Initial Evaluation of Prospective and Parallel Assessments of Cystic Fibrosis Newborn Screening Protocols in Eastern Andalusia: IRT/IRT versus IRT/PAP/IRT
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design and Cohort
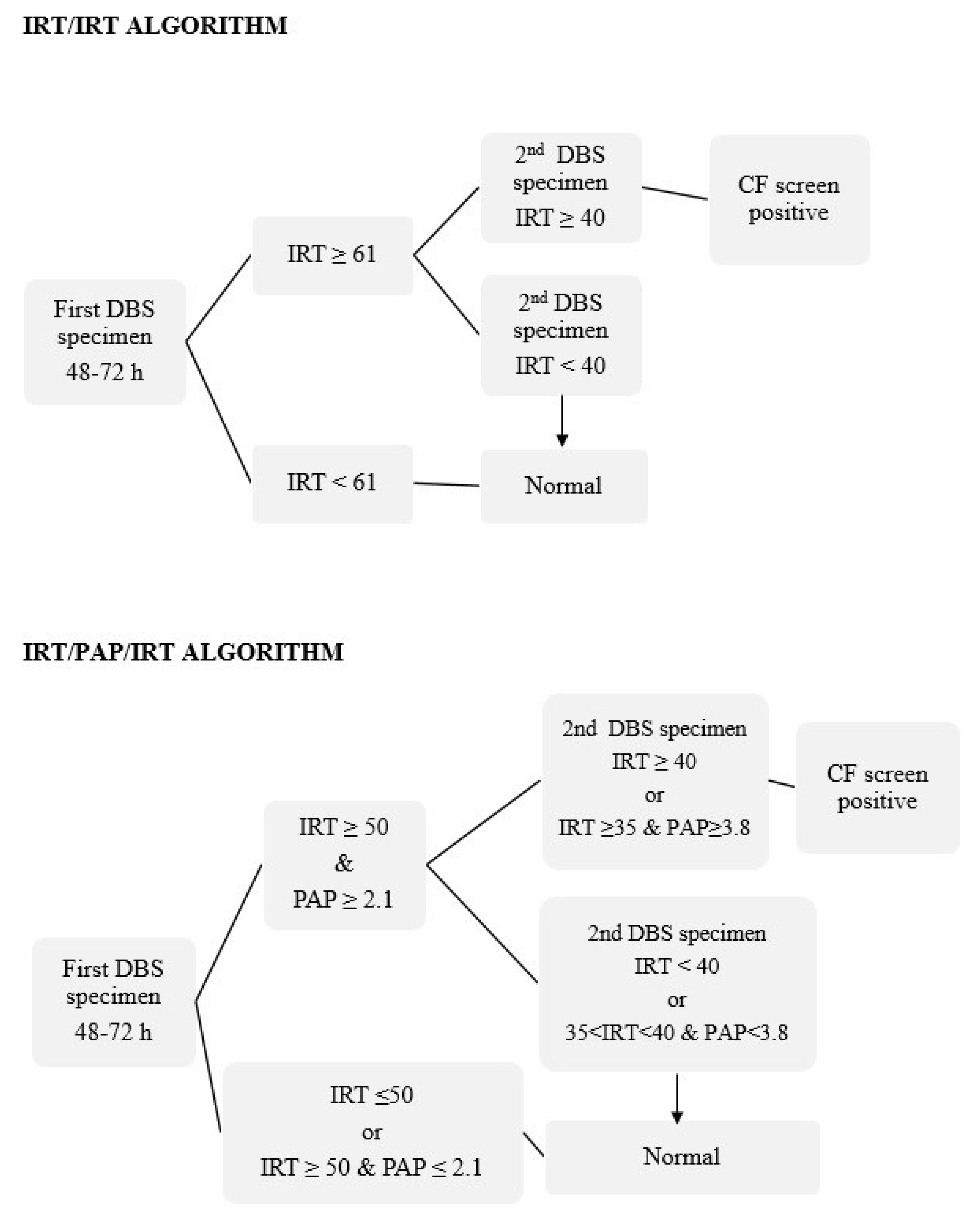
2.2. IRT and PAP Measurements and Screening Protocols
2.3. CF Diagnosis
2.4. PAP False Positive Values
2.5. Discrimination Analysis and Protocol Comparison
3. Results
3.1. Performance Metrics
3.2. Discrimination Analysis
3.3. PAP Results in Non-CF Newborns and Investigation of Sources of False Positives
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BW | birth weight |
| CI | confidence interval |
| CF | cystic fibrosis |
| CFSPID | cystic fibrosis screen positive with an inconclusive diagnosis |
| CFTR | cystic fibrosis transmembrane conductance regulator (gene) |
| DBS | dried blood spot |
| DNA | deoxyribonucleic acid |
| EGA | extended gene analysis |
| GA | gestational age |
| IRT | immunoreactive trypsinogen |
| NBS | newborn screening |
| NPV | negative predictive value |
| PAP | pancreatitis-associated protein |
| PPV | positive predictive value |
| SCT | sweat chloride test |
| SN | safety net |
References
- Zielenski, J.; Tsui, L.C. Cystic fibrosis: Genotypic and phenotypic variations. Annu. Rev. Genet. 1995, 29, 777–807. [Google Scholar] [CrossRef] [PubMed]
- Bear, C.E.; Li, C.H.; Kartner, N.; Bridges, R.J.; Jensen, T.J.; Ramjeesingh, M.; Riordan, J.R. Purification and functional reconstitution of the cystic fibrosis transmembrane conductance regulator (CFTR). Cell 1992, 68, 809–818. [Google Scholar] [CrossRef]
- Tsui, L.C.; Dorfman, R. The cystic fibrosis gene: A molecular genetic perspective. Cold Spring Harb. Perspect. Med. 2013, 3, a009472. [Google Scholar] [CrossRef] [PubMed]
- Sosnay, P.R.; Siklosi, K.R.; Van Goor, F.; Kaniecki, K.; Yu, H.; Sharma, N.; Ramalho, A.S.; Amaral, M.D.; Dorfman, R.; Zielenski, J.; et al. Defining the disease liability of variants in the cystic fibrosis transmembrane conductance regulator gene. Nat. Genet. 2013, 45, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Bronstein, M.N.; Sokol, R.J.; Abman, S.H.; Chatfield, B.A.; Hammond, K.B.; Hambidge, K.M.; Stall, C.D.; Accurso, F.J. Pancreatic insufficiency, growth, and nutrition in infants identified by newborn screening as having cystic fibrosis. J. Pediatr. 1992, 120, 533–540. [Google Scholar] [CrossRef]
- Lai, H.J.; Cheng, Y.; Cho, H.; Kosorok, M.R.; Farrell, P.M. Association between initial disease presentation, lung disease outcomes, and survival in patients with cystic fibrosis. Am. J. Epidemiol. 2004, 159, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.M.; Lai, H.J.; Li, Z.; Kosorok, M.R.; Laxova, A.; Green, C.G.; Collins, J.; Hoffman, G.; Laessig, R.; Rock, M.J.; et al. Evidence on improved outcomes with early diagnosis of cystic fibrosis through neonatal screening: Enough is enough! J. Pediatr. 2005, 147, S30–S36. [Google Scholar] [CrossRef]
- Castellani, C.; Massie, J.; Sontag, M.; Southern, K.W. Newborn screening for cystic fibrosis. Lancet Resp. Med. 2016, 4, 653–661. [Google Scholar] [CrossRef]
- Van der Ploeg, C.P.; van den Akker-van Marle, M.E.; Vernooij-van Langen, A.M.; Elvers, L.H.; Gille, J.J.; Verkek, P.H.; Dankert-Roelse, J.E.; CHOPIN Study Group. Cost-effectiveness of newborn screening for cystic fibrosis determined with real-life data. J. Cyst. Fibros. 2015, 14, 194–202. [Google Scholar] [CrossRef]
- Nshimyumukiza, L.; Bois, A.; Daigneault, P.; Lands, L.; Laberge, A.M.; Fournier, D.; Duplantie, J.; Giguère, Y.; Gekas, J.; Gagné, C.; et al. Cost effectiveness of newborn screening for cystic fibrosis: A simulation study. J. Cyst. Fibros. 2014, 13, 267–274. [Google Scholar] [CrossRef]
- Barben, J.; Castellani, C.; Dankert-Roelse, J.; Gartner, S.; Kashirskaya, N.; Linnane, B.; Mayell, S.; Munck, A.; Sands, D.; Sommerburg, O.; et al. The expansion and performance of national newborn screening programmes for cystic fibrosis in Europe. J. Cyst. Fibros. 2017, 16, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Castellani, C.; Southern, K.W.; Brownlee, K.; Dankert Roelse, J.; Duff, A.; Farrell, M.; Mehta, A.; Munck, A.; Pollitt, R.; Sermet-Gaudelus, I.; et al. European best practice guidelines for cystic fibrosis neonatal screening. J. Cyst. Fibros. 2009, 8, 153–173. [Google Scholar] [CrossRef]
- Brennan, M.L.; Schrijver, I. Cystic fibrosis: A review of associated phenotypes, use of molecular diagnostic approaches, genetic characteristics, progress, and dilemmas. J. Mol. Diagn. 2016, 18, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Crossley, J.R.; Elliott, R.B.; Smith, P.A. Dried-blood spot screening for cystic fibrosis in the newborn. Lancet 1979, 1, 472–474. [Google Scholar] [CrossRef]
- Rock, M.J.; Mischler, E.H.; Farrell, P.M.; Bruns, W.T.; Hassemer, D.J.; Laessig, R.H. Immunoreactive trypsinogen screening for cystic fibrosis: Characterization of infants with a false-positive screening test. Pediatr. Pulmonol. 1989, 6, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Delgado, I.; Pérez, E.; Álvarez, A.; Delgado, C.; Yahyaoui, R.; Carrasco, L.; Marcos, I.; Caro, P.; Moreno, M.J.; Quintana, M.E. Results of the Andalusian Cystic Fibrosis Neonatal Screening Program, 5 years after implementation. Arch. Bronconeumol. 2018, 54, 551–558. [Google Scholar]
- Schmidt, M.; Werbrouck, A.; Verhaeghe, N.; De Wachter, E.; Simoens, S.; Annemans, L.; Putman, K. Strategies for newborn screening for cystic fibrosis: A systematic review of health economic evaluations. J. Cyst. Fibros. 2018, 17, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Vernooij-van Langen, A.M.; Loeber, J.G.; Elvers, B.; CHOPIN Study Group. Novel strategies in newborn screening for cystic fibrosis: A prospective controlled study. Thorax 2012, 67, 289–295. [Google Scholar] [CrossRef]
- Wells, J.; Rosenberg, M.; Hoffman, G.; Anstead, M.; Farrell, P.M. A decision-tree approach to cost comparison of newborn screening strategies for cystic fibrosis. Pediatrics 2012, 129, e339–e347. [Google Scholar] [CrossRef]
- Wilcken, B. Newborn screening for cystic fibrosis: Techniques and strategies. J. Inherit. Metab. Dis. 2007, 30, 537–543. [Google Scholar] [CrossRef]
- Southern, K.W.; Barben, J.; Gartner, S.; Munck, A.; Castellani, C.; Mayell, S.J.; Davies, J.C.; Winters, V.; Murphy, J.; Salinas, D.; et al. Inconclusive diagnosis after a positive newborn bloodspot screening result for cystic fibrosis; clarification of the harmonized international definition. J. Cyst. Fibros. 2019. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Krulisova, V.; Hammermann, J.; Lindner, M.; Stahl, M.; Muckenthaler, M.; Kohlmueller, D.; Happich, M.; Kulozik, A.E.; Votava, F.; et al. Comparison of different IRT-PAP protocols to screen newborn for cystic fibrosis in three central European populations. J. Cyst. Fibros. 2014, 13, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Ross, L.F. Newborn screening for cystic fibrosis: A lesson in public health disparities. J. Pediatr. 2008, 153, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.B.; Lai, H.J.; Rock, M.J.; Farrell, P.M. Comparing age of cystic fibrosis diagnosis and treatment initiation after newborn screening with two common strategies. J. Cyst. Fibros. 2012, 11, 150–153. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kloosterboer, M.; Hoffman, G.; Rock, M.; Gershan, W.; Laxova, A.; Li, Z.; Farrell, P.M. Clarification of laboratory and clinical variables that influence cystic fibrosis newborn screening with initial analysis of immunoreactive trypsinogen. Pediatrics 2009, 123, e338–e346. [Google Scholar] [CrossRef]
- Sontag, M.K.; Lee, R.; Wright, D.; Freedenberg, D.; Sagel, S.D. Improving the sensitivity and positive predictive value in a cystic fibrosis newborn screening program using a repeat immunoreactive trypsinogen and genetic analysis. J. Pediatr. 2016, 175, 150–158. [Google Scholar] [CrossRef]
- Sarles, J.; Berthézéne, P.; Le Louarn, C.; Somma, C.; Perini, J.M.; Catheline, M.; Mrallé, S.; Luzet, K.; Roussey, M.; Farriaux, J.P.; et al. Combining immunoreactive trypsinogen and pancreatitis-associated protein assays, a method of newborn screening for cystic fibrosis that avoids DNA analysis. Pediatrics 2005, 10, 302–305. [Google Scholar] [CrossRef]
- Iovanna, J.L.; Férec, C.; Sarles, J.; Dagorn, J.C. The pancreatitis-associated protein (PAP). A new candidate for neonatal screening of cystic fibrosis. C. R. Acad. Sci. III 1994, 317, 561–564. [Google Scholar]
- Sarles, J.; Giorgi, R.; Berthézéne, P.; Munck, A.; Cheillan, D.; Dagorn, J.C.; Roussey, M. Neonatal screening for cystic fibrosis: Comparing the performances of IRT/DNA and IRT/PAP. J. Cyst. Fibros. 2014, 13, 384–390. [Google Scholar] [CrossRef]
- Sommerburg, O.; Hammermann, J.; Lindner, M.; Stahl, M.; Muckenthaler, M.; Kohlmueller, D.; Happich, M.; Kulozik, A.E.; Stopsack, M.; Gahr, M.; et al. Five years of experience with biochemical cystic fibrosis newborn screening based on IRT/PAP in Germany. Pediatr. Pulmonol. 2015, 50, 655–664. [Google Scholar] [CrossRef]
- Seror, V.; Cao, C.; Roussey, M.; Giorgi, R. PAP assays in newborn screening for cystic fibrosis: A population-based cost-effectiveness study. J. Med. Screen. 2016, 23, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Marcao, A.; Barreto, C.; Pereira, L.; Guedes, L.; Cavaco, J.; Casimiro, A.; Félix, M.; Reis, T.; Barbosa, T.; Freitas, C.; et al. Cystic fibrosis in newborn screening in Portugal: PAP values in populations with stringent rules for genetic studies. Int. J. Neonatal. Screen. 2018, 4, 22. [Google Scholar] [CrossRef]
- Dankert-Roelse, J.E.; Bouva, M.J.; Jakobs, B.S.; Janssens, H.M.; Winter-de Groot, K.M.; Schönbeck, Y.; Gille, J.J.P.; Gulmans, V.A.M.; Verschoof-Puite, R.K.; Schielen, P.C.J.I.; et al. Newborn blood spot screening for cystic fibrosis with a four-step screening estrategy in the Netherlands. J. Cystic Fibros. 2019, 18, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Castiñeiras, D.E.; Couce, M.L.; Marin, J.L.; González-Lamuño, D.; Rocha, H. Newborn screening for metabolic disorders in Spain and worldwide. An. Pediatr. 2019, 91, 128.e1–128.e14. [Google Scholar] [CrossRef]
- Weidler, S.; Stopsack, K.H.; Hammerman, J.; Sommerburg, O.; Mall, M.A.; Hoffmann, G.F.; Kohlmüller, D.; Okun, J.G.; Macek, M.; Votava, F.; et al. A product of immunoreactive trypsinogen and pancreatitis-associated protein as second-tier strategy in cystic fibrosis newborn screening. J. Cyst. Fibros. 2016, 15, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.M.; White, T.B.; Howenstine, M.S.; Munck, A.; Parad, R.B.; Rosenfeld, M.; Sommerburg, O.; Accurso, F.J.; Davies, J.C.; Rock, M.J.; et al. Diagnostic of cystic fibrosis in screened populations. J. Pediatr. 2017, 181, S33–S44.e2. [Google Scholar] [CrossRef] [PubMed]
- Hennekens, C.H.; Buring, J.E.; Mayrent, S.L. Epidemiology in Medicine; Little, Brown and Co: Boston, MA, USA, 1987; pp. 327–345. [Google Scholar]
- Cystic Fibrosis Foundation; Borowitz, D.; Parad, R.B.; Sharp, J.K.; Sabadosa, K.A.; Rock, M.J.; Farrell, P.M.; Sontag, M.K.; Rosenfeld, M.; Davis, S.D.; et al. Cystic Fibrosis Foundation practice guidelines for the management of infants with cystic fibrosis transmembrane conductance regulator-related metabolic syndrome during the first two years of life and beyond. J. Pediatr. 2009, 155, S106–S116. [Google Scholar] [CrossRef] [PubMed]
- Lebecque, P.; Leal, T.; De Boeck, C.; Jaspers, M.; Cuppens, H.; Cassiman, J.J. Mutations of the cystic fibrosis gene and intermediate sweat chloride levels in children. Am. J. Respir. Crit. Care Med. 2002, 165, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Savkovic, V.; Gaiser, S.; Iovanna, J.L.; Bödeker, H. The stress response of the exocrine pancreas. Dig. Dis. 2004, 22, 239–246. [Google Scholar] [CrossRef]
- Vernooij-van Langen, A.M.M.; Loeber, J.G.; Elvers, B.; Triepels, R.H.; Roefs, J.; Gille, J.J.; Reijntjens, S.; Dompeling, E.; Dankert-Roelse, J.E. The influence of sex, gestational age, birth weight, blood transfusion, and timing of the heel prick on the pancreatitis-associated protein concentration in newborn screening for cystic fibrosis. J. Inherit. Metab. Dis. 2013, 36, 147–154. [Google Scholar] [CrossRef]
| Patient ID (Days Old at 1st and 2nd DBS Collection) | BW (g) | GA (Weeks) | 1st IRT (ng/mL) | PAP (µg/L) | 2nd IRT (ng/mL) | SCT (mmol/L) | Fecal Elastase (µg/g) | PI | CFTR Gene Pathogenic Variants | |
|---|---|---|---|---|---|---|---|---|---|---|
| P1 (3,24) | 3400 | 40 | 115 | 5.23 | 41 | 73 | 464 | N | p.Trp1282* | c.3140-26A > G |
| P2 (4,28) | 2100 | 33 | 75 | 4.74 | 43 | 75 | 417 | N | p.Leu206Trpɸ | p.Arg285Glyɸ |
| P3 (4,25) | 2360 | 37 | 101 | 2.93 | 70 | 83 | 150 | Y | p.Phe508del | p.Phe508del |
| P4 (3,24) | 3400 | 40 | 110 | 2.32 | 90 | 101 | 24 | Y | p.Phe508del | # |
| P5 (3,25) | 3640 | 40 | 86 | 2.36 | 73 | 75 | 183 | Y | p.Phe508del | c.3140-26A > G |
| P6 (3,25) | 3350 | 40 | 114 | 5.13 | 80 | 90 | <15 | Y | p.Phe508del | p.Phe508del |
| P7 (3,24) | 3190 | 39 | 77 | 4.49 | 98 | 85 | 128 | Y | p.Phe508del | p.Phe508del |
| P8 (3,26) | 3020 | 39 | 131 | 6.17 | 106 | NA 1 | <15 | Y | p.Phe508del | p.Phe508del |
| P9 (6,28) | 3345 | 37 | 154 | >8.0 | 140 | 88 | NA | Y | p.Phe508del | p.Asn1303Lys |
| P10 (3,24) | 3000 | 38 | 153 | 7.64 | 150 | 94 | <15 | Y | p.Phe508del | p.Ile507del |
| P11 (4,25) | 1340 | 29 | 170 | 0.99 | 71 | NA | 141 | Y | p.Phe508del | p.Gln685ThrfsXɸ |
| P12 (3,26) | 2700 | 38 | 155 | 7.40 | 207 | 87 | <15 | Y | p.Gly673Terɸ | p.Asn1303Lys |
| P13 (5,25) | 2890 | 39 | 180 | 6.69 | 226 | 95 | <15 | Y | p.Phe508del | p.Phe508del |
| P14 (4,19) | 3200 | 37 | 137 | >8.0 | 255 | NA 2 | <15 | Y | p.Phe508del | p.Phe508del |
| Screening Protocol | IRT/IRT | IRT/PAP/IRT | IRT/PAP/IRT + SN | IRT × PAP/IRT |
|---|---|---|---|---|
| -CF-screen positives (% of infants screened) | 93 (0.13%) | 35 (0.05%) | 66 (0.09%) | 36 (0.05%) |
| -CF cases detected | 14 | 13 | 14 | 14 |
| -False positive cases (% of positive test cases with appropriate follow-up) | 79 (0.11%) | 22 (0.03%) | 52 (0.07%) | 22 (0.03%) |
| -False negative cases | 0 | 1 | 0 | 0 |
| -True negative cases | 68.409 | 68.466 | 68.436 | 68.466 |
| -Sensitivity (95% CI) | 100% [96.4–100] | 92.9% [75.7–100] | 100% [96.4–100] | 100% [96.4–100] |
| -Specificity (95% CI) | 99.9% [99.8–99.9] | 100% [99.9–100] | 99.9% [99.9–100] | 99.9% [99.9–100] |
| -PPV (95% CI) | 15.1% [7.2–22.8] | 37.1% [19.7–54.5] | 26.9% [10.5–31.8] | 39.0% [21.5–56.2] |
| -NPV (95% CI) | 100% [99.9–100] | 100% [99.9–100] | 100% [99.9–100] | 100% [99.9–100] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sadik, I.; Pérez de Algaba, I.; Jiménez, R.; Benito, C.; Blasco-Alonso, J.; Caro, P.; Navas-López, V.M.; Pérez-Frías, J.; Pérez, E.; Serrano, J.; et al. Initial Evaluation of Prospective and Parallel Assessments of Cystic Fibrosis Newborn Screening Protocols in Eastern Andalusia: IRT/IRT versus IRT/PAP/IRT. Int. J. Neonatal Screen. 2019, 5, 32. https://doi.org/10.3390/ijns5030032
Sadik I, Pérez de Algaba I, Jiménez R, Benito C, Blasco-Alonso J, Caro P, Navas-López VM, Pérez-Frías J, Pérez E, Serrano J, et al. Initial Evaluation of Prospective and Parallel Assessments of Cystic Fibrosis Newborn Screening Protocols in Eastern Andalusia: IRT/IRT versus IRT/PAP/IRT. International Journal of Neonatal Screening. 2019; 5(3):32. https://doi.org/10.3390/ijns5030032
Chicago/Turabian StyleSadik, Ilham, Inmaculada Pérez de Algaba, Rocío Jiménez, Carmen Benito, Javier Blasco-Alonso, Pilar Caro, Víctor M. Navas-López, Javier Pérez-Frías, Estela Pérez, Juliana Serrano, and et al. 2019. "Initial Evaluation of Prospective and Parallel Assessments of Cystic Fibrosis Newborn Screening Protocols in Eastern Andalusia: IRT/IRT versus IRT/PAP/IRT" International Journal of Neonatal Screening 5, no. 3: 32. https://doi.org/10.3390/ijns5030032
APA StyleSadik, I., Pérez de Algaba, I., Jiménez, R., Benito, C., Blasco-Alonso, J., Caro, P., Navas-López, V. M., Pérez-Frías, J., Pérez, E., Serrano, J., & Yahyaoui, R. (2019). Initial Evaluation of Prospective and Parallel Assessments of Cystic Fibrosis Newborn Screening Protocols in Eastern Andalusia: IRT/IRT versus IRT/PAP/IRT. International Journal of Neonatal Screening, 5(3), 32. https://doi.org/10.3390/ijns5030032






