Simultaneous Quantification of Bisphenol-A and 4-Tert-Octylphenol in the Live Aquaculture Feed Artemia franciscana and in Its Culture Medium Using HPLC-DAD
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Animals
2.3. Preparation of Stock and Standard Solutions
2.4. Sample Preparation
2.5. Pollutant Administration
2.6. Photodegradation Assay
2.7. Chromatography
3. Results and Discussion
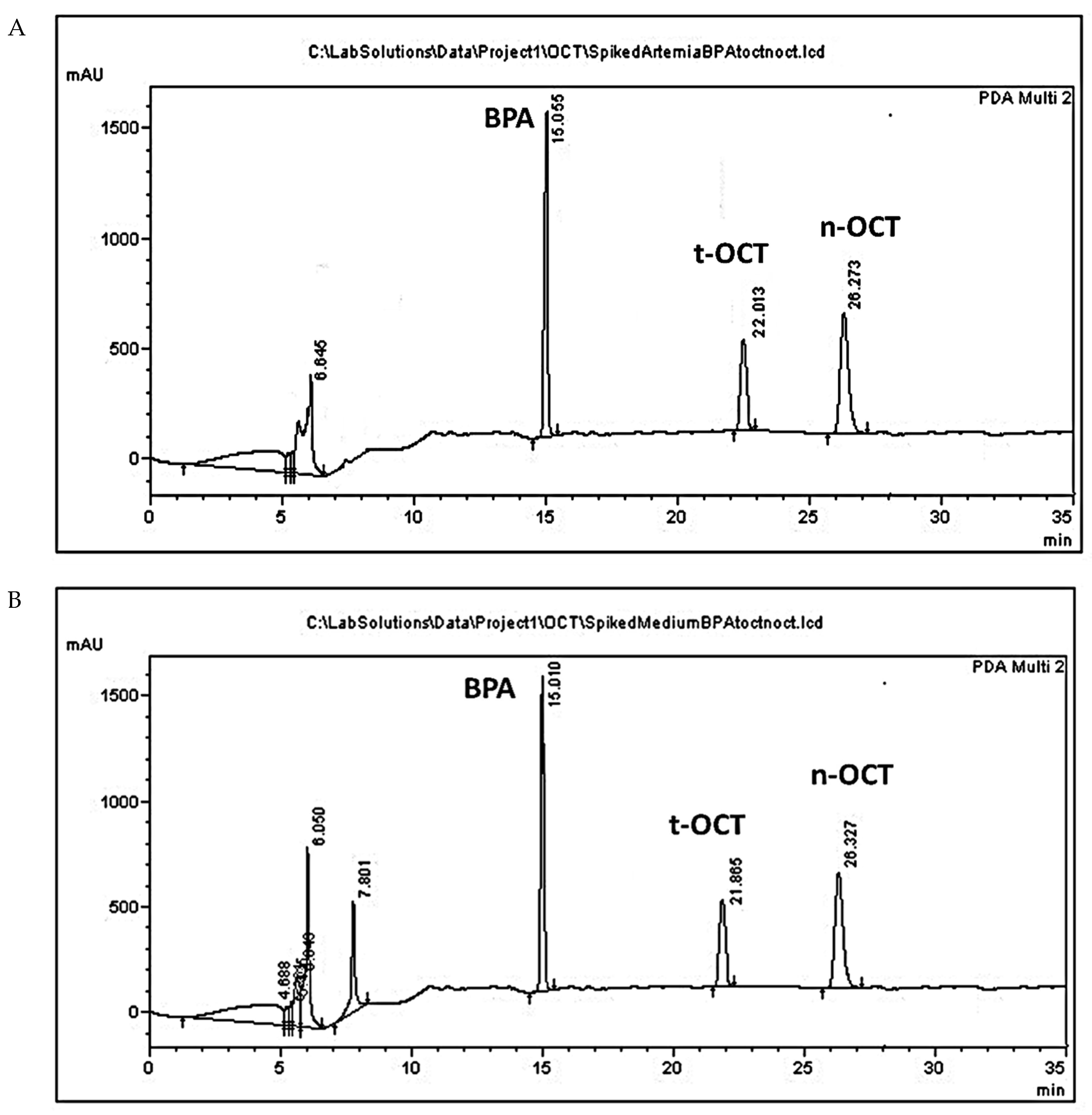
3.1. Analytes Extraction Efficiency
3.2. Method Validation
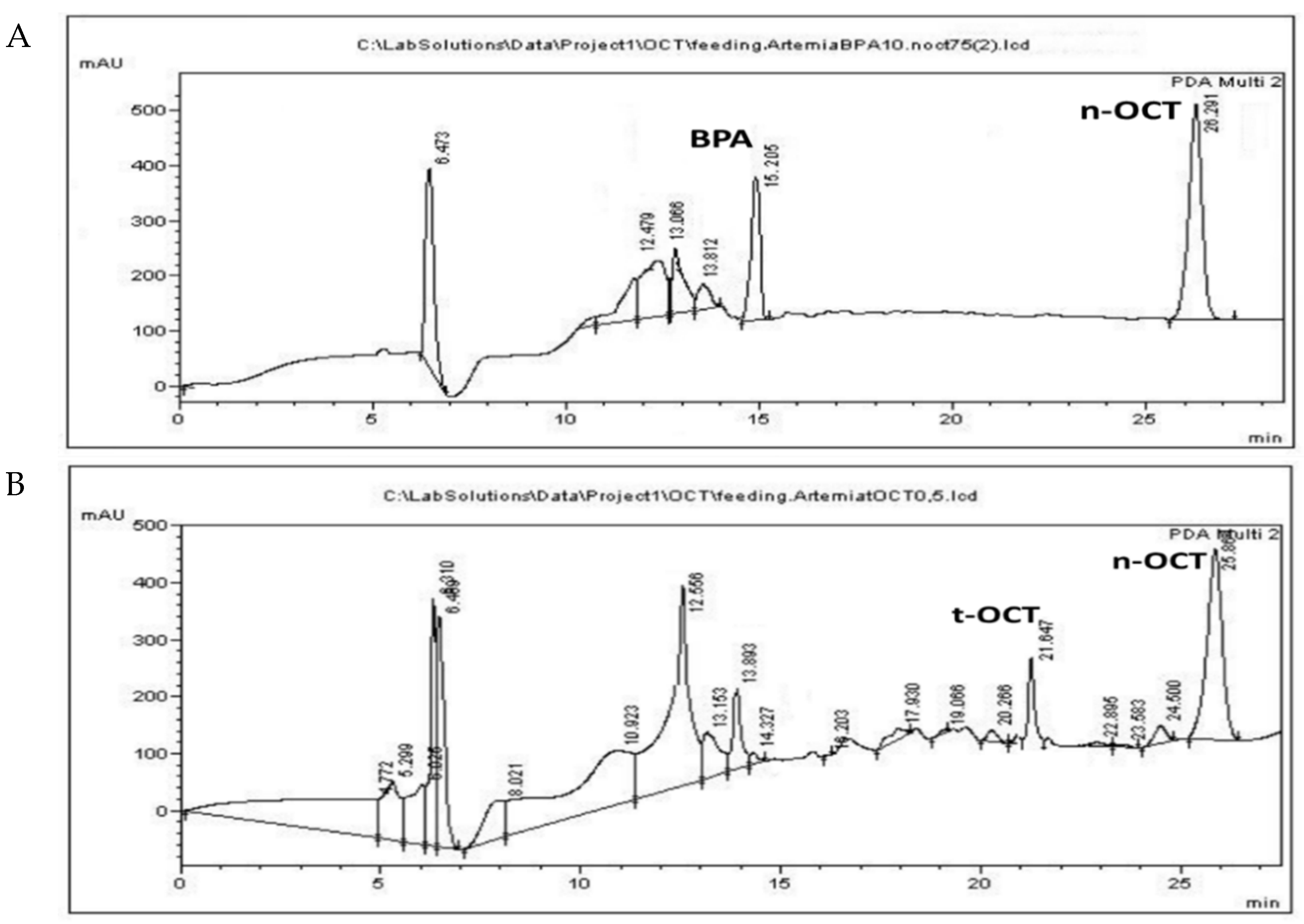
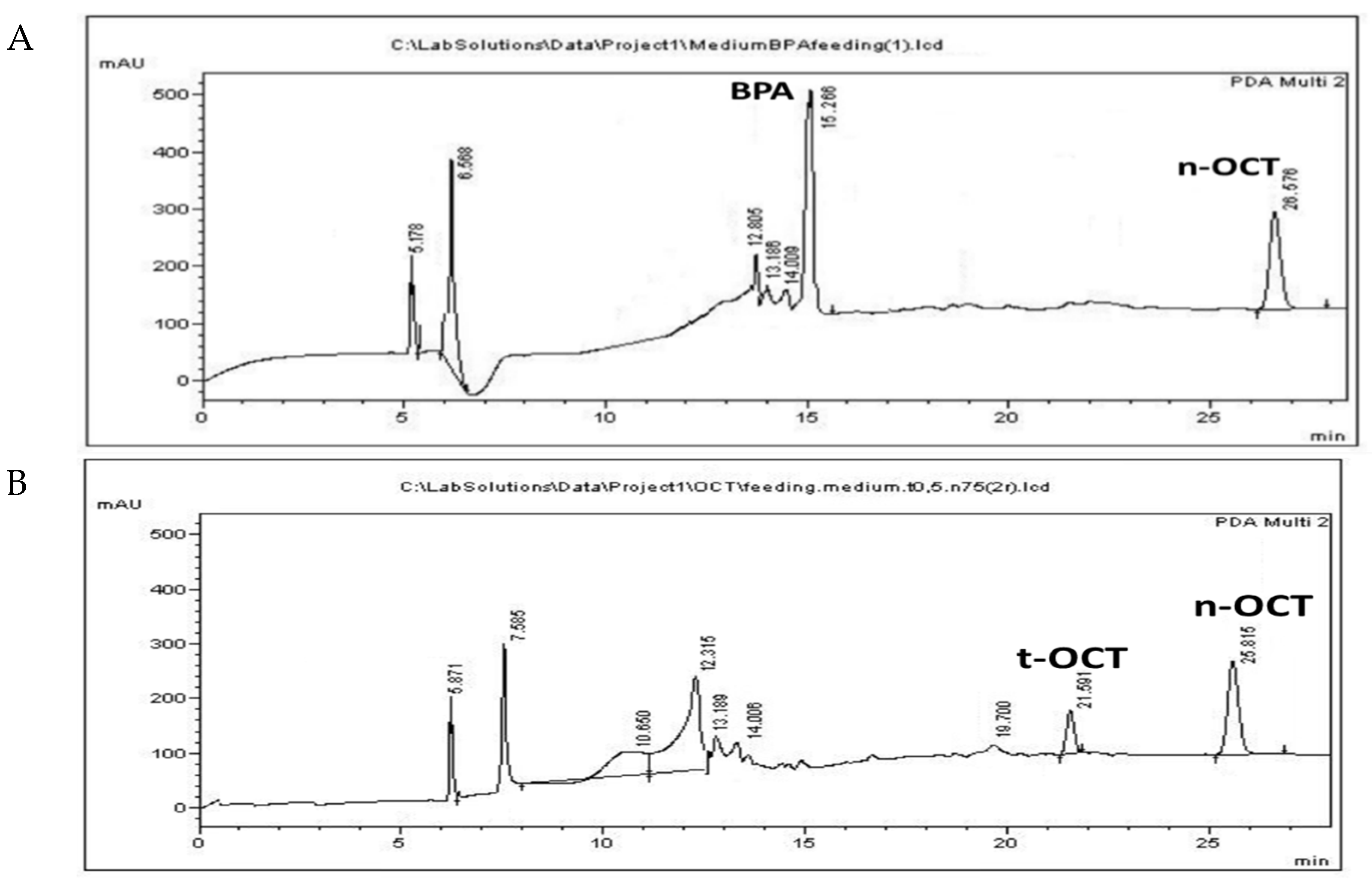
3.3. Administration of Pollutants
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Koelmans, A.A.; Besseling, E.; Foekema, E.M. Leaching of plastic additives to marine organisms. Environ. Pollut. 2014, 187, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Darbre, P.D. Chemical Components of Plastics as Endocrine Disruptors: Overview and Commentary. Birth Defects Res. 2020, 112, 1300–1307. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Allgeier, A.; Yin, D.; Hollert, H. Leaching of endocrine disrupting chemicals from marine microplastics and mesoplastics under common life stress conditions. Environ. Int. 2019, 130, 104938. [Google Scholar] [CrossRef] [PubMed]
- López-Cervantes, J.; Sánchez-Machado, D.; Paseiro-Losada, P.; Simal-Lozano, J. Effects of Compression, Stacking, Vacuum Packing and Temperature on the Migration of Bisphenol A from Polyvinyl Chloride Packaging Sheeting into Food Simulants. Chromatographia 2003, 58, 327–330. [Google Scholar] [CrossRef]
- Hahladakis, J.N.; Velis, C.A.; Weber, R.; Iacovidou, E.; Purnell, P. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater. 2018, 344, 179–199. [Google Scholar] [CrossRef]
- Hirai, H.; Takada, H.; Ogata, Y.; Yamashita, R.; Mizukawa, K.; Saha, M.; Kwan, C.; Moore, C.; Gray, H.; Laursen, D.; et al. Organic micropollutants in marine plastics debris from the open ocean and remote and urban beaches. Mar. Pollut. Bull. 2011, 62, 1683–1692. [Google Scholar] [CrossRef]
- European Chemicals Agency (ECHA). Candidate List of Substances of Very High Concern for Authorization (2020). Available online: https://echa.europa.eu/candidate-list-table (accessed on 3 April 2022).
- EFSA. Bisphenol, A. 2017. Available online: https://www.efsa.europa.eu/en/topics/topic/bisphenol (accessed on 2 April 2022).
- Available online: https://plasticseurope.org/wp-content/uploads/2021/10/20210301-BPA-Judgment-appeal.pdf (accessed on 1 April 2022).
- Vogel, S.A. The politics of plastics: The making and unmaking of bisphenol a “safety”. Am. J. Public Health 2009, 99 (Suppl. S3), S559–S566. [Google Scholar] [CrossRef]
- Ying, G.G.; Williams, B.; Kookana, R. Environmental fate of alkylphenols and alkylphenol ethoxylates—A review. Environ. Int. 2002, 28, 215–226. [Google Scholar] [CrossRef]
- Octylphenol. Available online: https://echa.europa.eu/el/substance-information/-/substanceinfo/100.060.634 (accessed on 2 April 2022).
- Kang, J.H.; Kondo, F.; Katayama, Y. Human exposure to bisphenol A. Toxicology 2006, 226, 79–89. [Google Scholar] [CrossRef]
- White, R.; Jobling, S.; Hoare, S.; Sumpter, J.; Parker, M. Environmentally persistent alkylphenolic compounds are estrogenic. Endocrinology 1994, 135, 175–182. [Google Scholar] [CrossRef]
- Olaniyan, L.; Okoh, O.O.; Mkwetshana, N.T.; Okoh, A.I. Environmental Water Pollution, Endocrine Interference and Ecotoxicity of 4-tert-Octylphenol: A Review. Rev. Environ. Contam. Toxicol. 2020, 248, 81–109. [Google Scholar] [CrossRef]
- Canesi, L.; Fabbri, E. Environmental Effects of BPA: Focus on Aquatic Species. Dose Response 2015, 13, 1559325815598304. [Google Scholar] [CrossRef] [PubMed]
- Nachman, R.M.; Hartle, J.C.; Lees, P.S.; Groopman, J.D. Early Life Metabolism of Bisphenol A: A Systematic Review of the Literature. Curr. Environ. Health Rep. 2014, 1, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Kyrila, G.; Katsoulas, A.; Schoretsaniti, V.; Rigopoulos, A.; Rizou, E.; Doulgeridou, S.; Sarli, V.; Samanidou, V.; Touraki, M. Bisphenol A removal and degradation pathways in microorganisms with probiotic properties. J. Hazard. Mater. 2021, 413, 125363. [Google Scholar] [CrossRef]
- Hayashi, O.; Kameshiro, M.; Masuda, M.; Satoh, K. Bioaccumulation and metabolism of [14C] bisphenol A in the brackish water bivalve Corbicula japonica. Biosci. Biotechnol. Biochem. 2008, 72, 3219–3224. [Google Scholar] [CrossRef][Green Version]
- Certa, H.; Fedtke, N.; Wiegand, H.J.; Müller, A.M.; Bolt, H.M. Toxicokinetics of p-tert-octylphenol in male Wistar rats. Arch. Toxicol. 1996, 71, 112–122. [Google Scholar] [CrossRef]
- Jing, X.; Bing, S.; Xiaoyan, W.; Xiaojie, S.; Yongning, W. A study on bisphenol A, nonylphenol, and octylphenol in human urine samples detected by SPE-UPLC-MS. Biomed. Environ. Sci. 2011, 24, 40–46. [Google Scholar] [CrossRef]
- Rajendran, R.K.; Huang, S.L.; Lin, C.C.; Kirschner, R. Biodegradation of the endocrine disrupter 4-tert-octylphenol by the yeast strain Candida rugopelliculosa RRKY5 via phenolic ring hydroxylation and alkyl chain oxidation pathways. Bioresour. Technol. 2017, 226, 55–64. [Google Scholar] [CrossRef]
- Pop, C.-E.; Draga, S.; Măciucă, R.; Niță, R.; Crăciun, N.; Wolff, R. Bisphenol A Effects in Aqueous Environment on Lemna minor. Processes 2021, 9, 1512. [Google Scholar] [CrossRef]
- Barboza, L.; Cunha, S.C.; Monteiro, C.; Fernandes, J.O.; Guilhermino, L. Bisphenol A and its analogs in muscle and liver of fish from the North East Atlantic Ocean in relation to microplastic contamination. Exposure and risk to human consumers. J. Hazard. Mater. 2020, 393, 122419. [Google Scholar] [CrossRef]
- Baralla, E.; Pasciu, V.; Varoni, M.V.; Nieddu, M.; Demuro, R.; Demontis, M.P. Bisphenols’ occurrence in bivalves as sentinel of environmental contamination. Sci. Total Environ. 2021, 785, 147263. [Google Scholar] [CrossRef] [PubMed]
- Staples, C.; van der Hoeven, N.; Clark, K.; Mihaich, E.; Woelz, J.; Hentges, S. Distributions of concentrations of bisphenol A in North American and European surface waters and sediments determined from 19 years of monitoring data. Chemosphere 2018, 201, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Staniszewska, M.; Falkowska, L.; Grabowski, P.; Kwaśniak, J.; Mudrak-Cegiołka, S.; Reindl, A.R.; Sokołowski, A.; Szumiło, E.; Zgrundo, A. Bisphenol A, 4-tert-octylphenol, and 4-nonylphenol in the Gulf of Gdańsk (Southern Baltic). Arch. Environ. Contam. Toxicol. 2014, 67, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Salgueiro-Gonzalez, N.; Turnes-Carou, I.; Besada, V.; Muniategui-Lorenzo, S.; Lopez-Mahia, P.; Prada-Rodriguez, D. Occurrence, distribution and bioaccumulation of endocrine disrupting compounds in water, sediment and biota samples from a European river basin. Sci. Total Environ. 2015, 529, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Diao, P.; Chen, Q.; Wang, R.; Sun, D.; Cai, Z.; Wu, H.; Duan, S. Phenolic endocrine-disrupting compounds in the Pearl River Estuary: Occurrence, bioaccumulation and risk assessment. Sci. Total Environ. 2017, 584–585, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Little, D.C.; Newton, R.W.; Beveridge, M.C. Aquaculture: A rapidly growing and significant source of sustainable food? Status, transitions and potential. Proc. Nutr. Soc. 2016, 75, 274–286. [Google Scholar] [CrossRef] [PubMed]
- Lusher, A.L.; Hollman, P.C.H.; Mendoza-Hill, J.J. Microplastics in Fisheries and Aquaculture: Status of Knowledge on Their Occurrence and Implications for Aquatic Organisms and Food Safety; FAO Fisheries and Aquaculture Technical Paper. No. 615; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017; ISBN 978-92-5-109882-0. Available online: https://www.fao.org/3/i7677e/i7677e.pdf (accessed on 20 March 2022).
- Vázquez-Rowe, I.; Ita-Nagy, D.; Kahhat, R. Microplastics in fisheries and aquaculture: Implications to food sustainability and safety. Curr. Opin. Green Sust. 2021, 29, 100464. [Google Scholar] [CrossRef]
- Kolkovski, S.; Curnow, J.; King, J. Intensive rearing system for fish larvae research II. Artemia hatching and enriching system. Aquac. Eng. 2004, 31, 309–317. [Google Scholar] [CrossRef]
- Turcihan, G.; Turgay, E.; Yardımcı, R.E.; Eryalçın, K.M. The effect of feeding with different microalgae on survival, growth, and fatty acid composition of Artemia franciscana metanauplii and on predominant bacterial species of the rearing water. Aquacult. Int. 2021, 29, 2223–2241. [Google Scholar] [CrossRef]
- Sargent, J.; McEvoy, L.; Estevez, A.; Bell, G.; Bell, M.; Henderson, J.; Tocher, D. Lipid nutrition of marine fish during early development: Current status and future directions. Aquaculture 1999, 179, 217–229. [Google Scholar] [CrossRef]
- Triantaphyllidis, G.V.; Abatzopoulos, T.J.; Sorgeloos, P. Review of the biogeography of the genus Artemia (Crustacea. Anostraca). J. Biogeogr. 1998, 25, 213–226. [Google Scholar] [CrossRef]
- Maldonado-Montiel, T.D.N.J.; Rodriguez-Canche, L.G.; Olveranova, M.A. Evaluation of Artemia biomass production in San Crisanto, Yucatan, Mexico, with the use of poultry manure as organic fertilizer. Aquaculture 2003, 219, 573–584. [Google Scholar] [CrossRef]
- Kokalj, A.J.; Kunej, U.; Skalar, T. Screening study of four environmentally relevant microplastic pollutants: Uptake and effects on Daphnia magna and Artemia franciscana. Chemosphere 2018, 208, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, H.; Wang, J.; Li, J.; Liu, S.; Tu, J.; Chen, Y.; Zong, Y.; Zhang, P.; Wang, Z.; et al. Influence of Microplastics on the Growth and the Intestinal Microbiota Composition of Brine Shrimp. Front. Microbiol. 2021, 12, 717272. [Google Scholar] [CrossRef] [PubMed]
- Batel, A.; Linti, F.; Scherer, M.; Erdinger, L.; Braunbeck, T. Transfer of benzo[a]pyrene from microplastics to Artemia nauplii and further to zebrafish via a trophic food web experiment: CYP1A induction and visual tracking of persistent organic pollutants. Environ. Toxicol. Chem. 2016, 35, 1656–1666. [Google Scholar] [CrossRef] [PubMed]
- Nunes, B.S.; Carvalho, F.D.; Guilhermino, L.M.; Van Stappen, G. Use of the genus Artemia in ecotoxicity testing. Environ. Pollut. 2006, 144, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Libralato, G. The case of Artemia spp. in nanoecotoxicology. Mar. Environ. Res. 2014, 101, 38–43. [Google Scholar] [CrossRef]
- Castritsi-Catharios, J.; Syriou, V.; Miliou, H.; Zouganelis, G.D. Toxicity effects of bisphenol A to the nauplii of the brine shrimp Artemia franciscana. J. Biol. Res. Thessalon. 2013, 19, 38–45. [Google Scholar]
- Ekonomou, G.; Lolas, A.; Castritsi-Catharios, J.; Neofitou, C.; Zouganelis, G.D.; Tsiropoulos, N.; Exadactylos, A. Mortality and Effect on Growth of Artemia franciscana Exposed to Two Common Organic Pollutants. Water 2019, 11, 1614. [Google Scholar] [CrossRef]
- Shaukat, A.; Liu, G.; Li, Z.; Xu, D.; Huang, T.; Chen, H. Toxicity of five phenolic compounds to brine shrimp Artemia sinica (Crustacea: Artemiidae). J. Ocean Univ. China 2014, 13, 141–145. [Google Scholar] [CrossRef]
- Kidd, K.A.; Paterson, M.J.; Rennie, M.D.; Podemski, C.L.; Findlay, D.L.; Blanchfield, P.J.; Liber, K. Direct and indirect responses of a freshwater food web to a potent synthetic oestrogen. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130578. [Google Scholar] [CrossRef]
- Jeong, T.Y.; Simpson, M.J. Endocrine Disruptor Exposure Causes Infochemical Dysregulation and an Ecological Cascade from Zooplankton to Algae. Environ. Sci. Technol. 2021, 55, 3845–3854. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros-Gómez, A.; Rubio, S.; Pérez-Bendito, D. Analytical methods for the determination of bisphenol A in food. J. Chromatogr. A 2009, 1216, 449–469. [Google Scholar] [CrossRef] [PubMed]
- Corrales, J.; Kristofco, L.A.; Steele, W.B.; Yates, B.S.; Breed, C.S.; Williams, E.S.; Brooks, B.W. Global Assessment of Bisphenol A in the Environment: Review and Analysis of Its Occurrence and Bioaccumulation. Dose-Response 2015, 13, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Li, Y.; Ouyang, H.; Xu, P.; Wu, D. High-performance liquid chromatographic analysis of bisphenol A and 4-nonylphenol in serum, liver and testis tissues after oral administration to rats and its application to toxicokinetic study. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2006, 830, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Samanidou, V.F.; Frysali, M.A.; Papadoyannis, I.N. Matrix solid phase dispersion for the extraction of bisphenol A from human breast milk prior to HPLC analysis. J. Liq. Chromatogr. Relat. Technol. 2014, 37, 247–258. [Google Scholar] [CrossRef]
- Samanidou, V.F.; Kerezoudi, C.; Tolika, E.; Palaghias, G. A Simple Isocratic HPLC Method for the Simultaneous Determination of the Five Most Common Residual Monomers Released from Resin-Based Dental Restorative Materials. J. Liq. Chromatogr. Relat. Technol. 2015, 38, 740–749. [Google Scholar] [CrossRef]
- Samanidou, V.; Hadjicharalampous, M.; Palaghias, G.; Papadoyannis, I. Development and validation of an isocratic HPLC method for the simultaneous determination of residual monomers released from dental polymeric materials in artificial saliva. J. Liq. Chromatogr. Relat. Technol. 2012, 35, 511–523. [Google Scholar] [CrossRef]
- Diamantopoulou, E.I.; Plastiras, O.E.; Mourouzis, P.; Samanidou, V. Validation of a Simple HPLC-UV Method for the Determination of Monomers Released from Dental Resin Composites in Artificial Saliva. Methods Protoc. 2020, 3, 35. [Google Scholar] [CrossRef]
- Liu, J.; Pan, X.; Huang, B.; Fang, K.; Wang, Y.; Gao, J. An improved method for simultaneous analysis of steroid and phenolic endocrine disrupting chemicals in biological samples. J. Environ. Anal. Chem. 2012, 92, 1135–1149. [Google Scholar] [CrossRef]
- Asimakopoulos, A.G.; Thomaidis, N.S.; Koupparis, M.A. Recent trends in biomonitoring of bisphenol A, 4-t-octylphenol, and 4-nonylphenol. Toxicol. Lett. 2012, 210, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Preindl, K.; Braun, D.; Aichinger, G.; Sieri, S.; Fang, M.; Marko, D.; Warth, B. A Generic Liquid Chromatography-Tandem Mass Spectrometry Exposome Method for the Determination of Xenoestrogens in Biological Matrices. Anal. Chem. 2019, 91, 11334–11342. [Google Scholar] [CrossRef] [PubMed]
- Aznar, R.; Albero, B.; Pérez, R.A.; Sánchez-Brunete, C.; Miguel, E.; Tadeo, J.L. Analysis of emerging organic contaminants in poultry manure by gas chromatography-tandem mass spectrometry. J. Sep. Sci. 2018, 41, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Staniszewska, M.; Koniecko, I.; Falkowska, L.; Krzymyk, E. Occurrence and distribution of bisphenol A and alkylphenols in the water of the gulf of Gdansk (southern Baltic). Mar. Pollut. Bull. 2015, 91, 372–379. [Google Scholar] [CrossRef]
- Staniszewska, M.; Graca, B.; Sokołowski, A.; Nehring, I.; Wasik, A.; Jendzul, A. Factors determining accumulation of bisphenol A and alkylphenols at a low trophic level as exemplified by mussels Mytilus trossulus. Environ. Pollut. 2017, 220, 1147–1159. [Google Scholar] [CrossRef]
- Staniszewska, M.; Nehring, I.; Mudrak- Cegiołka, S. Changes of concentrations and possibility of accumulation of bisphenol A and alkylphenols, depending on biomass and composition, in zooplankton of the Southern Baltic (Gulf of Gdansk). Environ. Pollut. 2016, 10, 489–501. [Google Scholar] [CrossRef]
- Rigopoulos, A.T.; Samanidou, V.F.; Touraki, M. Development and Validation of an HPLC-DAD Method for the Simultaneous Extraction and Quantification of Bisphenol-A, 4-Hydroxybenzoic Acid, 4-Hydroxyacetophenone and Hydroquinone in Bacterial Cultures of Lactococcus lactis. Separations 2018, 5, 12. [Google Scholar] [CrossRef]
- Samat, N.A.; Yusoff, F.M.; Rasdi, N.W.; Karim, M. Enhancement of Live Food Nutritional Status with Essential Nutrients for Improving Aquatic Animal Health: A Review. Animals 2020, 10, 2457. [Google Scholar] [CrossRef]
- Varó, I.; Serrano, R.; Pitarch, E.; Amat, F.; López, F.J.; Navarro, J.C. Bioaccumulation of chlorpyrifos through an experimental food chain: Study of protein HSP70 as biomarker of sublethal stress in fish. Arch. Environ. Contam. Toxicol. 2002, 42, 229–235. [Google Scholar] [CrossRef]
- Correa-Reyes, G.; Viana, M.T.; Marquez-Rocha, F.J.; Licea, A.F.; Ponce, E.; Vazquez-Duhalt, R. Nonylphenol algal bioaccumulation and its effect through the trophic chain. Chemosphere 2007, 68, 662–670. [Google Scholar] [CrossRef]
- Dorn, P.B.; Chou, C.S.; Getempo, J.J. Degradation of Bisphenol A in Natural Waters. Chemosphere 1987, 16, 1501–1507. [Google Scholar] [CrossRef]
- Chapin, R.E.; Adams, J.; Boekelheide, K.; Gray, L.E., Jr.; Hayward, S.W.; Lees, P.S.; McIntyre, B.S.; Portier, K.M.; Schnorr, T.M.; Selevan, S.G.; et al. NTP-CERHR expert panel report on the reproductive and developmental toxicity of bisphenol A. Birth Defects Res. B Dev. Reprod. Toxicol. 2008, 83, 157–395. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA J. 2015, 13, 3978. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 8814, 4-tert-Octylphenol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/4-tert-Octylphenol (accessed on 14 February 2022).
- Jonsson, B. Risk Assessment on Butylphenol, Octylphenol and Nonylphenol, and Estimated Human Exposure of Alkylphenols from Swedish Fish. Uppsala Uppsala University. 2006. Available online: https://www.uu.se/digitalAssets/177/c_177024-l_3-k_jonsson-beatrice-report.pdf (accessed on 22 April 2022).
- Profiles of the Initial Environmental Risk Assessment of Chemicals Vol. 7, Ministry of the Environment, Government. Available online: https://www.env.go.jp/en/chemi/chemicals/profile_erac/index.html#vol7 (accessed on 22 April 2022).
- Touraki, M.; Karamanlidou, G.; Karavida, P.; Chrysi, K. Evaluation of the probiotics Bacillus subtilis and Lactobacillus plantarum bioencapsulated in Artemia nauplii against vibriosis in European sea bass larvae (Dicentrarchus labrax, L.). World J. Microbiol. Biotechnol. 2012, 28, 2425–2433. [Google Scholar] [CrossRef]
- Giarma, E.; Amanetidou, E.; Toufexi, A.; Touraki, M. Defense systems in developing Artemia franciscana nauplii and their modulation by probiotic bacteria offer protection against a Vibrio anguillarum challenge. Fish Shellfish Immunol. 2017, 66, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.N.; Lindholst, C. Quantification of the xenoesterogens 4-tert-octylphenol and bisphenol A in water and in fish tissue based on microwave assisted extraction, solid-phase extraction and liquid chromatography-mass spectrometry. J. Chromatogr. A 1999, 864, 17–24. [Google Scholar] [CrossRef]
- Singh, B.; Kumar, A.; Malik, A.K. Recent advances in sample preparation methods for analysis of endocrine disruptors from various matrices. Crit. Rev. Anal. Chem. 2014, 44, 255–269. [Google Scholar] [CrossRef]
- Cerkvenik-Flajs, V.; Fonda, I.; Gombač, M. Analysis and Occurrence of Bisphenol A in Mediterranean Mussels (Mytilus galloprovincialis) Sampled from the Slovenian Coastal Waters of the North Adriatic Sea. Bull. Environ. Contam. Toxicol. 2018, 101, 439–445. [Google Scholar] [CrossRef]
- Völkel, W.; Colnot, T.; Csanady, G.; Filser, J.G.; Dekant, W. Metabolism and kinetics of bisphenol A in humans at low doses following oral administration. Chem. Res. Toxicol. 2002, 15, 1281–1287. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.W.; Chen, C.F. Seasonal and spatial distribution of 4-nonylphenol and 4-tert-octylphenol in the sediment of Kaohsiung Harbor, Taiwan. Chemosphere 2015, 134, 588–597. [Google Scholar] [CrossRef]
- Pisciottano, I.D.M.; Mita, G.D.; Gallo, P. Bisphenol A, octylphenols and nonylphenols in fish muscle determined by LC/ESI-MS/MS after affinity chromatography clean up. Food Addit. Contam. Part B 2020, 13, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Cañadas, R.; Garrido Gamarro, E.; Garcinuño Martínez, R.M.; Paniagua González, G.; Fernández Hernando, P. Occurrence of common plastic additives and contaminants in mussel samples: Validation of analytical method based on matrix solid-phase dispersion. Food Chem. 2021, 349, 129169. [Google Scholar] [CrossRef] [PubMed]
- Basheer, C.; Lee, H.K.; Tan, K.S. Endocrine disrupting alkylphenols and bisphenol-A in coastal waters and supermarket seafood from Singapore. Mar. Pollut. Bull. 2004, 48, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Uhrovčík, J. Strategy for determination of LOD and LOQ values—Some basic aspects. Talanta 2014, 119, 178–180. [Google Scholar] [CrossRef] [PubMed]
- McMillan, J. Principles of analytical validation. In Proteomic Profiling and Analytical Chemistry: The Crossroads, 2nd ed.; Ciborowski, P., Silberring, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 205–215. [Google Scholar] [CrossRef]
- Shaukat, A.; Guangxing, L.; Zhengyan, L. The Acute Toxicity of Phenolic Compounds and Heavy Metals on Brine Shrimp Artemia sinica (Crustacea: Artemiidae). Sindh Univ. Res. J. 2013, 45, 21–24. [Google Scholar]
- Rajasree, S.R.R.; Kumar, V.G.; Abraham, L.S.; Manoharan, N. Assessment on the toxicity of engineered nanoparticles on the life stages of marine aquatic invertebrate Artemia salina. Int. J. Nanosci. 2011, 10, 1153–1159. [Google Scholar] [CrossRef]
- Mtibaà, R.; Ezzanad, A.; Aranda, E.; Pozo, C.; Ghariani, B.; Moraga, J.; Nasri, M.; Manuel Cantoral, J.; Garrido, C.; Mechichi, T. Biodegradation and toxicity reduction of nonylphenol, 4-tert-octylphenol and 2,4-dichlorophenol by the ascomycetous fungus Thielavia sp HJ22: Identification of fungal metabolites and proposal of a putative pathway. Sci. Total Environ. 2020, 708, 135129. [Google Scholar] [CrossRef]
- Olaniyan, L.W.B.; Okoh, A.I. Determination and ecological risk assessment of two endocrine disruptors from River Buffalo, South Africa. Environ. Monit. Assess. 2020, 192, 750. [Google Scholar] [CrossRef]
- Gurban, A.M.; Burtan, D.; Rotariu, L.; Bala, C. Manganese oxide based screen-printed sensor for xenooestrogens detection. Sens. Actuators B Chem. 2015, 210, 273–280. [Google Scholar] [CrossRef]
- Neamtu, M.; Frimmel, F.H. Degradation of endocrine disrupting bisphenol A by 254 nm irradiation in different water matrices and effect on yeast cells. Water Res. 2006, 40, 3745–3750. [Google Scholar] [CrossRef]
- Wu, Y.; Yuan, H.; Wei, G.; Zhang, S.; Li, H.; Dong, W. Photodegradation of 4-tert octylphenol in aqueous solution promoted by Fe (III). Environ. Sci. Pollut. Res. Int. 2013, 20, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, K.; Nakajima, N. Improvement of marine environmental pollution using eco-system: Decomposition and recovery of endocrine disrupting chemicals by marine phyto- and zooplanktons. J. Mol. Catal. B 2003, 23, 419–424. [Google Scholar] [CrossRef]
- Ocharoen, Y.; Boonphakdee, C.; Boonphakdee, T.; Shinn, A.P.; Moonmangmee, S. High levels of the endocrine disruptors bisphenol-A and 17β-estradiol detected in populations of green mussel, Perna viridis, cultured in the Gulf of Thailand. Aquaculture 2018, 497, 348–356. [Google Scholar] [CrossRef]
- Lu, I.-C.; Chao, H.-R.; Mansor, W.-N.-W.; Peng, C.-W.; Hsu, Y.-C.; Yu, T.-Y.; Chang, W.-H.; Fu, L.-M. Levels of Phthalates, Bisphenol-A, Nonylphenol, and Microplastics in Fish in the Estuaries of Northern Taiwan and the Impact on Human Health. Toxics 2021, 9, 246. [Google Scholar] [CrossRef]
- Yap, C.K. Contamination of Heavy Metals and Other Organic Pollutants in Perna Viridis from the Coastal Waters of Malaysia: A Review Based On 1998 Data. J. Sci. Res. Rep. 2014, 3, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Arditsoglou, A.; Voutsa, D. Occurrence and partitioning of endocrine-disrupting compounds in the marine environment of Thermaikos Gulf, Northern Aegean Sea, Greece. Mar. Pollut. Bull. 2012, 64, 2443–2452. [Google Scholar] [CrossRef]
- Giebichenstein, J.; Giebichenstein, J.; Hasler, M.; Schulz, C.; Ueberschär, B. Comparing the performance of four commercial microdiets in an early weaning protocol for European seabass larvae (Dicentrarchus labrax). Aquac. Res. 2022, 53, 544–558. [Google Scholar] [CrossRef]
- Capó, X.; Alomar, C.; Compa, M.; Sole, M.; Sanahuja, I.; Soliz Rojas, D.L.; González, G.P.; Garcinuño Martínez, R.M.; Deudero, S. Quantification of differential tissue biomarker responses to microplastic ingestion and plasticizer bioaccumulation in aquaculture reared sea bream Sparus aurata. Environ. Res. 2022, 211, 113063. [Google Scholar] [CrossRef]
| Compound (Abbreviation, CAS#, Formula) | Structure | MW | Solubility in Water (mg L−1) | LD50 g kg−1 | TDI (Tolerable Daily Intake) ng/kg bw/day |
|---|---|---|---|---|---|
| Bisphenol-A BPA, 80-05-7 C15H16O2 | 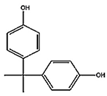 | 240.20 | 120 [66] | 3-5 (rat) [67] | 4000 (human) [68] |
| 4-tert-Octylphenol t-OCT, 140-66-9 C14H22O | 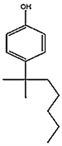 | 206.32 | 5.1 [69] | 4.6 (rat) [69] | 0.067 (men) 33.3 (women) [70] |
| 4-n-Octylphenol n-OCT, 1806-26-4 C14H22O | 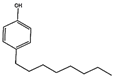 | 206.32 | 3.1 [71] | 87.8 µg L−1 (fish) [71] | - |
| Analyte Sample Std. Curve /R2 | Nominal Conc. (μg/mL) | Calculated Conc. (μg/mL) (Mean ± SD) | Relative Bias (%) | Precision | Recovery (%) | ||
|---|---|---|---|---|---|---|---|
| Intra- (n = 3) | Inter- (n = 2 × 3) | (Mean ± SD) | RSD% | ||||
| BPA culture medium y = 0.0027x − 0.024 0.9998 | 1 | 0.94 ± 0.002 | −5.3 | 0.2 | 0.3 | 94.8 ± 0.3 | 0.3 |
| 25 | 24.86 ± 0.1 | −0.6 | 0.4 | 0.5 | 99.4 ± 0.4 | 0.4 | |
| 50 | 50.6 ± 0.3 | 1.2 | 0.6 | 0.7 | 101.2 ± 0.6 | 0.6 | |
| 75 | 74.8 ± 0.09 | −0.2 | 0.1 | 0.2 | 99.8 ± 0.1 | 0.1 | |
| 100 | 100.2 ± 0.6 | 0.2 | 0.6 | 0.7 | 100.2 ± 0.6 | 0.6 | |
| 125 | 123.7 ± 1.8 | −1.1 | 0.4 | 1.4 | 98.9 ± 1.4 | 1.4 | |
| BPA Artemia y = 0.045x − 0.0887 0.999 | 2.5 | 2.64 ± 0.02 | 5.9 | 0.4 | 0.6 | 105.6 ± 0.6 | 0.6 |
| 5 | 4.81 ± 0.05 | −3.8 | 0.8 | 1.0 | 96.3 ± 0.9 | 0.9 | |
| 7.5 | 7.61 ± 0.02 | 1.3 | 0.1 | 0.3 | 101.4 ± 0.3 | 0.3 | |
| 10 | 9.86 ± 0.02 | 0.1 | 0.2 | 0.7 | 98.6 ± 0.2 | 0.2 | |
| 12.5 | 12.45 ± 0.03 | −0.4 | 0.1 | 0.2 | 99.6 ± 0.2 | 0.2 | |
| 15 | 15.11 ± 0.02 | 0.7 | 0.03 | 0.1 | 100.7 ± 0.1 | 0.1 | |
| t-OCT culture medium y = 0.018x + 0.037 0.999 | 1 | 1.03 ± 0.04 | 3.1 | 0.6 | 4.3 | 103.1 ± 4.5 | 4.3 |
| 2.5 | 2.60 ± 0.04 | 4.1 | 0.5 | 1.6 | 104.1 ± 1.7 | 1.6 | |
| 5 | 5.05 ± 0.03 | 1.1 | 0.03 | 0.7 | 101.1 ± 0.7 | 0.7 | |
| 7.5 | 7.69 ± 0.02 | 2.5 | 0.1 | 0.2 | 102.5 ± 0.2 | 0.2 | |
| 10 | 9.87 ± 0.03 | −1.2 | 0.1 | 0.3 | 98.8 ± 0.3 | 0.3 | |
| 12.5 | 12.75 ± 0.1 | 2.0 | 0.7 | 0.9 | 102.0± 0.9 | 0.9 | |
| t-OCT Artemia y = 0.019x + 0.012 0.9989 | 1 | 1.08 ± 0.02 | 8.7 | 0.01 | 1.9 | 108.7 ± 2.0 | 1.9 |
| 2.5 | 2.41 ± 0.01 | −3.4 | 0.5 | 0.6 | 96.6 ± 0.6 | 0.6 | |
| 5 | 4.85 ± 0.09 | −2.8 | 0.4 | 1.8 | 97.2 ± 1.8 | 1.8 | |
| 7.5 | 7.59 ± 0.03 | 1.3 | 0.1 | 0.4 | 101.3 ± 0.4 | 0.4 | |
| 10 | 9.98 ± 0.02 | −0.2 | 0.3 | 0.3 | 99.8 ± 0.2 | 0.2 | |
| 12.5 | 12.49± 0.2 | 0.2 | 0.4 | 1.8 | 100.2 ± 1.8 | 1.8 | |
| Std. Curve | Retention Time (min) Mean ± SD (RSD %) | LOD (μg/mL) | LOQ (μg/mL) |
|---|---|---|---|
| BPA in Artemia | 15.200 ± 0.12 (0.82) | 0.21 | 0.65 |
| BPA in culture medium | 15.310 ± 0.10 (0.69) | 0.63 | 1.92 |
| t-OCT in Artemia | 21.876 ± 0.11 (0.49) | 0.17 | 0.52 |
| t-OCT in culture medium | 21.597 ± 0.31 (1.46) | 0.41 | 1.25 |
| Compound | BPA | t-OCT |
|---|---|---|
| Administered in 1000 mL culture (μg) | 10,000.0 | 500.0 |
| Found in Artemia (μg/g wet weight) | 68.3 | 5.9 |
| Found in culture medium (μg/mL) | 6.68 | 0.3 |
| Total found in Artemia (μg) | 40.9 | 3.6 |
| Total found in 1000 mL medium (μg) | 6680 | 319.3 |
| Total found (μg) | 6720.9 | 322.9 |
| Concentration reduction in cultures (%) | 32.8 | 35.4 |
| Decline due to Photolysis (%) | 9.2 | 19.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giamaki, D.; Dindini, K.; Samanidou, V.F.; Touraki, M. Simultaneous Quantification of Bisphenol-A and 4-Tert-Octylphenol in the Live Aquaculture Feed Artemia franciscana and in Its Culture Medium Using HPLC-DAD. Methods Protoc. 2022, 5, 38. https://doi.org/10.3390/mps5030038
Giamaki D, Dindini K, Samanidou VF, Touraki M. Simultaneous Quantification of Bisphenol-A and 4-Tert-Octylphenol in the Live Aquaculture Feed Artemia franciscana and in Its Culture Medium Using HPLC-DAD. Methods and Protocols. 2022; 5(3):38. https://doi.org/10.3390/mps5030038
Chicago/Turabian StyleGiamaki, Despoina, Konstantina Dindini, Victoria F. Samanidou, and Maria Touraki. 2022. "Simultaneous Quantification of Bisphenol-A and 4-Tert-Octylphenol in the Live Aquaculture Feed Artemia franciscana and in Its Culture Medium Using HPLC-DAD" Methods and Protocols 5, no. 3: 38. https://doi.org/10.3390/mps5030038
APA StyleGiamaki, D., Dindini, K., Samanidou, V. F., & Touraki, M. (2022). Simultaneous Quantification of Bisphenol-A and 4-Tert-Octylphenol in the Live Aquaculture Feed Artemia franciscana and in Its Culture Medium Using HPLC-DAD. Methods and Protocols, 5(3), 38. https://doi.org/10.3390/mps5030038








