Comparison between PSMA PET/CT and MRI for Characterizing Hepatocellular carcinoma: A Real-World Study
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
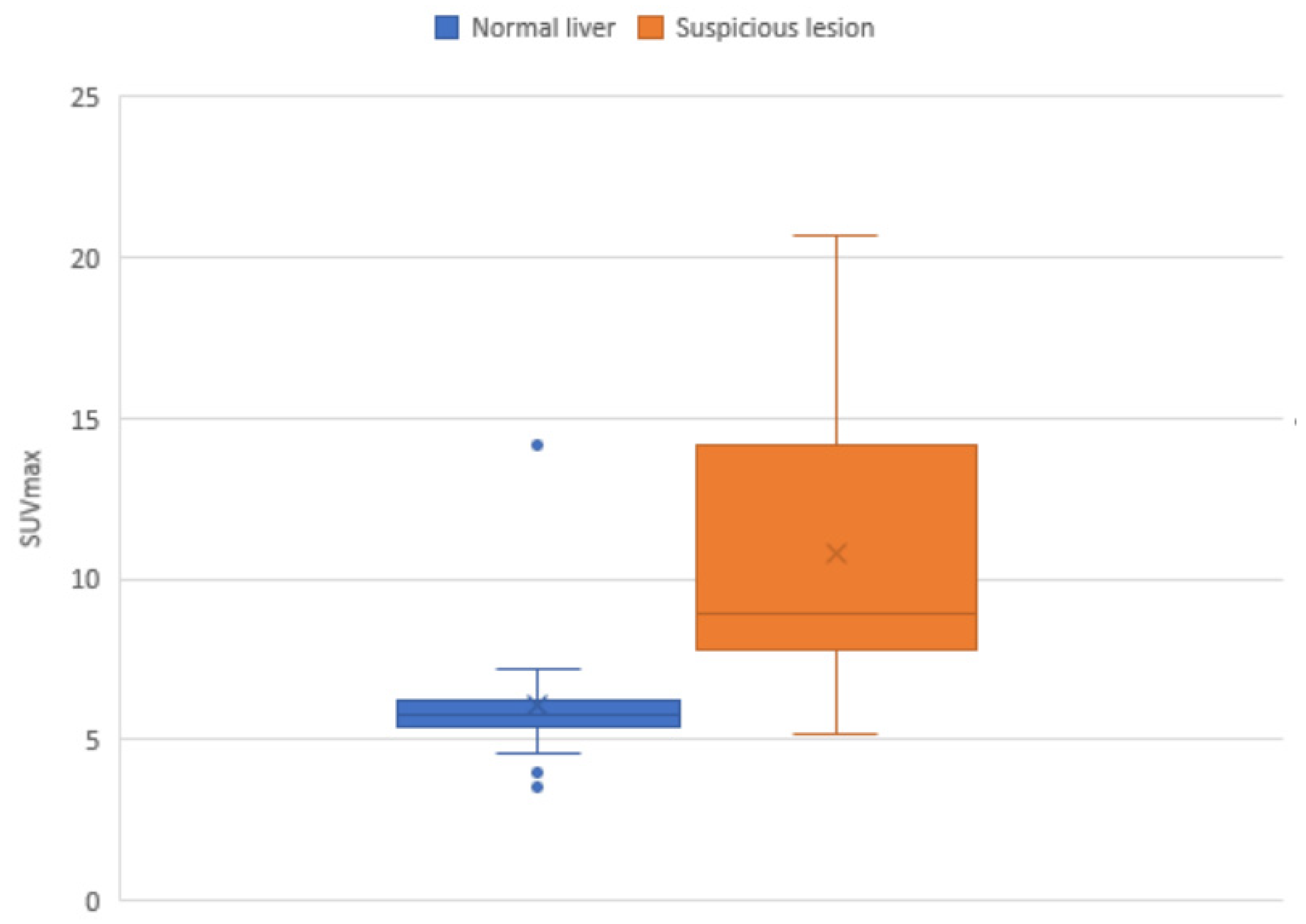
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer 2020. Available online: https://gco.iarc.fr/ (accessed on 5 November 2020).
- Villanueva, A. Hepatocellular Carcinoma. N. Engl. J. Med. 2019, 380, 1450–1462. [Google Scholar] [CrossRef] [PubMed]
- Kansagara, D.; Papak, J.; Pasha, A.S.; O’Neil, M.; Freeman, M.; Relevo, R.; Quiñones, A.; Motu’apuaka, M.; Jou, J.H. Screening for Hepatocellular Carcinoma in Chronic Liver Disease. Ann. Intern. Med. 2014, 161, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Sherman, M. Surveillance for hepatocellular carcinoma. Best Pract. Res. Clin. Gastroenterol. 2014, 28, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.-H.; Yang, B.-H.; Tang, Z.-Y. Randomized controlled trial of screening for hepatocellular carcinoma. J. Cancer Res. Clin. Oncol. 2004, 130, 417–422. [Google Scholar] [CrossRef]
- Patel, D.; Loh, H.; Le, K.; Stevanovic, A.; Mansberg, R. Incidental Detection of Hepatocellular Carcinoma on Ga-68-Labeled Prostate-Specific Membrane Antigen PET/CT. Clin. Nucl. Med. 2017, 42, 881–884. [Google Scholar] [CrossRef]
- Kesler, M.; Levine, C.; Hershkovitz, D.; Mishani, E.; Menachem, Y.; Lerman, H.; Zohar, Y.; Shibolet, O.; Even-Sapir, E. 68 Ga-labeled prostate-specific membrane antigen is a novel PET/CT tracer for imaging of hepatocellular carcinoma: A prospective pilot study. J. Nucl. Med. 2019, 60, 185–191. [Google Scholar] [CrossRef]
- Berger, I.; Annabattula, C.; Lewis, J.; Shetty, D.V.; Kam, J.; Maclean, F.; Arianayagam, M.; Canagasingham, B.; Ferguson, R.; Khadra, M.; et al. (68)Ga-PSMA PET/CT vs. mpMRI for locoregional prostate cancer staging: Correlation with final histopathology. Prostate Cancer Prostatic Dis. 2018, 21, 204–211. [Google Scholar] [CrossRef]
- Sinn, D.H.; Choi, G.-S.; Park, H.C.; Kim, J.M.; Kim, H.; Song, K.D.; Kang, T.W.; Lee, M.W.; Rhim, H.; Hyun, D.; et al. Multidisciplinary approach is associated with improved survival of hepatocellular carcinoma patients. PLoS ONE 2019, 14, e0210730. [Google Scholar] [CrossRef]
- Zhou, J.; Sun, H.; Wang, Z.; Cong, W.; Wang, J.; Zeng, M.; Zhou, W.; Bie, P.; Liu, L.; Wen, T.; et al. Guidelines for the Diagnosis and Treatment of Hepatocellular Carcinoma (2019 Edition). Liver Cancer 2020, 9, 682–720. [Google Scholar] [CrossRef]
- Shetty, D.; Patel, D.; Le, K.; Bui, C.; Mansberg, R. Pitfalls in Gallium-68 PSMA PET/CT Interpretation—A Pictorial Review. Tomography 2018, 4, 182–193. [Google Scholar] [CrossRef]
- Jiao, D.; Li, Y.; Yang, F.; Han, D.; Wu, J.; Shi, S.; Tian, F.; Guo, Z.; Xi, W.; Li, G.; et al. Expression of Prostate-Specific Membrane Antigen in Tumor-Associated Vasculature Predicts Poor Prognosis in Hepatocellular Carcinoma. Clin. Transl. Gastroenterol. 2019, 10, e00041. [Google Scholar] [CrossRef] [PubMed]
- Tolkach, Y.; Goltz, D.; Kremer, A.; Ahmadzadehfar, H.; Bergheim, D.; Essler, M.; Lam, M.; de Keizer, B.; Fischer, H.P.; Kristiansen, G. Prostate-specific membrane antigen expression in hepatocellular carcinoma: Potential use for prognosis and diagnostic imaging. Oncotarget 2019, 10, 4149–4160. [Google Scholar] [CrossRef]
- Chen, L.X.; Zou, S.J.; Li, D.; Zhou, J.Y.; Cheng, Z.T.; Zhao, J.; Zhu, Y.L.; Kuang, D.; Zhu, X.H. Prostate-specific membrane antigen expression in hepatocellular carcinoma, cholangiocarcinoma, and liver cirrhosis. World J. Gastroenterol. 2020, 26, 7664–7678. [Google Scholar] [CrossRef] [PubMed]
- Kuyumcu, S.; Has-Simsek, D.; Iliaz, R.; Sanli, Y.; Buyukkaya, F.; Akyuz, F.; Turkmen, C. Evidence of Prostate-Specific Membrane Antigen Expression in Hepatocellular Carcinoma Using 68Ga-PSMA PET/CT. Clin. Nucl. Med. 2019, 44, 702–706. [Google Scholar] [CrossRef]
- Roberts, L.R.; Sirlin, C.B.; Zaiem, F.; Almasri, J.; Prokop, L.J.; Heimbach, J.K.; Murad, M.H.; Mohammed, K. Imaging for the diagnosis of hepatocellular carcinoma: A systematic review and meta-analysis. Hepatology 2018, 67, 401–421. [Google Scholar] [CrossRef] [PubMed]
- Chernyak, V.; Fowler, K.J.; Kamaya, A.; Kielar, A.Z.; Elsayes, K.M.; Bashir, M.R.; Kono, Y.; Do, R.K.; Mitchell, D.G.; Singal, A.G.; et al. Liver Imaging Reporting and Data System (LI-RADS) Version 2018: Imaging of Hepatocellular Carcinoma in At-Risk Patients. Radiology 2018, 289, 816–830. [Google Scholar] [CrossRef]
- Hong, T.P.; Gow, P.J.; Fink, M.; Dev, A.; Roberts, S.K.; Nicoll, A.; Lubel, J.S.; Kronborg, I.; Arachchi, N.; Ryan, M.; et al. Surveillance improves survival of patients with hepatocellular carcinoma: A prospective population-based study. Med. J. Aust. 2018, 209, 348–354. [Google Scholar] [CrossRef]
- Sacks, A.; Peller, P.J.; Surasi, D.S.; Chatburn, L.; Mercier, G.; Subramaniam, R.M. Value of PET/CT in the Management of Primary Hepatobiliary Tumors, Part 2. Am. J. Roentgenol. 2011, 197, W260–W265. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Nishiyama, Y.; Kameyama, R.; Okano, K.; Kashiwagi, H.; Deguchi, A.; Kaji, M.; Ohkawa, M. Detection of Hepatocellular Carcinoma Using 11C-Choline PET: Comparison with 18F-FDG PET. J. Nucl. Med. 2008, 49, 1245. [Google Scholar] [CrossRef]
- Filippi, L.; Schillaci, O.; Bagni, O. Recent advances in PET probes for hepatocellular carcinoma characterization. Expert Rev. Med. Devices 2019, 16, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Haug, A.R. Imaging of primary liver tumors with positron-emission tomography. Q. J. Nucl. Med. Mol. Imaging 2017, 61, 292–300. [Google Scholar] [CrossRef]
- Goenka, A. 68 Ga PSMA PET/MRI for Hepatocellular Carcinoma—Phase 2 Study Protocol. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT03982407 (accessed on 1 December 2021).
- Dondi, F.; Albano, D.; Cerudelli, E.; Gazzilli, M.; Giubbini, R.; Treglia, G.; Bertagna, F. Radiolabelled PSMA PET/CT or PET/MRI in hepatocellular carcinoma (HCC): A systematic review. Clin. Transl. Imaging 2020, 8, 461–467. [Google Scholar] [CrossRef]
- Lu, Q.; Long, Y.; Fan, K.; Shen, Z.; Gai, Y.; Liu, Q.; Jiang, D.; Cai, W.; Wan, C.; Lan, X. PET imaging of hepatocellular carcinoma by targeting tumor-associated endothelium using [68Ga]Ga-PSMA-617. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 4000–4013. [Google Scholar] [CrossRef] [PubMed]

| Mean age (years) | 65 (SD 7.2) |
| Male | 18/19 (95%) |
| Average BMI (kg/m2) | 31.5 (SD 6.7) |
| Aetiology of liver disease (/19 patients) | Hepatitis C (HCV): 5 (26%) Non-alcoholic steatohepatosis (NASH): 4 (21%) Alcoholic liver disease: 1 (5%) Mixed (HCV and NASH): 1 (5%) Mixed (HCV and alcoholic liver disease): 2 (11%) Unknown: 6 (32%) |
| Lesions | Total assessed: 49 Average lesion/patient: 2.6 * |
| Average PSMA activity administered (MBq) | 285.7 |
| Median AFP at time of study (normal reference ≤ 8 IU/mL) | 5 |
| Previously treated patients | 12/19 (63%) |
| Median follow-up period | 204 days |
| (n) | PSMA (49) | MRI (30) * | CT (49) | Serum AFP (19) * | Average AFP Measurement (19) * |
|---|---|---|---|---|---|
| True negative | 19 | 11 | 27 | 7 | |
| True positive | 20 | 13 | 7 | 5 | |
| False negative | 2 | 2 | 15 | 6 | |
| False positive | 8 | 4 | 0 | 1 | |
| Sensitivity | 91 | 87 | 32 | 45 | |
| Specificity | 70 | 73 | 100 | 88 | |
| PPV | 71 | 76 | 100 | 83 | |
| NPV | 90 | 85 | 64 | 54 |
| Lesion/Patient Initials | Size (Segment) | Previous Treatment | PSMA Uptake | MRI (Performed within 3 Months) | CT | AFP at Time of Scan (Peak) (RR ≤ 8) | Diagnosis (Obtained at MDT) | Progress (Months of Follow-Up Post Study) |
|---|---|---|---|---|---|---|---|---|
| 1 BB | 34 mm (7) | MWA | N | - | A+, PV+, D+ | 4 (30) | SD | SD (11.3) |
| 2 BB | <5 mm (5) | N | N | - | A+, PV+, D+ | SD | ||
| 3 GB | 9 mm (7) | N | N | A+, DWI− | NS | 6 (511) | SD | SD (0.5) |
| 4 GB | 9 mm (5/8) | MWA | N | A-, DWI− | NS | SD | ||
| 5 GB | 19 mm (3) | MWA | N | A−, PV− | Hypodense | SD | ||
| 6 GB | <5 mm (7) | N | N | A+ (sus) | NS | SD | ||
| 7 HB | 47 mm (8) | TACE | Heterogeneous | T2+, T1−, DWI+, A+, progressive washout (sus) | Hypodense, mild PV enhancement (sus) | 197 (214) | PD | PD, palliative, deceased (12.5) |
| 8 HB | 17 mm (2) | N | Y (SUVmax 8.3) | T2+, progressive washout (sus) | NS | PD | ||
| 9 HB | 11 mm (4A) | N | Y (SUVmax 8.9) | T2+, progressive washout (sus) | NS | PD | ||
| 10 HB | 7 mm (6) | N | Y (SUVmax 10.1) | T2+, progressive washout (sus) | NS | PD | ||
| 11 HB | 6 mm (8) | N | Heterogeneous | T2+, progressive washout (sus) | NS | PD | ||
| 12 JB | 17 mm (7/8) | TACE | Y (SUVmax 5.2) | - | Isodense, areas of PV washout (sus) | 23 (44) | PD | PD (9.6), retreated |
| 13 KC | 10 mm (4A/B) | Resected | Y (SUVmax 9.3) | - | NS | 2 (3) | ? PD | SD, false positive (6.4) |
| 14 RC | 38 mm (2) | MWA | N | - | Hypodense | 7 (18) | SD | PD, false negative (6.9) |
| 15 RC | 30 mm (2/3) | TACE | Heterogeneous, SUVmax up to 14.2 | - | Hypodense | SD | ||
| 16 DD | 25 mm (4A) | TACE | Y (SUVmax 8.0) | T1−, T2+, A+, progressive washout (sus) | Hyperdense | 326 (443) | ? PD | PD (6.8) |
| 17 DD | 33 mm (7) | TACE | N | Heterogeneous, T1+, T2+, progressive washout, some peripheral enhancement (sus) | Hypodense | SD | ||
| 18 DD | 37 mm (6) | - | Y (SUVmax 20.5) | T1−, T2+, DWI+, A+, progressive washout (sus) | NS | PD | ||
| 19 DD | 12 mm (4B) | - | Y (SUVmax 5.9) | T1−, T2+, DWI+, A+, progressive washout (sus) | NS | PD | ||
| 20 GE | 31 mm (5) | TACE | N | T1+, T2−, DWI−, A− | Hypodense, subtle arterial enhancement, no significant washout | 1 (2) | SD | SD (3.5) |
| 21 GE | Adjacent to lesion above (5/8) | - | Y (SUVmax 7.2) | T1+, T2−, DWI−, A− | Hypodense, subtle arterial enhancement, no significant washout | PD | ||
| 22 JF | 20 mm (7) | TACE | N | No significant contrast enhancement | Hyperdense, A− | 2 (7) | SD | PD (11.9) |
| 23 JF | 17 mm (1) | - | Y (SUVmax 8.1) | A+ (sus) | NS | PD | ||
| 24 JF | 5 mm (5) | - | N | A+, rapid washout | NS | SD | ||
| 25 JF | 22 mm (6) | MWA | N | A+, rapid washout | A+, no significant washout | SD | ||
| 26 JF | 5 mm (7) | - | N | A+, rapid washout | NS | SD | ||
| 27 RH | 25 mm (7) | SBRT | Y (SUVmax 12.7) | - | Hypodense | 35 (1196) | SD | SD, false positive (2.3) |
| 28 NK | 37 mm (3) | MWA | Y (SUVmax 11.0) | - | A−, no significant portal venous enhancement | 3 (3) | SD | SD, false positive (6.0) |
| 29 NK | 10 mm (7) | - | Y (SUVmax 10.7) | - | NS | SD | ||
| 30 RL | 50 mm (8) | MWA | N | Areas of peripheral T1+ with central T1 isointensity, central T2+ and peripheral T2−, subtle restricted diffusion (sus) | Low attenuation | 2 (4) | SD | SD (6.3) |
| 31 RM | 23 mm (6) | TACE | Y (SUVmax 6.4) | No enhancement | No significant enhancement | 3 (220) | SD | SD, false positive (12.5) |
| 32 RM | 8.4 mm (6/7) | - | N | A+, washout with normalisation | No significant enhancement | SD | ||
| 33 JM | 18 mm (8) | MWA | N | - | No significant enhancement | 15 (15) | SD | PD, false negative (4.5) |
| 34 JM | 13 mm (6) | TACE | N | - | NS | PD | ||
| 35 JM | 12 mm (2) | - | N | - | NS | SD | ||
| 36 JM | 5 mm (4A) | - | N | - | NS | SD | ||
| 37 JM | 5 mm (4B) | - | N | - | NS | PD | ||
| 38 JM | 5 mm (4A/8) | MWA | Y (SUVmax 8.4) | Delayed hypointensity | NS | 5 (7) | PD | PD (9.9) |
| 39 JM | 32 mm (7) | MWA | N | Small T1+ hyperintensity, A- | NS | SD | ||
| 40 FP | 33 mm (8) | MWA | Y (SUVmax 11.6) | A−, hypointense on PV and hepatocyte phase imaging | No significant enhancement | 3 (4) | PD | PD (7.0) |
| 41 RS | 19 mm (2) | - | Y (SUVmax 10.0) | Mildly T2−, isointense T1 FS, DWI+, peripheral A+ and early washout (sus) | Enlarging, low attenuation with A+ and washout (sus) | 18 (30) | PD | PD (8.1) |
| 42 RS | 38 mm (5) | - | Y (SUVmax 16.7) | Mildly T2-, isointense T1 FS, DWI+, peripheral A+ and early washout (sus) | Low attenuation with A+ and washout (sus) | PD | ||
| 43 RS | 19 mm (6) | - | Y (SUVmax 9.6) | Mildly T2−, isointense T1 FS, DWI+, peripheral A+ and early washout (sus) | Low attenuation with A+ and washout (sus) | PD | ||
| 44 RS | 27 mm (5/8) | - | Y (SUVmax 17.4) | Mildly T2−, isointense T1 FS, DWI+, peripheral A+ and early washout (sus) | Enlarging, low attenuation with A+ and washout (sus) | PD | ||
| 45 KU | 10 mm (6) | - | Y (SUVmax 7.6) | - | Subtle arterial enhancement with washout on delayed phase (sus) | 2 (32) | PD | PD (3.6) |
| 46 KU | 19 mm (6) | MWA | Y (SUVmax 8.1) | - | Hypointense, non-enhancing | SD | ||
| 47 KU | 20 mm (4A) | - | Y (SUVmax 7.5) | - | Hypointense, non-enhancing | SD | ||
| 48 KU | 34 mm (5/8) | MWA | Y (SUVmax 7.4) | - | Hypointense, non-enhancing | SD | ||
| 49 SW | 29 mm (7) | MWA | Y (SUVmax 20.7) | - | Non-enhancing | 1 (2) | PD | PD (5.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wong, V.C.K.; Yip, J.; Fragomeli, V.; Weltman, M.; Loh, H.; Le, K.; Nguyen, D.; Bui, C.; Mansberg, R. Comparison between PSMA PET/CT and MRI for Characterizing Hepatocellular carcinoma: A Real-World Study. Tomography 2023, 9, 130-138. https://doi.org/10.3390/tomography9010011
Wong VCK, Yip J, Fragomeli V, Weltman M, Loh H, Le K, Nguyen D, Bui C, Mansberg R. Comparison between PSMA PET/CT and MRI for Characterizing Hepatocellular carcinoma: A Real-World Study. Tomography. 2023; 9(1):130-138. https://doi.org/10.3390/tomography9010011
Chicago/Turabian StyleWong, Veronica Chi Ken, Joshua Yip, Vincenzo Fragomeli, Martin Weltman, Han Loh, Ken Le, Diep Nguyen, Chuong Bui, and Robert Mansberg. 2023. "Comparison between PSMA PET/CT and MRI for Characterizing Hepatocellular carcinoma: A Real-World Study" Tomography 9, no. 1: 130-138. https://doi.org/10.3390/tomography9010011
APA StyleWong, V. C. K., Yip, J., Fragomeli, V., Weltman, M., Loh, H., Le, K., Nguyen, D., Bui, C., & Mansberg, R. (2023). Comparison between PSMA PET/CT and MRI for Characterizing Hepatocellular carcinoma: A Real-World Study. Tomography, 9(1), 130-138. https://doi.org/10.3390/tomography9010011






