Dermoid Cysts of the Asterion: An Unusual Location for Unusual Dermoids, Radiological Findings and Neurosurgical Implications
Abstract
1. Introduction
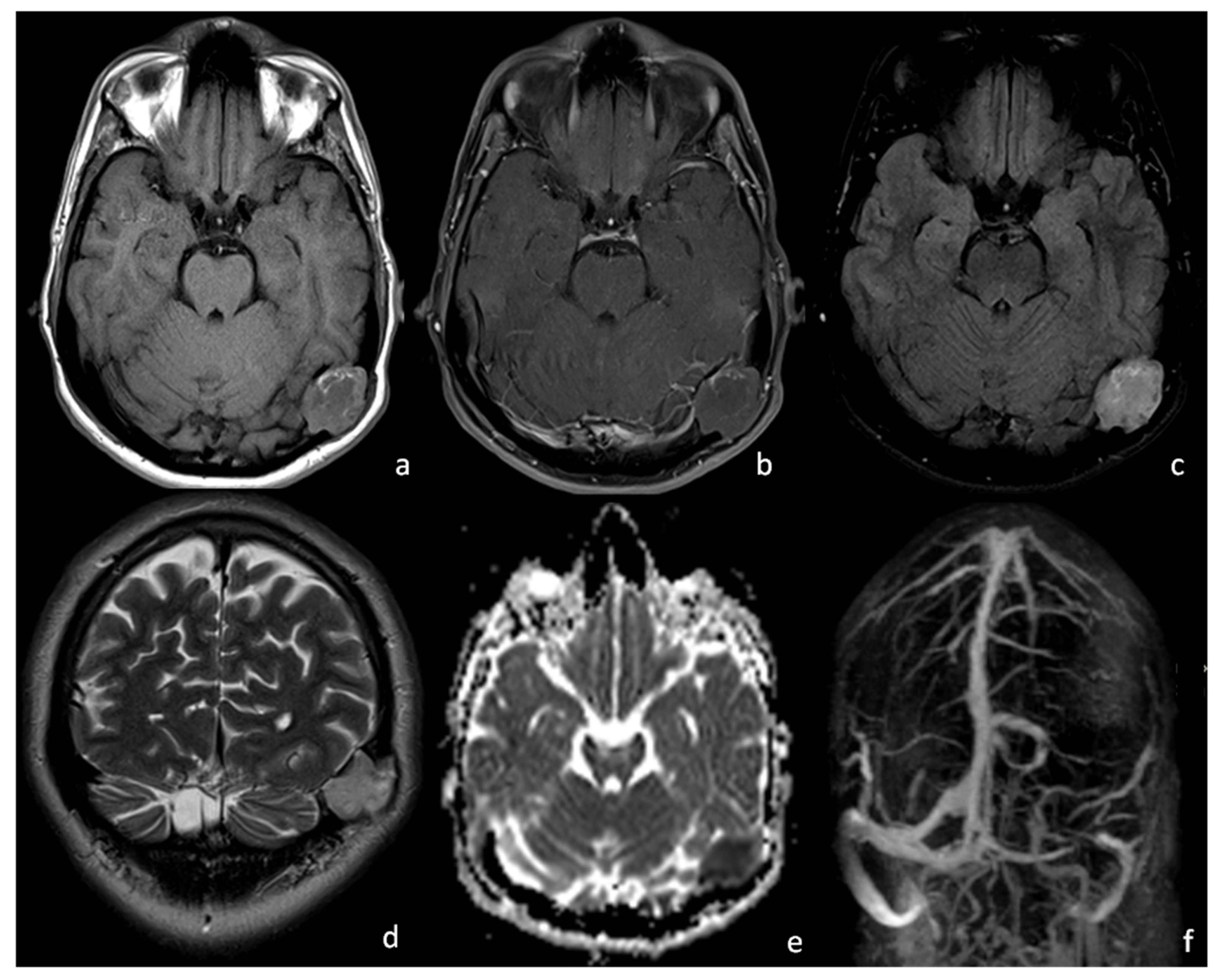
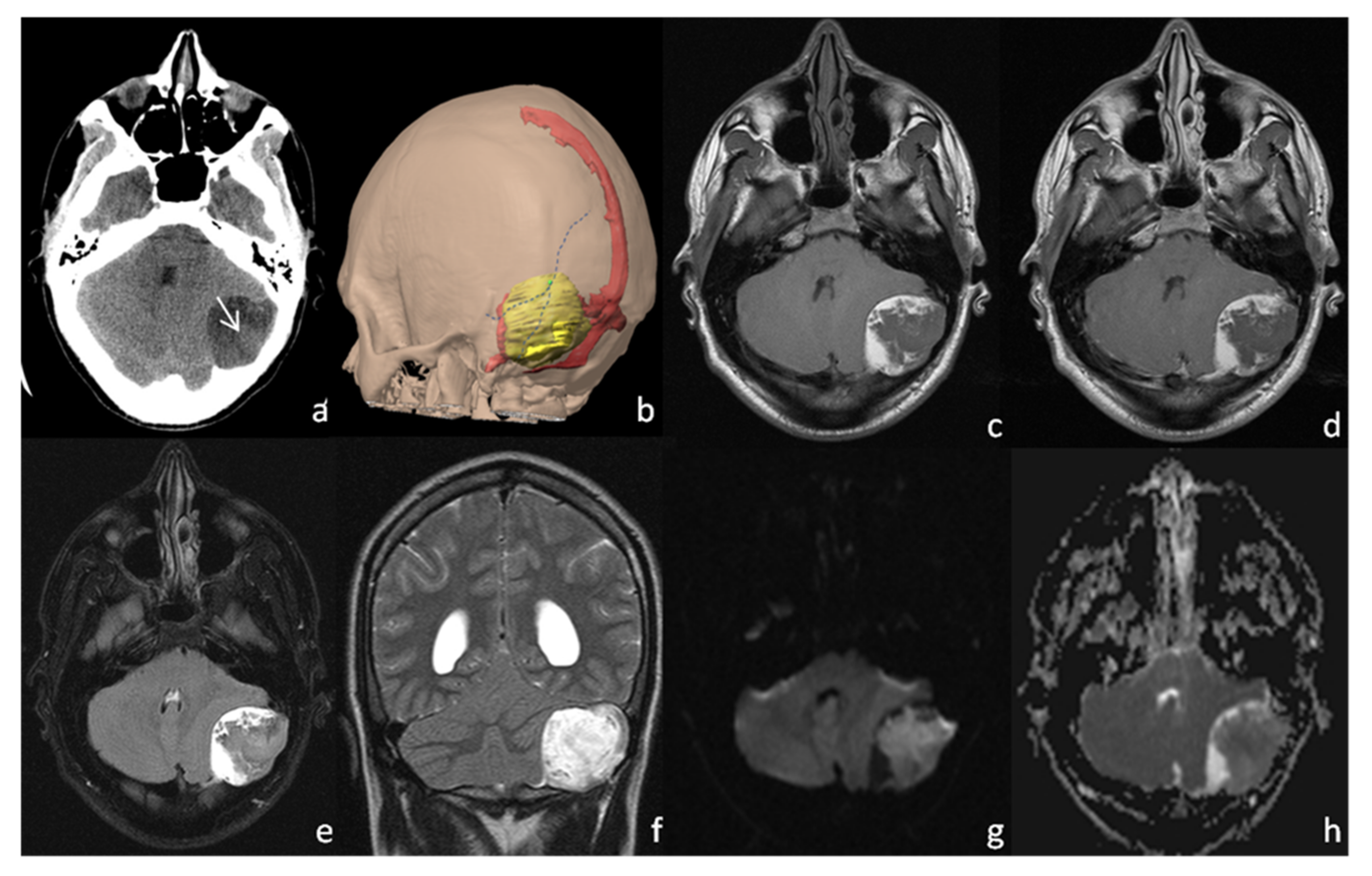
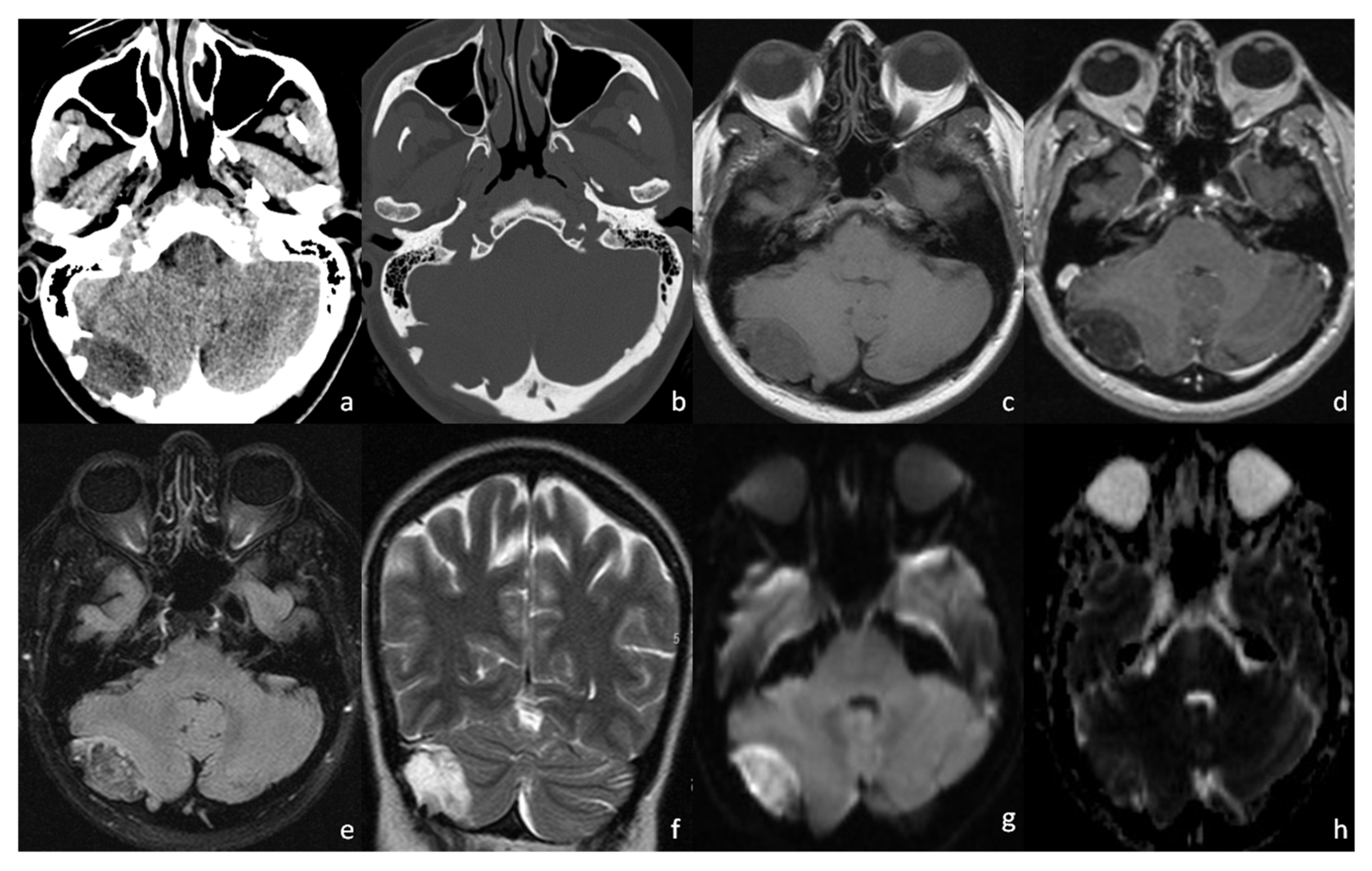
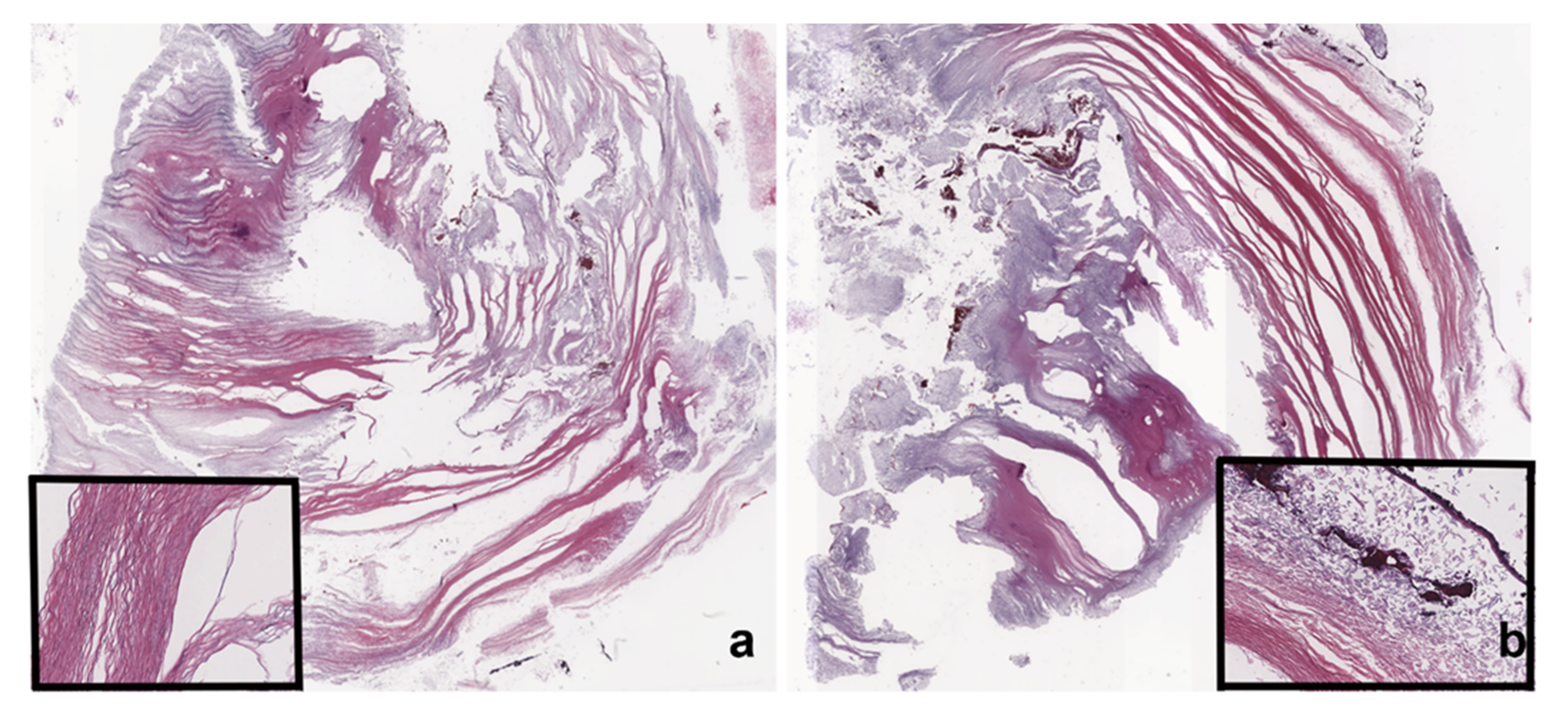
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Day, J.D.; Tschabitscher, M. Anatomic Position of the Asterion. Neurosurgery 1998, 42, 198–199. [Google Scholar] [CrossRef] [PubMed]
- Duangthongpon, P.; Thanapaisal, C.; Kitkhuandee, A.; Chaiciwamongkol, K.; Morthong, V. The Relationships between Asterion, the Transverse-Sigmoid Junction, the Superior Nuchal Line and the Transverse Sinus in Thai Cadavers: Surgical Relevance. J. Med. Assoc. Thail. 2016, 99 (Suppl. S5), S127–S131. [Google Scholar] [PubMed]
- Ucerler, H.; Govsa, F. Asterion as a Surgical Landmark for Lateral Cranial Base Approaches. J. Cranio-Maxillofac. Surg. 2006, 34, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Joharapurkar, S.R.; Shahapurkar, V.V. Giant Asterional Dermoid Cyst with Dermal Sinus; a Case Report. Iran. J. Pediatrics 2009, 19, 439–441. [Google Scholar]
- Osborn, A.G.; Hedlund, G.L.; Salzman, K.L. Osborn’s Brain: Imaging, Pathology, and Anatomy; Taylor & Francis: Salt Lake City, UT, USA, 2018; ISBN 9780323553179. [Google Scholar]
- Saito, A.; Ishida, T.; Inoue, T.; Inoue, T.; Suzuki, S.; Ezura, M.; Uenohara, H. Infantile Dermoid Cyst in the Lateral Wall of the Cavernous Sinus: A Case Report and Literature Review. NMC Case Rep. J. 2018, 5, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Smirniotopoulos, J.G.; Chiechi, M.V. Teratomas, Dermoids, and Epidermoids of the Head and Neck. Radiographics 1995, 15, 1437–1455. [Google Scholar] [CrossRef] [PubMed]
- Pannell, B.W.; Hendrick, E.B.; Hoffman, H.J.; Humphreys, R.P. Dermoid Cysts of the Anterior Fontanelle. Neurosurgery 1982, 10, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Ray, M.J.; Jordan Ray, M.; Barnett, D.W.; Snipes, G.J.; Layton, K.F.; Opatowsky, M.J. Ruptured Intracranial Dermoid Cyst. Bayl. Univ. Med. Cent. Proc. 2012, 25, 23–25. [Google Scholar] [CrossRef] [PubMed]
- Teranishi, Y.; Kohno, M.; Sora, S.; Sato, H. Determination of the Keyhole Position in a Lateral Suboccipital Retrosigmoid Approach. Neurol. Med. Chir. 2014, 54, 261–266. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schaefer, P.W.; Ellen Grant, P.; Gilberto Gonzalez, R. Diffusion-Weighted MR Imaging of the Brain. Radiology 2000, 217, 331–345. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.Y.; Morokoff, A.P.; Mitchell, P.J.; Gonzales, M.F. Unusual Imaging Appearance of an Intracranial Dermoid Cyst. AJNR Am. J. Neuroradiol. 2001, 22, 1970–1972. [Google Scholar] [PubMed]
- Wallace, D.; Tress, B.; Kwan, P.F. Radiologically Atypical Congenital Posterior Fossa Dermoid Cyst Presenting Late in Life. J. Clin. Neurosci. 2008, 15, 835–838. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, S.P.; Srinivasa, R.; Ghosal, N. Unusual Radiological Presentation of Intracranial Dermoid Cyst: A Case Series. Asian J. Neurosurg. 2019, 14, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Balasundaram, P.; Garg, A.; Prabhakar, A.; Joseph Devarajan, L.S.; Gaikwad, S.B.; Khanna, G. Evolution of Epidermoid Cyst into Dermoid Cyst: Embryological Explanation and Radiological-Pathological Correlation. Neuroradiol. J. 2019, 32, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Day, J.D.; Diaz Day, J.; Kellogg Jordi, X.; Manfred, T.; Takanori, F. Surface and Superficial Surgical Anatomy of the Posterolateral Cranial Base: Significance for Surgical Planning and Approach. Neurosurgery 1996, 38, 1079–1084. [Google Scholar] [PubMed]
- Kim, Y.-S.; Jung, S.-H.; Lim, D.-H.; Kim, T.-S.; Kim, J.-H.; Lee, J.-K. Traumatic Dural Venous Sinus Injury. Korean J. Neurotrauma 2015, 11, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Ironside, J.W. Pathology of Tumours of the Nervous System, 5th ed.; Russell, D.S., Rubinstein, L.J., Eds.; Edward Arnold: London, UK, 1989. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarnera, A.; Trasimeni, G.; Romano, A.; Stoppacciaro, A.; Serio, M.; Miscusi, M.; Bozzao, A. Dermoid Cysts of the Asterion: An Unusual Location for Unusual Dermoids, Radiological Findings and Neurosurgical Implications. Tomography 2022, 8, 1141-1147. https://doi.org/10.3390/tomography8020093
Guarnera A, Trasimeni G, Romano A, Stoppacciaro A, Serio M, Miscusi M, Bozzao A. Dermoid Cysts of the Asterion: An Unusual Location for Unusual Dermoids, Radiological Findings and Neurosurgical Implications. Tomography. 2022; 8(2):1141-1147. https://doi.org/10.3390/tomography8020093
Chicago/Turabian StyleGuarnera, Alessia, Guido Trasimeni, Andrea Romano, Antonella Stoppacciaro, Mattia Serio, Massimo Miscusi, and Alessandro Bozzao. 2022. "Dermoid Cysts of the Asterion: An Unusual Location for Unusual Dermoids, Radiological Findings and Neurosurgical Implications" Tomography 8, no. 2: 1141-1147. https://doi.org/10.3390/tomography8020093
APA StyleGuarnera, A., Trasimeni, G., Romano, A., Stoppacciaro, A., Serio, M., Miscusi, M., & Bozzao, A. (2022). Dermoid Cysts of the Asterion: An Unusual Location for Unusual Dermoids, Radiological Findings and Neurosurgical Implications. Tomography, 8(2), 1141-1147. https://doi.org/10.3390/tomography8020093






