rossi, R.; Page, C.P.; Gresele, P. Allergen Induces the Migration of Platelets to Lung Tissue in Allergic Asthma. Am. J. Respir. Crit. Care Med. 2008, 177, 604–612. [Google Scholar] [CrossRef]
- Jiang, M.P.; Xu, C.; Guo, Y.W.; Luo, Q.J.; Li, L.; Liu, H.L.; Jiang, J.; Chen, H.X.; Wei, X.Q. Beta-arrestin 2 attenuates lipopolysaccharide-induced liver injury via inhibition of TLR4/NF-kappaB signaling pathway-mediated inflammation in mice. World J. Gastroenterol. 2018, 24, 216–225. [Google Scholar] [CrossRef]
- Chiarot, E.; Spagnuolo, A.; Maccari, S.; Naimo, E.; Acquaviva, A.; Cecchi, R.; Galletti, B.; Fabbrini, M.; Mori, E.; Ruggiero, P.; et al. Protective effect of Group B Streptococcus type-III polysaccharide conjugates against maternal colonization, ascending infection and neonatal transmission in rodent models. Sci. Rep. 2018, 8, 2593. [Google Scholar] [CrossRef]
- Amison, R.T.; O’Shaughnessy, B.G.; Arnold, S.; Cleary, S.J.; Nandi, M.; Pitchford, S.C.; Bragonzi, A.; Page, C.P. Platelet Depletion Impairs Host Defense to Pulmonary Infection with Pseudomonas aeruginosa in Mice. Am. J. Respir. Cell Mol. Biol. 2018, 58, 331–340. [Google Scholar] [CrossRef]
- Wu, X.; Schwacha, M.G.; Dubick, M.A.; Cap, A.P.; Darlington, D.N. Trauma-Related Acute Lung Injury Develops Rapidly Irrespective of Resuscitation Strategy in the Rat. Shock 2016, 46 (Suppl. S1), 108–114. [Google Scholar] [CrossRef]
- Arnold, S.; Shah, S.A.; Cleary, S.J.; O’Shaughnessy, B.G.; Amison, R.T.; Arkless, K.; Page, C.P.; Pitchford, S.C. Red Blood Cells Elicit Platelet-Dependent Neutrophil Recruitment into Lung Airspaces. Shock 2020, 56, 278–286. [Google Scholar] [CrossRef]
- Alam, M.S. Proximity Ligation Assay (PLA). Curr. Protoc. Immunol. 2018, 123, e58. [Google Scholar] [CrossRef]
- Parra, E.R.; Uraoka, N.; Jiang, M.; Cook, P.; Gibbons, D.; Forget, M.-A.; Bernatchez, C.; Haymaker, C.; Wistuba, I.I.; Rodriguez-Canales, J. Validation of multiplex immunofluorescence panels using multispectral microscopy for immune-profiling of formalin-fixed and paraffin-embedded human tumor tissues. Sci. Rep. 2017, 7, 13380. [Google Scholar] [CrossRef]
- Simonson, P.D.; Valencia, I.; Patel, S.S. Tyramide-conjugated DNA barcodes enable signal amplification for multiparametric CODEX imaging. Commun. Biol. 2022, 5, 627. [Google Scholar] [CrossRef]
- Page, C.; Pitchford, S. Neutrophil and platelet complexes and their relevance to neutrophil recruitment and activation. Int. Immunopharmacol. 2013, 17, 1176–1184. [Google Scholar] [CrossRef]
- Kornerup, K.N.; Salmon, G.P.; Pitchford, S.C.; Liu, W.L.; Page, C.P. Circulating platelet-neutrophil complexes are important for subsequent neutrophil activation and migration. J. Appl. Physiol. 2010, 109, 758–776. [Google Scholar] [CrossRef]

| Target Antigen | Supplier | Catalogue Number | Antibody Raised In | Dilutions Tested | Success | Optimum HIER Buffer |
|---|---|---|---|---|---|---|
| Ly-6G | Bioss, USA | bs-2576R | rabbit | 1/100, 1/500 | × | N/A |
| MPO | Abcam, UK | ab9535 | rabbit | 1/25 1/50, 1/100 1/500 | 1/50 in frozen and FFPE | EDTA pH 8 |
| MPO | Abcam, UK | ab90810 | mouse | 1/50, 1/100 1/500 | × | N/A |
| Neutrophil Elastase | Abcam, UK | ab68672 | rabbit | 1/50, 1/100 1/200, 1/500 | × | N/A |
| Target Antigen | Supplier | Catalogue Number | Species Raised In | Dilutions Tested | Success |
|---|---|---|---|---|---|
| CD41 | Santa Cruz Biotechnologies, USA | sc-6602 | goat | 1/100 1/500 | × |
| CD41 | Santa Cruz Biotechnologies, USA | sc-6604 | goat | 1/100 1/500 | × |
| CD41 | Abcam, UK | ab93983 | rabbit | 1/50 1/100 1/500 | × |
| CD42b | Abcam, UK | ab183345 | rabbit | 1/50 1/100 1/200 1/500 | × |
| CD42c | Santa Cruz Biotechnologies, USA. | sc-7073 | goat | 1/50 1/100 1/500 | × |
| CD49b | Abcam, UK | ab181548 | rabbit | 1/100 1/200 1/500 | × |
| CD61 | BD Biosciences, USA | MCA2263GA | mouse | 1/50 1/100 1/500 | × |
| CD61 | Boster, USA | PB9647 | rabbit | 1/50 1/100 1/500 | × |
| CD61 | BD Biosciences, USA | 554951 | mouse | 1/50 1/100 1/500 | 1/50 in frozen tissue |
| Target Antigen | Supplier | Catalogue Number | Antibody Raised In | Dilutions Tested | Success | Optimum HIER Buffer |
|---|---|---|---|---|---|---|
| Ly-6G | BioXCell, USA | BE00751 | rat | 1/100 1/1000 | × | N/A |
| Ly-6G | Bioss, USA | bs-2576R | rabbit | 1/100 1/500 | × | N/A |
| MPO | Abcam, UK | ab9535 | rabbit | 1/25 1/50 1/100 1/500 | 1/50 in frozen and FFPE | EDTA pH 8 |
| MPO | Abcam, UK | ab90810 | mouse | 1/50, 1/100 1/500 | × | N/A |
| Neutrophil (NIMP-R14) | Abcam, UK | ab2557 | rat | 1/100 1/500 | × | N/A |
| PSGL-1 | Abcam, UK | ab110096 | rat | 1/50, 1/500 | × | N/A |
| Target Antigen | Supplier | Catalogue Number | Species Raised In | Dilutions Tested | Success | Optimum HIER Buffer |
|---|---|---|---|---|---|---|
| CD41 | Santa Cruz Biotechnologies, USA | sc-6602 | goat | 1/100 1/500 | × | N/A |
| CD41 | Santa Cruz Biotechnologies, USA | sc-6604 | goat | 1/100 1/500 | × | N/A |
| CD41 | Abcam, UK | ab33661 | rat | 1/100 1/500 | × | N/A |
| CD41 | Abcam, UK | ab93983 | rabbit | 1/50, 1/100 1/500 | × | N/A |
| CD42b | Abcam, UK | ab183345 | rabbit | 1/50, 1/100 1/200 1/500 | 1/100 FFPE and Frozen | Tris-EDTA pH 9.0 |
| CD42c | Santa Cruz Biotechnologies, USA | sc-7073 | goat | 1/50, 1/100 1/500 | × | N/A |
| CD49b | Abcam, UK | ab181548 | rabbit | 1/100, 1/200 1/500 | 1/100 FFPE | Tris-EDTA pH 9.0 |
| CD61 | BD Biosciences, USA | JM2E5 | mouse | 1/50, 1/100 1/500 | × | N/A |
| Target Species | Supplier | Catalogue Number | Antibody Raised In | Dilutions Tested |
|---|---|---|---|---|
| Goat | Santa Cruz, USA | sc-3854 | donkey | 1/100, 1/200 |
| Mouse | Vector, USA | BA2001 | horse | 1/200 |
| Rabbit | Vector, USA | BA1000 | horse | 1/200 |
| Cell Type | Fixation Method | Primary Antibody | Secondary Antibody | Tertiary Reagent | Colour Substrate |
|---|---|---|---|---|---|
| Neutrophils | Frozen | Rabbit anti-human myeloperoxidase (MPO) ab9535 (Abcam, UK) Concentration: 1/25 (1 h) | ImmPRESS-AP Anti-Rabbit IgG (alkaline phosphatase) Polymer Detection Kit MP-5401 (Vector Laboratories, USA) (30 min) | N/A | Vector Blue Alkaline Phosphatase (AP) substrate kit SK-5300 (Vector Laboratories, USA) (10 min) |
| Platelets | Frozen | Mouse anti-rat CD61 PB9647 (Boster, USA) Concentration: 1/100 (1 h) | Biotinylated horse anti-mouse (BA-2001), (Vector Laboratories, USA) Concentration: 1/200 (1 h) | Avadin and biotinylated HRP enzymes VECTASTAIN elite ABC kit PK-6100, Vector Laboratories, USA (30 min) | NovaRED Peroxidase (HRP) substrate kit SK-4800 (Vector Laboratories, USA) (20 min) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arnold, S.; Watts, S.; Kirkman, E.; Page, C.P.; Pitchford, S.C. Single and Multiplex Immunohistochemistry to Detect Platelets and Neutrophils in Rat and Porcine Tissues. Methods Protoc. 2022, 5, 71. https://doi.org/10.3390/mps5050071
Arnold S, Watts S, Kirkman E, Page CP, Pitchford SC. Single and Multiplex Immunohistochemistry to Detect Platelets and Neutrophils in Rat and Porcine Tissues. Methods and Protocols. 2022; 5(5):71. https://doi.org/10.3390/mps5050071
Chicago/Turabian StyleArnold, Stephanie, Sarah Watts, Emrys Kirkman, Clive P. Page, and Simon C. Pitchford. 2022. "Single and Multiplex Immunohistochemistry to Detect Platelets and Neutrophils in Rat and Porcine Tissues" Methods and Protocols 5, no. 5: 71. https://doi.org/10.3390/mps5050071
APA StyleArnold, S., Watts, S., Kirkman, E., Page, C. P., & Pitchford, S. C. (2022). Single and Multiplex Immunohistochemistry to Detect Platelets and Neutrophils in Rat and Porcine Tissues. Methods and Protocols, 5(5), 71. https://doi.org/10.3390/mps5050071





