Effect of Different Anthropometric Body Indexes on Radiation Exposure in Patients Undergoing Cardiac Catheterisation and Percutaneous Coronary Intervention
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smilowitz, N.R.; Balter, S.; Weisz, G. Occupational hazards of interventional cardiology. Cardiovasc. Revasc. Med. 2013, 14, 223–228. [Google Scholar] [CrossRef] [PubMed]
- The 2007 Recommendations of the International Commission on Radiological Protection; ICRP Publication 103; SAGE Publications Ltd.: London, UK, 2007; Volume 37, pp. 1–332.
- Linet, M.S.; Slovis, T.L.; Miller, D.L.; Kleinerman, R.; Lee, C.; Rajaraman, P.; De Gonzalez, A.B. Cancer risks associated with external radiation from diagnostic imaging procedures. CA A Cancer J. Clin. 2012, 62, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Karatasakis, A.; Brilakis, H.S.; Danek, B.A.; Karacsonyi, J.; Martinez-Parachini, J.R.; Nguyen-Trong, P.-K.J.; Alame, A.J.; Roesle, M.K.; Rangan, B.V.; Rosenfield, K.; et al. Radiation-associated lens changes in the cardiac catheterization laboratory: Results from the IC-CATARACT (CATaracts Attributed to RAdiation in the CaTh lab) study. Catheter. Cardiovasc. Interv. 2017, 91, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; AbAziz, A.; Yusof, A.K. Radiation-Induced Noncancer Risks in Interventional Cardiology: Optimisation of Procedures and Staff and Patient Dose Reduction. BioMed Res. Int. 2013, 2013, 976962. [Google Scholar] [CrossRef] [PubMed]
- Madder, R.D.; VanOosterhout, S.; Mulder, A.; Brock, T.T.; Clarey, A.T.; Parker, J.L.; Jacoby, M.E. Patient Body Mass Index and Physician Radiation Dose During Coronary Angiography. Circ. Cardiovasc. Interv. 2019, 12, e006823. [Google Scholar] [CrossRef] [PubMed]
- Crowhurst, J.A.; Whitby, M.; Savage, M.; Murdoch, D.; Robinson, B.; Shaw, E.; Gaikwad, N.; Saireddy, R.; Hay, K.; Walters, D.L. Factors contributing to radiation dose for patients and operators during diagnostic cardiac angiography. J. Med Radiat. Sci. 2018, 66, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Delewi, R.; Hoebers, L.P.; Råmunddal, T.; Henriques, J.P.; Angerås, O.; Stewart, J.; Robertsson, L.; Wahlin, M.; Petursson, P.; Piek, J.J.; et al. Clinical and Procedural Characteristics Associated with Higher Radiation Exposure during Percutaneous Coronary Interventions and Coronary Angiography. Circ. Cardiovasc. Interv. 2013, 6, 501–506. [Google Scholar] [CrossRef]
- Pancholy, S.B.; Joshi, P.; Shah, S.; Rao, S.V.; Bertrand, O.F.; Patel, T.M. Effect of Vascular Access Site Choice on Radiation Exposure during Coronary Angiography: The Revere Trial (Randomized Evaluation of Vascular Entry Site and Radiation Exposure). JACC Cardiovasc. Interv. 2015, 8, 1189–1196. [Google Scholar] [CrossRef]
- Fung, A.; Soundappan, D.; Loewenstein, D.; Playford, D.; Strange, G.; Kozor, R.; Otton, J.; Ugander, M. Prognostic Association Supports Indexing Size Measures in Echocardiography by Body Surface Area. Heart Lung Circ. 2022, 31, S174. [Google Scholar] [CrossRef]
- Kawel-Boehm, N.; Hetzel, S.J.; Ambale-Venkatesh, B.; Captur, G.; Francois, C.J.; Jerosch-Herold, M.; Salerno, M.; Teague, S.D.; Valsangiacomo-Buechel, E.; van der Geest, R.J.; et al. Reference ranges (“normal values”) for cardiovascular magnetic resonance (CMR) in adults and children: 2020 update. J. Cardiovasc. Magn. Reson. 2020, 22, 1–63. [Google Scholar] [CrossRef]
- Fancher, K.M.; Sacco, A.J.; Gwin, R.C.; Gormley, L.K.; Mitchell, C.B. Comparison of two different formulas for body surface area in adults at extremes of height and weight. J. Oncol. Pharm. Pract. 2016, 22, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Choi, J.-W.; Kim, H. Determination of Body Surface Area and Formulas to Estimate Body Surface Area Using the Alginate Method. J. Physiol. Anthropol. 2008, 27, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Adler, A.C.; Nathanson, B.H.; Raghunathan, K.; McGee, W.T. Effects of Body Surface Area-Indexed Calculations in the Morbidly Obese: A Mathematical Analysis. J. Cardiothorac. Vasc. Anesth. 2013, 27, 1140–1144. [Google Scholar] [CrossRef] [PubMed]
- Stub, D.; Lefkovits, J.; Brennan, A.L.; Dinh, D.; Brien, R.; Duffy, S.J.; Cox, N.; Nadurata, V.; Clark, D.J.; Andrianopoulos, N.; et al. The Establishment of the Victorian Cardiac Outcomes Registry (VCOR): Monitoring and Optimising Outcomes for Cardiac Patients in Victoria. Heart Lung Circ. 2017, 27, 451–463. [Google Scholar] [CrossRef]
- Bloom, J.E.; Dinh, D.T.; Noaman, S.; Martin, C.; Lim, M.; Batchelor, R.; Zheng, W.; Reid, C.; Rn, A.B.; Lefkovits, J.; et al. Adverse impact of chronic kidney disease on clinical outcomes following percutaneous coronary intervention. Catheter. Cardiovasc. Interv. 2020, 97, E801–E809. [Google Scholar] [CrossRef]
- Agarwal, S.; Parashar, A.; Ellis, S.G.; Heupler, F.A.; Lau, E.; Tuzcu, E.M.; Kapadia, S.R. Measures to Reduce Radiation in a Modern Cardiac Catheterization Laboratory. Circ. Cardiovasc. Interv. 2014, 7, 447–455. [Google Scholar] [CrossRef]
- Shah, A.; Das, P.; Subkovas, E.; Buch, A.N.; Rees, M.; Bellamy, C. Radiation Dose during Coronary Angiogram: Relation to Body Mass Index. Heart Lung Circ. 2015, 24, 21–25. [Google Scholar] [CrossRef]
- Bor, D.; Sancak, T.; Olgar, T.; Elcim, Y.; Adanali, A.; Sanlıdilek, U.; Akyar, S. Comparison of effective doses obtained from dose–area product and air kerma measurements in interventional radiology. Br. J. Radiol. 2004, 77, 315–322. [Google Scholar] [CrossRef]
- Huda, W. Kerma-Area Product in Diagnostic Radiology. Am. J. Roentgenol. 2014, 203, W565–W569. [Google Scholar] [CrossRef]
- Kotowycz, M.A.; Johnston, K.W.; Ivanov, J.; Asif, N.; Almoghairi, A.M.; Choudhury, A.; Nagy, C.D.; Sibbald, M.; Chan, W.; Seidelin, P.H.; et al. Predictors of Radial Artery Size in Patients Undergoing Cardiac Catheterization: Insights from the Good Radial Artery Size Prediction (GRASP) Study. Can. J. Cardiol. 2014, 30, 211–216. [Google Scholar] [CrossRef]
- Yoo, B.-S.; Yoon, J.; Ko, J.-Y.; Kim, J.-Y.; Lee, S.-H.; Hwang, S.O.; Choe, K.-H. Anatomical consideration of the radial artery for transradial coronary procedures: Arterial diameter, branching anomaly and vessel tortuosity. Int. J. Cardiol. 2005, 101, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Fetterly, K.A.; Lennon, R.J.; Bell, M.R.; Holmes, D.R.; Rihal, C.S. Clinical Determinants of Radiation Dose in Percutaneous Coronary Interventional Procedures: Influence of Patient Size, Procedure Complexity, and Performing Physician. JACC Cardiovasc. Interv. 2011, 4, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Memon, S.; Larsen, T.R.; Mathew, J. Effect of a low-dose interventional X-ray system on radiation exposure in the higher body surface area patient population. Proc. Bayl. Univ. Med. Cent. 2018, 31, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Mantis, C.; Papadakis, E.; Anadiotis, A.; Kafkas, N.; Patsilinakos, S. Factors affecting radiation exposure during transradial cardiac catheterisation and percutaneous coronary intervention. Clin. Radiol. 2022, 77, e387–e393. [Google Scholar] [CrossRef]
- Sullivan, P.M.; Harrison, D.; Badran, S.; Takao, C.M.; Ing, F.F. Reduction in Radiation Dose in a Pediatric Cardiac Catheterization Lab Using the Philips AlluraClarity X-ray System. Pediatric Cardiol. 2017, 38, 1583–1591. [Google Scholar] [CrossRef]
- Moore, J.D.; Shim, D.; Sweet, J.; Arheart, K.L.; Beekman, R.H., 3rd. Radiation exposure to children during coil occlusion of the patent ductus arteriosus. Catheter. Cardiovasc. Interv. 1999, 47, 449–454. [Google Scholar] [CrossRef]
- Wang, F.; Lu, Y.; Yuan, C.; Bai, J.R.; Yang, H.S.; Wu, L. Evaluation of a low dose imaging protocol on radiation exposure reduction in pediatric supraventricular tachycardia ablation procedure. Zhonghua Er Ke Za Zhi Chin. J. Pediatrics 2017, 55, 272–276. [Google Scholar]
- Nickoloff, E.L.; Lu, Z.F.; Dutta, A.K.; So, J.C. Radiation Dose Descriptors: BERT, COD, DAP, and Other Strange Creatures. RadioGraphics 2008, 28, 1439–1450. [Google Scholar] [CrossRef]
- Smith, K.; Crowhurst, J.; Walters, D.; Starkey, D. Bi-plane and single plane angiography: A study to compare contrast usage and radiation doses for adult cardiac patients in diagnostic studies. Br. J. Radiol. 2018, 92, 20180367. [Google Scholar] [CrossRef]
- Agache, P.; Fanian, F. Body surface area. In Agache’s Measuring the Skin: Non-Invasive Investigations, Physiology, Normal Constants, 2nd ed.; Springer: Cham, Switzerland, 2016; pp. 1565–1566. [Google Scholar]
- Schueler, B.A. The AAPM/RSNA Physics Tutorial for Residents General Overview of Fluoroscopic Imaging. RadioGraphics 2000, 20, 1115–1126. [Google Scholar] [CrossRef]
- Christopoulos, G.; Papayannis, A.C.; Alomar, M.; Kotsia, A.; Michael, T.T.; Rangan, B.V.; Roesle, M.; Shorrock, D.; Makke, L.; Layne, R.; et al. Effect of a real-time radiation monitoring device on operator radiation exposure during cardiac catheterization: The radiation reduction during cardiac catheterization using real-time monitoring study. Circ. Cardiovasc. Interv. 2014, 7, 744–750. [Google Scholar] [CrossRef] [Green Version]
- Chon, M.K.; Chun, K.J.; Lee, D.S.; Lee, S.Y.; Hwang, J.; Lee, S.H.; Hwang, K.W.; Kim, J.S.; Park, Y.H.; Kim, J.H. Radiation reduction during percutaneous coronary intervention: A new protocol with a low frame rate and selective fluoroscopic image storage. Medicine 2017, 96, e7517. [Google Scholar] [CrossRef] [PubMed]
- Musallam, A.; Volis, I.; Dadaev, S.; Abergel, E.; Soni, A.; Yalonetsky, S.; Kerner, A.; Roguin, A. A randomized study comparing the use of a pelvic lead shield during trans-radial interventions: Threefold decrease in radiation to the operator but double exposure to the patient. Catheter. Cardiovasc. Interv. 2015, 85, 1164–1170. [Google Scholar] [CrossRef] [PubMed]
- Pantos, I.; Patatoukas, G.; Katritsis, D.G.; Efstathopoulos, E. Patient Radiation Doses in Interventional Cardiology Procedures. Curr. Cardiol. Rev. 2009, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Le, N.T.T.; Robinson, J.; Lewis, S.J. Obese patients and radiography literature: What do we know about a big issue? J. Med. Radiat. Sci. 2015, 62, 132–141. [Google Scholar] [CrossRef]
- Marcusohn, E.; Postnikov, M.; Musallam, A.; Yalonetsky, S.; Mishra, S.; Kerner, A.; Poliakov, A.; Roguin, A. Usefulness of Pelvic Radiation Protection Shields during Transfemoral Procedures—Operator and Patient Considerations. Am. J. Cardiol. 2018, 122, 1098–1103. [Google Scholar] [CrossRef]
- Politi, L.; Biondi-Zoccai, G.; Nocetti, L.; Costi, T.; Monopoli, D.; Rossi, R.; Sgura, F.; Modena, M.G.; Sangiorgi, G.M. Reduction of scatter radiation during transradial percutaneous coronary angiography: A randomized trial using a lead-free radiation shield. Catheter. Cardiovasc. Interv. 2012, 79, 97–102. [Google Scholar] [CrossRef]
- Blankenship, J.C.; Gigliotti, O.S.; Feldman, D.N.; Mixon, T.A.; Patel, R.A.; Sorajja, P.; Yakubov, S.J.; Chambers, C.E. Ad Hoc percutaneous coronary intervention: A consensus statement from the society for cardiovascular angiography and interventions. Catheter. Cardiovasc. Interv. 2012, 81, 748–758. [Google Scholar] [CrossRef]
- Gierach, M.; Gierach, J.; Ewertowska, M.; Arndt, A.; Junik, R. Correlation between Body Mass Index and Waist Circumference in Patients with Metabolic Syndrome. ISRN Endocrinol. 2014, 2014, 514589. [Google Scholar] [CrossRef] [Green Version]
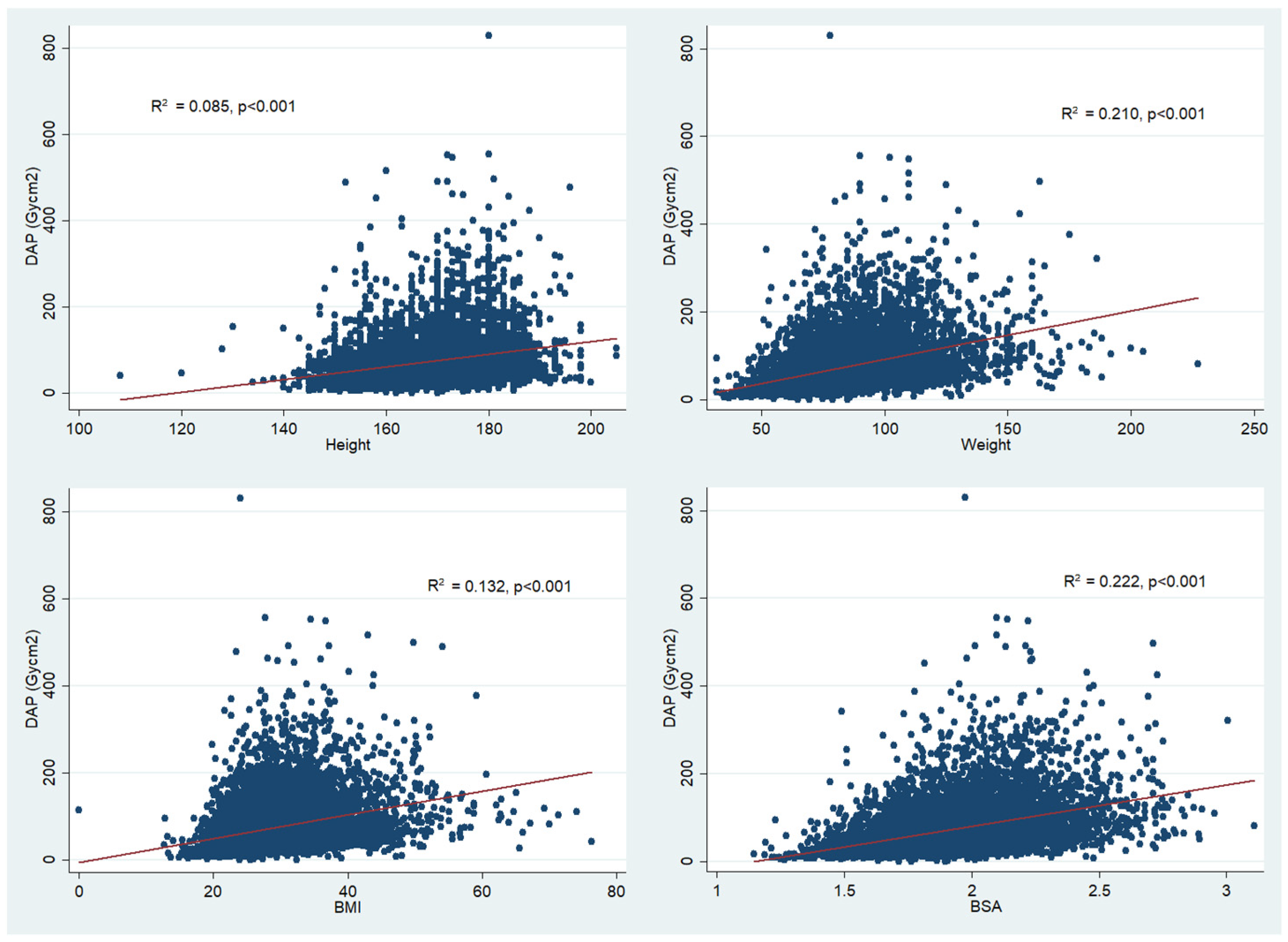
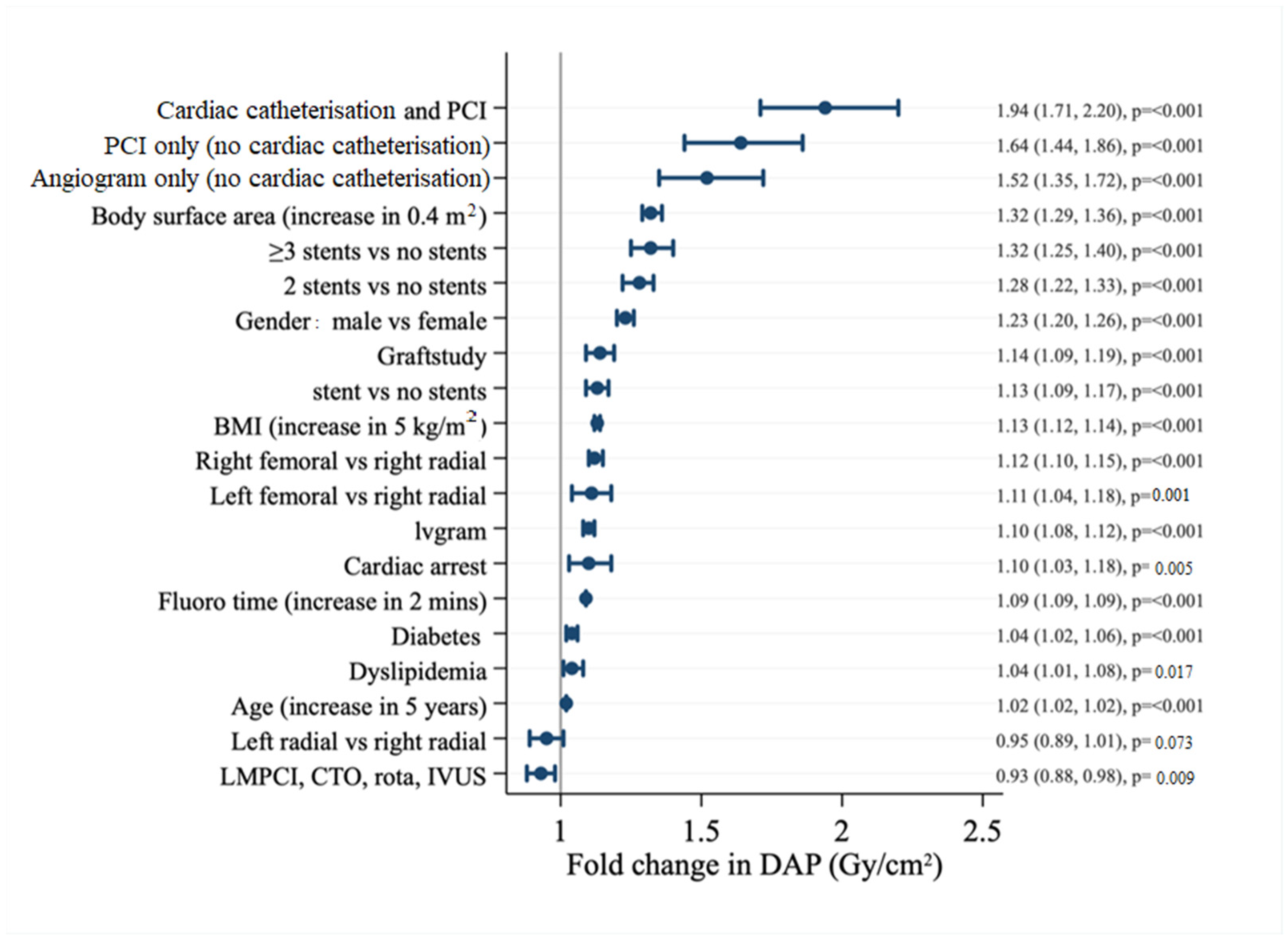
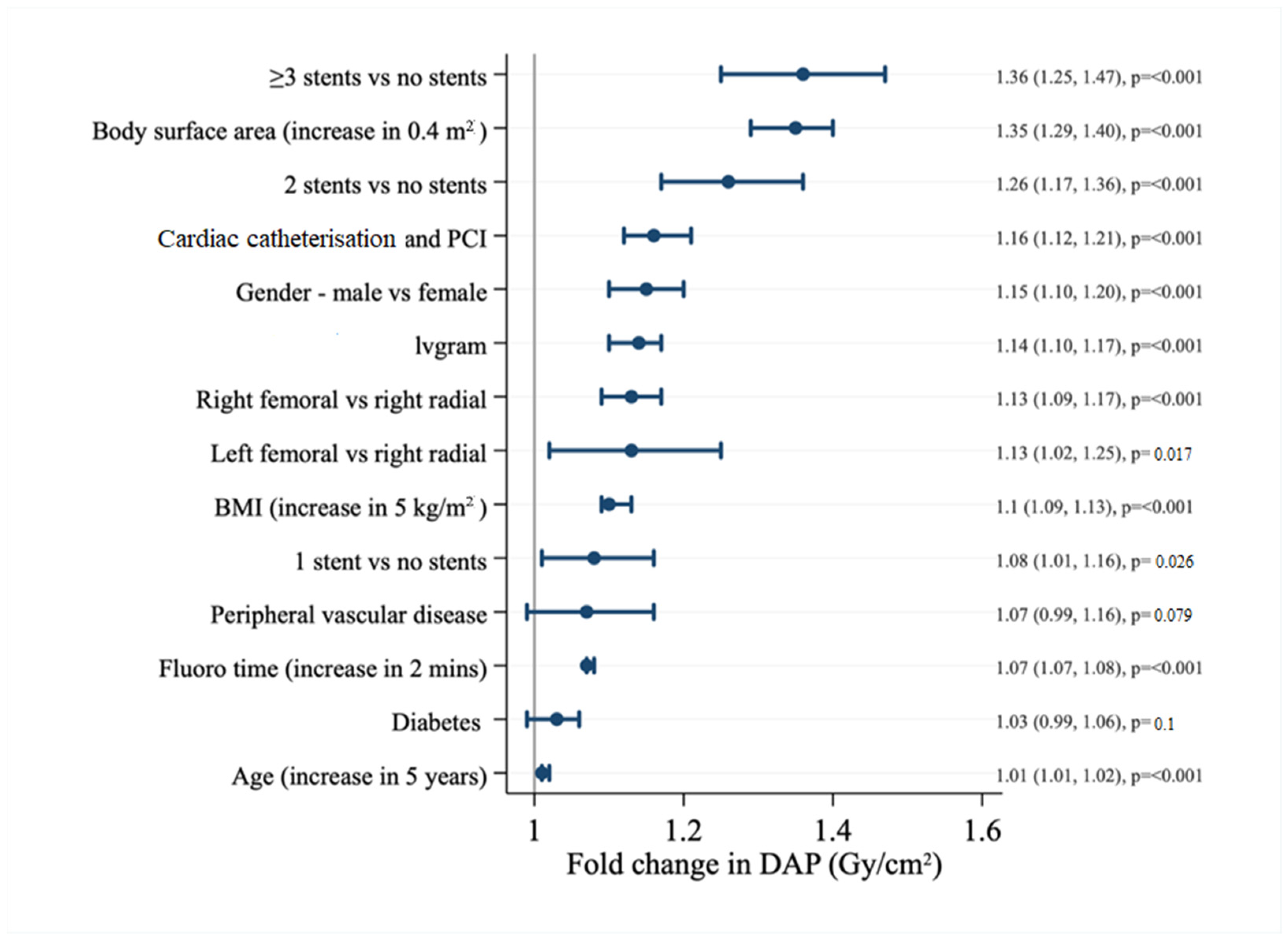
| Factor | BSA < 1.8 m2 | BSA 1.8–2.0 m2 | BSA > 2.0 m2 | p-Value |
|---|---|---|---|---|
| No. of patients | 2322 | 3079 | 2699 | |
| Age, mean (SD) | 70 (12.2) | 66 (12.1) | 61 (12.0) | <0.001 |
| Female | 1397 (60.2%) | 724 (26.8%) | 400 (13.0%) | <0.001 |
| Height, mean (SD) | 155 (7.3) | 170 (7.2) | 176 (7.4) | <0.001 |
| Weight, median (IQR) | 64 (59–70) | 80 (75–84) | 100 (91–110) | <0.001 |
| Body Mass Index (BMI), median (IQR) | 24.6 (22.2–27.5) | 27.6 (25.1–30.8) | 32.4 (29.3–36.8) | <0.001 |
| Peripheral vascular disease | 76 (3.3%) | 104 (3.9%) | 85 (2.8%) | 0.067 |
| Diabetes | 745 (32.1%) | 909 (33.7%) | 1117 (36.3%) | 0.004 |
| Dyslipidaemia | 114 (4.9%) | 203 (7.5%) | 219 (7.1%) | <0.001 |
| Hypertension | 594 (25.6%) | 606 (22.5%) | 704 (22.9%) | 0.02 |
| Ever smoker | 537 (23.1%) | 726 (26.9%) | 859 (27.9%) | <0.001 |
| Cardiac arrest | 17 (0.73%) | 49 (1.82%) | 64 (2.08%) | <0.001 |
| Cardiac catheterisation only (no PCI) | 1463 (63.0%) | 1478 (54.8%) | 1710 (55.5%) | <0.001 |
| PCI only (no cardiac catheterisation) | 128 (5.5%) | 167 (6.2%) | 219 (7.1%) | 0.137 |
| Ad hoc PCI | 600 (25.8%) | 890 (33.0%) | 972 (31.6%) | <0.001 |
| Access site | <0.001 | |||
| Right radial | 1615 (69.6%) | 1970 (73%) | 2360 (76.7%) | |
| Left radial | 51 (2.2%) | 54 (2.0%) | 62 (2.0%) | |
| Right Femoral | 597 (25.7%) | 609 (22.6%) | 597 (19.4%) | |
| Left Femoral | 52 (2.2%) | 55 (2.0%) | 52 (1.7%) | |
| Other | 7 (0.30%) | 11 (0.41%) | 7 (0.23%) | |
| Left ventriculography | 1659 (71.5%) | 1924 (71.3%) | 2242 (72.8%) | 0.364 |
| Factor | BSA < 1.8 m2 | BSA 1.8–2.0 m2 | BSA > 2.0 m2 | p-Value |
|---|---|---|---|---|
| No. of patients | 728 | 1057 | 1191 | |
| Age, mean (SD) | 68.2 (12.5) | 63.4 (12.0) | 60 (11.3) | 0.009 |
| Female | 363 (49.9%) | 199 (18.8%) | 107 (9.0%) | <0.001 |
| Height, mean (SD) | 161 (6.9%) | 170 (6.8%) | 176 (7.0%) | <0.001 |
| Weight, median (IQR) | 65 (60–69) | 80 (75–83) | 98 (90–110) | 0.0001 |
| Body Mass Index (BMI), median (IQR) | 24.2 (22.1–27.0) | 27.0 (25.0–30.1) | 31.7 (28.9–35.9) | 0.0001 |
| Peripheral vascular disease | 26 (3.6%) | 42 (4.0%) | 35 (2.9%) | 0.400 |
| Diabetes | 205 (28.2%) | 270 (25.5%) | 316 (26.5%) | 0.465 |
| Dyslipidaemia | 42 (5.8%) | 96 (9.1%) | 103 (8.7%) | 0.023 |
| Hypertension | 185 (25.4%) | 213 (20.2%) | 242 (20.3%) | 0.014 |
| Ever smoker | 199 (27.3%) | 339 (32.1%) | 377 (31.7%) | 0.069 |
| Cardiac arrest | 17 (2.3%) | 45 (4.3%) | 60 (5.0%) | 0.01 |
| Acute coronary syndrome type | ||||
| Unstable angina | 26 (3.6%) | 32 (3.0%) | 69 (5.8%) | 0.001 |
| NSTEMI | 247 (33.9%) | 324 (30.7%) | 333 (28.0%) | |
| STEMI | 182 (25.0%) | 302 (28.6%) | 339 (28.5%) | |
| Elective | 273 (37.5%) | 399 (37.8%) | 450 (37.8%) | |
| Ad hoc PCI | 600 (82.4%) | 890 (84.2%) | 972 (81.6%) | 0.260 |
| Complex PCI | 55 (7.6%) | 63 (6.0%) | 68 (5.7%) | 0.239 |
| Left main PCI | 11 (0.15%) | 21 (0.20%) | 13 (0.11%) | 0.221 |
| CTO | 30 (4.12%) | 31 (1.15%) | 49 (1.59%) | 0.261 |
| Rotablation | 11 (0.47%) | 10 (0.37%) | 7 (0.23%) | 0.127 |
| IVUS | 15 (0.65%) | 26 (0.96%) | 22 (0.71%) | 0.598 |
| Graft study | 20 (2.8%) | 30 (2.8%) | 23 (1.9%) | 0.321 |
| Access site | 0.001 | |||
| Right radial | 486 (66.8%) | 792 (75.9%) | 908 (76.2%) | |
| Left radial | 10 (1.4%) | 11 (1.0%) | 18 (1.5%) | |
| Right Femoral | 212 (29.1%) | 228 (21.6%) | 235 (19.7%) | |
| Left Femoral | 17 (2.3%) | 22 (2.1%) | 25 (2.1%) | |
| Other | 3 (0.41%) | 4 (0.38%) | 5 (0.42%) | |
| Left ventriculography | 451 (62%) | 697 (65.9%) | 749 (62.9%) | 0.165 |
| Number of stents | ||||
| 0 | 32 (4.4%) | 46 (4.4%) | 57 (4.8%) | 0.814 |
| 1 | 430 (59.7%) | 627 (59.3%) | 732 (61.5%) | |
| 2 | 191 (26.2%) | 269 (25.5%) | 277 (23.3%) | |
| ≥3 | 75 (10.3%) | 115 (10.9%) | 125 (10.5%) |
| Factor | BSA < 1.8 m2 | BSA 1.8–2.0 m2 | BSA > 2.0 m2 | p-Value |
|---|---|---|---|---|
| Fluoroscopy Time Indexed to BSA (min/m2), median (IQR) | 3.5 (1.9–6.3) | 3.6 (1.9–5.9) | 3.2 (1.7–5.5) | 0.0001 |
| DAP Gycm2, median (IQR) | 36.7 (23.6–59.2) | 56.2 (37.1–86.0) | 79.9 (52.3–123) | <0.001 |
| Air Kerma mGy, median (IQR) | 506 (323–840) | 758 (497–1240) | 1070 (689–1740) | <0.001 |
| Peak Skin Dose mGy, median (IQR) | 200 (120–410) | 320 (180–640) | 440 (250–890) | <0.001 |
| Factor | BSA < 1.8 m2 | BSA 1.8–2.0 m2 | BSA > 2.0 m2 | p-Value |
|---|---|---|---|---|
| Fluoroscopy Time Indexed to BSA (min/m2), median (IQR) | 6.8 (4.7–9.8) | 5.7 (4.0–8.3) | 5.3 (3.7–8.0) | 0.0001 |
| DAP Gycm2, median (IQR) | 63.8 (44.6–94.1) | 82.4 (60.7–120) | 119 (85.0–169) | <0.001 |
| Air Kerma mGy, median (IQR) | 91 (673–1490) | 1260 (880–1820) | 1800 (1270–2560) | <0.001 |
| Peak Skin Dose mGy, median (IQR) | 520 (350–800) | 680 (460–970) | 940 (650–1380) | <0.001 |
| R2 | p-Value | |
|---|---|---|
| Model without anthropometric measures | 0.531 | |
| Model + height | 0.544 | 1.000 |
| Model + weight | 0.686 | ≤0.001 |
| Model + BMI | 0.673 | ≤0.001 |
| Model + BSA | 0.672 | ≤0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koh, Y.; Vogrin, S.; Noaman, S.; Lam, S.; Pham, R.; Clark, A.; Biffin, L.; Hanson, L.B.; Bloom, J.E.; Stub, D.; et al. Effect of Different Anthropometric Body Indexes on Radiation Exposure in Patients Undergoing Cardiac Catheterisation and Percutaneous Coronary Intervention. Tomography 2022, 8, 2256-2267. https://doi.org/10.3390/tomography8050189
Koh Y, Vogrin S, Noaman S, Lam S, Pham R, Clark A, Biffin L, Hanson LB, Bloom JE, Stub D, et al. Effect of Different Anthropometric Body Indexes on Radiation Exposure in Patients Undergoing Cardiac Catheterisation and Percutaneous Coronary Intervention. Tomography. 2022; 8(5):2256-2267. https://doi.org/10.3390/tomography8050189
Chicago/Turabian StyleKoh, Youlin, Sara Vogrin, Samer Noaman, Simon Lam, Raymond Pham, Andrew Clark, Leah Biffin, Laura B. Hanson, Jason E. Bloom, Dion Stub, and et al. 2022. "Effect of Different Anthropometric Body Indexes on Radiation Exposure in Patients Undergoing Cardiac Catheterisation and Percutaneous Coronary Intervention" Tomography 8, no. 5: 2256-2267. https://doi.org/10.3390/tomography8050189
APA StyleKoh, Y., Vogrin, S., Noaman, S., Lam, S., Pham, R., Clark, A., Biffin, L., Hanson, L. B., Bloom, J. E., Stub, D., Brennan, A. L., Reid, C., Dinh, D. T., Lefkovits, J., Cox, N., & Chan, W. (2022). Effect of Different Anthropometric Body Indexes on Radiation Exposure in Patients Undergoing Cardiac Catheterisation and Percutaneous Coronary Intervention. Tomography, 8(5), 2256-2267. https://doi.org/10.3390/tomography8050189





