Considerations for Newborn Screening for Critical Congenital Heart Disease in Low- and Middle-Income Countries
Abstract
1. Introduction
2. CCHD Screening in LMICs
3. Recommendations for CCHD Screening in LMICs
- Without ready access to definitive diagnosis, safe transportation and high-quality surgical and interventional cardiology services, newborn screening cannot save as many lives as it should. It will help detect cases, but, unless these babies can access surgical services, many will not survive or will live a life with serious disability. It is critical for policy implementation to include an assessment of the existing infrastructure to assure effective utilization of all available resources, rather than considering making investments in new infrastructure. This would involve holding consultations with a broad representation of the whole health sector, including public and private healthcare providers, patients and families, policymakers and the biomedical industry.
- Prior to implementation, it is necessary to thoroughly investigate what the process of birth delivery in the country is and who would be best positioned to administer the test. This includes developing a good understanding of what the healthcare workers administering the test’s usual scope of practice is and what their upskilling and training needs would be.
- It is critical to collect data on the new process and to study it as it is being implemented, as well as on the quality of the surgical outcomes of the children detected through pulse oximetry screening. One of the biggest influences of newborn CCHD screening is that it finds children who were invisible before, providing a more accurate picture of the burden of disease, and this can create a strong impetus for downstream capacity development and infrastructure investments, which will ultimately benefit many more children with CHD. In the case of Kerala, the government developed a state-wide registration system, which helped with the subsequent data analysis.
- The implementing parties need to back their plans with robust budgetary considerations, as this is not a simple clinical practice change but rather a health policy decision affecting lifelong care for children with CCHD. We have found that this implementation is more successful when it is led by the government, given that health policy officials have the ability and experience mandating screenings for other diseases in the newborn period.
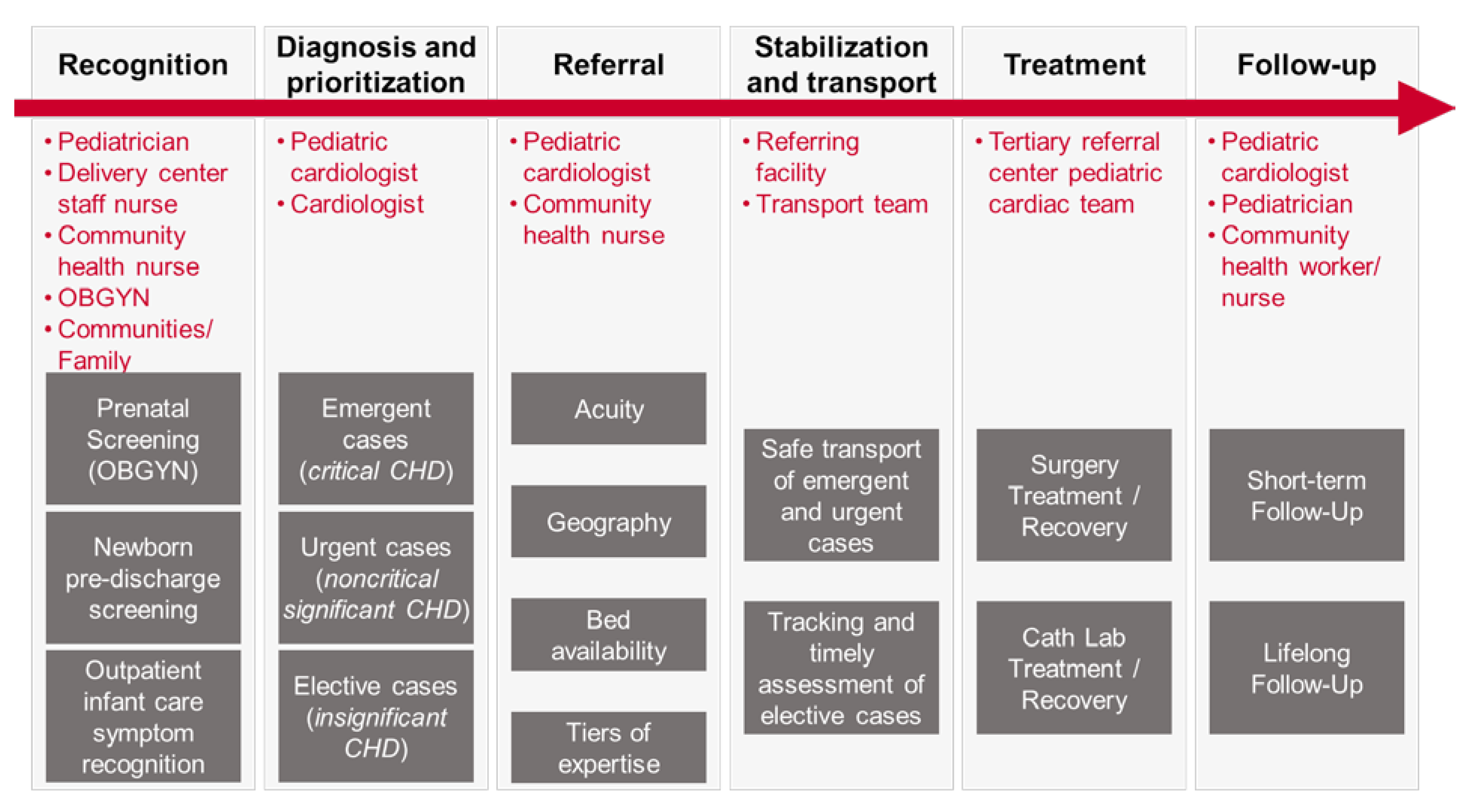
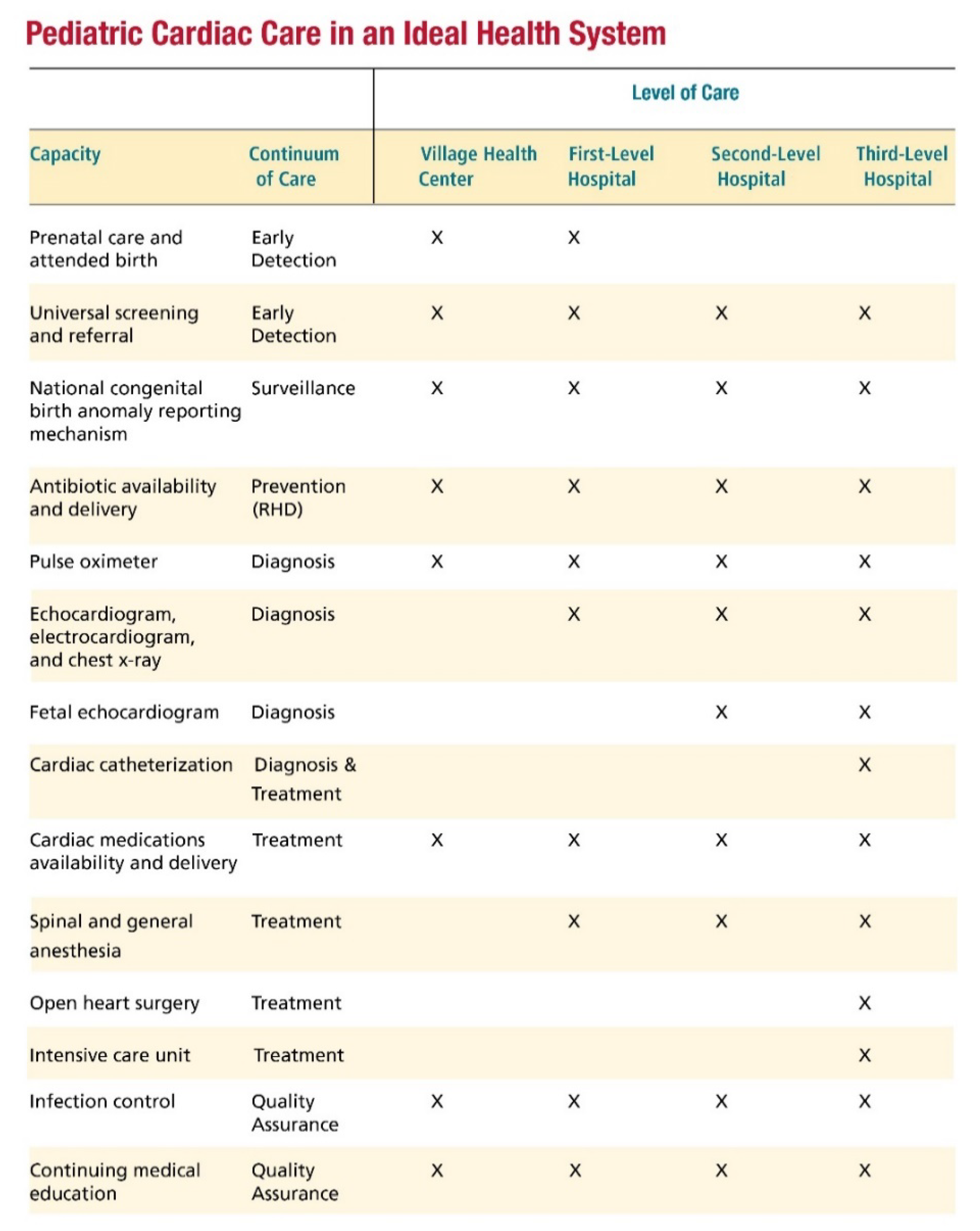
- Pulse oximetry CCHD screening in newborns should also ideally be considered as one of the steps to screen for CHD, given that there is no one method that can detect it all. A population health approach to CHD screening should also include antenatal screening and follow up screening at different points in the first several years of life, taking advantage of the frequent primary care interactions for regular vaccinations, for example. This recommendation is especially important for LMICs, where there are still significant numbers of undetected children with CHD. It is also critical to look at where CHD care is needed in the different levels of the healthcare system (Figure 2) [7].
Author Contributions
Funding
Conflicts of Interest
References
- Zimmerman, M.S.; Smith, A.G.C.; Sable, C.A.; Echko, M.M.; Wilner, L.B.; Olsen, H.E.; Atalay, H.T.; Awasthi, A.; Bhutta, Z.A.; Boucher, J.L.; et al. Global, Regional, and National Burden of Congenital Heart Disease, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet Child Adolesc. Health 2020, 4, 185–200. [Google Scholar] [CrossRef]
- United Nations Sustainable Development Goals (UN SDGs). Available online: https://www.un.org/sustainabledevelopment/sustainable-development-goals/ (accessed on 17 May 2020).
- Zühlke, L.; Vaidyanathan, B. Is It Time for Developing Countries to Adopt Neonatal Pulse Oximetry Screening for Critical Congenital Heart Disease? Cardiovasc. J. S. Afr. 2013, 10. [Google Scholar] [CrossRef]
- Van Niekerk, A.M.; Cullis, R.M.; Linley, L.L.; Zühlke, L. Feasibility of Pulse Oximetry Pre-Discharge Screening Implementation for Detecting Critical Congenital Heart Lesions in Newborns in a Secondary Level Maternity Hospital in the Western Cape, South Africa: The ‘POPSICLe’ Study. S. Afr. Med. J. 2016, 106, 817. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Ma, X.; Ge, X.; Liu, F.; Yan, W.; Wu, L.; Ye, M.; Liang, X.; Zhang, J.; Gao, Y.; et al. Pulse Oximetry with Clinical Assessment to Screen for Congenital Heart Disease in Neonates in China: A Prospective Study. Lancet 2014, 384, 747–754. [Google Scholar] [CrossRef]
- Hu, X.; Ma, X.; Zhao, Q.; Yan, W.; Ge, X.; Jia, B.; Liu, F.; Wu, L.; Ye, M.; Liang, X.; et al. Pulse Oximetry and Auscultation for Congenital Heart Disease Detection. Pediatrics 2017, 140, e20171154. [Google Scholar] [CrossRef] [PubMed]
- The Invisible Child. Finding the Invisible Child: Childhood Heart Disease and the Global Health Agenda. Available online: https://childrensheartlink.org/wp-content/uploads/2017/06/The-Invisible-Child-Brief-2.pdf (accessed on 17 May 2020).
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheleva, B.; Nair, S.M.; Dobrzycka, A.; Saarinen, A. Considerations for Newborn Screening for Critical Congenital Heart Disease in Low- and Middle-Income Countries. Int. J. Neonatal Screen. 2020, 6, 49. https://doi.org/10.3390/ijns6020049
Zheleva B, Nair SM, Dobrzycka A, Saarinen A. Considerations for Newborn Screening for Critical Congenital Heart Disease in Low- and Middle-Income Countries. International Journal of Neonatal Screening. 2020; 6(2):49. https://doi.org/10.3390/ijns6020049
Chicago/Turabian StyleZheleva, Bistra, Sreehari M. Nair, Adriana Dobrzycka, and Annamarie Saarinen. 2020. "Considerations for Newborn Screening for Critical Congenital Heart Disease in Low- and Middle-Income Countries" International Journal of Neonatal Screening 6, no. 2: 49. https://doi.org/10.3390/ijns6020049
APA StyleZheleva, B., Nair, S. M., Dobrzycka, A., & Saarinen, A. (2020). Considerations for Newborn Screening for Critical Congenital Heart Disease in Low- and Middle-Income Countries. International Journal of Neonatal Screening, 6(2), 49. https://doi.org/10.3390/ijns6020049




