Left Ventricular Thrombi: Insights from Cardiac Magnetic Resonance Imaging
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stratton, J.R.; Resnick, A.D. Increased embolic risk in patients with left ventricular thrombi. Circulation 1987, 75, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Asinger, R.W.; Mikell, F.L.; Elsperger, J.; Hodges, M. Incidence of left ventricular thrombosis after acute transmural myocardial infarction. Serial evaluation by two-dimensional echocardiography. N. Engl. J. Med. 1981, 305, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Gottdiener, J.S.; Massie, B.; Ammons, S.B.; Egher, C. Prevalence of left ventricular thrombus in dilated cardiomyopathy: The WATCH trial (abstr). J. Am. Coll. Cardiol. 2003, 41, 202A. [Google Scholar] [CrossRef][Green Version]
- Sharma, N.D.; McCullough, P.A.; Philbin, E.F.; Weaver, W.D. Left ventricular thrombus and subsequent thromboembolism in patients with severe systolic dysfunction. Chest 2000, 117, 314–320. [Google Scholar] [CrossRef]
- Adelborg, K.; Szepligeti, S.; Sundboll, J.; Horvath-Puho, E.; Henderson, V.W.; Ording, A.; Pedersen, L.; Sørensen, H.T. Risk of stroke in patients with heart failure: A population-based 30-year cohort study. Stroke 2017, 48, 1161–1168. [Google Scholar] [CrossRef] [PubMed]
- Driesman, A.; Hyder, O.; Lang, C.; Stockwell, P.; Poppas, A.; Abbott, J.D. Incidence and predictors of left ventricular thrombus after primary percutaneous coronary intervention for anterior ST-segment elevation myocardial infarction. Clin. Cardiol. 2015, 38, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Khoury, S.; Carmon, S.; Margolis, G.; Karen, G.; Shacham, Y. Incidence and outcomes of early left ventricular thrombus following ST-elevation myocardial infarction treated with primary percutaneous coronary intervention. Clin. Res. Cardiol. 2017, 106, 695–701. [Google Scholar] [CrossRef]
- Berger, A.K.; Gottdiener, J.S.; Yohe, M.A.; Guerrero, J.L. Epidemiological approach to quality assessment in echocardiographic diagnosis. J. Am. Coll. Cardiol. 1999, 34, 1831–1836. [Google Scholar] [CrossRef]
- Platts, D.G.; Fraser, J.F. Contrast echocardiography in critical care: Echoes of the future? A review of the role of microsphere contrast echocardiography. Crit. Care Resusc. 2011, 13, 44–55. [Google Scholar]
- Pazos-López, P.; Pozo, E.; Siqueira, M.E.; García-Lunar, I.; Cham, M.; Jacobi, A.; Macaluso, F.; Fuster, V.; Narula, J.; Sanz, J. Value of CMR for the differential diagnosis of cardiac masses. JACC Cardiovasc. Imaging 2014, 7, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Roifman, I.; Connelly, K.A.; Wright, G.A.; Wijeysundera, H.C. Echocardiography vs. Cardiac Magnetic Resonance Imaging for the diagnosis of left ventricular thrombus: A systematic review. Can. J. Cardiol. 2015, 31, 785–791. [Google Scholar] [CrossRef]
- Srichai, M.B.; Junor, C.; Rodriguez, L.L.; Stillman, A.E.; Grimm, R.A.; Lieber, M.L.; Weaver, J.A.; Smedira, N.G.; White, R.D. Clinical, imaging, and pathologic characteristics of left ventricular thrombus: A comparison of contrast enhanced magnetic resonance imaging, transthoracic echocardiography and transesophageal echocardiography with surgical or pathological validation. Am. Heart J. 2006, 152, 75–84. [Google Scholar] [CrossRef]
- Kramer, C.M.; Barkhausen, J.; Flamm, S.D.; Kim, R.J.; Nagel, E. Standardized cardiovascular magnetic resonance (CMR) protocols 2013 update. J. Cardiovasc. Magn. Reson. 2013, 15, 91. [Google Scholar] [CrossRef]
- Shah, D.J.; Judd, R.M.; Kim, R.J. Myocardial viability. In Clinical Magnetic Resonance Imaging, 3rd ed.; Edelman, R.R., Hesselink, J.R., Zlatkin, M.B., Eds.; Elsevier: New York, NY, USA, 2006. [Google Scholar]
- Weinsaft, J.W.; Kim, H.W.; Crowley, A.L.; Klem, I.; Shenoy, C.; Assche, L.V.; Brosnan, R.; Shah, D.J.; Velazquez, E.J.; Parker, M.; et al. Left ventricular thrombus detection by routine echocardiography: Insights into performance characteristics using delayed enhancement CMR. JACC Cardiovasc. Imaging 2011, 4, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Jungehulsing, M.; Sechtem, U.; Theissen, P.; Hilger, H.H.; Schicha, H. Left ventricular thrombi: Evaluation with spin-echo and gradient-echo MR imaging. Radiology 1992, 182, 225–229. [Google Scholar] [CrossRef]
- Semelka, R.C.; Shoenut, J.P.; Wilson, M.E.; Pellech, A.E.; Patton, J.N. Cardiac masses: Signal intensity features on spin-echo, gradient-echo, gadolinium-enhanced spin-echo, and TurboFLASH images. J. Magn. Reson. Imaging 1992, 2, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Fujita, N.; Caputo, G.R.; Higgins, C.B. Diagnosis and characterization of intracardiac masses by magnetic resonance imaging. Am. J. Card. Imaging 1994, 8, 69–80. [Google Scholar] [PubMed]
- Weinsaft, J.W.; Kim, R.J.; Ross, M.; Krauser, D.; Manoushagian, S.; LaBounty, T.M.; Cham, M.D.; Min, J.K.; Healy, K.; Wang, Y.; et al. Contrast-enhanced anatomic imaging as compared to contrast-enhanced tissue characterization for detection of left ventricular thrombus. JACC Cardiovasc. Imaging 2009, 2, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Haeusler, K.G.; Wollboldt, C.; Bentheim, L.Z.; Herm, J.; Jäger, S.; Kunze, C.; Eberle, H.-C.; Deluigi, C.C.; Bruder, O.; Malsch, C.; et al. Feasibility and diagnostic value of cardiovascular magnetic resonance imaging after acute ischemic stroke of undetermined origin. Stroke 2017, 48, 1241–1247. [Google Scholar] [CrossRef]
- Hong, Y.J.; Hur, J.; Kim, Y.J.; Lee, H.J.; Nam, J.E.; Kim, H.Y.; Choe, K.O.; Choi, B.W. The usefulness of delayed contrast-enhanced cardiovascular magnetic resonance imaging in differentiating cardiac tumors from thrombi in stroke patients. Int. J. Cardiovasc. Imaging 2011, 27, 89–95. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, D.H.; Abbara, S.; Chaithiraphan, V.; Yaraed, K.; Killeen, R.P.; Cury, R.C.; Dodd, J.D. Cardiac tumors: Optimal cardiac MR sequences and spectrum of imaging appearances. Am. J. Roentgenol. 2009, 193, 377–387. [Google Scholar] [CrossRef]
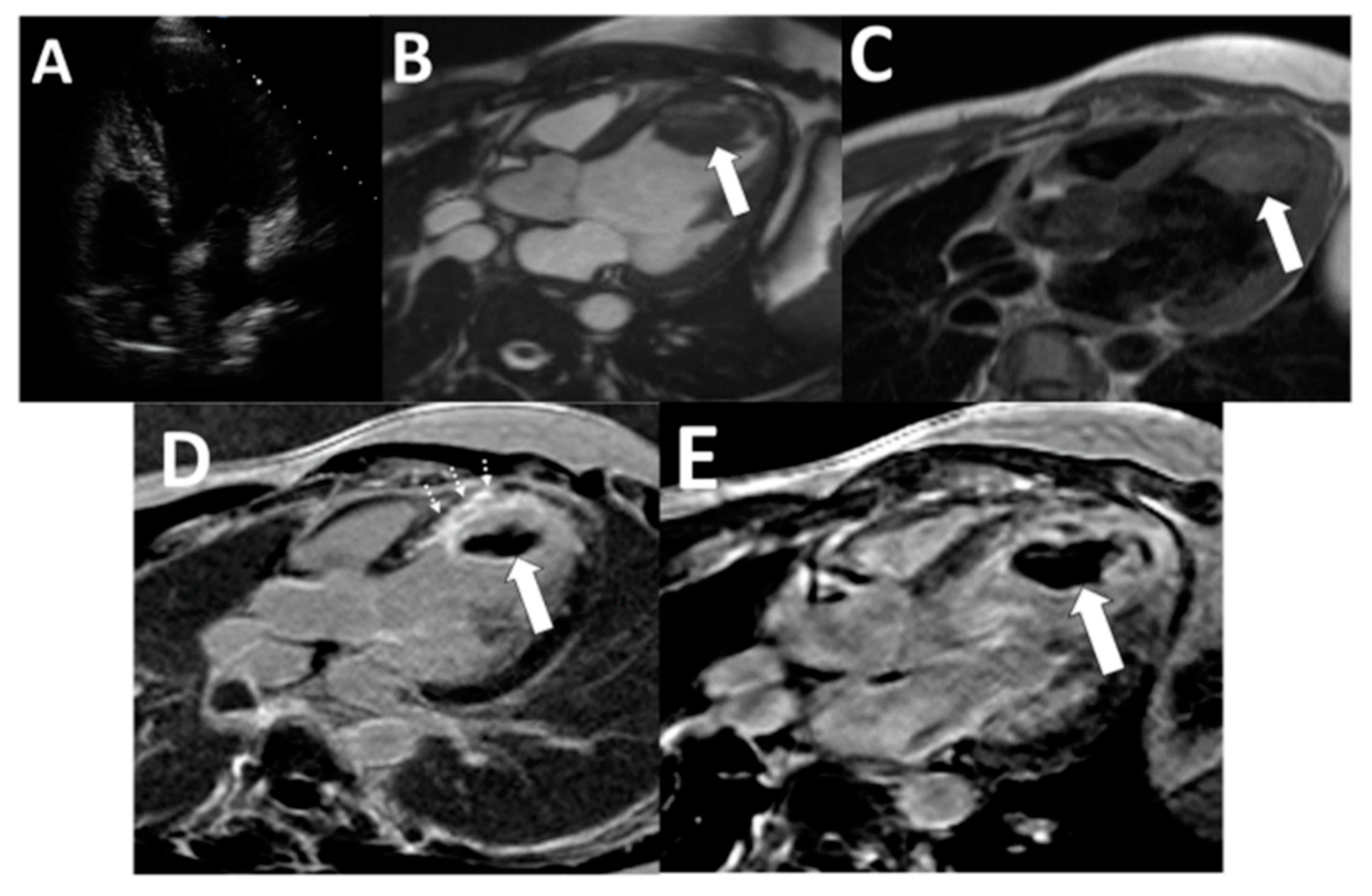
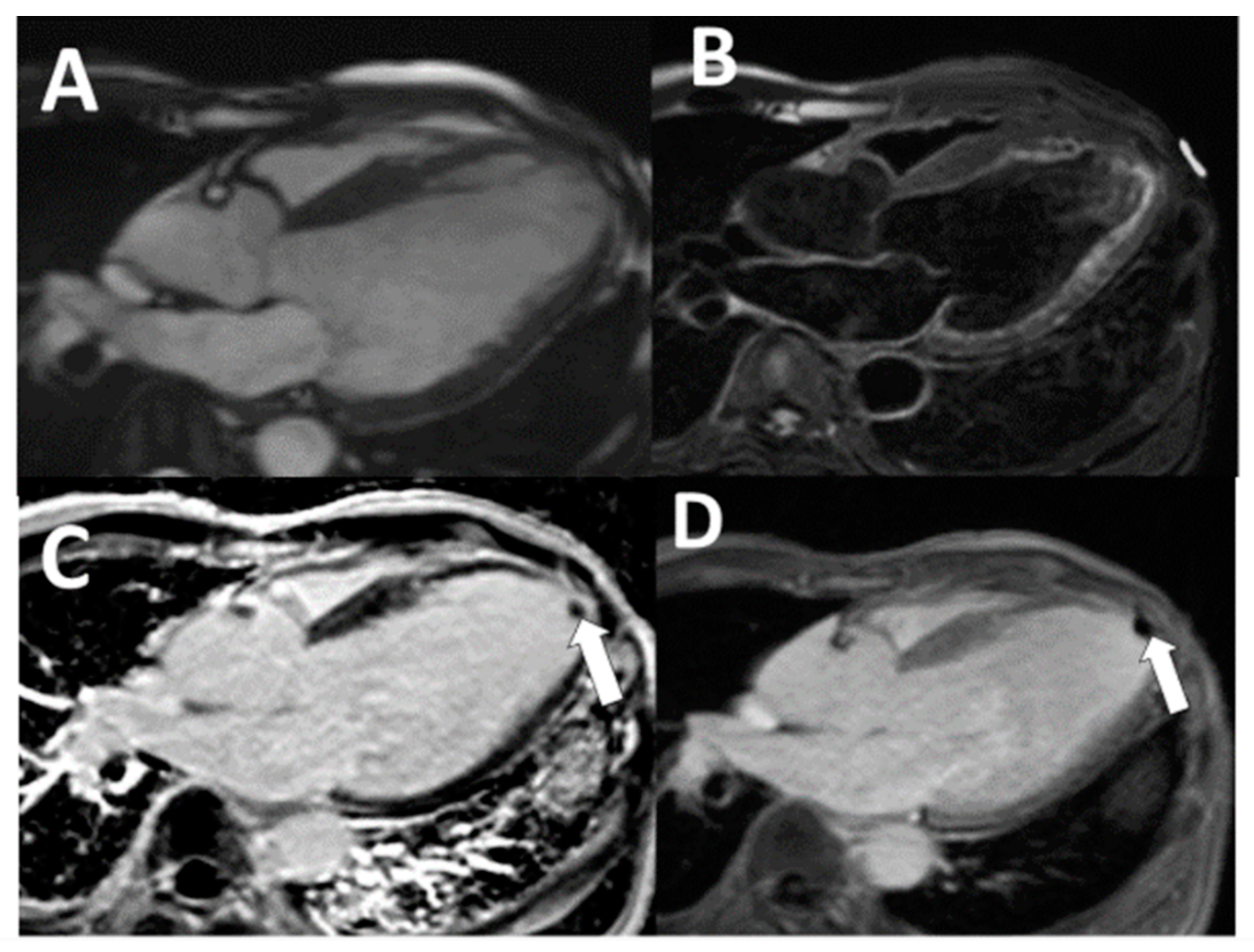
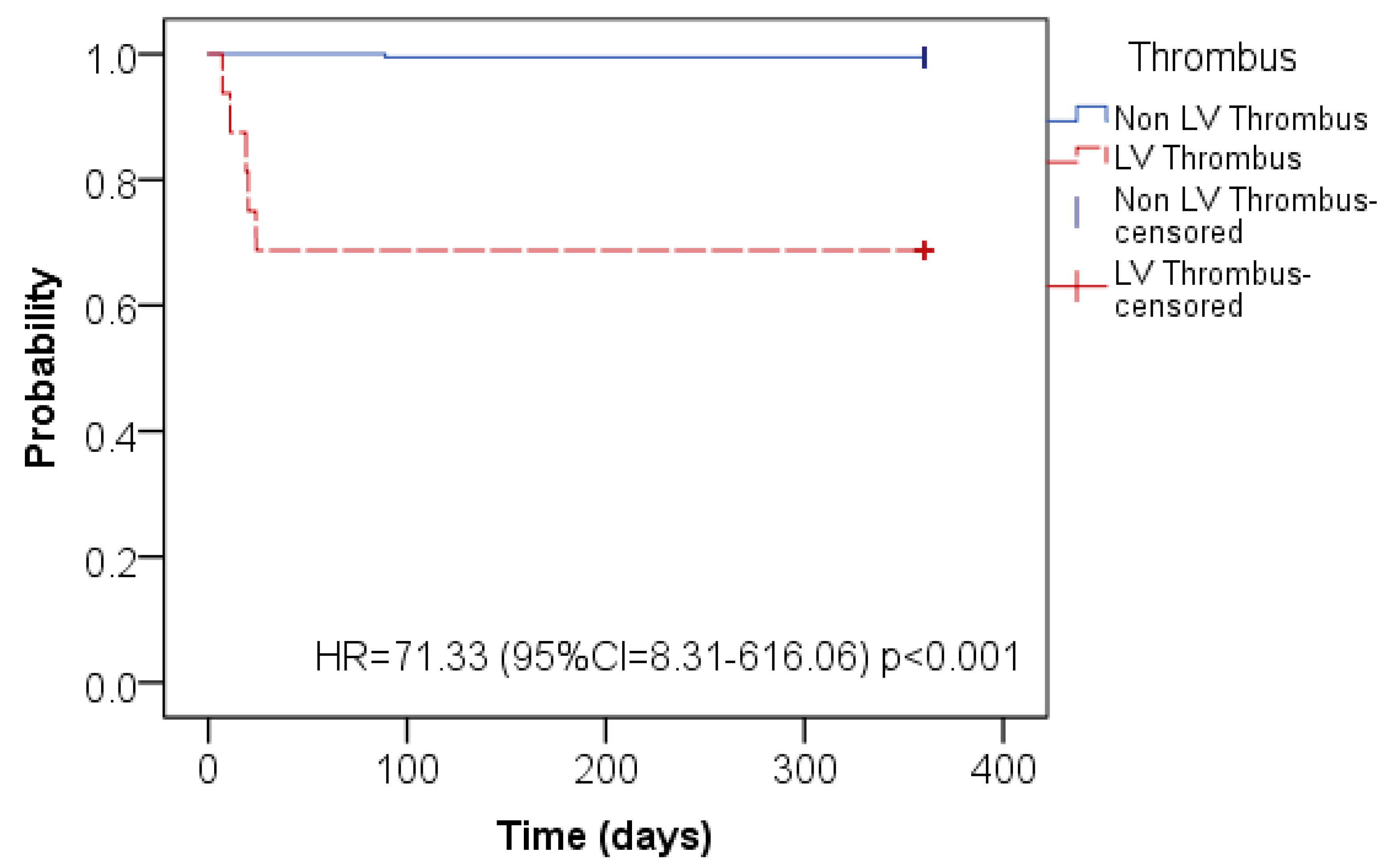
| All (n = 206) | Thrombus Present on LGE-CMR (n = 16) | Thrombus Absent on LGE-CMR (n = 190) | p-Value | |
|---|---|---|---|---|
| Age | 60.2 ± 14.2 | 62.5 ± 15.9 | 59.2 ± 13.1 | 0.34 |
| Male gender | 68.4% (141) | 87.5% (14) | 66.8% (127) | 0.03 |
| Prior myocardial infarction | 77.7% (160) | 87.5% (14) | 76.8% (146) | 0.65 |
| Coronary revascularization | 58.7% (121) | 75% (12) | 57.4% (109) | 0.004 |
| History of embolic events | 2.9% (6) | 31.3% (5) | 0.5% (1) | <0.0001 |
| Antithrombotic medications such as aspirin, warfarin | 76.2% (157) | 100% (16) | 74.2% (141) | 0.006 |
| Echocardiography | ||||
| LV function | ||||
| LVEF (%) | 40.5 ± 22.1 | 37.6 ± 18.9 | 41.1 ± 15.6 | 0.39 |
| End-diastolic diameter (cm) | 5.8 ± 0.8 | 5.9 ± 0.9 | 5.7 ± 1.0 | 0.44 |
| End-systolic diameter (cm) | 4.7 ± 0.9 | 4.9 ± 0.8 | 4.7 ± 1.0 | 0.34 |
| LV Aneurysm | 7.8% (16) | 31.3% (5) | 5.8% (11) | <0.001 |
| CMR | ||||
| LV function | ||||
| LVEF (%) | 39.9 ± 16.6 | 35.9 ± 11.5 | 41.5 ± 12.2 | 0.07 |
| End-diastolic volume (mL) | 189.2 ± 81.9 | 204.5 ± 99.8 | 169.9 ± 78.9 | 0.10 |
| End-systolic volume (mL) | 121.1 ± 99.1 | 137.6 ± 82.5 | 115 ± 79.6 | 0.28 |
| LV Aneurysm | 8.7% (18) | 37.5% (6) | 6.3% (12) | <0.001 |
| LV infarct size (% LV) | 18.7 ± 11.6 | 25.6 ± 9.8 | 14.6 ± 8.7 | <0.001 |
| Parameter | Regression Coefficient | Standard Error | p-Value |
|---|---|---|---|
| LV end-diastolic volume | 205.6 | 99.9 | 0.42 |
| LV end-systolic volume | 139.8 | 77.8 | 0.58 |
| LV infarct size | 24.9 | 8.7 | 0.018 |
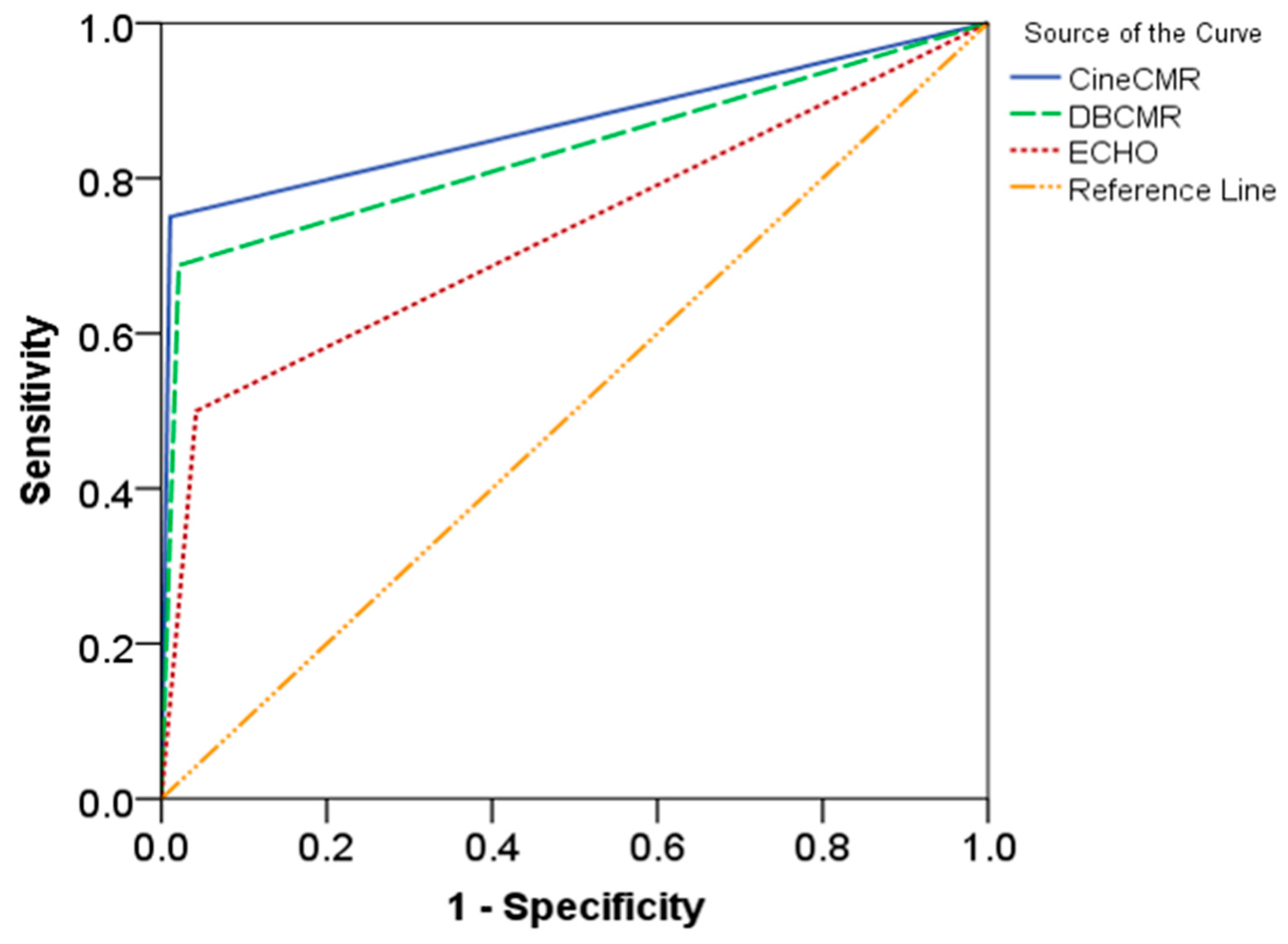
| Sensitivity (%) (95% CI) | Specificity (%) (95% CI) | Accuracy (%) (95% CI) | PPV (%) (95% CI) | NPV (%) (95% CI) | |
|---|---|---|---|---|---|
| Echocardiography | 50% (24.65–75.35) | 96.84% (93.25–98.83) | 93.09% (88.73–96.15) | 57.93% (32.26–77.68) | 95.7% (93.17–97.32) |
| Cine-CMR | 75.0% (47.6–92.7) | 98.95% (96.25–99.87) | 97.03% (93.69–98.89) | 86.1% (60.26–96.2) | 97.85% (95.1–99.1) |
| DB-CMR | 68.75% (41.3–88.9) | 97.9% (94.7–99.4) | 95.56% (91.78–97.94) | 73.96% (50.48–88.78) | 97.4% (94.57–97.94) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaosuwannakit, N.; Makarawate, P. Left Ventricular Thrombi: Insights from Cardiac Magnetic Resonance Imaging. Tomography 2021, 7, 180-188. https://doi.org/10.3390/tomography7020016
Chaosuwannakit N, Makarawate P. Left Ventricular Thrombi: Insights from Cardiac Magnetic Resonance Imaging. Tomography. 2021; 7(2):180-188. https://doi.org/10.3390/tomography7020016
Chicago/Turabian StyleChaosuwannakit, Narumol, and Pattarapong Makarawate. 2021. "Left Ventricular Thrombi: Insights from Cardiac Magnetic Resonance Imaging" Tomography 7, no. 2: 180-188. https://doi.org/10.3390/tomography7020016
APA StyleChaosuwannakit, N., & Makarawate, P. (2021). Left Ventricular Thrombi: Insights from Cardiac Magnetic Resonance Imaging. Tomography, 7(2), 180-188. https://doi.org/10.3390/tomography7020016





