Retrospective Motion Artifact Reduction by Spatial Scaling of Liver Diffusion-Weighted Images
Abstract
:1. Introduction
2. Materials and Methods
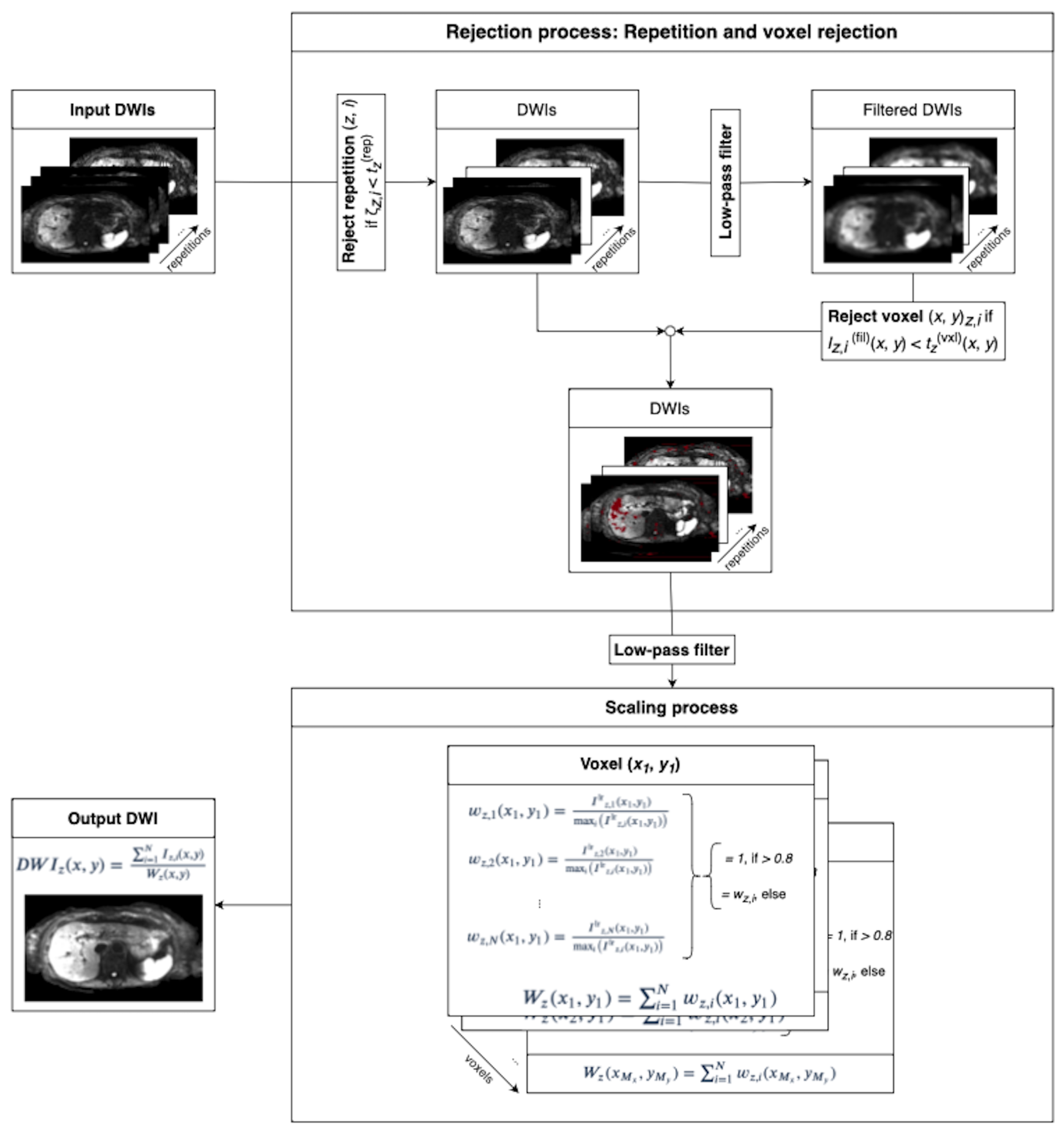
2.1. Implicit Weighted Averaging
2.2. Spatial Scaling for the Reduction of Motion-Induced Signal Loss
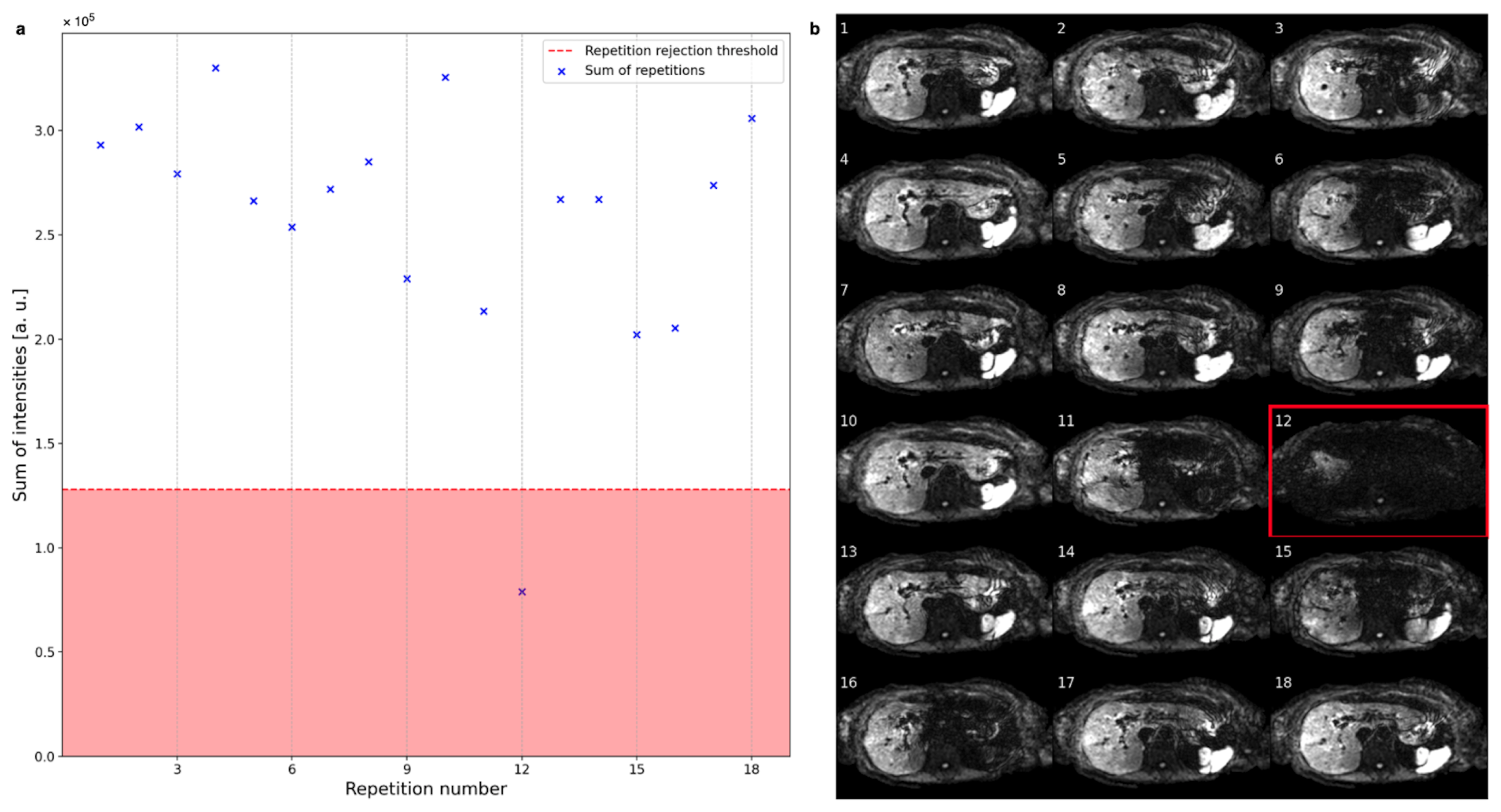
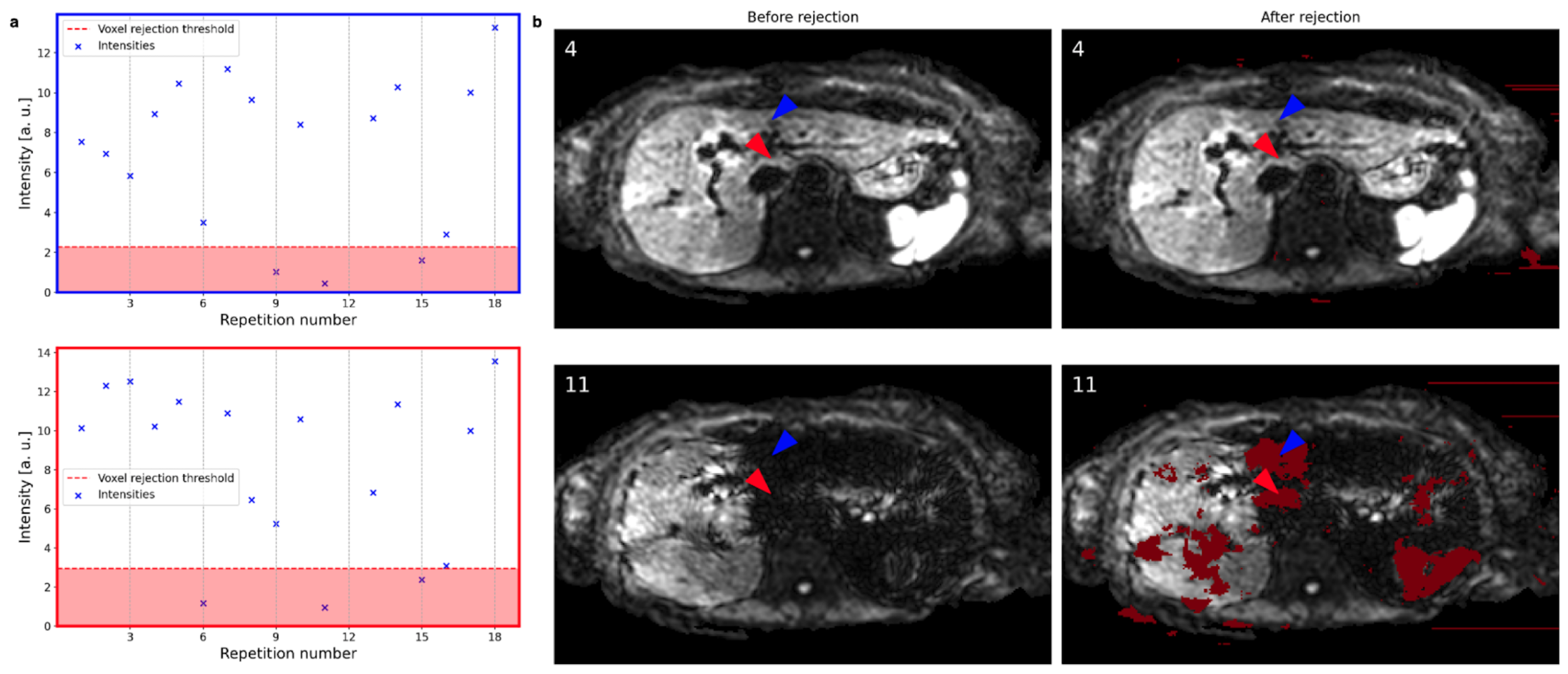
2.2.1. Rejection Process
2.2.2. Spatial Scaling of Average Diffusion-Weighted Images
2.3. Data Acquisition
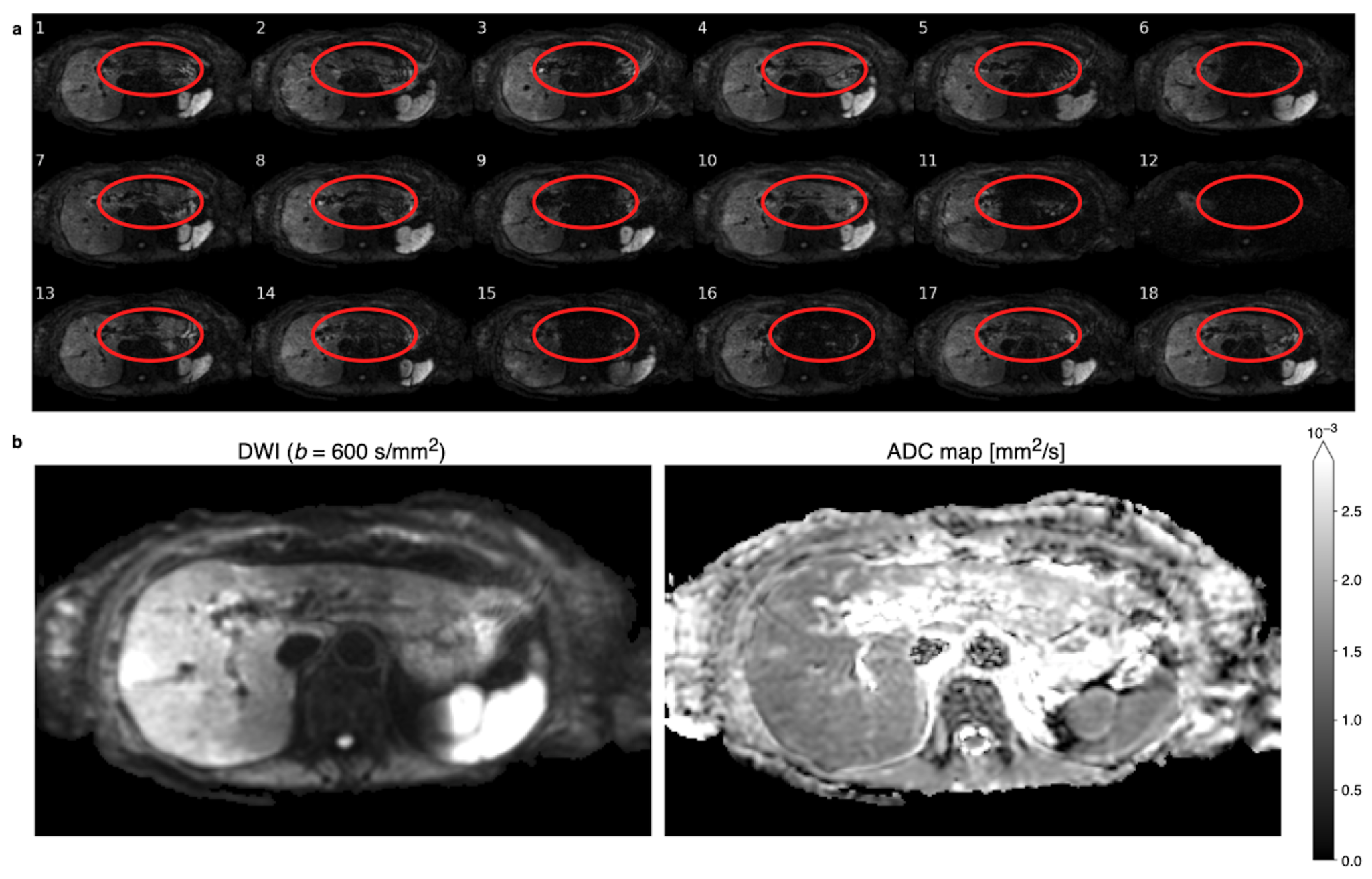
2.4. Image Reconstruction and Analysis
2.4.1. ADC Comparison
2.4.2. Clinical Evaluation
- Overall image quality: the gross appearance of the whole image volume;
- Liver homogeneity: the contrast between the signal of the liver parenchyma of the left lobe versus the right lobe;
- Perceived signal-to-noise ratio (SNR): the visual perception of the noise performance;
- Quality of lesion detection: the possibility to distinguish healthy liver parenchyma from lesions.
2.4.3. Statistical Evaluation
3. Results
3.1. Repetition and Voxel Rejection
3.2. Spatial Scaling of Average DWI
3.3. Quantitative ADC Measurement
3.4. Radiological Reading
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADC | Apparent Diffusion Coefficient |
| ALR | Apparent diffusion coefficient liver Lobe Ratio |
| DW | Diffusion-Weighted |
| DWI | Diffusion-Weighted (magnetic resonance) Imaging |
| EPI | Echo-Planar Imaging |
| FOV | Field Of View |
| MCDE | Motion-Compensated Diffusion Encoding |
| MRI | Magnetic Resonance Imaging |
| ROI | Region Of Interest |
| SNR | Signal-to-Noise Ratio |
| TE | Echo Time |
| T2 | T2 relaxation time |
References
- Stejskal, E.O.; Tanner, J.E. Spin Diffusion Measurements: Spin Echoes in the Presence of a Time-Dependent Field Gradient. J. Chem. Phys. 1965, 42, 288–292. [Google Scholar] [CrossRef]
- Frahm, J.; Merboldt, K.D.; Hänicke, W.; Haase, A. Stimulated Echo Imaging. J. Magn. Reson. 1985, 64, 81–93. [Google Scholar] [CrossRef]
- Le Bihan, D.; Breton, E.; Lallemand, D.; Aubin, M.L.; Vignaud, J.; Laval-Jeantet, M. Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology 1988, 168, 497–505. [Google Scholar] [CrossRef]
- Le Bihan, D. Looking into the functional architecture of the brain with diffusion MRI. Nat. Rev. Neurosci. 2003, 4, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Le Bihan, D. Diffusion, confusion and functional MRI. NeuroImage 2012, 62, 1131–1136. [Google Scholar] [CrossRef]
- Le Bihan, D.; Johansen-Berg, H. Diffusion MRI at 25: Exploring brain tissue structure and function. Neuroimage 2012, 61, 324–341. [Google Scholar] [CrossRef]
- Alexander, D.C.; Dyrby, T.B.; Nilsson, M.; Zhang, H. Imaging brain microstructure with diffusion MRI: Practicality and applications. NMR Biomed. 2017, 32, e3841. [Google Scholar] [CrossRef] [PubMed]
- Koh, D.M.; Collins, D.J. Diffusion-Weighted MRI in the Body: Applications and Challenges in Oncology. Am. J. Roentgenol. 2007, 188, 1622–1635. [Google Scholar] [CrossRef]
- Kele, P.G.; van der Jagt, E.J. Diffusion weighted imaging in the liver. World J. Gastroentorol. 2010, 16, 1567–1576. [Google Scholar] [CrossRef]
- Narquin, S.; Ingrand, P.; Azais, I.; Delwail, V.; Vialle, R.; Boucecbi, S.; Tasu, J.P. Comparison of whole-body diffusion MRI and conventional radiological assessment in the staging of myeloma. Diagn. Interv. Imaging 2013, 94, 629–636. [Google Scholar] [CrossRef]
- Ayuso, C.; Rimola, J.; Vilana, R.; Burrel, M.; Darnell, A.; García-Criado, Á.; Bianchi, L.; Belmonte, E.; Caparroz, C.; Barrufet, M.; et al. Diagnosis and staging of hepatocellular carcinoma (HCC): Current guidelines. Eur. J. Radiol. 2018, 101, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, T.; Haradome, H.; Hachiya, J.; Nitatori, T.; Araki, T. Diffusion-weighted MR imaging with a single-shot echoplanar sequence: Detection and characterization of focal hepatic lesions. Am. J. Roentgenol. 1998, 170, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Parikh, T.; Drew, S.J.; Lee, V.S.; Wong, S.; Hecht, E.M.; Babb, J.S.; Taouli, B. Focal liver lesion detection and characterization with diffusion-weighted MR imaging: Comparison with standard breath-hold T2-weighted imaging. Radiology 2008, 246, 812–822. [Google Scholar] [CrossRef]
- Yoon, J.H.; Lee, J.M.; Yu, M.H.; Kiefer, B.; Han, J.K.; Choi, B.I. Evaluation of hepatic focal lesions using diffusion-weighted MR imaging: Comparison of apparent diffusion coefficient and intravoxel incoherent motion-derived parameters. J. Magn. Reson. Imaging 2014, 39, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Joo, I.; Kim, T.Y.; Han, S.W.; Kim, Y.J.; Lee, J.M.; Han, J.K. Diffusion-Related MRI Parameters for Assessing Early Treatment Response of Liver Metastases to Cytotoxic Therapy in Colorectal Cancer. Am. J. Roentgenol. 2016, 207, W26–W32. [Google Scholar] [CrossRef]
- Koinuma, M.; Ohashi, I.; Hanafusa, K.; Shibuya, H. Apparent diffusion coefficient measurements with diffusion-weighted magnetic resonance imaging for evaluation of hepatic fibrosis. J. Magn. Reson. Imaging 2005, 22, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Amano, Y.; Kumazaki, T.; Ishihara, M. Single-shot diffusion-weighted echo-planar imaging of normal and cirrhotic livers using a phased-array multicoil. Acta Radiol. 2010, 39, 440–442. [Google Scholar] [CrossRef]
- Meier, C.; Zwanger, M.; Feiweier, T.; Porter, D. Concomitant field terms for asymmetric gradient coils: Consequences for diffusion, flow, and echo-planar imaging. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2008, 60, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Malyarenko, D.I.; Newitt, D.; Wilmes, L.J.; Tudorica, A.; Helmer, K.G.; Arlinghaus, L.R.; Jacobs, M.A.; Jajamovich, G.; Taouli, B.; Yankeelov, T.E.; et al. Demonstration of nonlinearity bias in the measurement of the apparent diffusion coefficient in multicenter trials. Magn. Reson. Med. 2016, 75, 1312–1323. [Google Scholar] [CrossRef]
- McTavish, S.; Van, A.T.; Peeters, J.M.; Weiss, K.; Makowski, M.R.; Braren, R.F.; Karampinos, D.C. Gradient nonlinearity correction in liver DWI using motion-compensated diffusion encoding waveforms. Magn. Reson. Mater. Phys. Biol. Med. 2021, 35, 827–841. [Google Scholar] [CrossRef]
- Krzyżak, A.T.; Olejniczak, Z. Improving the accuracy of PGSE DTI experiments using the spatial distribution of b matrix. Magn. Reson. Imaging 2015, 33, 286–295. [Google Scholar] [CrossRef]
- Koh, D.M.; Takahara, T.; Imai, Y.; Collins, D.J. Practical Aspects of Assessing Tumors Using Clinical Diffusion-weighted Imaging in the Body. Magn. Reson. Med. Sci. 2007, 6, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Kwee, T.C.; Takahara, T.; Niwa, T.; Ivancevic, M.K.; Herigault, G.; van Cauteren, M.; Luijten, P.R. Influence of cardiac motion on diffusion-weighted magnetic resonance imaging of the liver. Magn. Reson. Mater. Phys. Biol. Med. 2009, 22, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Roberts, N.T.; Hinshaw, L.A.; Colgan, T.J.; Ii, T.; Hernando, D.; Reeder, S.B. B0 and B1 inhomogeneities in the liver at 1.5 T and 3.0 T. Magn. Reson. Med. 2021, 85, 2212–2220. [Google Scholar] [CrossRef] [PubMed]
- Luna, A.; Ribes, R.; Soto, J.A. Diffusion MRI Outside the Brain: A Cased-Based Review and Clinical Applications; Springer Science+Business Media: Berlin/Heidelberg, Germany; University of British Columbia: Vancouver, BC, Canada, 2012; Chapter 1. [Google Scholar] [CrossRef]
- De Bazelaire, C.M.J.; Duhamel, G.D.; Rofsky, N.M.; Alsop, D.C. MR Imaging Relaxation Times of Abdominal and Pelvic Tissues Measured in Vivo at 3.0 T: Preliminary Results. Radiology 2021, 230, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Delshad, S.; Woo, M.A.; Macey, P.M.; Harper, R.M. Age-related regional brain T2-relaxation changes in healthy adults. J. Magn. Reson. Imaging 2012, 35, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Naganawa, S.; Kawai, H.; Fukatsu, H.; Sakurai, Y.; Aoki, I.; Miura, S.; Mimura, T.; Kanazawe, H.; Ishigaki, T. Diffusion-weighted Imaging of the Liver: Technical Challenges and Prospects for the Future. Magn. Reson. Med. Sci. 2005, 4, 175–186. [Google Scholar] [CrossRef]
- Liau, J.; Lee, J.; Schroeder, M.E.; Sirlin, C.B.; Bydder, M. Cardiac motion in diffusion-weighted MRI of the liver: Artifact and a method of correction. J. Magn. Reson. Imaging 2011, 35, 318–327. [Google Scholar] [CrossRef]
- Kwee, T.C.; Takahara, T.; Koh, D.M.; Nievelstein, R.A.J.; Luijten, P.R. Comparison and reproducibility of ADC measurements in breathhold, respiratory triggered, and free-breathing diffusion-weighted MR imaging of the liver. J. Magn. Reson. Imaging 2008, 28, 1141–1148. [Google Scholar] [CrossRef]
- Rauh, S.S.; Riexinger, A.J.; Ohlmeyer, S.; Hammon, M.; Saake, M.; Stemmer, A.; Uder, M.; Hensel, B.; Laun, F.B. A mixed waveform protocol for reduction of the cardiac motion artifact in black-blood diffusion-weighted imaging of the liver. Magn. Reson. Imaging 2020, 67, 59–68. [Google Scholar] [CrossRef]
- Nasu, K.; Kuroki, Y.; Sekiguchi, R.; Kazama, T.; Nakajima, H. Measurement of the apparent diffusion coefficient in the liver: Is it a reliable index for hepatic disease diagnosis? Radiat. Med. 2006, 24, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Kandpal, H.; Sharma, R.; Madhusudhan, K.S.; Kapoor, K.S. Respiratory-Triggered Versus Breath-Hold Diffusion-Weighted MRI of Liver Lesions: Comparison of Image Quality and Apparent Diffusion Coefficient Values. Am. J. Roentgenol. 2009, 192, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Taouli, B.; Sandberg, A.; Stemmer, A.; Parikh, T.; Wong, S.; Xu, J.; Lee, V.S. Diffusion-weighted imaging of the liver: Comparison of navigator triggered and breathhold acquisitions. J. Magn. Reson. Imaging 2009, 30, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Dyvorne, H.A.; Galea, N.; Nevers, T.; Fiel, M.I.; Carpenter, D.; Wong, E.; Orton, M.; de Oliveira, A.; Feiweier, T.; Vachon, M.L.; et al. Diffusion-weighted Imaging of the Liver with Multiple b Values: Effect of Diffusion Gradient Polarity and Breathing Acquisition on Image Quality and Intravoxel Incoherent Motion Parameters—A Pilot Study. Radiology 2013, 266, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Mürtz, P.; Flacke, S.; Träber, F.; van den Brink, J.S.; Gieseke, J.; Schild, H.H. Abdomen: Diffusion-weighted MR imaging with pulse-triggered single-shot sequences. Radiology 2002, 224, 258–264. [Google Scholar] [CrossRef]
- Aliotta, E.; Wu, H.H.; Ennis, D.B. Convex optimized diffusion encoding (CODE) gradient waveforms for minimum echo time and bulk motion-compensated diffusion MRI. Magn. Reson. Med. 2017, 77, 717–729. [Google Scholar] [CrossRef]
- Peña-Nogales, Ó.; Zhang, Y.; Wang, X.; de Luis-Garcia, R.; Aja-Fernández, S.; Holmes, J.H.; Hernando, D. Optimized Diffusion-Weighting Gradient Waveform Design (ODGD) formulation for motion compensation and concomitant gradient nulling. Magn. Reson. Med. 2019, 81, 989–1003. [Google Scholar] [CrossRef]
- Zhang, Y.; Peña-Nogales, Ó.; Holmes, J.H.; Hernando, D. Motion-robust and blood-suppressed M1-optimized diffusion MR imaging of the liver. Magn. Reson. Med. 2019, 82, 302–311. [Google Scholar] [CrossRef]
- Chang, L.C.; Walker, L.; Pierpaoli, C. Informed RESTORE: A method for robust estimation of diffusion tensor from low redundancy datasets in the presence of physiological noise artifacts. Magn. Reson. Med. 2012, 68, 1654–1663. [Google Scholar] [CrossRef]
- Ichikawa, S.; Motosugi, I.; Tamada, D.; Wakayama, T.; Sato, K.; Funayama, S.; Onishi, H. Improving the Quality of Diffusion-weighted Imaging of the Left Hepatic Lobe Using Weighted Averaging of Signals from Multiple Excitations. Magn. Reson. Med. Sci. 2019, 18, 225–232. [Google Scholar] [CrossRef]
- Führes, T.; Saake, M.; Lorenz, J.; Seuss, H.; Stemmer, A.; Benkert, T.; Uder, M.; Laun, F.B. Reduction of the cardiac pulsation artifact and improvement of lesion conspicuity in flow-compensated diffusion images in the liver—A quantitative evaluation of postprocessing algorithms. Magn. Reson. Med. 2023, 89, 423–439. [Google Scholar] [CrossRef] [PubMed]
- Gadjimuradov, F.; Benkert, T.; Nickel, M.D.; Maier, A. Deep Learning-based Adaptive Image Combination for Signal-Dropout Suppression in Liver DWI. In Proceedings of the 2021 ISMRM & SMRT Annual Meeting and Exhibition, Virtual, 15–20 May 2021. [Google Scholar]
- Gadjimuradov, F.; Benkert, T.; Nickel, M.D.; Führes, T.; Saake, M.; Maier, A. Deep learning–guided weighted averaging for signal dropout compensation in DWI of the liver. Magn. Reson. Med. 2022, 88, 2679–2693. [Google Scholar] [CrossRef] [PubMed]
- Raspe, J.; Van, A.T.; Harder, F.; McTavish, S.; Peeters, J.; Weiss, K.; Makowski, M.R.; Braren, R.F.; Karampinos, D.C. Spatial Scaling of Respiratory-Triggered Liver Diffusion Weighted Imaging. In Proceedings of the 2022 ISMRM & SMRT Annual Meeting and Exhibition, London, UK, 7–12 May 2022. [Google Scholar]
- Taouli, B.; Vilgrain, V.; Dumont, E.; Daire, J.L.; Fan, B.; Menu, Y. Evaluation of Liver Diffusion Isotropy and Characterization of Focal Hepatic Lesions with Two Single-Shot Echo-Planar MR Imaging Sequences: Prospective Study in 66 Patients. Radiology 2003, 226, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Daintith, J. Dictionary of Physics, 6th ed.; Oxford University Press: Oxford, UK, 2009. [Google Scholar]
- Gauß, C.F. Bestimmung der Genauigkeit der Beobachtungen. In Zeitschrift für Astronomie und Verwandte Wissenschaften; J.G. Cotta’sche Buchhandlung: Tübingen, Germany, 1816; Volume 1, pp. 185–197. [Google Scholar]
- Leys, C.; Ley, C.; Klein, O.; Bernard, P.; Licata, L. Detecting outliers: Do not use standard deviation around the mean, use absolute deviation around the median. J. Exp. Soc. Psychol. 2013, 49, 764–766. [Google Scholar] [CrossRef]
- MATLAB, Version 9.10.0.1684407 (R2021a) Update3; The MathWorks: Natick, MA, USA, 2021.
- Van Rossum, G.; Drake, F.L. Python 3 Reference Manual; CreateSpace: Scotts Valley, CA, USA, 2009. [Google Scholar]
- Harris, C.R.; Millman, K.J.; van der Walt, S.J.; Gommers, R.; Virtanen, P.; Cournapeau, D.; Wieser, E.; Taylor, J.; Berg, S.; Smith, N.J.; et al. Array programming with NumPy. Nature 2020, 585, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental Algorithms for Scientific Computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef]
- Astropy Collaboration; Robitaille, T.P.; Tollerud, E.J.; Greenfield, P.; Droettboom, M.; Bray, E.; Aldcroft, T.; Davis, M.; Ginsburg, A.; Price-Whelan, A.M.; et al. Astropy: A community Python package for astronomy. Astron. Astrophys. 2013, 558, A33. [Google Scholar] [CrossRef]
- Astropy Collaboration; Price-Whelan, A.M.; Sipőcz, B.M.; Günther, H.M.; Lim, P.L.; Crawford, S.M.; Conseil, S.; Shupe, D.L.; Craig, M.W.; Dencheva, N.; et al. The Astropy Project: Building an Open-science Project and Status of the v2.0 Core Package. Astron. J. 2018, 156, 123. [Google Scholar] [CrossRef]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Collette, A. Python and HDF5; O’Reilly: Sebastopol, CA, USA, 2013. [Google Scholar]
- Juza, R.M.; Pauli, E.M. Clinical and surgical anatomy of the liver: A review for clinicians. Clin. Anat. 2014, 27, 764–769. [Google Scholar] [CrossRef]
- Sibulesky, L. Normal liver anatomy. Clin. Liver Dis. 2013, 2, S1–S3. [Google Scholar] [CrossRef] [PubMed]
- Student. The Probable Error of a Mean. Biometrika 1908, 6, 4–25. [Google Scholar] [CrossRef]
- Fisher, R.A. III. The influence of rainfall on the yield of wheat at Rothamsted. Philos. Trans. R. Soc. Lond. Ser. B Contain. Pap. A Biol. Character 1925, 213, 89–142. [Google Scholar] [CrossRef]
- Merriman, M. A List of Writings Relating to the Method of Least Squares: With Historical and Critical Notes; Academic Press: Cambridge, MA, USA, 1877; Volume 4. [Google Scholar]
- Pearson, K. VII. Note on regression and inheritance in the case of two parents. Proc. R. Soc. Lond. 1895, 58, 240–242. [Google Scholar] [CrossRef]
- Wright, S. Correlation and causation. J. Agric. Res. 1921, 20, 557–585. [Google Scholar]
- Sandrasegaran, K.; Akisik, F.M.; Lin, C.; Tahir, B.; Rajan, J.; Saxena, R.; Aisen, A.M. Value of Diffusion-Weighted MRI for Assessing Liver Fibrosis and Cirrhosis. Am. J. Roentgenol. 2009, 193, 1556–1560. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, N.; Gourtsoyianni, S.; Yarmenitis, S.; Maris, T.; Gourtsoyiannis, N. Comparison between two-point and four-point methods for quantification of normal liver parenchyma and focal lesions. Value of normalization with spleen. Eur. J. Radiol. 2010, 73, 305–309. [Google Scholar] [CrossRef]
- Chen, X.; Qin, L.; Pan, D.; Huang, Y.; Yan, L.; Wang, G.; Liu, Y.; Liang, C.; Liu, Z. Liver diffusion-weighted MR imaging: Reproducibility comparison of ADC measurements obtained with multiple breath-hold, free-breathing, respiratory-triggered, and navigator-triggered techniques. Radiology 2014, 271, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Le Bihan, D.; Poupon, C.; Amadon, A.; Lethimonnier, F. Artifacts and Pitfalls in Diffusion MRI. J. Magn. Reson. Imaging 2006, 24, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Borkowski, K.; Krzyżak, A.T. Assessment of the systematic errors caused by diffusion gradient inhomogeneity in DTI-computer simulations. NMR Biomed. 2019, 32, e4130. [Google Scholar] [CrossRef]
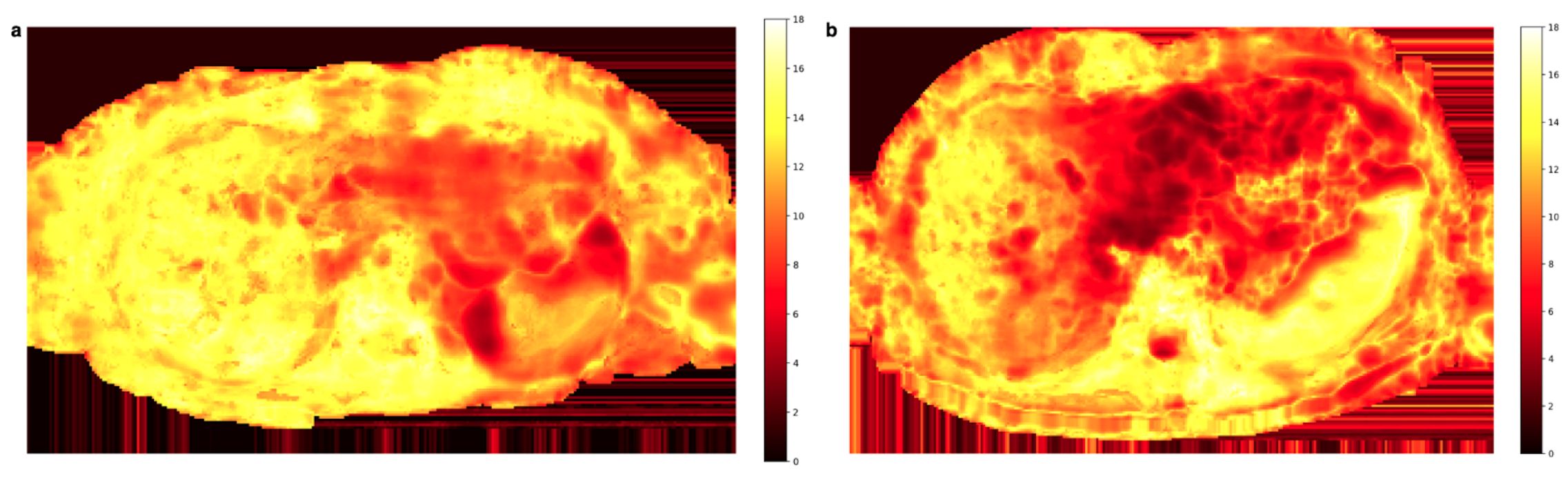
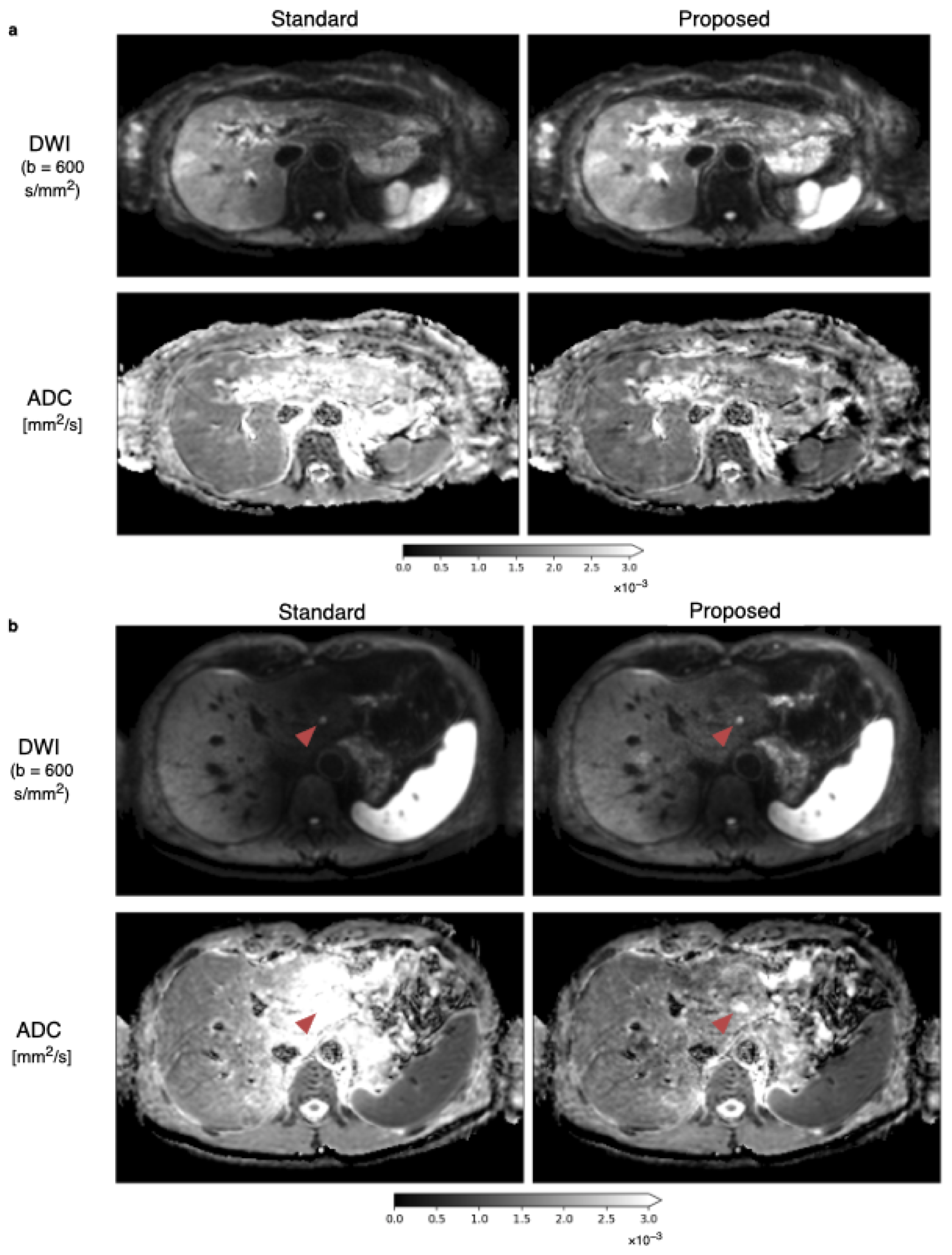
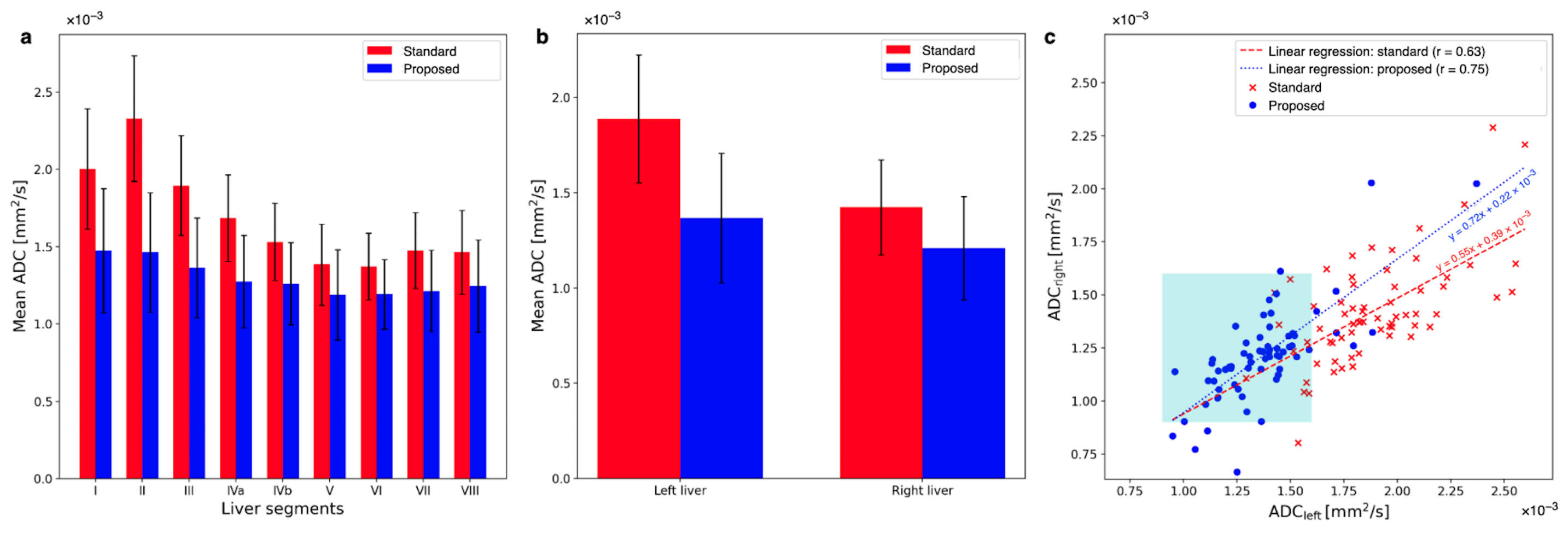

| Liver Segment | ADCstandard [mm2/s] | ADCproposed [mm2/s] | p-Value |
|---|---|---|---|
| I | 1.97 ± 0.35 | 1.71 ± 0.36 | 9.0 × 10 |
| II | 2.25 ± 0.33 | 1.82 ± 0.33 | 2.5 × 10 |
| III | 1.87 ± 0.27 | 1.60 ± 0.28 | 1.3 × 10 |
| IVa | 1.70 ± 0.25 | 1.48 ± 0.25 | 1.7 × 10 |
| IVb | 1.58 ± 0.22 | 1.43 ± 0.23 | 1.2 × 10 |
| V | 1.43 ± 0.23 | 1.32 ± 0.24 | 5.9 × 10 |
| VI | 1.42 ± 0.18 | 1.31 ± 0.18 | 2.4 × 10 |
| VII | 1.54 ± 0.20 | 1.40 ± 0.21 | 3.8 × 10 |
| VIII | 1.51 ± 0.23 | 1.38 ± 0.24 | 8.4 × 10 |
| Image Quality | Liver Homogeneity | Perceived SNR | Lesion Detection | |
|---|---|---|---|---|
| Better | 1 | 24 | 13 | 3 |
| Same | 66 | 39 | 48 | 25 |
| Worse | 0 | 4 | 6 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raspe, J.; Harder, F.N.; Rupp, S.; McTavish, S.; Peeters, J.M.; Weiss, K.; Makowski, M.R.; Braren, R.F.; Karampinos, D.C.; Van, A.T. Retrospective Motion Artifact Reduction by Spatial Scaling of Liver Diffusion-Weighted Images. Tomography 2023, 9, 1839-1856. https://doi.org/10.3390/tomography9050146
Raspe J, Harder FN, Rupp S, McTavish S, Peeters JM, Weiss K, Makowski MR, Braren RF, Karampinos DC, Van AT. Retrospective Motion Artifact Reduction by Spatial Scaling of Liver Diffusion-Weighted Images. Tomography. 2023; 9(5):1839-1856. https://doi.org/10.3390/tomography9050146
Chicago/Turabian StyleRaspe, Johannes, Felix N. Harder, Selina Rupp, Sean McTavish, Johannes M. Peeters, Kilian Weiss, Marcus R. Makowski, Rickmer F. Braren, Dimitrios C. Karampinos, and Anh T. Van. 2023. "Retrospective Motion Artifact Reduction by Spatial Scaling of Liver Diffusion-Weighted Images" Tomography 9, no. 5: 1839-1856. https://doi.org/10.3390/tomography9050146
APA StyleRaspe, J., Harder, F. N., Rupp, S., McTavish, S., Peeters, J. M., Weiss, K., Makowski, M. R., Braren, R. F., Karampinos, D. C., & Van, A. T. (2023). Retrospective Motion Artifact Reduction by Spatial Scaling of Liver Diffusion-Weighted Images. Tomography, 9(5), 1839-1856. https://doi.org/10.3390/tomography9050146








