Evaluation of Regional Lung Function in Pulmonary Fibrosis with Xenon-129 MRI
Abstract
:1. Introduction
2. Materials and Methods
3. Results
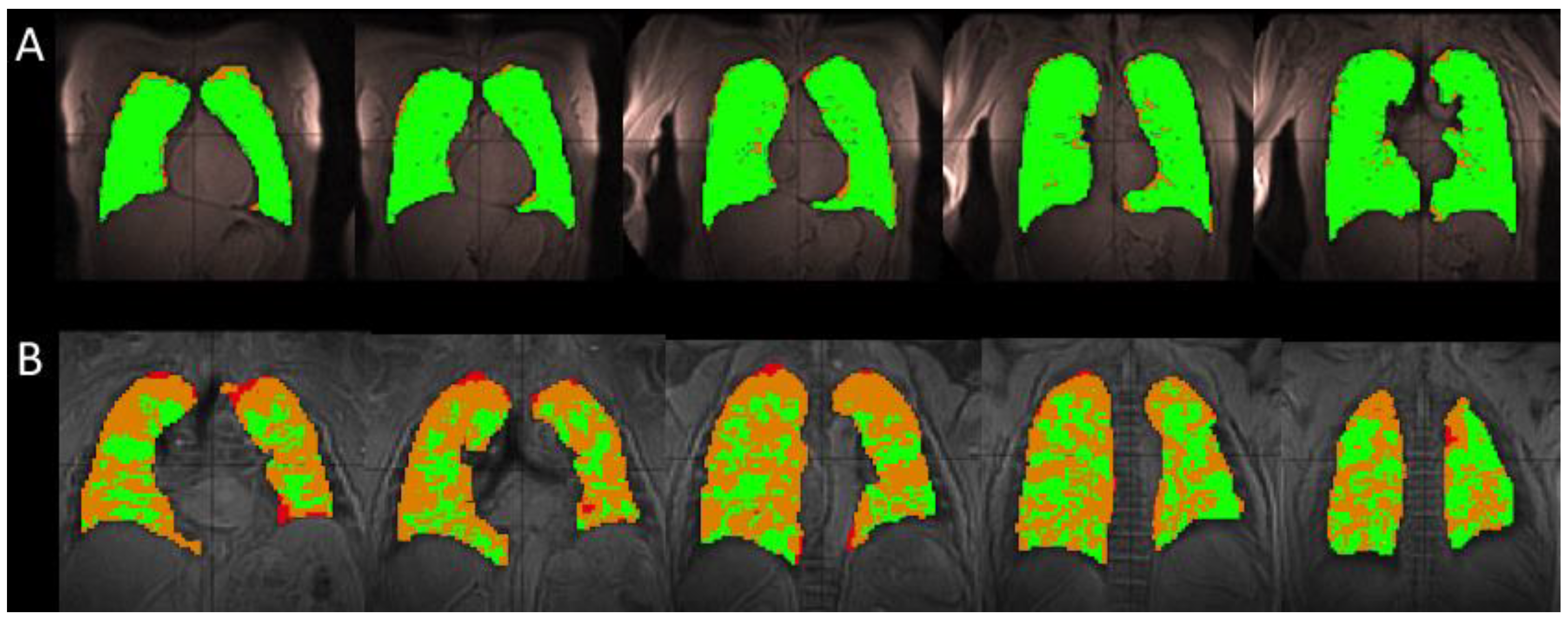
3.1. Comparison of Healthy and IPF Ventilation Images
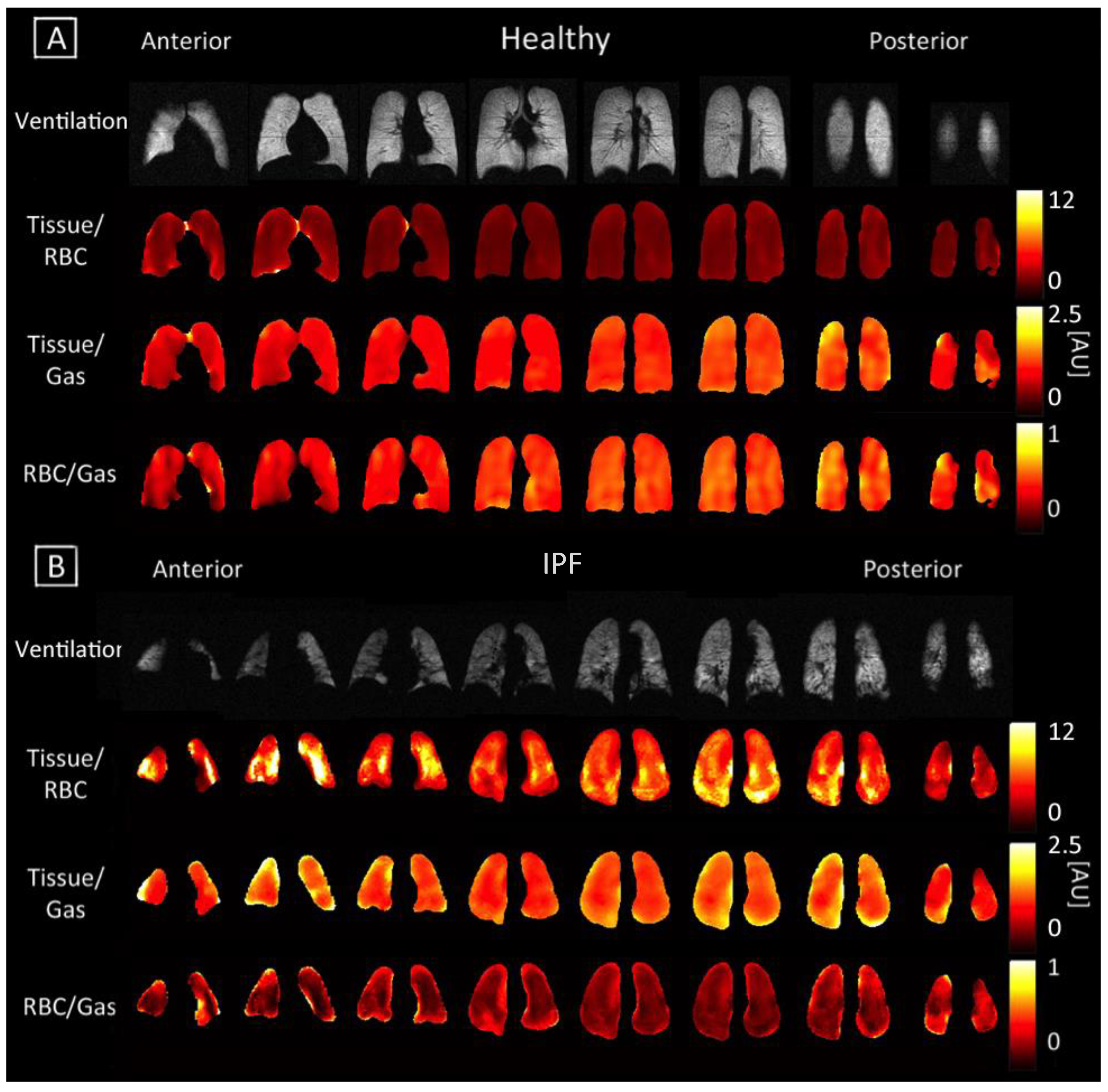
3.2. Comparing Ratio Maps between Healthy and IPF
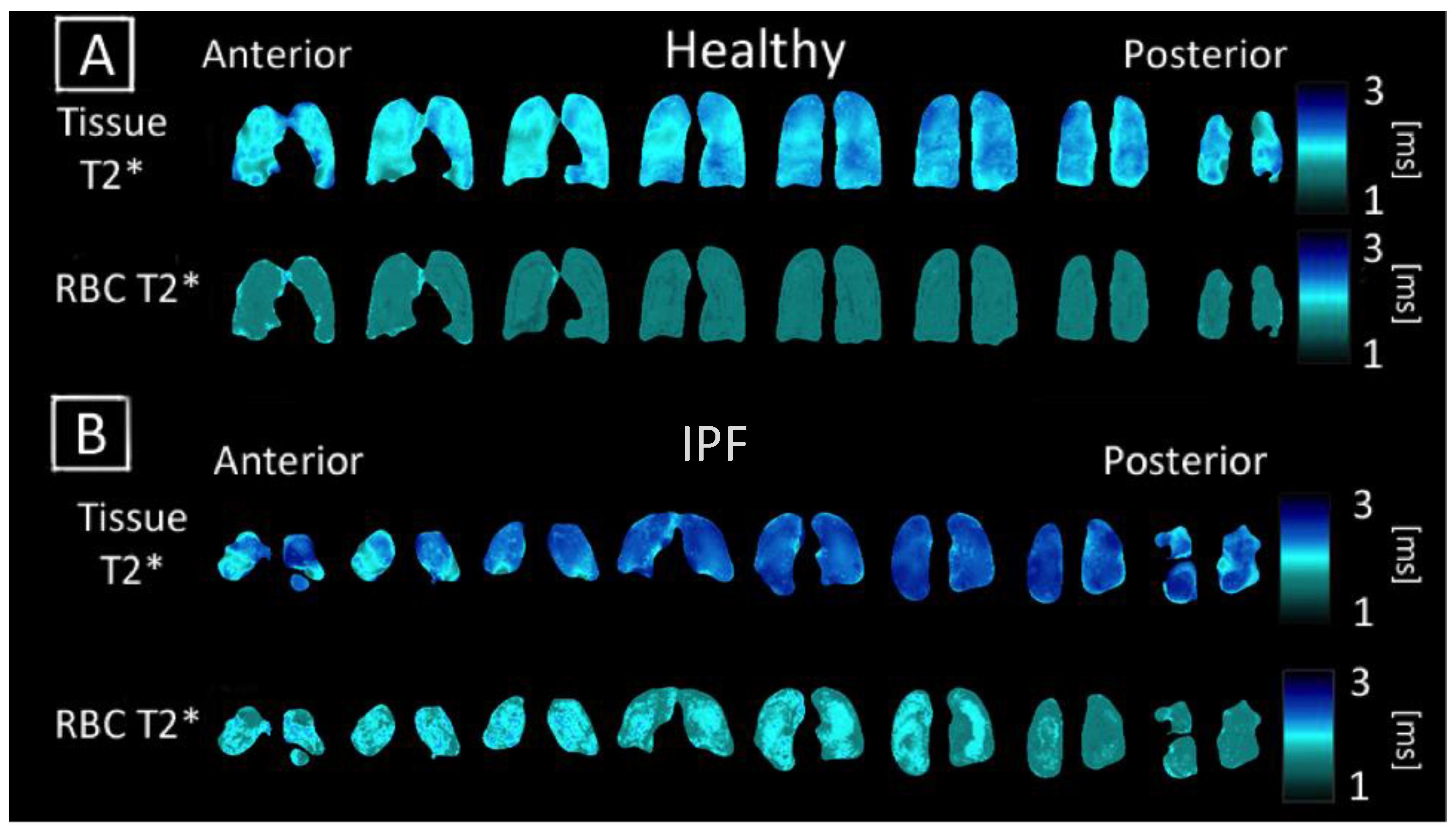
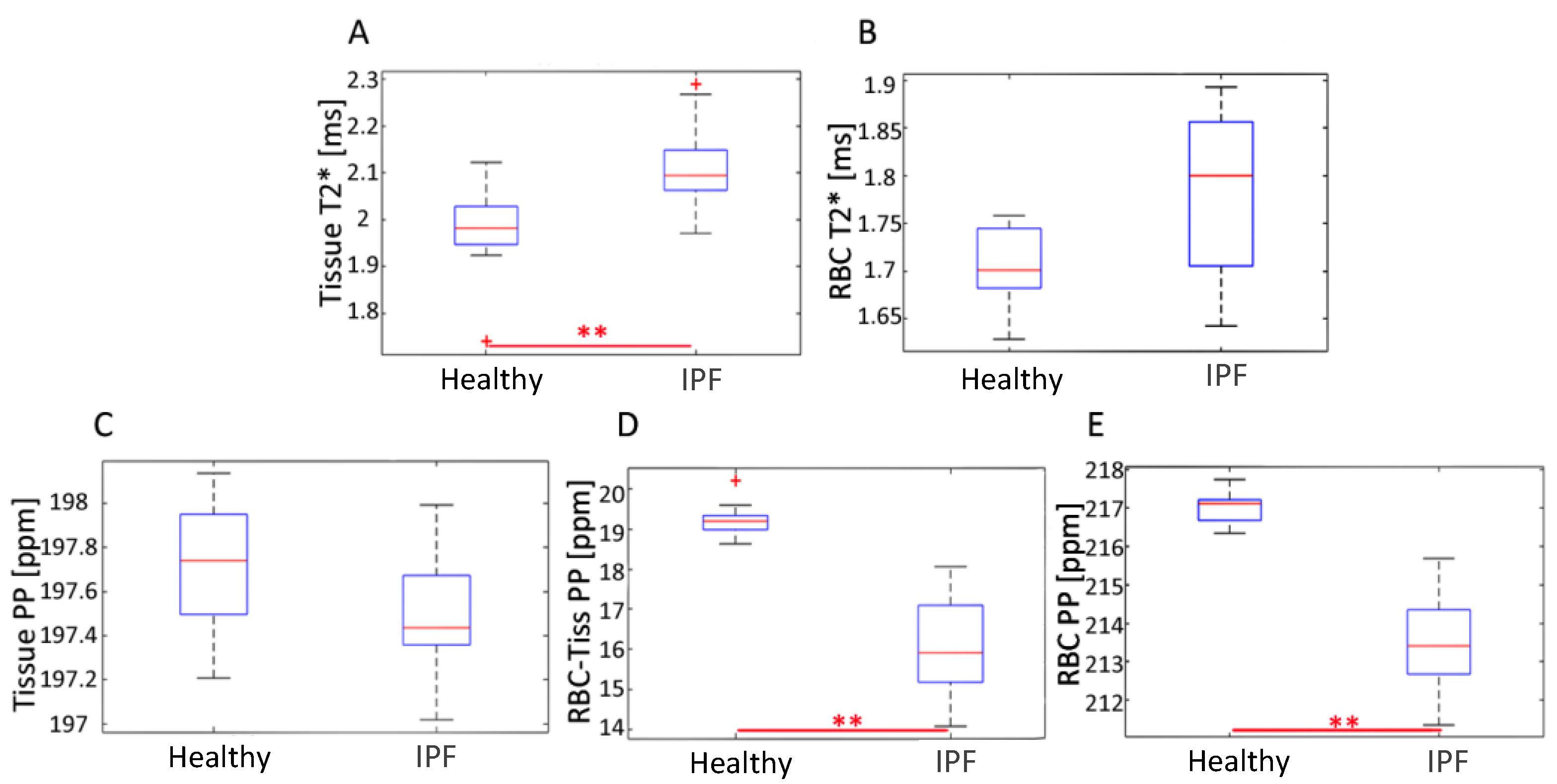
3.3. Comparing T2* and Chemical Shift Maps
3.4. Subject Characterization with PFTs and HRCT
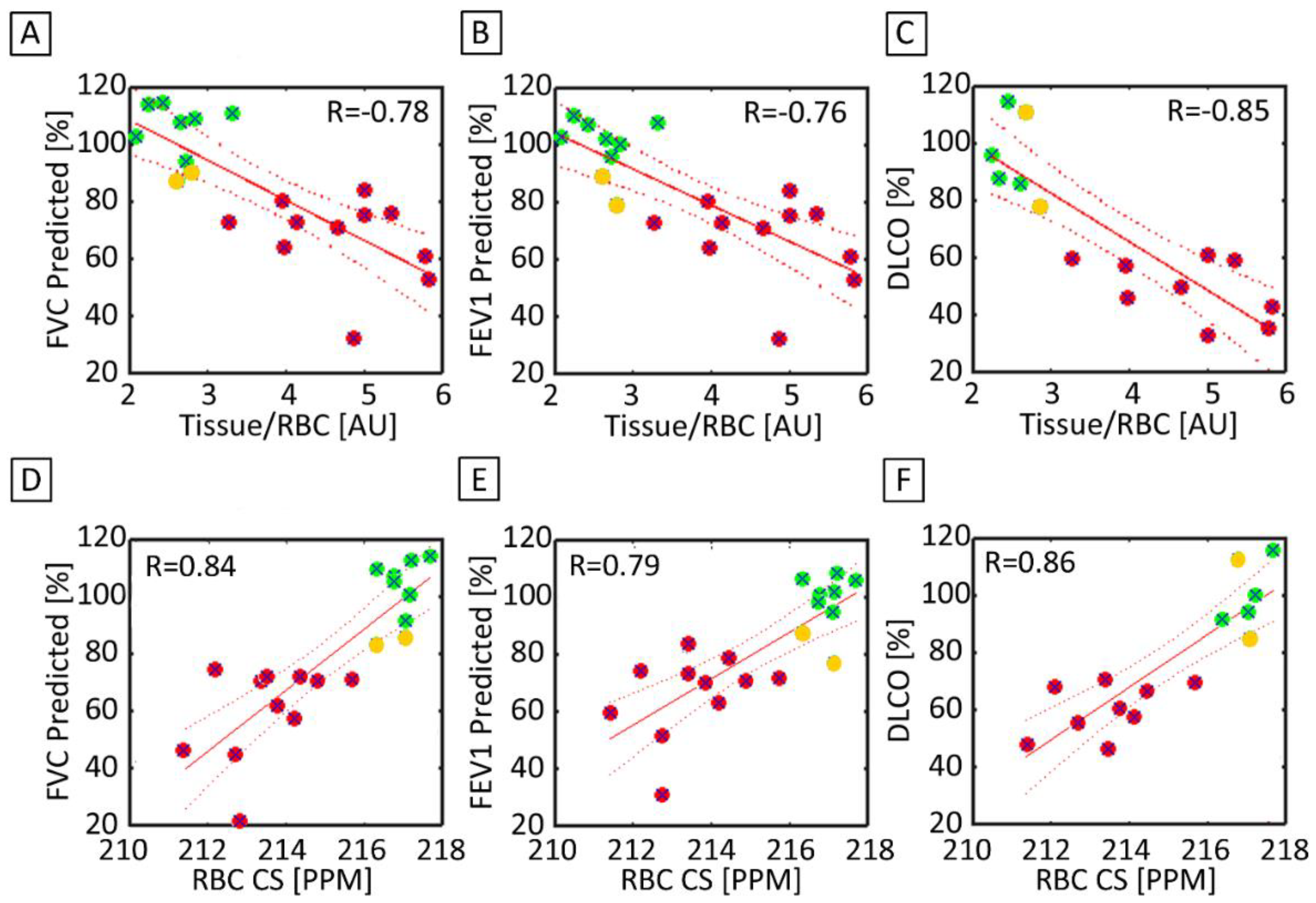
3.5. Correlation between 3D-SBCSI with PFTs and HRCT
4. Discussion
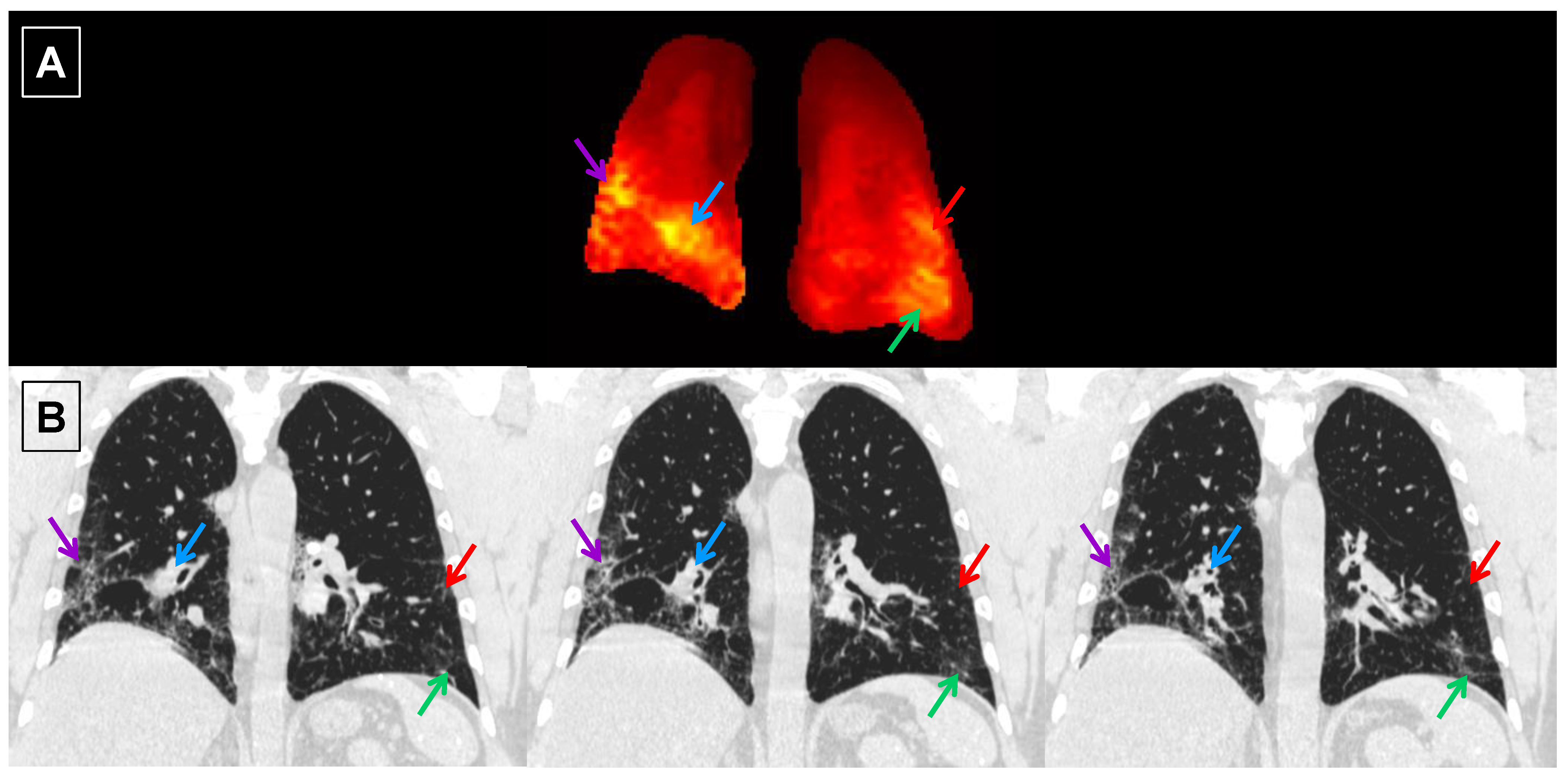
4.1. Ventilation Images
4.2. Ratio Maps
4.3. Tissue and RBC T2* Maps
4.4. Correlations with PFTs and HRCT
4.5. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wallis, A.; Spinks, K. The diagnosis and management of interstitial lung diseases. BMJ 2015, 350, h2072. [Google Scholar] [CrossRef] [Green Version]
- Raghu, G.; Remy-Jardin, M.; Myers, J.L.; Richeldi, L.; Ryerson, C.J.; Lederer, D.J.; Behr, J.; Cottin, V.; Danoff, S.K.; Morell, F.; et al. Diagnosis of Idiopathic Pulmonary Fibrosis. An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2018, 198, e44–e68. [Google Scholar] [CrossRef]
- Hutchinson, J.; Fogarty, A.; Hubbard, R.; McKeever, T. Global incidence and mortality of idiopathic pulmonary fibrosis: A systematic review. Eur. Respir. J. 2015, 46, 795–806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barratt, S.L.; Creamer, A.; Hayton, C.; Chaudhuri, N. Idiopathic Pulmonary Fibrosis (IPF): An Overview. J. Clin. Med. 2015, 7, 201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lederer, D.J.; Martinez, F.J. Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2018, 378, 1811–1823. [Google Scholar] [CrossRef] [PubMed]
- Carlos, W.G.; Strek, M.E.; Wang, T.S.; Patel, H.; Raghu, G.; Wilson, K.C.; Thomson, C.C. Treatment of Idiopathic Pulmonary Fibrosis. Ann. Am. Thorac. Soc. 2016, 13, 115–117. [Google Scholar] [CrossRef]
- Schmidt, S.L.; Tayob, N.; Han, M.K.; Zappala, C.; Kervitsky, D.; Murray, S.; Wells, A.U.; Brown, K.K.; Martinez, F.J.; Flaherty, K.R. Predicting pulmonary fibrosis disease course from past trends in pulmonary function. Chest 2014, 145, 579–585. [Google Scholar] [CrossRef] [Green Version]
- Collard, H.R.; Ryerson, C.J.; Corte, T.J.; Jenkins, G.; Kondoh, Y.; Lederer, D.J.; Lee, J.S.; Maher, T.M.; Wells, A.U.; Antoniou, K.M.; et al. Acute Exacerbation of Idiopathic Pulmonary Fibrosis. An International Working Group Report. Am. J. Respir. Crit. Care Med. 2016, 194, 265–275. [Google Scholar] [CrossRef]
- Cottin, V.; Richeldi, L. Neglected evidence in idiopathic pulmonary fibrosis and the importance of early diagnosis and treatment. Eur. Respir. Rev. 2014, 23, 106–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sgalla, G.; Biffi, A.; Richeldi, L. Idiopathic pulmonary fibrosis: Diagnosis, epidemiology and natural history. Respirology 2016, 21, 427–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; Araki, T.; Okajima, Y.; Albert, M.; Hatabu, H. Pulmonary hyperpolarized noble gas MRI: Recent advances and perspectives in clinical application. Eur. J. Radiol. 2014, 83, 1282–1291. [Google Scholar] [CrossRef] [PubMed]
- Mugler, J.P., 3rd; Altes, T.A.; Ruset, I.C.; Dregely, I.M.; Mata, J.F.; Miller, G.W.; Ketel, S.; Ketel, J.; Hersman, F.W.; Ruppert, K. Simultaneous magnetic resonance imaging of ventilation distribution and gas uptake in the human lung using hyperpolarized xenon-129. Proc. Natl. Acad. Sci. USA 2010, 107, 21707–21712. [Google Scholar] [CrossRef] [Green Version]
- Mugler, J.P., 3rd; Altes, T.A. Hyperpolarized 129Xe MRI of the human lung. J. Magn. Reson. Imaging 2013, 37, 313–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakai, K.; Bilek, A.M.; Oteiza, E.; Walsworth, R.L.; Balamore, D.; Jolesz, F.A.; Albert, M.S. Temporal dynamics of hyperpolarized 129Xe resonances in living rats. J. Magn. Reson. B 1996, 111, 300–304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, K.W.; Reo, N.V.; Schoot Uiterkamp, A.J.; Stengle, D.P.; Stengle, T.R.; Williamson, K.L. Xenon NMR: Chemical shifts of a general anesthetic in common solvents, proteins, and membranes. Proc. Natl. Acad. Sci. USA 1981, 78, 4946–4949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruppert, K.; Brookeman, J.R.; Hagspiel, K.D.; Mugler, J.P., 3rd. Probing lung physiology with xenon polarization transfer contrast (XTC). Magn. Reson. Med. 2000, 44, 349–357. [Google Scholar] [CrossRef]
- Wagshul, M.E.; Button, T.M.; Li, H.F.; Liang, Z.; Spring, C.S.; Zhong, K.; Wishnia, A. In vivo MR imaging and spectroscopy using hyperpolarized 129Xe. Magn. Reson. Med. 1996, 36, 183–191. [Google Scholar] [CrossRef]
- Fernandes, C.; Ruppert, K.; Altes, T.; Mugler, J., 3rd; Ruset, I.; Miller, W.; Hersman, W.; Mata, J. Hyperpolarized xenon-129 3D-Chemical Shift Imaging of the lung in subjects with a history of smoke exposure. In Proceedings of the International Society of Magnetic Resonance in Medicine, Salt Lake City, UT, USA, 20–26 April 2013. [Google Scholar]
- Guan, S.; Qing, K.; Altes, T.; Mugler, J.P., 3rd; Fernandes, C.; Ruppert, K.; Ruset, I.; Hersman, W.F.; Froh, D.; Teague, W.; et al. Regional Quantification of Lung Function in Cystic Fibrosis using 3D Single-Breath Chemical Shift Imaging. In Proceedings of the International Society of Magnetic Resonance in Medicine, Milan, Italy, 10–16 May 2014. [Google Scholar]
- Guan, S.; Qing, K.; Altes, T.; Mugler, J.P., 3rd; Ruset, I.; Froh, D.; Grady, M.; Brookeman, J.; Mata, J.F. T2* and Frequency Shift Maps of Healthy and CF Subjects. In Proceedings of the International Society of Magnetic Resonance in Medicine, Toronto, ON, Canada, 30 May–5 June 2015. [Google Scholar]
- Mata, J.; Ruppert, K.; Dregely, I.; Altes, T.A.; Miller, W.; Sylvester, P.; Ketel, S.; Ketel, J.; Ruset, I.; Hersman, W.F.; et al. Quantification and temporal study of physiologic lung changes in animal models of lung disease using 2D and 3D-CSI with Xe-129. In Proceedings of the International Society of Magnetic Resonance in Medicine, Stockholm, Sweden, 1–7 May 2010. [Google Scholar]
- Mata, J.; Ruppert, K.; Sylvester, P.; Dregely, I.; Altes, T.A.; Ruset, I.; Hersman, W.F.; Miller, G.; Ketel, S.; Ketal, J.; et al. Improved Separation and Quantification of Xe-129 Dissolved-Phase Resonances in the Lung. In Proceedings of the International Society of Magnetic Resonance in Medicine, Montreal, QC, Canada, 7–13 May 2011. [Google Scholar]
- Mata, J.; Sheng, K.; Read, P.; Ruppert, K.; Hagspiel, K.; Altes, T.A.; Tobias, W.; Cates, G.; Brookeman, J.; Mugler, J.P., 3rd. Chemical Shift Imaging of the lungs with hyperpolarized Xe-129: Results from a rabbit model of stereotactic lung radiosurgery with and without a radioprotector. In Proceedings of the International Society of Magnetic Resonance in Medicine, Honolulu, HI, USA, 18–24 April 2009. [Google Scholar]
- Mata, J.; Bolzar, U.; Ruppert, K.; Hagspieal, K.; Altes, T.A.; Soltis, A.; Miller, W.; Tobias, W.; Cates, G.; Brookeman, J.; et al. High-Resolution Chemical Shift Imaging of the Lungs with Xe-129 during a Single 6 Second Breath-Hold: Results from a Rabbit Model of Pulmonary Embolism. In Proceedings of the International Society of Magnetic Resonance in Medicine, Toronto, ON, Canada, 3–9 May 2008. [Google Scholar]
- Avants, B.B.; Tustison, N.; Song, G. Advanced Normalization Tools (ANTS). Insight J. 2009, 2, 1–35. [Google Scholar]
- Norquay, G.; Leung, G.; Stewart, N.J.; Wolber, J.; Wild, J.M. 129 Xe chemical shift in human blood and pulmonary blood oxygenation measurement in humans using hyperpolarized 129 Xe NMR. Magn. Reson. Med. 2017, 77, 1399–1408. [Google Scholar] [CrossRef] [Green Version]
- Wolber, J.; Cherubini, A.; Leach, M.O.; Bifone, A. Hyperpolarized 129Xe NMR as a probe for blood oxygenation. Magn. Reson. Med. 2000, 43, 491–496. [Google Scholar] [CrossRef]
- Kaushik, S.S.; Freeman, M.S.; Yoon, S.W.; Liljeroth, M.G.; Stiles, J.V.; Roos, J.E.; Foster, W.; Rackley, C.R.; McAdams, H.P.; Driehuys, B. Measuring diffusion limitation with a perfusion-limited gas--hyperpolarized 129Xe gas-transfer spectroscopy in patients with idiopathic pulmonary fibrosis. J. Appl. Physiol. 2014, 117, 577–585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, Y.V.; Quirk, J.D.; Ruset, I.C.; JJAtkinson Hersman, F.W.; Woods, J.C. Quantification of human lung structure and physiology using hyperpolarized 129Xe. Magn. Reson. Med. 2014, 71, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Best, A.C.; Meng, J.; Lynch, A.M.; Bozic, C.M.; Miller, D.; Grunwald, G.K.; Lynch, D.A. Idiopathic pulmonary fibrosis: Physiologic tests, quantitative CT indexes, and CT visual scores as predictors of mortality. Radiology 2008, 246, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, A. Imaging: How to recognise idiopathic pulmonary fibrosis. Eur. Respir. Rev. 2014, 23, 215–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cleveland, Z.I.; Cofer, G.P.; Metz, G.; Beaver, D.; Nouls, J.; Kaushik, S.S.; Kraft, M.; Wolber, J.; Kelly, K.T.; McAdams, H.P. Hyperpolarized Xe MR imaging of alveolar gas uptake in humans. PLoS ONE 2010, 5, e12192. [Google Scholar] [CrossRef]
- Keeler, J. Understanding NMR Spectroscopy, 1st ed.; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Misumi, S.; Lynch, D.A. Idiopathic pulmonary fibrosis/usual interstitial pneumonia: Imaging diagnosis, spectrum of abnormalities, and temporal progression. Proc. Am. Thorac. Soc. 2006, 3, 307–314. [Google Scholar] [CrossRef]
- Wang, J.M.; Robertson, S.H.; Wang, Z.; He, M.; Virgincar, R.S.; Schrank, G.M.; Smigla, R.M.; O’Riordan, T.G.; Sundy, J.; Ebner, L.; et al. Using hyperpolarized 129Xe MRI to quantify regional gas transfer in idiopathic pulmonary fibrosis. Thorax 2018, 73, 21–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolber, J.; Cherubini, A.; Santoro, D.; Payne, G.S.; Leach, M.O.; Bifone, A. Linewidths of Hyperpolarized 129Xe NMR Spectra in Human Blood at 1.5T. In Proceedings of the International Society of Magnetic Resonance in Medicine, Denver, CO, USA, 1–7 April 2000. [Google Scholar]
- Guan, S.; Qing, K.; Altes, T.; Mugler, J.P., 3rd; Mehrad, B.; Shim, M.; Chen, Q.; Read, P.; Larner, J.; Ruset, I.; et al. Hyperpolarized Xenon-129 Lung 3D-CSI at 1.5 and 3 Tesla. In Proceedings of the International Society of Magnetic Resonance in Medicine, Singapore, 7–13 May 2016. [Google Scholar]


| Subject No. | Diagnosis | Sex | FVC Pred. [%] | FEV1 Pred. [%] | Age (Years) |
|---|---|---|---|---|---|
| H1 | Healthy | M | 103 | 103 | 26 |
| H2 | Healthy | F | 115 | 107 | 29 |
| H3 | Healthy | F | 111 | 108 | 52 |
| H4 | Healthy | F | 108 | 102 | 24 |
| H5 | Healthy | M | 90 | 79 | 31 |
| H6 | Healthy | M | 114 | 110 | 29 |
| H7 | Healthy | M | 109 | 100 | 24 |
| H8 | Healthy | F | 87 | 89 | 21 |
| H9 | Healthy | M | 94 | 96 | 24 |
| I1 | IPF | M | 76 | 84 | 74 |
| I2 | IPF | M | 77 | 80 | 70 |
| I3 | IPF | M | 76 | 73 | 67 |
| I4 | IPF | F | 31 | 32 | 38 |
| I5 | IPF | M | 77 | 75 | 80 |
| I6 | IPF | M | 79 | 76 | 68 |
| I7 | IPF | M | 76 | 73 | 56 |
| I8 | IPF | M | 64 | 64 | 68 |
| I9 | IPF | F | 53 | 53 | 64 |
| I10 | IPF | M | 68 | 71 | 64 |
| I11 | IPF | F | 54 | 61 | 79 |
| Healthy | IPF | p-Value | ||
|---|---|---|---|---|
| 3D-SBCSI | Tissue/RBC | 2.63 ± 0.37 | 4.71 ± 0.81 | <0.01 |
| Tissue/Gas | 0.93 ± 0.22 | 1.31 ± 0.26 | <0.01 | |
| RBC/Gas | 0.37 ± 0.09 | 0.28 ± 0.06 | <0.05 | |
| Tissue T2* [ms] | 1.97 ± 0.11 | 2.12 ± 0.09 | <0.01 | |
| RBC T2* [ms] | 1.71 ± 0.04 | 1.79 ± 0.09 | 0.06 | |
| Tissue Chemical Shift [PPM] | 197.7 ± 0.29 | 197.5 ± 0.29 | 0.07 | |
| RBC Chemical Shift [PPM] | 217.0 ± 0.44 | 213.5 ± 1.25 | <0.01 | |
| RBC-Tissue Chemical Shift [PPM] | 19.2 ± 0.45 | 16.1 ± 1.21 | <0.01 | |
| PFT | FEV1 [%] | 99.3 ± 10.00 | 67.5 ± 14.68 | <0.01 |
| FVC [%] | 103.4 ± 10.57 | 66.5 ± 15.02 | <0.01 | |
| DLCO [%] | 96 ± 14.7 | 48 ± 12.1 | <0.01 | |
| Hematocrit [%] | 43 ±1.5 | 41 ± 2.3 | 0.07 | |
| HRCT Score | Reticulation | - | 29 ± 13.7 | - |
| Traction Bronchiectasis | - | 15 ± 12.8 | - | |
| Ground-Glass opacities | - | 21 ± 10.6 | - | |
| Honeycombing | - | 12 ± 10.8 | - | |
| Emphysema | - | 0.8 ± 1.5 | - | |
| Ventilation | Total Volume [L] | 4.1 ± 0.56 | 2.6 ± 0.70 | <0.05 |
| Relative Defect Volume [%] | 9.6 ± 7.37 | 34.1 ± 6.44 | <0.05 | |
| Reticulation | Traction Bronchiectasis | Ground Glass Opacity | Honeycombing | Emphysema | |
|---|---|---|---|---|---|
| Right Upper | 20.5 ± 1.41 | 5.8 ± 2.47 | 12.8 ± 4.59 | 3.0 ± 0.71 | 3.3 ± 0.35 |
| Left Upper | 15.8 ±1.77 | 2.8 ± 1.77 | 8.8 ± 4.59 | 1.5 ± 2.12 | 3.3 ± 0.35 |
| Right Mid | 29.5 ± 1.41 | 12.8 ± 3.89 | 24.0 ± 0.00 | 12.3 ± 1.06 | 3.0 ± 0.71 |
| Left Mid | 27.8 ± 3.89 | 10.8 ± 2.47 | 19.5 ± 7.07 | 9.8 ± 0.35 | 3.0 ± 0.71 |
| Right Lower | 43.3 ± 7.42 | 28.3 ± 3.18 | 35.0 ± 7.01 | 26.3 ± 0.35 | 0.5 ± 0.81 |
| Left Lower | 41.3 ± 5.30 | 27.0 ± 0.00 | 32.3 ± 8.13 | 25.0 ± 0.00 | 0.5 ± 0.71 |
| Tissue/RBC [AU] | Tissue/Gas [AU] | RBC/Gas [AU] | Tissue T2* [ms] | Tissue-RBC T2* [ms] | Tissue CS [PPM] | RBC T2* [ms] | RBC-Tissue CS [PPM] | RBC CS [PPM] | |
|---|---|---|---|---|---|---|---|---|---|
| Right Upper | 4.88 | 1.37 | 0.28 | 2.18 | 0.36 | 197.72 | 1.81 | 16.19 | 213.84 |
| Left Upper | 4.92 | 1.50 | 0.32 | 2.17 | 0.36 | 197.82 | 1.82 | 16.18 | 213.92 |
| Right Mid | 4.71 | 1.24 | 0.27 | 2.10 | 0.32 | 197.40 | 1.78 | 16.18 | 213.49 |
| Left Mid | 4.65 | 1.40 | 0.31 | 2.11 | 0.30 | 197.53 | 1.81 | 16.15 | 213.64 |
| Right Lower | 4.86 | 1.22 | 0.26 | 2.10 | 0.38 | 197.28 | 1.75 | 16.00 | 212.93 |
| Left Lower | 4.71 | 1.41 | 0.30 | 2.08 | 0.35 | 197.35 | 1.78 | 15.93 | 213.14 |
| Reticulation | Traction Bronchiectasis | Ground Glass Opacity | Honeycombing | Emphysema | |
|---|---|---|---|---|---|
| Tissue/RBC [AU] | −0.39 | −0.29 | −0.40 | -0.33 | 0.12 |
| Tissue/Gas [AU] | −0.57 | −0.51 | −0.63 | −0.52 | 0.34 |
| RBC/Gas [AU] | −0.51 | −0.47 | −0.56 | −0.47 | 0.35 |
| Tissue T2* [ms] | −0.85 | −0.80 | −0.87 | −0.84 | 0.67 |
| Tissue-RBC T2* [ms] | 0.23 | 0.34 | 0.20 | 0.29 | −0.50 |
| RBC T2* [ms] | −0.88 | −0.87 | −0.92 | −0.87 | 0.77 |
| Tissue CS [PPM] | −0.95 | −0.91 | −0.97 | −0.93 | 0.78 |
| RBC-Tissue CS [PPM] | −0.87 | −0.91 | −0.83 | −0.90 | 0.97 |
| RBC CS [PPM] | −0.99 | −0.99 | −0.99 | −0.99 | 0.94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mata, J.; Guan, S.; Qing, K.; Tustison, N.; Shim, Y.; Mugler, J.P., III; Altes, T.; Huaromo, J.; Mehrad, B. Evaluation of Regional Lung Function in Pulmonary Fibrosis with Xenon-129 MRI. Tomography 2021, 7, 452-465. https://doi.org/10.3390/tomography7030039
Mata J, Guan S, Qing K, Tustison N, Shim Y, Mugler JP III, Altes T, Huaromo J, Mehrad B. Evaluation of Regional Lung Function in Pulmonary Fibrosis with Xenon-129 MRI. Tomography. 2021; 7(3):452-465. https://doi.org/10.3390/tomography7030039
Chicago/Turabian StyleMata, Jaime, Steven Guan, Kun Qing, Nicholas Tustison, Yun Shim, John P. Mugler, III, Talissa Altes, Jhosep Huaromo, and Borna Mehrad. 2021. "Evaluation of Regional Lung Function in Pulmonary Fibrosis with Xenon-129 MRI" Tomography 7, no. 3: 452-465. https://doi.org/10.3390/tomography7030039
APA StyleMata, J., Guan, S., Qing, K., Tustison, N., Shim, Y., Mugler, J. P., III, Altes, T., Huaromo, J., & Mehrad, B. (2021). Evaluation of Regional Lung Function in Pulmonary Fibrosis with Xenon-129 MRI. Tomography, 7(3), 452-465. https://doi.org/10.3390/tomography7030039






