Influence of Spatial Resolution and Compressed SENSE Acceleration Factor on Flow Quantification with 4D Flow MRI at 3 Tesla
Abstract
:1. Introduction
2. Materials and Methods
2.1. Flow Models and Circulation Setup
2.2. Magnetic Resonance Imaging
2.3. Time-Resolved Hemodynamic Simulations
2.4. Data Processing
- The linear offset phase correction was conducted on each slice individually to correct for the presence of eddy currents. The fit was calculated at the reference heart phase (at the peak flow time) and then applied to all heart phases. A phase correction to compensate for concomitant gradients (Maxwell terms) and geometry correction to compensate for inhomogeneities of the main magnetic field and non-linearity of the gradient fields was performed on MR systems as part of the standard phase-contrast MR image reconstruction.
- Velocities in voxels outside of the flow lumen were nulled based on a magnitude intensity threshold.
- The data were inspected against phase-aliasing and manually corrected if necessary.
- ROIs were created manually on MRI magnitude data. First, contours around the tube’s lumen were drawn using a b-spline curve (feature in GTflow) on 2D flow MRI. Note that the 2D flow MRI acquisition planes are already perpendicular to the flow direction. Next, the resulting 2D flow ROIs were translated to the 4D flow MRI data, ensuring identical placement of ROIs on the 2D and 4D flow datasets.
- In a given ROI, the flow of all voxels was summed up for each time point , where i indicates the voxel and t the temporal point. The flow was spatially averaged over ROI A-C, as follows: .
- The number of voxels per ROI diameter (nROI) was calculated to obtain a measurement not depending on the voxel size and vessel diameter as .
- The time-dependent difference between flow values obtained with 4D and 2D flow MRI was calculated as . Similarly, the difference between flow values obtained with 4D flow MRI and US sensor was calculated.
- The normalized root-mean-square (RMS) error was used to assess the accuracy of flow quantification. RMS was calculated as the sum of squared differences between 2D and 4D flow MRI data over the time steps and normalized by the time-averaged flow acquired with 2D flow MRI: , where t indicates the temporal measurement point and Num is the number of temporal points. Similarly, RMS between flow values obtained with 4D flow MRI and US sensor was calculated.
- Time-averaged velocity magnitude in the evaluation plane across the aneurysm was visualized pixel-wise on a color-coded representation (MATLAB R2019a, MathWork, Natick, MA, USA).
- Repeatability of flow measurements with 2D flow MRI was assessed with repeatability coefficient (RC) as follows: (1) 2D flow MRI was measured five times in parental vessel and at the aneurysm sac; (2) RC was calculated for each time point using the equation adapted from Raunig et al. [39] , where SD is the standard deviation and is the mean flow rate over five measurements; (3) time-depended RC were time-averaged as , where t indicates the temporal measurement point and Num is the number of temporal points.
2.5. Statistical Analysis
3. Results
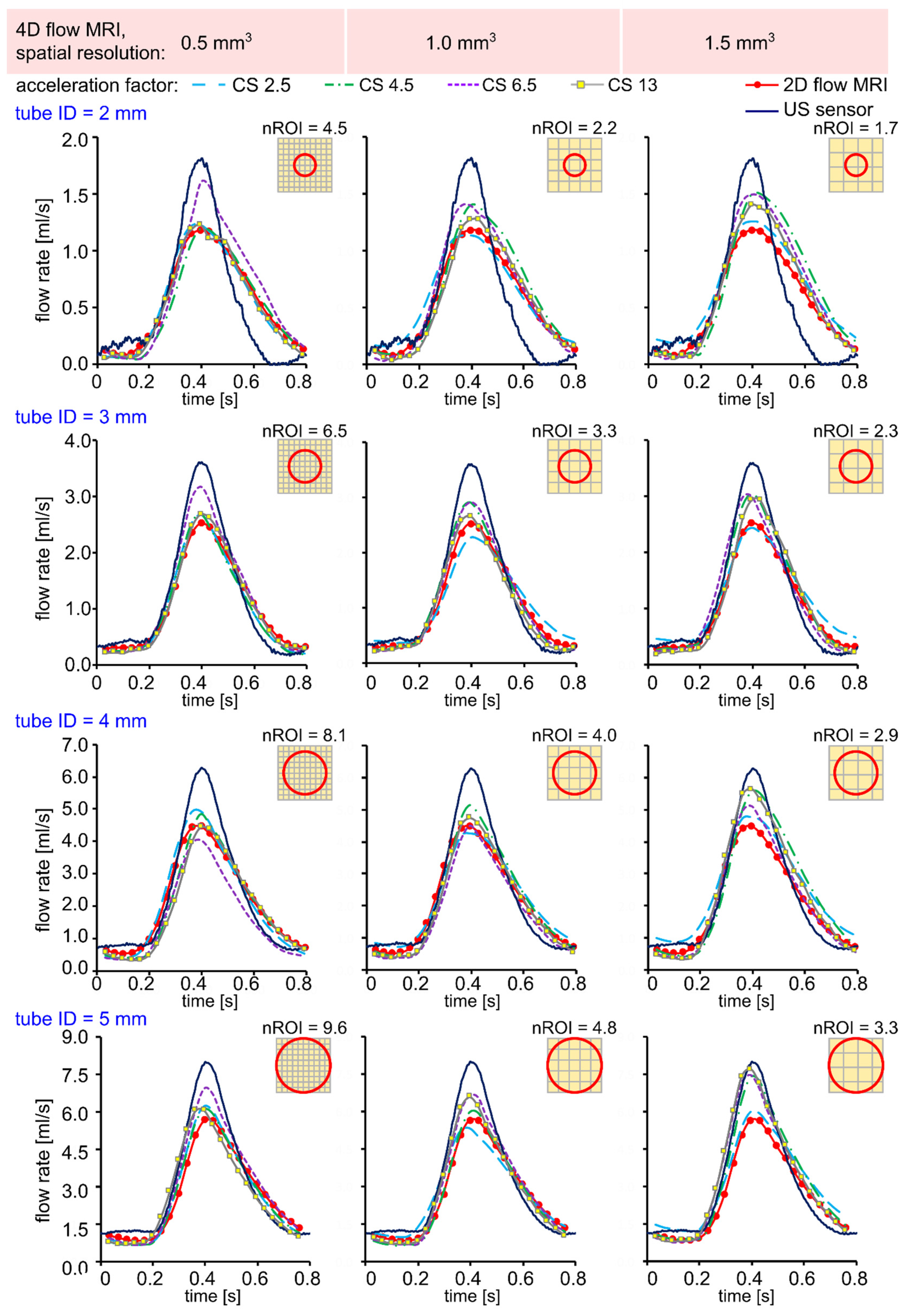
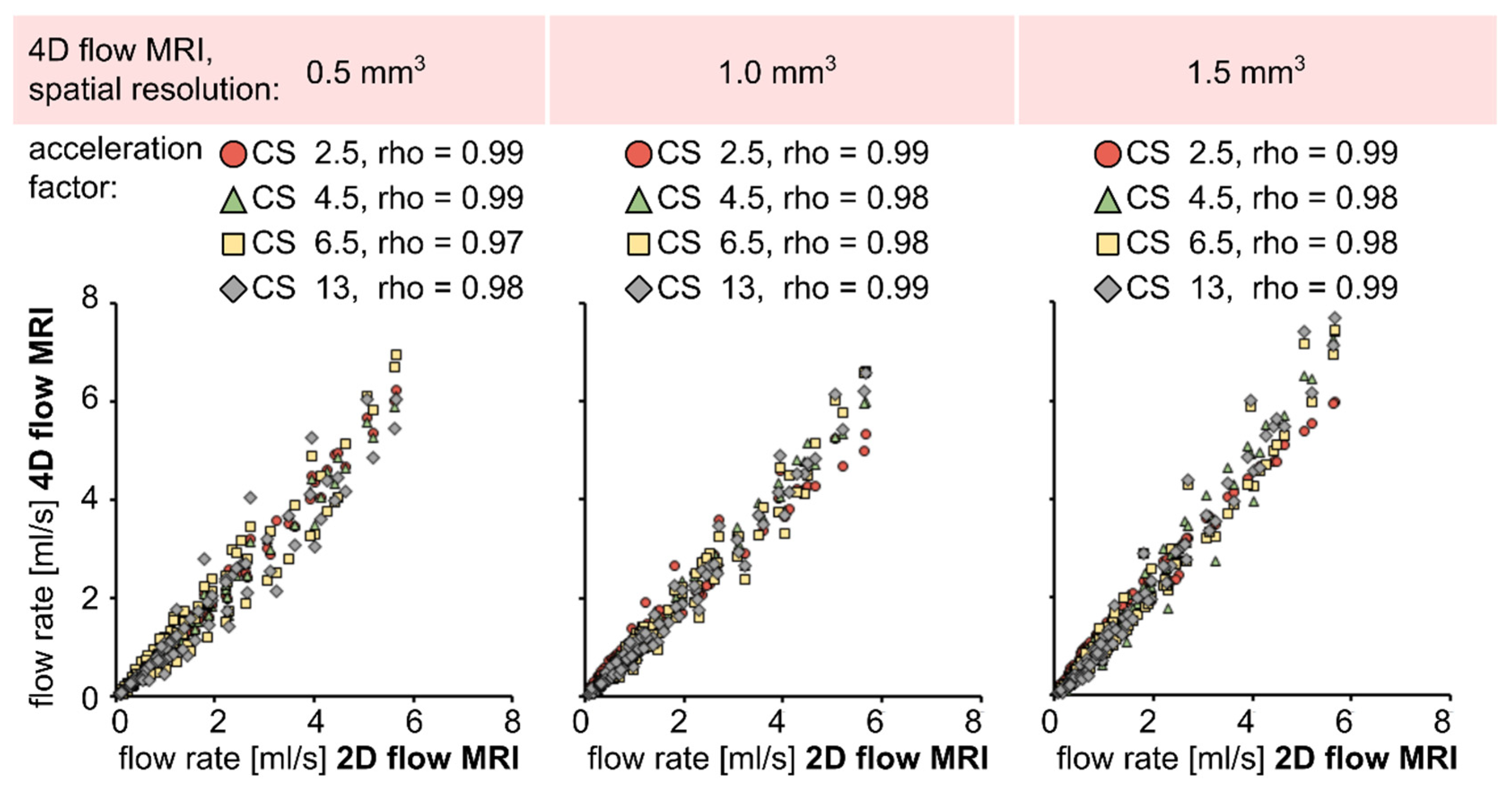
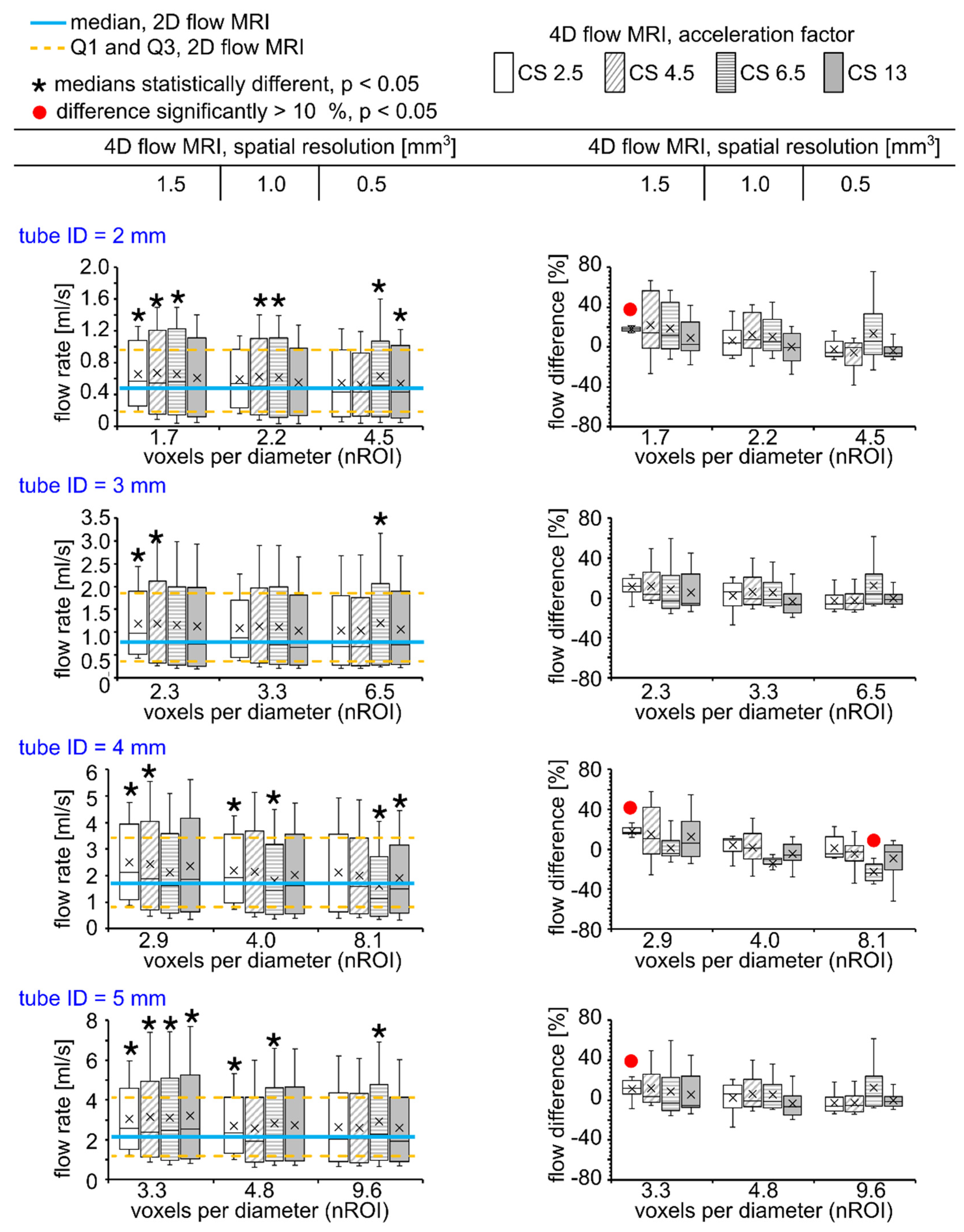
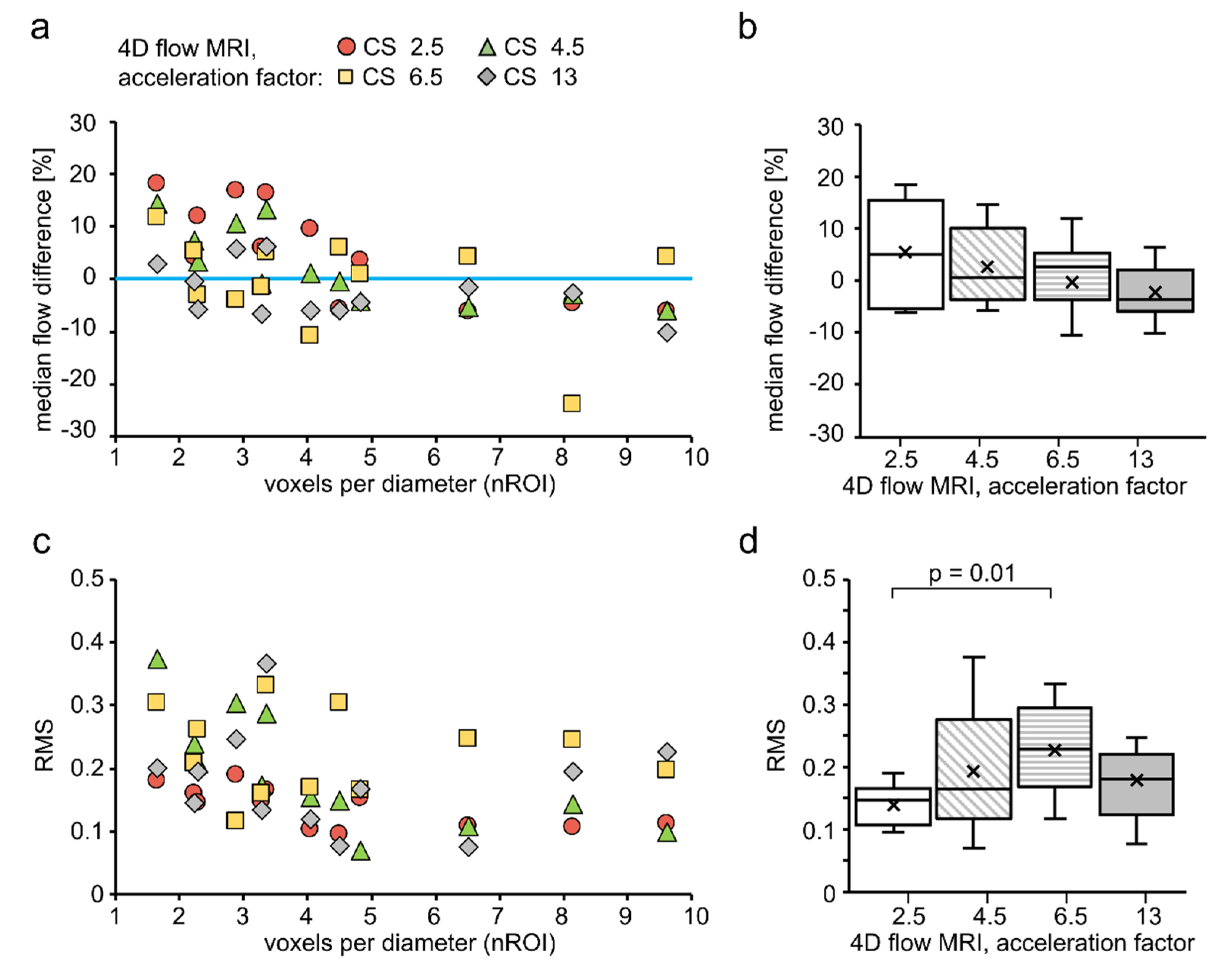
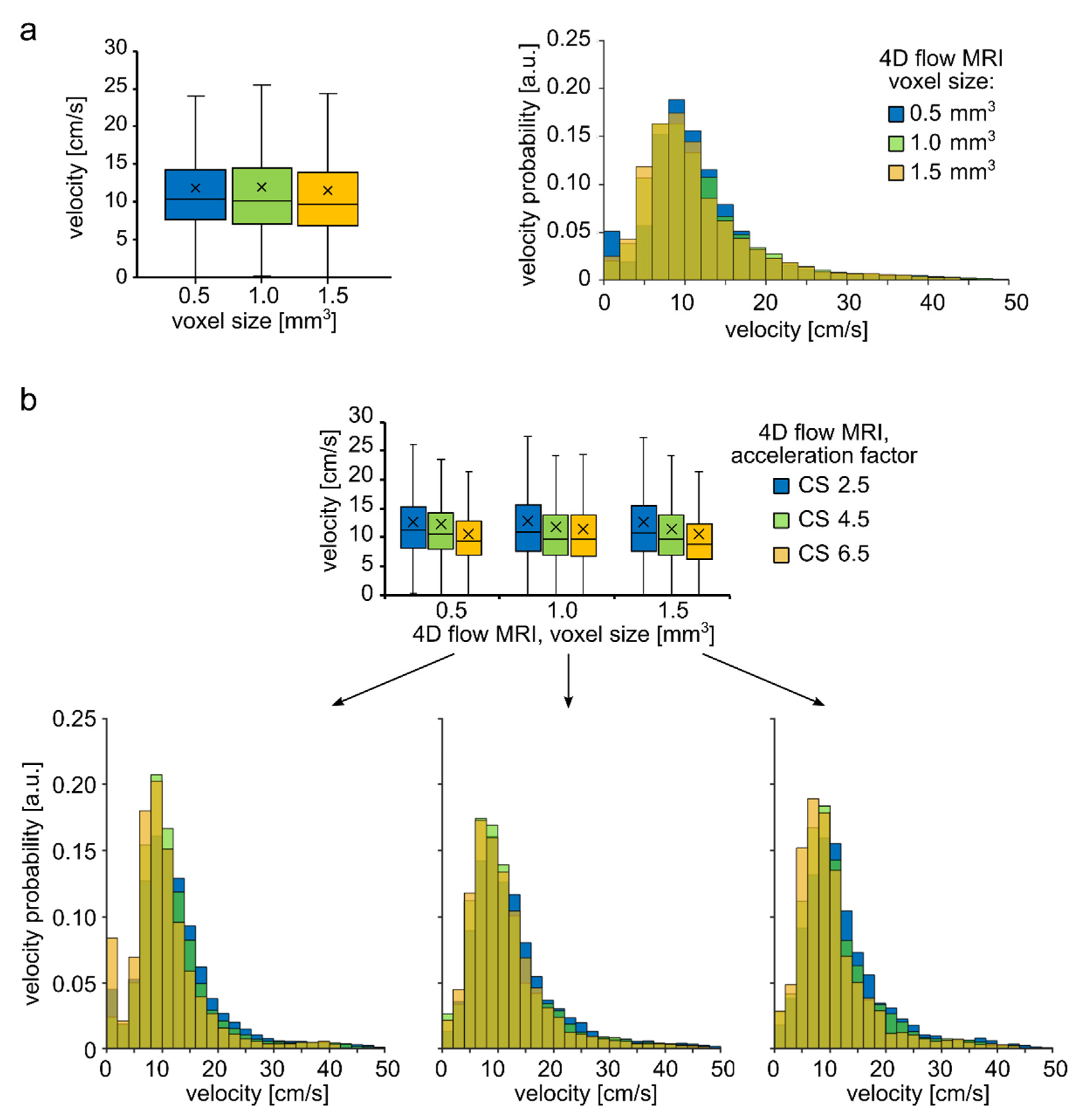
3.1. Flow in Silicone Tubes
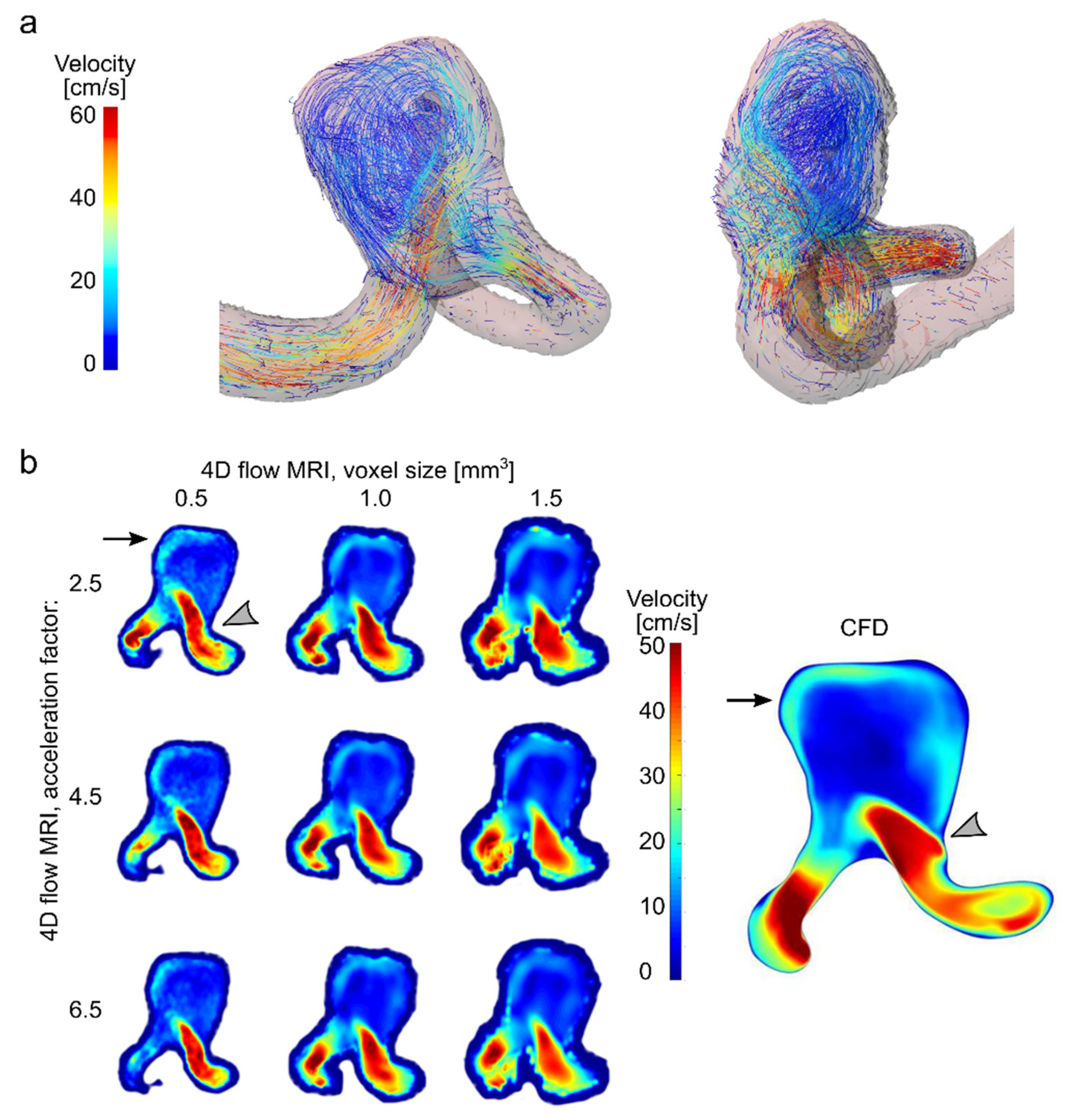
3.2. Velocity in an Aneurysm Model
4. Discussion
4.1. Effect of Spatial Resolution and MR Acceleration on the Flow in Silicone Tubes
4.2. Velocity in an Aneurysm Model
4.3. Limitation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, L.; Barlinn, K.; Sharma, V.K.; Tsivgoulis, G.; Cava, L.F.; Vasdekis, S.N.; Teoh, H.L.; Triantafyllou, N.; Chan, B.P.; Sharma, A.; et al. Velocity criteria for intracranial stenosis revisited. Stroke 2011, 42, 3429–3434. [Google Scholar] [CrossRef] [PubMed]
- Hope, T.A.; Hope, M.D.; Purcell, D.D.; von Morze, C.; Vigneron, D.B.; Alley, M.T.; Dillon, W.P. Evaluation of intracranial stenoses and aneurysms with accelerated 4D flow. Magn. Reason. Imaging 2010, 28, 41–46. [Google Scholar] [CrossRef]
- Sforza, D.M.; Putman, C.M.; Cebral, J.R. Hemodynamics of cerebral aneurysms. Annu. Rev. Fluid Mech. 2009, 41, 91–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meckel, S.; Stalder, A.F.; Santini, F.; Radü, E.-W.; Rüfenacht, D.A.; Markl, M.; Wetzel, S.G. In vivo visualization and analysis of 3-D hemodynamics in cerebral aneurysms with flow-sensitized 4-D MR imaging at 3 T. Neuroradiology 2008, 50, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Markl, M.; Wu, C.; Hurley, M.C.; Ansari, S.A.; Carroll, T.J.; Rahme, R.J.; Aoun, S.G.; Carr, J.; Batjer, H.; Bendok, B.R. Cerebral arteriovenous malformation: Complex 3D hemodynamics and 3D blood flow alterations during staged embolization. J. Magn. Reson. Imaging 2013, 38, 946–950. [Google Scholar] [CrossRef] [PubMed]
- Markl, M.; Chan, F.P.; Alley, M.T.; Wedding, K.L.; Draney, M.T.; Elkins, C.J.; Parker, D.W.; Wicker, R.; Taylor, C.A.; Herfkens, R.J. Time-resolved three-dimensional phase-contrast MRI. J. Magn. Reson. Imaging 2003, 17, 499–506. [Google Scholar] [CrossRef] [Green Version]
- Boussel, L.; Rayz, V.; Martin, A.; Acevedo-Bolton, G.; Lawton, M.T.; Higashida, R.; Higashida, R.; Smith, W.S.; Young, W.L.; Saloner, D. Phase-contrast magnetic resonance imaging measurements in intracranial aneurysms in vivo of flow patterns, velocity fields, and wall shear stress: Comparison with computational fluid dynamics. Magn. Reson. Med. 2009, 61, 409–417. [Google Scholar] [CrossRef] [Green Version]
- Van Ooij, P.; Potters, W.V.; Guédon, A.; Schneiders, J.J.; Marquering, H.A.; Majoie, C.B.; van Bavel, E.; Nederveen, A.J. Wall shear stress estimated with phase contrast MRI in an in vitro and in vivo intracranial aneurysm. J. Magn. Reson. Imaging 2013, 38, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Sotelo, J.; Urbina, J.; Valverde, I.; Tejos, C.; Irarrázaval, P.; Andia, M.E.; Uribe, S.; Hurtado, D.E. 3D Quantification of wall shear stress and oscillatory shear index using a finite-element method in 3D CINE PC-MRI data of the thoracic aorta. IEEE Trans. Med. Imaging 2016, 35, 1475–1487. [Google Scholar] [CrossRef]
- Stalder, A.F.; Russe, M.F.; Frydrychowicz, A.; Bock, J.; Hennig, J.; Markl, M. Quantitative 2D and 3D phase contrast MRI: Optimized analysis of blood flow and vessel wall parameters. Magn. Reson. Med. 2008, 60, 1218–1231. [Google Scholar] [CrossRef]
- Morgan, A.G.; Thrippleton, M.J.; Wardlaw, J.M.; Marshall, I. 4D flow MRI for non-invasive measurement of blood flow in the brain: A systematic review. J. Cereb. Blood Flow Metab. 2021, 41, 206–218. [Google Scholar] [CrossRef] [PubMed]
- Brina, O.; Bouillot, P.; Reymond, P.; Luthman, A.S.; Santarosa, C.; Fahrat, M.; Lovblad, K.O.; Machi, P.; Delattre, B.M.A.; Pereira, V.M.; et al. How Flow Reduction Influences the Intracranial Aneurysm Occlusion: A Prospective 4D Phase-Contrast MRI Study. Am J Neuroradiology. 2019, 40, 2117–2123. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.M.; Brina, O.; Delattre, B.M.A.; Ouared, R.; Bouillot, P.; Erceg, G.; Schaller, K.; Lovblad, K.O.; Vargas, M.I. Assessment of intra-aneurysmal flow modification after flow diverter stent placement with four-dimensional flow MRI: A feasibility study. J. NeuroInterventional Surg. 2015, 7, 913–999. [Google Scholar] [CrossRef] [PubMed]
- Sindeev, S.; Arnold, P.G.; Frolov, S.; Prothmann, S.; Liepsch, D.; Balasso, A.; Berg, P.; Kaczmarz, S.; Kirschke, J.S. Phase-contrast MRI versus numerical simulation to quantify hemodynamical changes in cerebral aneurysms after flow diverter treatment. PLoS ONE 2018, 13, e0190696. [Google Scholar] [CrossRef] [Green Version]
- Turski, P.; Scarano, A.; Hartman, E.; Clark, Z.; Schubert, T.; Rivera, L.; Wu, Y.; Wieben, O.; Johnson, K. Neurovascular 4DFlow MRI (phase contrast MRA): Emerging clinical applications. Neurovascular Imaging 2016, 2, 8. [Google Scholar] [CrossRef] [Green Version]
- Schnell, S.; Wu, C.; Ansari, S.A. 4D MRI flow examinations in cerebral and extracerebral vessels. Ready for clinical routine? Curr. Opin. Neurol. 2016, 29, 419–428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slesnick, T.C.; Hashemi, S. 4-dimensional phase contrast imaging in congenital heart disease: How we do it. MAGNETOM Flash 2018, 70, 84–97. [Google Scholar]
- Bollache, E.; Barker, A.J.; Dolan, R.S.; Carr, J.C.; van Ooij, P.; Ahmadian, R.; Powell, A.; Collins, J.D.; Geiger, J.; Markl, M. k-t accelerated aortic 4D flow MRI in under two minutes: Feasibility and impact of resolution, k-space sampling patterns, and respiratory navigator gating on hemodynamic measurements. Magn. Reson. Med. 2018, 79, 195–207. [Google Scholar] [CrossRef]
- Bollache, E.; Knott, K.D.; Jarvis, K.; Boubertakh, R.; Dolan, R.S.; Camaioni, C.; Collins, L.; Scully, P.; Rabin, S.; Treibel, T.; et al. Two-Minute k-Space and Time–Accelerated Aortic Four-Dimensional Flow MRI: Dual-Center Study of Feasibility and Impact on Velocity and Wall Shear Stress Quantification. Radiol. Cardiothorac. Imaging 2019, 1, e180008. [Google Scholar] [CrossRef]
- Giese, D.; Wong, J.; Greil, G.F.; Buehrer, M.; Schaeffter, T.; Kozerke, S. Towards highly accelerated Cartesian time-resolved 3D flow cardiovascular magnetic resonance in the clinical setting. J. Cardiovasc. Magn. Reson. 2014, 16, 42. [Google Scholar] [CrossRef] [Green Version]
- Hsiao, A.; Lustig, M.; Alley, M.T.; Murphy, M.; Chan, F.P.; Herfkens, R.J.; Vasanawala, S.S. Rapid pediatric cardiac assessment of flow and ventricular volume with compressed sensing parallel imaging volumetric cine phase-contrast MRI. Am. J. Roentgenol. 2012, 198, W250–W259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- David, A.; Le Touze, D.; Warin-Fresse, K.; Paul-Gilloteaux, P.; Bonnefoy, F.; Idier, J.; Moussaoui, S.; Guerin, P.; Serfaty, J.M. In-vitro validation of 4D flow MRI measurements with an experimental pulsatile flow model. Diagn. Interv. Imaging 2019, 100, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Gabbour, M.; Rigsby, C.; Markl, M.; Schnell, S.; Jarvis, K.B.; de Freitas, R.A.; Popescu, A.R.; Robinson, J.D. Comparison of 4D flow and 2D PC MRI blood flow quantification in children and young adults with congenital heart disease. J. Cardiovasc. Magn. Reson. 2013, 15, E90. [Google Scholar] [CrossRef] [Green Version]
- Jiang, J.; Strother, C.; Johnson, K.; Baker, S.; Consigny, D.; Wieben, O.; Zagzebski, J. Comparison of blood velocity measurements between ultrasound Doppler and accelerated phase-contrast MR angiography in small arteries with disturbed flow. Phys. Med. Biol. 2011, 56, 1755–1773. [Google Scholar] [CrossRef] [PubMed]
- Medero, R.; Hoffman, C.; Roldán-Alzate, A. Comparison of 4D flow MRI and particle image velocimetry using an in vitro carotid bifurcation model. Ann. Biomed. Eng. 2018, 46, 2112–2122. [Google Scholar] [CrossRef] [PubMed]
- Berg, P.; Stucht, D.; Janiga, G.; Beuing, O.; Speck, O.; Thévenin, D. Cerebral blood flow in a healthy circle of Willis and two intracranial aneurysms: Computational fluid dynamics versus four-dimensional phase-contrast magnetic resonance imaging. J. Biomech. Eng. 2014, 136, 041003. [Google Scholar] [CrossRef] [PubMed]
- Roloff, C.; Stucht, D.; Beuing, O.; Berg, P. Comparison of intracranial aneurysm flow quantification techniques: Standard PIV vs stereoscopic PIV vs tomographic PIV vs phase-contrast MRI vs. CFD. J. Neurointerv. Surg. 2019, 11, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Hofman, M.B.M.; Visser, F.C.; Rossum, A.C.V.; Vink, G.Q.M.; Sprenger, M.; Westerhof, N. In vivo validation of magnetic resonance blood volume flow measurements with limited spatial resolution in small vessels. Magn. Reson. Med. 1995, 33, 778–784. [Google Scholar] [CrossRef]
- Wollschlaeger, G.; Wollschlaeger, P.B.; Lucas, F.V.; Lopez, V.F. Experience and result with postmortem cerebral angiography performed as routine procedure of the autopsy. Am. J. Roentgenol. 1967, 7101, 68–87. [Google Scholar] [CrossRef]
- Zarrinkoob, L.; Ambarki, K.; Wåhlin, A.; Birgander, R.; Eklund, A.; Malm, J. Blood flow distribution in cerebral arteries. J. Cereb. Blood Flow Metab. 2015, 35, 648–654. [Google Scholar] [CrossRef] [Green Version]
- Pravdivtseva, M.S.; Peschke, E.; Lindner, T.; Wodarg, F.; Hensler, J.; Gabbert, D.; Voges, I.; Berg, P.; Barker, A.J.; Jansen, O.; et al. 3D-printed, patient-specific intracranial aneurysm models: From clinical data to flow experiments with endovascular devices. Med Phys. 2021, 48, 1469–1484. [Google Scholar] [CrossRef] [PubMed]
- Haselhoff, E. Understanding How Compressed SENSE Makes MRI Faster; Koninklijke Philips N.V.: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Geerts-Ossevoort, L.; de Weerdt, E.; Duijndam, A.; van IJperen, G.; Peeters, H.; Doneva, M.; Nijenhuis, M.; Huang, A. Compressed SENSE. Speed Done Right. Every Time; Koninklijke Philips N.V.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Donoho, D.L. Compressed sensing. IEEE Trans. Inf. Theory 2006, 52, 1289–1306. [Google Scholar] [CrossRef]
- Pruessmann, K.P.; Weiger, M.; Scheidegger, M.B.; Boesiger, P. SENSE: Sensitivity encoding for fast MRI. Magnetic Resonance in Medicine 1999, 42, 952–962. [Google Scholar] [CrossRef]
- Pelc, N.J.; Bernstein, M.A.; Shimakawa, A.; Glover, G.H. Encoding strategies for three-direction phase-contrast MR imaging of flow. J. Magn. Reson. Imaging 1991, 1, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Liu, J.; Tarumi, T.; Lawley, J.S.; Liu, P.; Zhu, D.C.; Lu, H.; Zhang, R. Measurement of cerebral blood flow using phase contrast magnetic resonance imaging and duplex ultrasonography. J. Cereb. Blood Flow Metab. 2017, 37, 541–549. [Google Scholar] [CrossRef]
- Schnell, S.; Ansari, S.A.; Vakil, P.; Wasielewski, M.; Carr, M.L.; Hurley, M.C.; Bendok, B.R.; Batjer, H.; Carroll, T.J.; Carr, J.; et al. Three-dimensional hemodynamics in intracranial aneurysms: Influence of size and morphology. J. Magn. Reson. Imaging. 2014, 39, 120–131. [Google Scholar] [CrossRef] [Green Version]
- Raunig, D.L.; McShane, L.M.; Pennello, G.; Gatsonis, C.; Carson, P.L.; Voyvodic, J.T.; Wahl, R.L.; Kurland, B.F.; Schwarz, A.J.; Gönen, M.; et al. Quantitative imaging biomarkers: a review of statistical methods for technical performance assessment. Stat. Methods Med. Res. 2015, 24, 27–67. [Google Scholar] [CrossRef] [Green Version]
- Shapiro-Wilk and Shapiro-Francia Normality Tests. Available online: https://www.mathworks.com/matlabcentral/fileexchange/13964-shapiro-wilk-and-shapiro-francia-normality-tests (accessed on 3 January 2022).
- Doyle, C.M.; Orr, J.; Greenwood, J.P.; Plein, S.; Tsoumpas, C.; Bissell, M.M. Four-dimensional flow magnetic resonance imaging in the assessment of blood flow in the heart and great vessels: A systematic review. J. Magn. Reson. Imaging 2021. [Google Scholar] [CrossRef]
- Gottwald, L.M.; Peper, E.S.; Zhang, Q.; Coolen, B.F.; Strijkers, G.J.; Nederveen, A.J.; van Ooij, P. Pseudo-Spiral Sampling and Compressed Sensing Reconstruction Provides Flexibility of Temporal Resolution in Accelerated Aortic 4D Flow MRI: A Comparison With k-t Principal Component Analysis. 2020, 33, e4255.
- Stam, K.; Chelu, R.G.; van der Velde, N.; van Duin, R.; Wielopolski, P.; Nieman, K.; Merkus, D.; Hirsch, A. Validation of 4D flow CMR against simultaneous invasive hemodynamic measurements: A swine study. Int. J. Cardiovasc. Imaging 2019, 35, 1111–1118. [Google Scholar] [CrossRef] [Green Version]
- Wolf, R.L.; Ehman, R.L.; Riederer, S.J.; Rossman, P.J. Analysis of systematic and random error in MR volumetric flow measurements. Magn. Reson. Med. 1993, 30, 82–91. [Google Scholar] [CrossRef]
- Aristova, M.; Vali, A.; Ansari, S.A.; Shaibani, A.; Alden, T.D.; Hurley, M.C.; Jahromi, B.S.; Potts, M.B.; Markl, M.; Schnell, S. Standardized evaluation of cerebral arteriovenous malformations using flow distribution network graphs AND dual- venc 4D Flow MRI. J. Magn. Reson. Imaging 2019, 50, 1718–1730. [Google Scholar] [CrossRef] [PubMed]
- Bouillot, P.; Delattre, B.M.A.; Brina, O.; Ouared, R.; Farhat, M.; Chnafa, C.; Steinman, D.A.; Lovblad, K.O.; Pereira, V.M.; Vargas, M.I. 3D phase contrast MRI: Partial volume correction for robust blood flow quantification in small intracranial vessels. Magn. Reson. Med. 2018, 79, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Blatter, D.D.; Parker, D.L. Correction of partial-volume effects in phase-contrast flow measurements. J. Magn. Reson. Imaging 1995, 5, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Hoogeveen, R.M.; Bakker, C.J.G.; Viergever, M.A. MR phase-contrast flow measurement with limited spatial resolution in small vessels: Value of model-based image analysis. Magn. Reson. Med. 1999, 41, 520–528. [Google Scholar] [CrossRef]
- Liu, X.; Kao, E.; Haraldsson, H.; Ballweber, M.; Martin, A.; Li, Y.; Wang, Y.; Saloner, D. Identification of Intra-Individual Variation in Intracranial Arterial Flow by MRI and the Effect on Computed Hemodynamic Descriptors. MAGMA 2021, 34, 659–666. [Google Scholar] [CrossRef]
- Neuhaus, E.; Weiss, K.; Bastkowski, R.; Koopmann, J.; Maintz, D.; Giese, D. Accelerated Aortic 4D Flow Cardiovascular Magnetic Resonance Using Compressed Sensing: Applicability, Validation and Clinical Integration. J. Cardiovasc. Magn. Reson. 2019, 21, 65. [Google Scholar] [CrossRef]
- Peper, E.S.; Gottwald, L.M.; Zhang, Q.; Coolen, B.F.; van Ooij, P.; Nederveen, A.J.; Strijkers, G.J. Highly accelerated 4D flow cardiovascular magnetic resonance using a pseudo-spiral Cartesian acquisition and compressed sensing reconstruction for carotid flow and wall shear stress. J. Cardiovasc. Magn. Reson. 2020, 22, 7. [Google Scholar] [CrossRef]
- Pathrose, A.; Ma, L.; Berhane, H.; Scott, M.B.; Chow, K.; Forman, C.; Jin, N.; Serhal, A.; Avery, R.; Carr, J. Highly accelerated aortic 4D flow MRI using compressed sensing: Performance at different acceleration factors in patients with aortic disease. Magn. Reson. Med. 2021, 85, 2174–2187. [Google Scholar] [CrossRef]
- Brisman, J.L.; Song, J.K.; Newell, D.W. Cerebral aneurysms. N. Engl. J. Med. 2006, 355, 928–939. [Google Scholar] [CrossRef] [Green Version]
- Medero, R.; Ruedinger, K.; Rutkowski, D.; Johnson, K.; Roldán-Alzate, A. In vitro assessment of flow variability in an intracranial aneurysm model using 4D flow MRI and tomographic PIV. Ann. Biomed. Eng. 2020, 48, 2484–2493. [Google Scholar] [CrossRef]
- Brindise, M.C.; Rothenberger, S.; Dickerhoff, B.; Schnell, S.; Markl, M.; Saloner, D.; Rays, V.L.; Vlachos, P.P. Multi-modality cerebral aneurysm haemodynamic analysis: In vivo 4D flow MRI, in vitro volumetric particle velocimetry and in silico computational fluid dynamics. J. R. Soc. Interface 2019, 16, 20190465. [Google Scholar] [CrossRef] [Green Version]
- Marlevi, D.; Schollenberger, J.; Aristova, M.; Ferdian, E.; Ma, Y.; Young, A.A.; Edelman, E.R.; Schnell, S.; Figueroa, C.A.; Nordsletten, D.A. Noninvasive quantification of cerebrovascular pressure changes using 4D flow MRI. Magn. Reson. Med. 2021, 86, 3096–3110. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Koskas, L.; Faraji, F.; Kao, E.; Wang, Y.; Haraldsson, H.; Kefayati, S.; Zhu, C.; Ahn, S.; Laub, G.; et al. Highly accelerated intracranial 4D flow MRI: Evaluation of healthy volunteers and patients with intracranial aneurysms. MAGMA 2018, 31, 295–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cibis, M.; Potters, W.V.; Gijsen, F.J.; Marquering, H.; van Ooij, P.; van Bavel, E.; Wentzel, J.J.; Nederveen, A.J. The effect of spatial and temporal resolution of cine phase contrast MRI on wall shear stress and oscillatory shear index assessment. PLoS ONE 2016, 11, e0163316. [Google Scholar]
- Gottwald, L.M.; Töger, J.; Bloch, K.M.; Peper, E.S.; Coolen, B.F.; Strijkers, G.J.; van Ooij, P.; Nederveen, A.J. High Spatiotemporal Resolution 4D Flow MRI of Intracranial Aneurysms at 7T in 10 Minutes. Am. J. Neuroradiol. 2020, 41, 1201–1208. [Google Scholar] [CrossRef] [PubMed]


| MRI Protocol | P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 |
|---|---|---|---|---|---|---|---|---|
| MRI sequence | 2D flow | 4D flow | TOF | |||||
| model | tubes, aneurysm | tubes | aneurysm | aneurysm | ||||
| TR/TE [ms] | 9.4/6.2 | 10/6.3 | 7.5/4.6 | 6.6/4.0 | 10.6/6.4 | 7.2/4.4 | 6.5/3.9 | 25/5.8 |
| vox. size [mm3] | 0.5 × 0.5 × 4 | 0.5 × 0.5 × 0.5 | 1 × 1 × 1 | 1.5 × 1.5 × 1.5 | 0.5 × 0.5 × 0.5 | 1 × 1 × 1 | 1.5 × 1.5 × 1.5 | 0.25 × 0.40 × 050 |
| FOV [mm3] | 180 × 180 | 110 × 78 × 30 | 100 × 100 × 20 | 180 × 180 × 160 | ||||
| CS factor | 2.5 | 2.5; 4.5; 6.5; 13 | 2.5; 4.5; 6.5 | 4.7 | ||||
| acq. Time [min] | 2 | 11.2–57.5 | 2.7–14.2 | 1.2–6.2 | 28.5–73.2 | 7.4–18.8 | 3.2–8.2 | 20 |
| card. Phase | 24 | - | ||||||
| Venc | 60, 80 | 60 | 80 | - | ||||
| Acquisition Parameters of 4D Flow MRI | A Linear Fit | ||
|---|---|---|---|
| Spatial Resolution [mm3] | Acceleration Factor | Linear Slope | R2 |
| 0.5 | 2.5 | 1.08 | 0.99 |
| 4.5 | 1.05 | 0.99 | |
| 6.5 | 1.07 | 0.97 | |
| 13 | 1.01 | 0.96 | |
| 1.0 | 2.5 | 0.93 | 0.97 |
| 4.5 | 1.06 | 0.97 | |
| 6.5 | 1.08 | 0.97 | |
| 13 | 1.09 | 0.97 | |
| 1.5 | 2.5 | 1.05 | 0.97 |
| 4.5 | 1.25 | 0.97 | |
| 6.5 | 1.21 | 0.97 | |
| 13 | 1.29 | 0.97 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pravdivtseva, M.S.; Gaidzik, F.; Berg, P.; Ulloa, P.; Larsen, N.; Jansen, O.; Hövener, J.-B.; Salehi Ravesh, M. Influence of Spatial Resolution and Compressed SENSE Acceleration Factor on Flow Quantification with 4D Flow MRI at 3 Tesla. Tomography 2022, 8, 457-478. https://doi.org/10.3390/tomography8010038
Pravdivtseva MS, Gaidzik F, Berg P, Ulloa P, Larsen N, Jansen O, Hövener J-B, Salehi Ravesh M. Influence of Spatial Resolution and Compressed SENSE Acceleration Factor on Flow Quantification with 4D Flow MRI at 3 Tesla. Tomography. 2022; 8(1):457-478. https://doi.org/10.3390/tomography8010038
Chicago/Turabian StylePravdivtseva, Mariya S., Franziska Gaidzik, Philipp Berg, Patricia Ulloa, Naomi Larsen, Olav Jansen, Jan-Bernd Hövener, and Mona Salehi Ravesh. 2022. "Influence of Spatial Resolution and Compressed SENSE Acceleration Factor on Flow Quantification with 4D Flow MRI at 3 Tesla" Tomography 8, no. 1: 457-478. https://doi.org/10.3390/tomography8010038
APA StylePravdivtseva, M. S., Gaidzik, F., Berg, P., Ulloa, P., Larsen, N., Jansen, O., Hövener, J.-B., & Salehi Ravesh, M. (2022). Influence of Spatial Resolution and Compressed SENSE Acceleration Factor on Flow Quantification with 4D Flow MRI at 3 Tesla. Tomography, 8(1), 457-478. https://doi.org/10.3390/tomography8010038






