Newborn Screening with (C16 + C18:1)/C2 and C14/C3 for Carnitine Palmitoyltransferase II Deficiency throughout Japan Has Revealed C12/C0 as an Index of Higher Sensitivity and Specificity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Screening Test for CPT II Deficiency
2.2. Confirmatory Tests for CPT II Deficiency
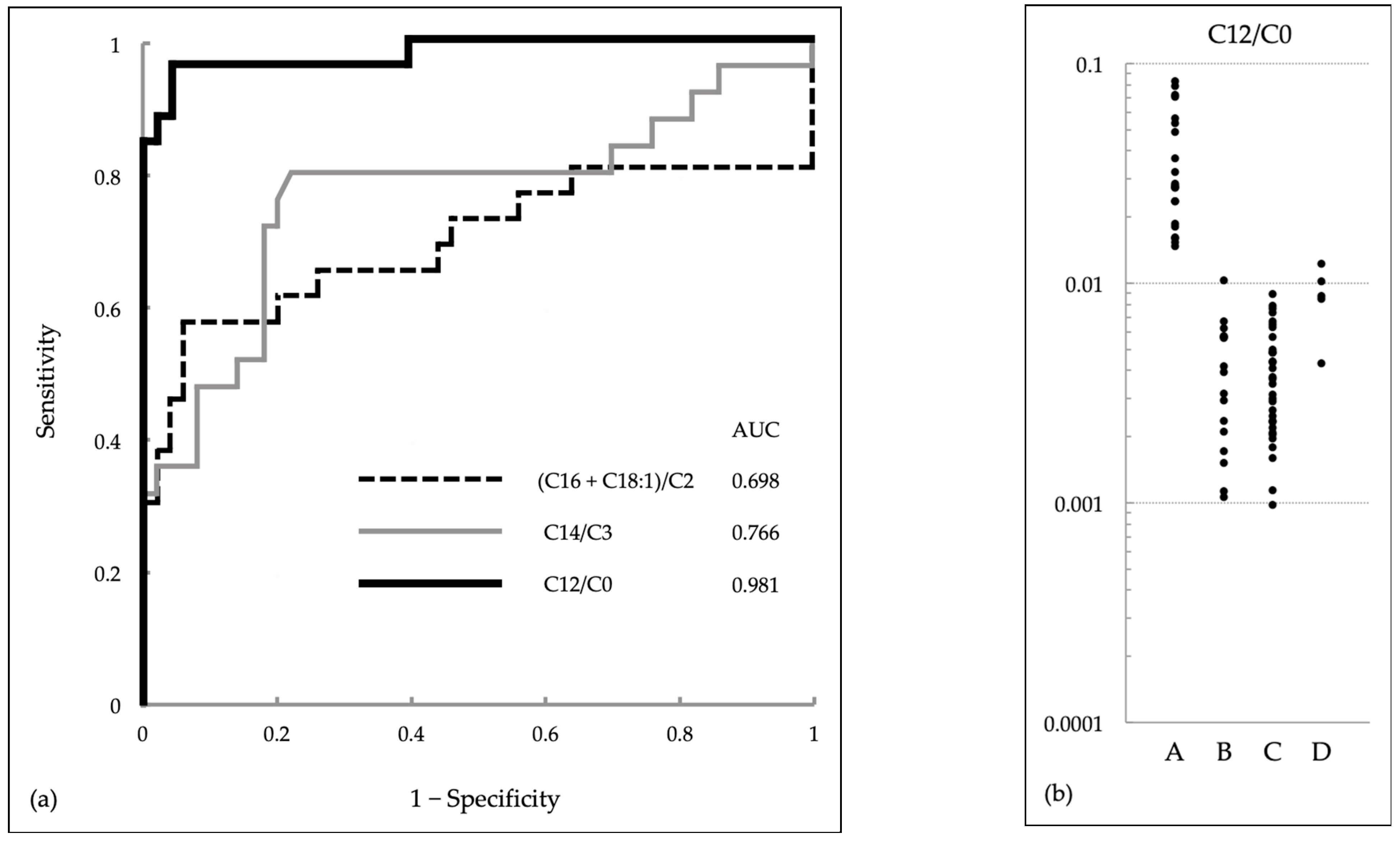
2.3. Receiver Operating Characteristic Analysis
3. Results
3.1. Diagnoses of NBS Positivity and Levels of the Current Indices
3.2. Patients with Symptoms of CPT II Deficiency but Normal NBS Results
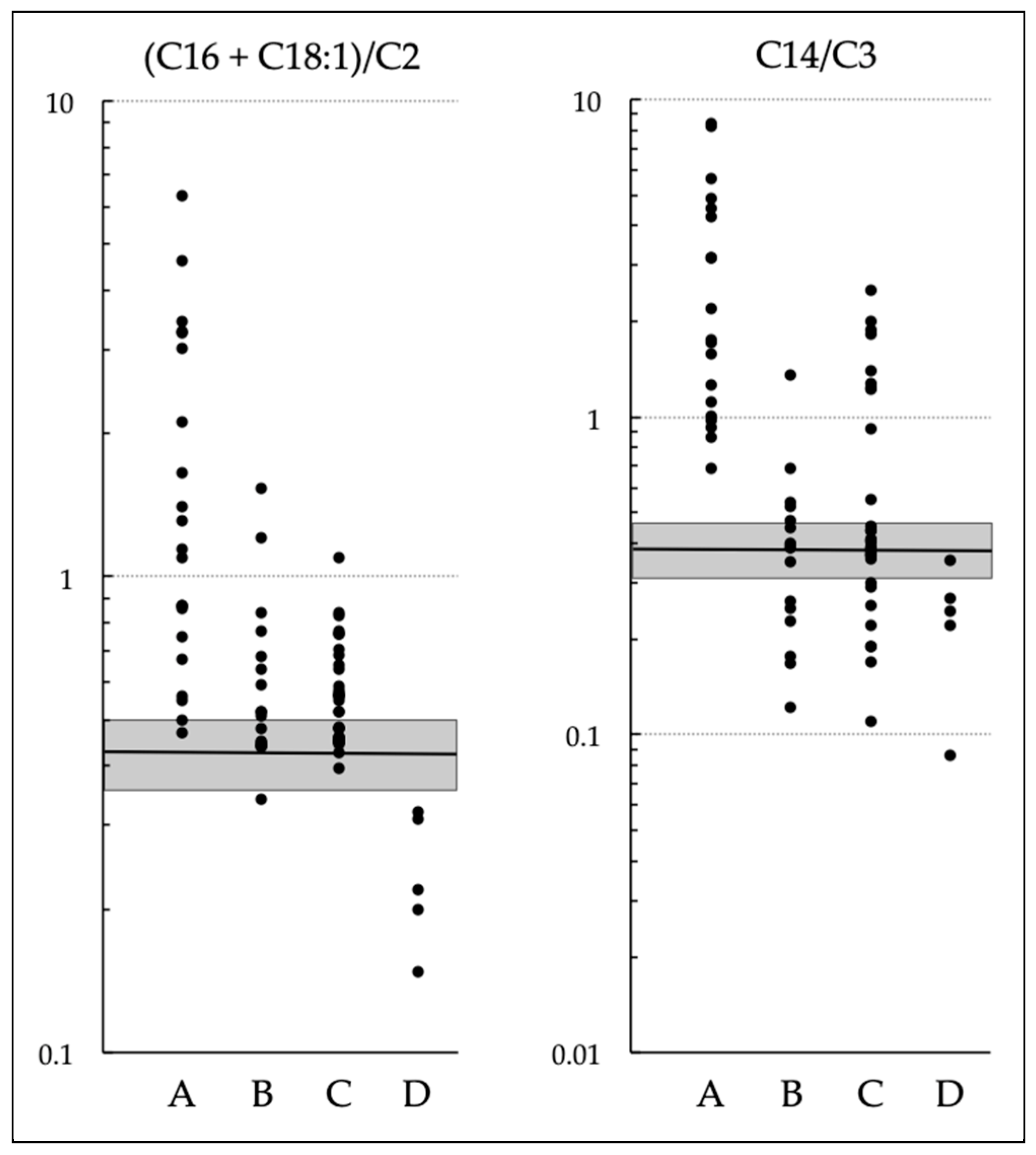
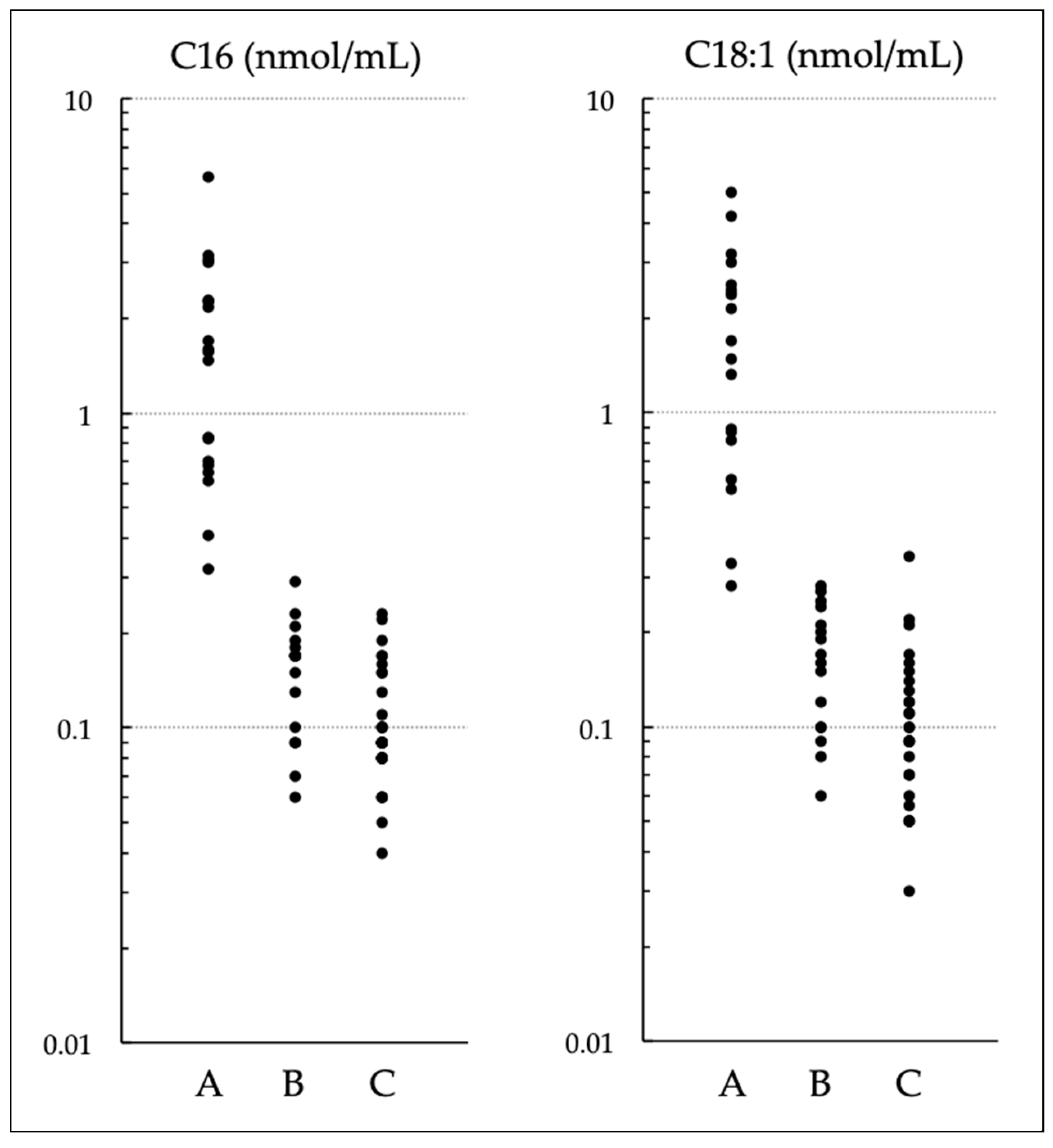
3.3. Comparison of (C16 + C18:1)/C2 and C14/C3 with Various Acylcarnitines and Their Ratios
3.4. Results of Serum Acylcarnitine Analysis at the First Medical Examination
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Joshi, P.R.M.; Zierz, S. Carnitine palmitoyltransferase II (CPT II) deficiency: Genotype–phenotype analysis of 50 patients. J. Neurol. Sci. 2014, 338, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Yasuno, T.; Kaneoka, H.; Tokuyasu, T.; Aoki, J.; Yoshida, S.; Takayanagi, M.; Ohtake, A.; Kanazawa, M.; Ogawa, A.; Tojo, K.; et al. Mutations of carnitine palmitoyltransferase II (CPT II) in Japanese patients with CPT II deficiency. Clin. Genet. 2008, 73, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Ishikawa, N.; Tsumura, M.; Fjii, Y.; Okada, S.; Shigematsu, Y.; Kobayashi, M. Acute severe encephalopathy related to human herpesvirus-6 infection in a patient with carnitine palmitoyltransferase 2 deficiency carrying thermolabile variants. Brain Dev. 2013, 35, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Tajima, G.; Hara, K.; Tsumura, M.; Kagawa, R.; Okada, S.; Sakura, N.; Maruyama, S.; Noguchi, A.; Awaya, T.; Ishige, M.; et al. Newborn screening for carnitine palmitoyltransferase II deficiency using (C16 + C18:1)/C2: Evaluation of additional indices for adequate sensitivity and lower false-positivity. Mol. Genet. Metab. 2017, 122, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, A.C.; Salant, J.; Ierardi-Curto, L.A.; Ficicioglu, C. Missed newborn screening case of carnitine palmitoyltransferase-II deficiency. JIMD Rep. 2017, 33, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Tajima, G.; Hara, K.; Yuasa, M. Carnitine palmitoyltransferase II deficiency with a focus on newborn screening. J. Hum. Genet. 2019, 64, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, M.; Hata, I.; Sugihara, K.; Isozaki, Y.; Ohshima, Y.; Hara, K.; Tajima, G.; Shigematsu, Y. Evaluation of metabolic defects in fatty acid oxidation using peripheral blood mononuclear cells loaded with deuterium-labeled fatty acids. Dis. Markers 2019, 2984747. [Google Scholar] [CrossRef] [PubMed]
- Marsden, D.; Bedrosian, C.L.; Vockley, J. Impact of newborn screening on the reported incidence and clinical outcomes associated with medium- and long-chain fatty acid oxidation disorders. Genet. Med. 2021, 23, 816–829. [Google Scholar] [CrossRef] [PubMed]
- Lindner, M.; Hoffmann, G.F.; Matern, D. Newborn screening for disorders of fatty-acid oxidation: Experience and recommendations from an expert meeting. J. Inherit. Metab. Dis. 2010, 33, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Mador-House, R.; Liu, Z.; Dyack, S. Detection of early onset carnitine palmitoyltransferase II deficiency by newborn screening: Should CPT II deficiency be a primary disease target? Int. J. Neonatal Screen. 2021, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Cheng, Y.; Yin, X.; Miao, H.; Hu, Z.; Yang, J.; Zhang, Y.; Wu, B.; Huang, X. Newborn screening for mitochondrial carnitine-acylcarnitine cycle disorders in Zhejiang Province, China. Front. Genet. 2022, 13, 823687. [Google Scholar] [CrossRef] [PubMed]
- Tamaoki, Y.; Kimura, M.; Hasegawa, Y.; Iga, M.; Inoue, M.; Yamaguchi, S. A survey of Japanese patients with mitochondrial fatty acid b-oxidation and related disorders as detected from 1985 to 2000. Brain Dev. 2002, 24, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Wataya, K.; Akanuma, J.; Cavadini, P.; Aoki, Y.; Kure, S.; Invernizzi, F.; Yoshida, I.; Kira, J.; Taroni, F.; Matsubara, Y.; et al. Two CPT2 mutations in three Japanese patients with carnitine palmitoyltransferase II deficiency: Functional analysis and associateon with polymorphic haplotypes and two clinical phenotypes. Hum. Mutat. 1998, 11, 377–386. [Google Scholar] [CrossRef]
- Osawa, Y.; Kobayashi, H.; Tajima, G.; Hara, K.; Yamada, K.; Fukuda, S.; Hasegawa, Y.; Aisaki, J.; Yuasa, M.; Hata, I.; et al. The frequencies of very long-chain acyl-CoA dehydrogenase deficiency genetic variants in Japan have changed since the implementation of expanded newborn screening. Mol. Genet. Metab. 2022, 136, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.J.; Burton, B.K. Diagnosis and clinical management of long-chain fatty-acid oxidation disorders: A review. touchREV Endocrinol. 2021, 17, 108–111. [Google Scholar] [CrossRef] [PubMed]
| Groups and Descriptions | n | Case ID * | CPT II Activity |
|---|---|---|---|
| Group A: NBS-positive, patients with CPT II deficiency | 21 | S-01, N-01 to N-20 | 1.2–27.9% |
| Group B-1: NBS-positive, carriers heterozygous for pathogenic CPT2 variants | 2 | N-21, N-37 | 31.8%, 70.8% |
| Group B-2: NBS-positive, homozygous for p.F352C | 3 | N-22, N-26, N-35 | 34.0%, 52.0%, 63.8% |
| Group B-3: NBS-positive, CPT II activity between 31.8% and 70.8% without genetic confirmation of either heterozygosity for pathogenic variants or homozygosity for p.F352C | 12 | N-23 to N-25, N-27 to N-34, N-36 | 37.7–67.4% |
| Group C: NBS-positive infants with normal-level CPT II activity | 33 | N-38 to N-70 | 78.1–410.4% (mean value = 152.2% ± 83.8%) |
| Group D: Patients with symptoms of CPT II deficiency but normal results in NBS | 5 | S-02 to S-06 | 8.1–30.2% |
| Characteristic | Acylcarnitine and Free Carnitine (C0) Levels | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C16 | C18:1 | C18 | C16-OH | C14 | C14:1 | C12 | C10 | C8 | C6 | C4 | C3 | C2 | C0 | |
| Optimal cutoff (nmol/mL) | 9.8 | 2.56 | 1.695 | 0.027 | 0.295 | 0.106 | 0.155 | 0.085 | 0.095 | 0.02 | 0.111 | 0.55 | 5.61 | 20.225 |
| Sensitivity | 0.231 | 0.346 | 0.308 | 0.600 | 0.640 | 0.400 | 0.885 | 0.846 | 0.192 | 0.696 | 0.750 | 0.808 | 0.769 | 0.885 |
| Specificity | 1 | 0.980 | 0.980 | 0.872 | 0.900 | 0.851 | 0.938 | 0.646 | 0.917 | 0.425 | 0.475 | 0.480 | 0.640 | 0.620 |
| Area under the curve | 0.526 | 0.576 | 0.603 | 0.727 | 0.822 | 0.644 | 0.960 | 0.784 | 0.524 | 0.526 | 0.600 | 0.622 | 0.706 | 0.733 |
| Characteristic | Acylcarnitine Ratios | |||||||
|---|---|---|---|---|---|---|---|---|
| (C16 + C18:1)/C2 | C14/C3 | C14/C2 | C14/C0 | C12/C2 | C12/C0 | C10/C2 | C10/C0 | |
| Optimal cutoff | 0.857 | 0.620 | 0.066 | 0.018 | 0.020 | 0.008 | 0.017 | 0.005 |
| Sensitivity | 0.577 | 0.800 | 0.760 | 0.800 | 0.885 | 0.962 | 0.808 | 0.962 |
| Specificity | 0.940 | 0.780 | 1 | 0.940 | 0.979 | 0.958 | 0.896 | 0.667 |
| Area under the curve | 0.698 | 0.766 | 0.832 | 0.908 | 0.932 | 0.981 | 0.895 | 0.885 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tajima, G.; Hara, K.; Tsumura, M.; Kagawa, R.; Sakura, F.; Sasai, H.; Yuasa, M.; Shigematsu, Y.; Okada, S. Newborn Screening with (C16 + C18:1)/C2 and C14/C3 for Carnitine Palmitoyltransferase II Deficiency throughout Japan Has Revealed C12/C0 as an Index of Higher Sensitivity and Specificity. Int. J. Neonatal Screen. 2023, 9, 62. https://doi.org/10.3390/ijns9040062
Tajima G, Hara K, Tsumura M, Kagawa R, Sakura F, Sasai H, Yuasa M, Shigematsu Y, Okada S. Newborn Screening with (C16 + C18:1)/C2 and C14/C3 for Carnitine Palmitoyltransferase II Deficiency throughout Japan Has Revealed C12/C0 as an Index of Higher Sensitivity and Specificity. International Journal of Neonatal Screening. 2023; 9(4):62. https://doi.org/10.3390/ijns9040062
Chicago/Turabian StyleTajima, Go, Keiichi Hara, Miyuki Tsumura, Reiko Kagawa, Fumiaki Sakura, Hideo Sasai, Miori Yuasa, Yosuke Shigematsu, and Satoshi Okada. 2023. "Newborn Screening with (C16 + C18:1)/C2 and C14/C3 for Carnitine Palmitoyltransferase II Deficiency throughout Japan Has Revealed C12/C0 as an Index of Higher Sensitivity and Specificity" International Journal of Neonatal Screening 9, no. 4: 62. https://doi.org/10.3390/ijns9040062
APA StyleTajima, G., Hara, K., Tsumura, M., Kagawa, R., Sakura, F., Sasai, H., Yuasa, M., Shigematsu, Y., & Okada, S. (2023). Newborn Screening with (C16 + C18:1)/C2 and C14/C3 for Carnitine Palmitoyltransferase II Deficiency throughout Japan Has Revealed C12/C0 as an Index of Higher Sensitivity and Specificity. International Journal of Neonatal Screening, 9(4), 62. https://doi.org/10.3390/ijns9040062







