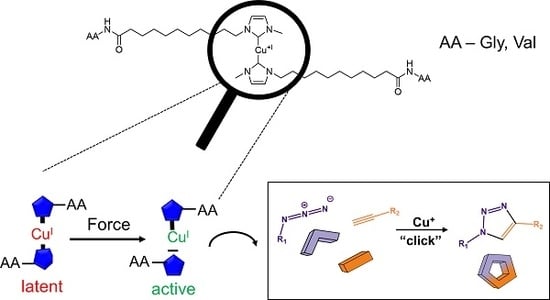
Synthesis and Mechanochemical Activity of Peptide-Based Cu(I) Bis(N-heterocyclic carbene) Complexes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
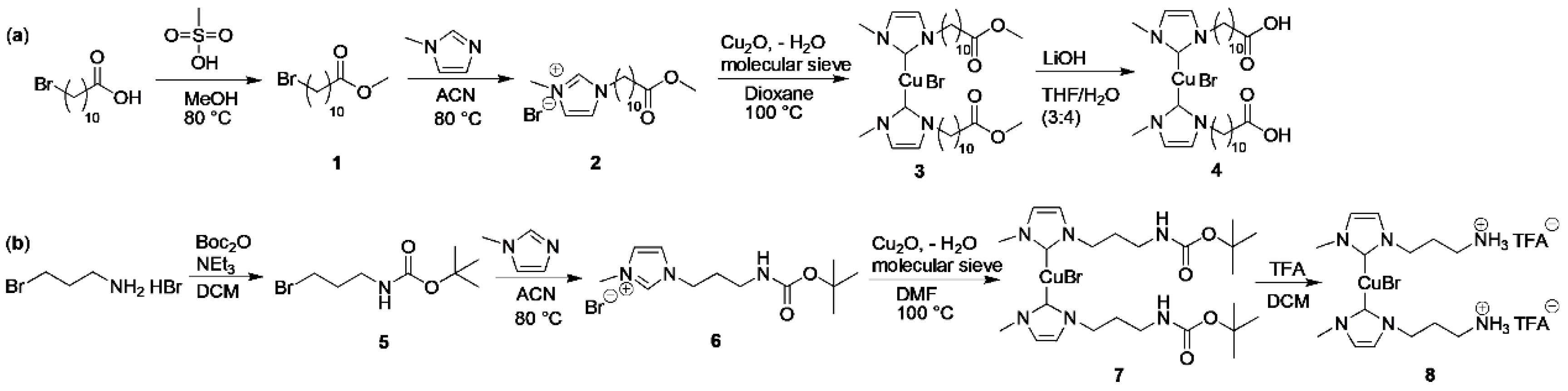
2.3. Synthesis
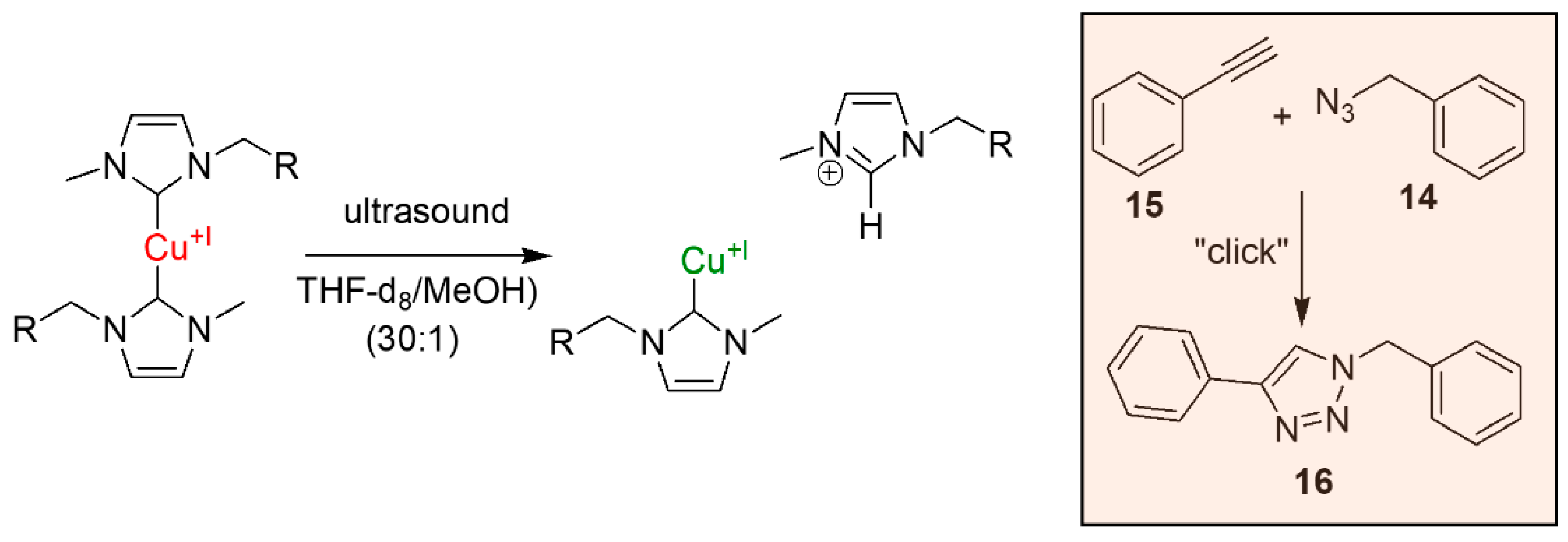
2.4. Mechanochemical Activation
3. Results and Discussion
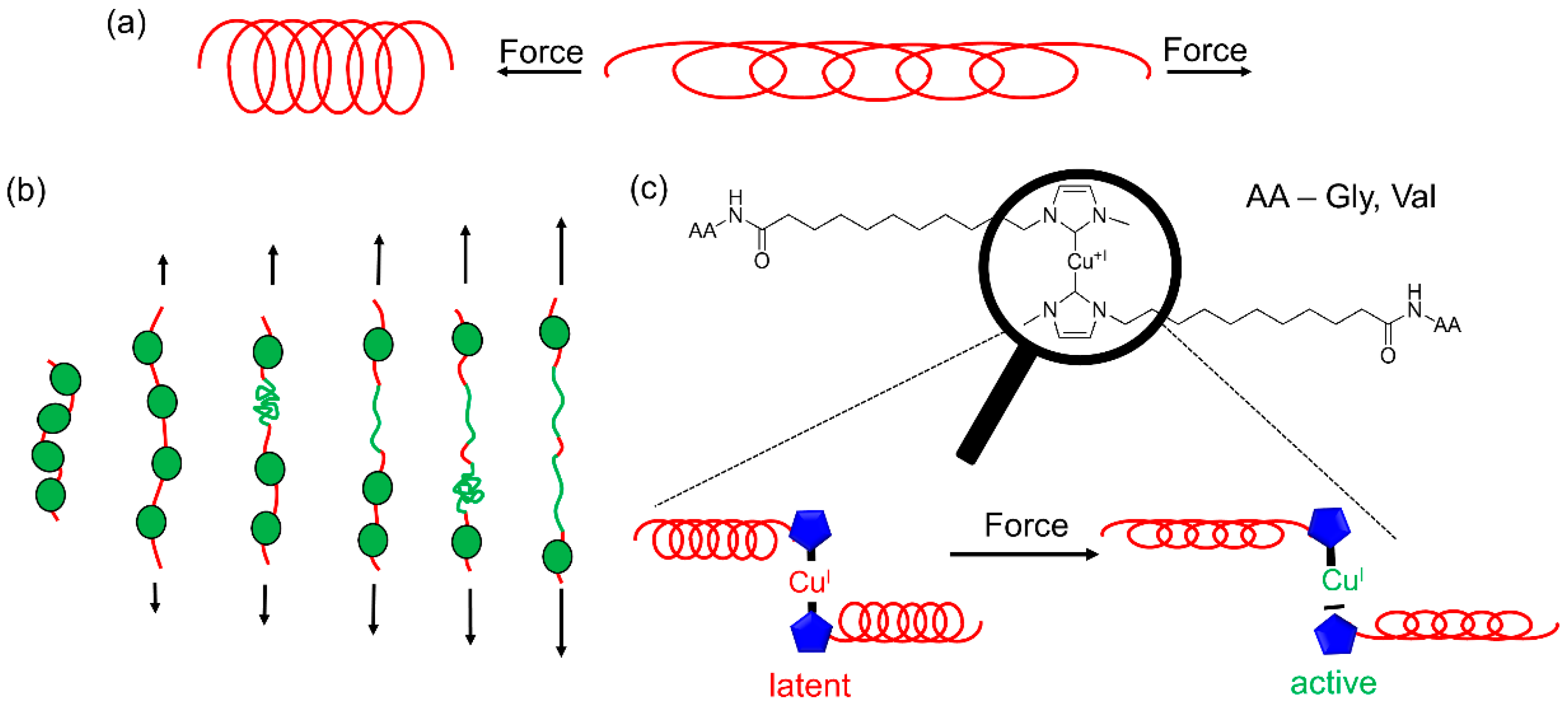
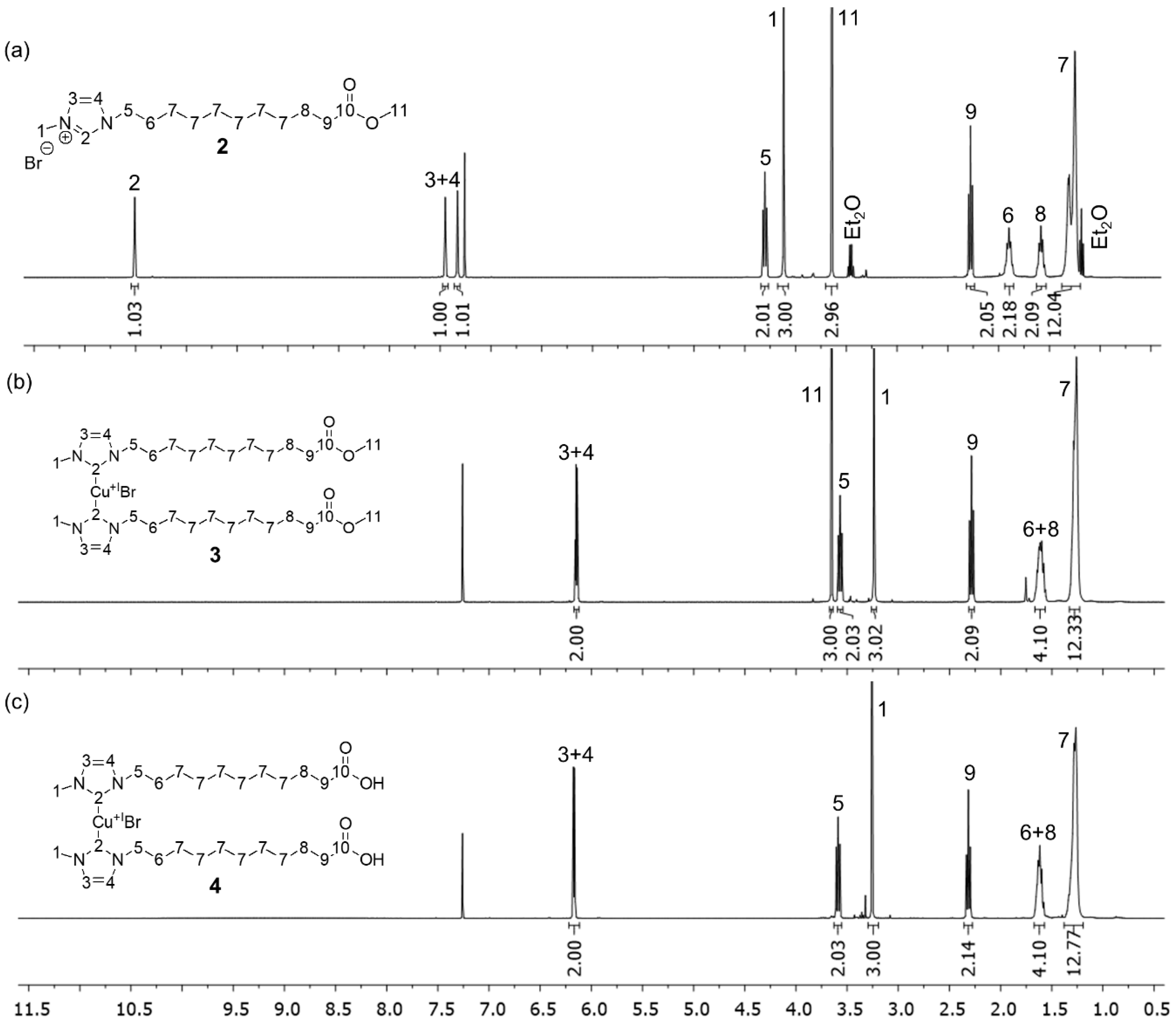
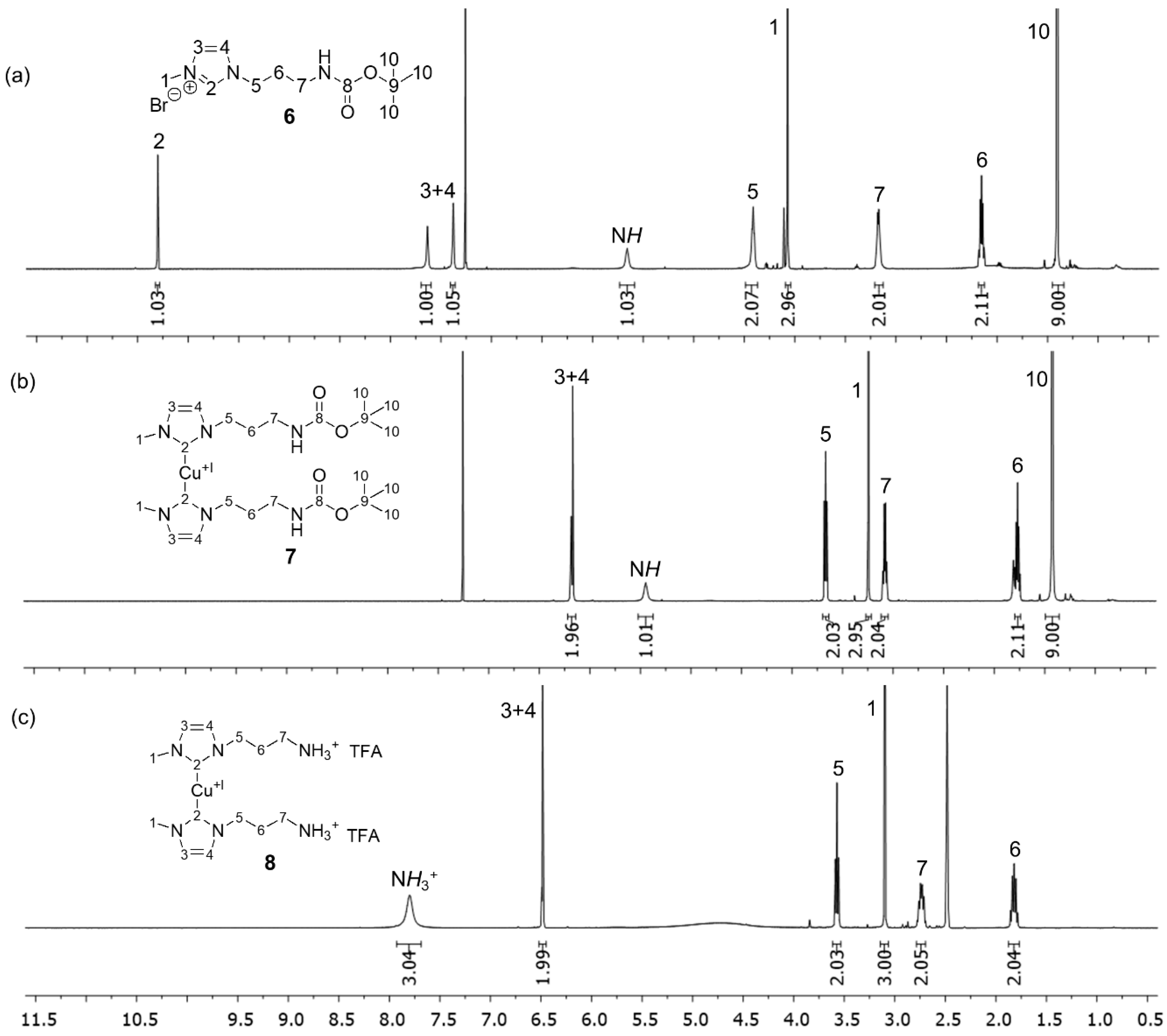
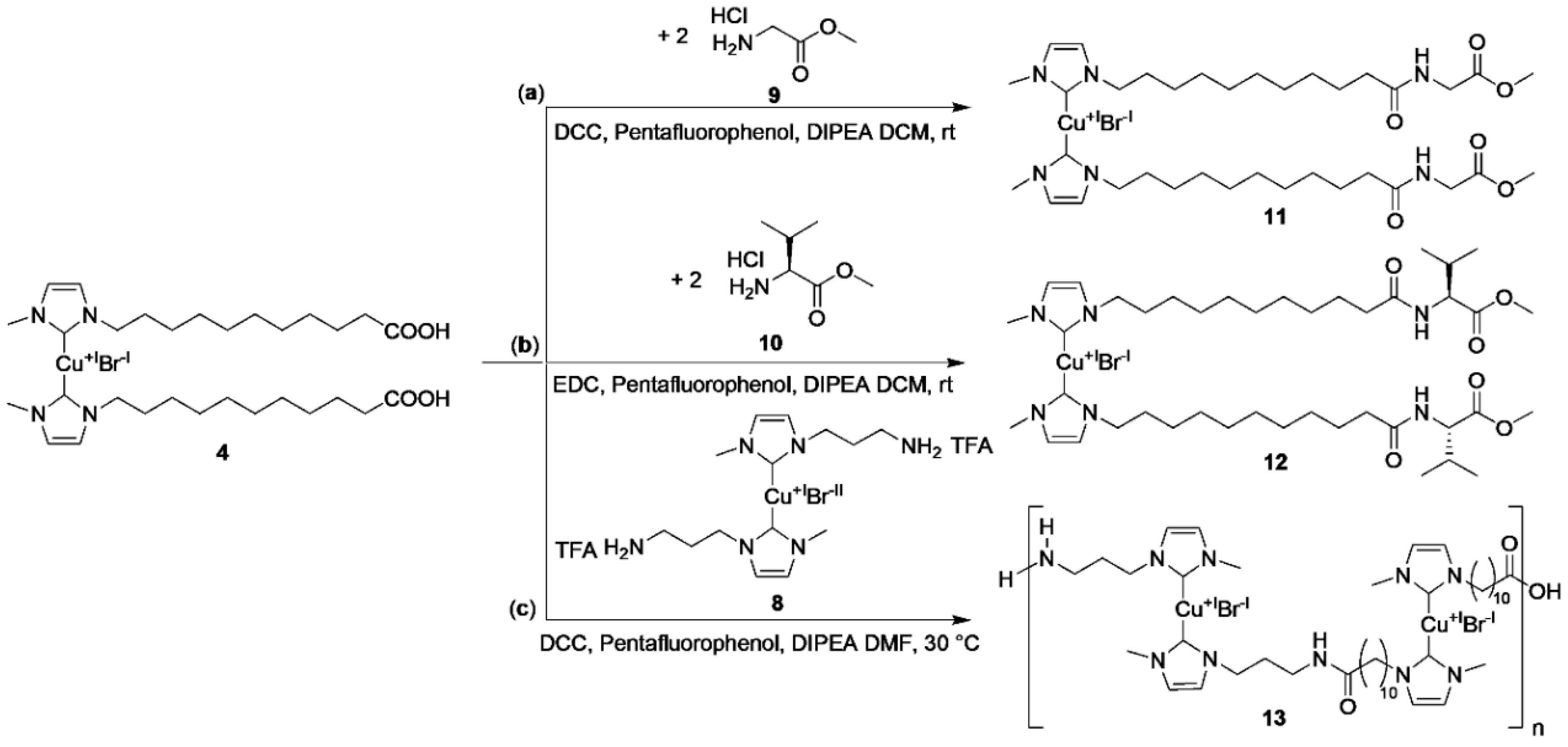
3.1. Synthesis of the Mechanophoric Catalysts
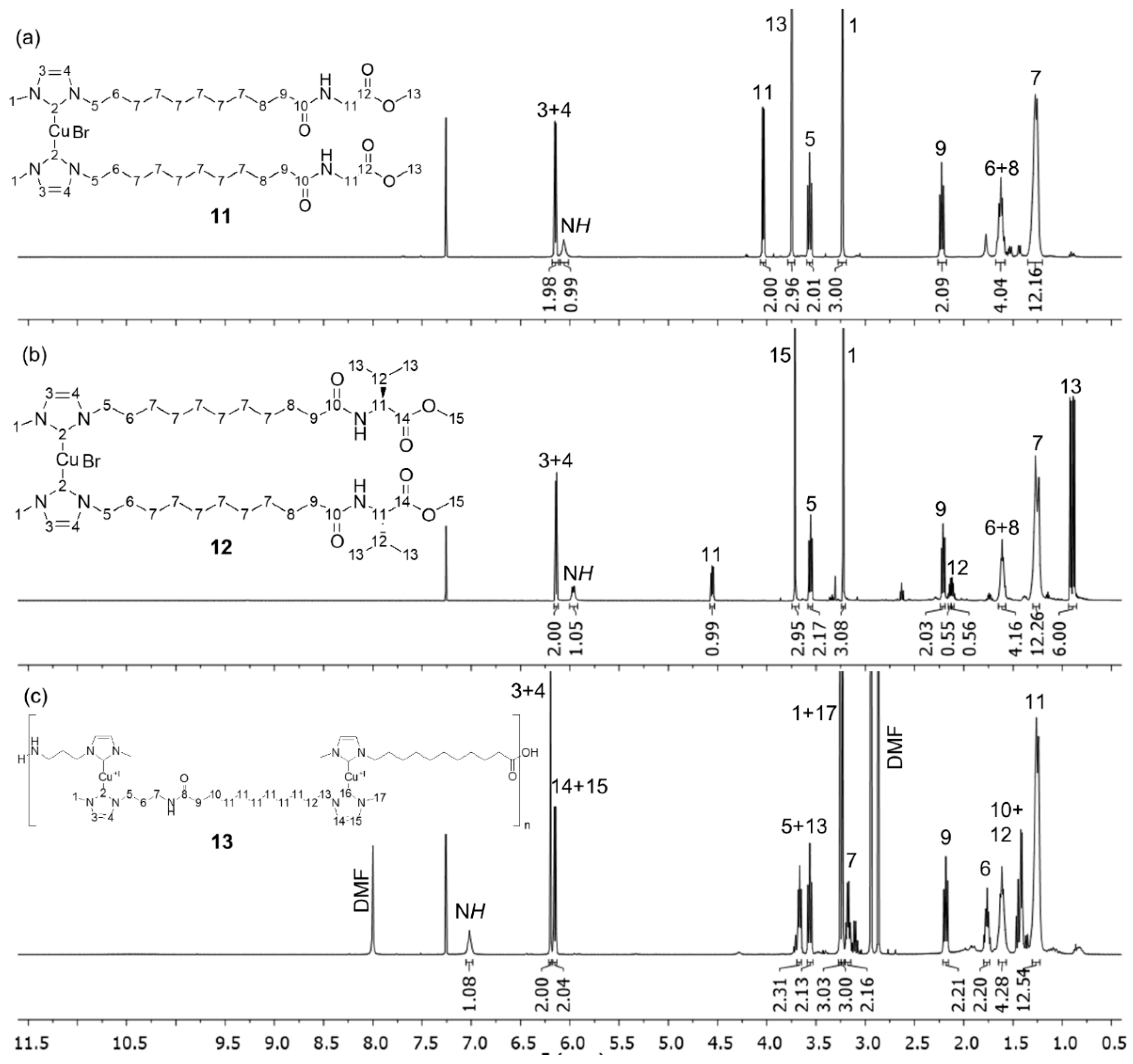
3.2. Peptide Coupling Reactions
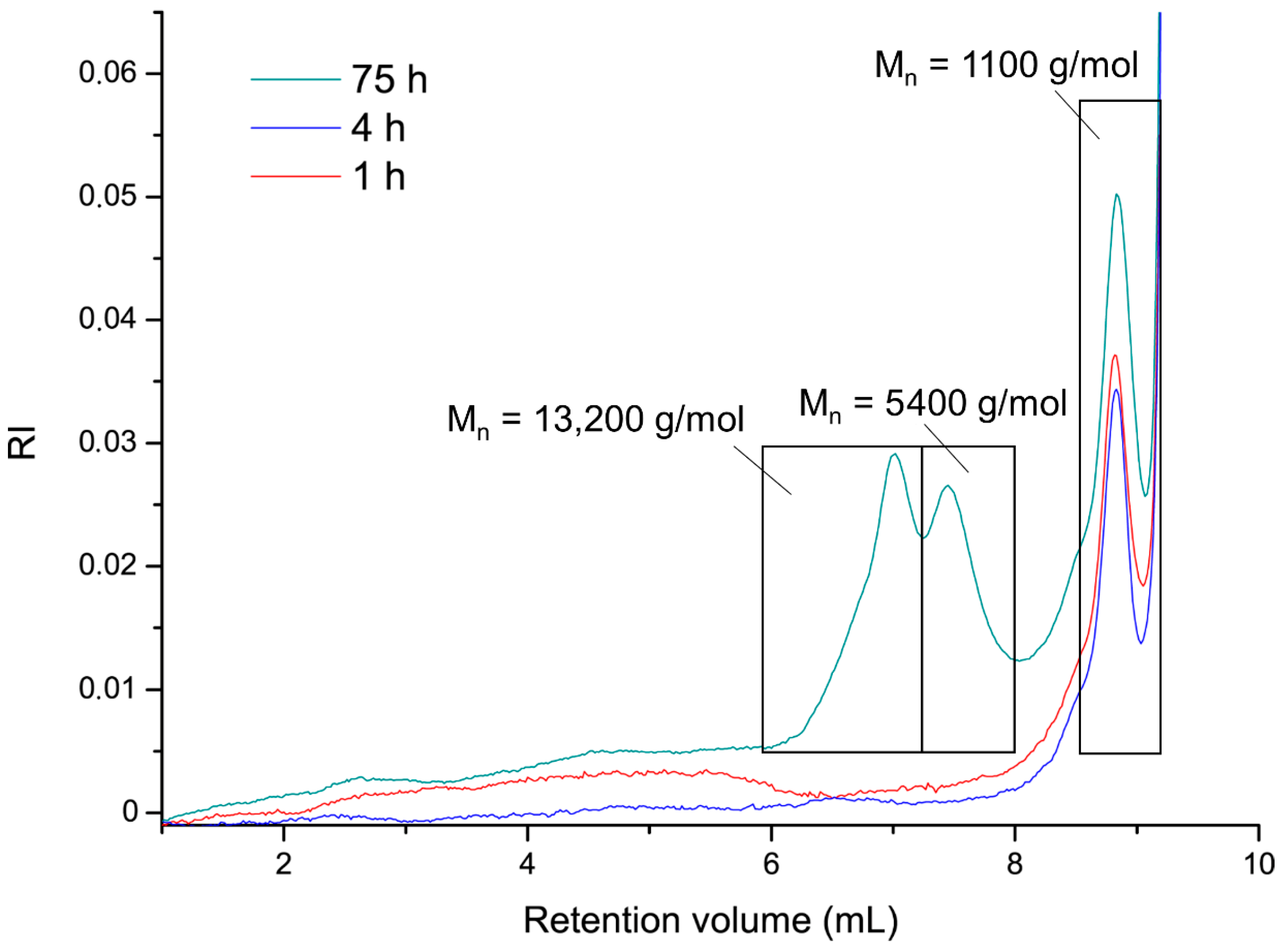
3.3. Investigation of Catalytic Activity of the Cu(I) bis(NHC) Complexes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Staudinger, H.; Bondy, H.F. Über Isopren und Kautschuk, 19. Mitteil.: Über die Molekülgröße des Kautschuks und der Balata. Chem. Ber. 1930, 63, 734–736. [Google Scholar] [CrossRef]
- Kauzmann, W.; Eyring, H. The viscous flow of large molecules. J. Am. Chem. Soc. 1940, 62, 3113–3125. [Google Scholar] [CrossRef]
- Caruso, M.M.; Davis, D.A.; Shen, Q.; Odom, S.A.; Sottos, N.R.; White, S.R.; Moore, J.S. Mechanically-induced chemical changes in polymeric materials. Chem. Rev. 2009, 109, 5755–5798. [Google Scholar] [CrossRef]
- Groote, R.; Jakobs, R.T.M.; Sijbesma, R.P. Mechanocatalysis: Forcing latent catalysts into action. Polym. Chem. 2013, 4, 4846–4859. [Google Scholar] [CrossRef]
- Boulatov, R. Polymer Mechanochemistry; Springer International Publishing: Heidelberg, Germany, 2015; Volume 369. [Google Scholar]
- Hickenboth, C.R.; Moore, J.S.; White, S.R.; Sottos, N.R.; Baudry, J.; Wilson, S.R. Biasing reaction pathways with mechanical force. Nature 2007, 446, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.A.; Hamilton, A.; Yang, J.; Cremar, L.D.; Van Gough, D.; Potisek, S.L.; Ong, M.T.; Braun, P.V.; Martínez, T.J.; White, S.R.; et al. Force-induced activation of covalent bonds in mechanoresponsive polymeric materials. Nature 2009, 459, 68. [Google Scholar] [CrossRef]
- Chen, Y.; Spiering, A.J.H.; Karthikeyan, S.; Peters, G.W.M.; Meijer, E.W.; Sijbesma, R.P. Mechanically induced chemiluminescence from polymers incorporating a 1,2-dioxetane unit in the main chain. Nat. Chem. 2012, 4, 559. [Google Scholar] [CrossRef]
- Michael, P.; Binder, W.H. A mechanochemically triggered “click” catalyst. Angew. Chem. Int. Ed. 2015, 54, 13918–13922. [Google Scholar] [CrossRef]
- Clough, J.M.; Balan, A.; van Daal, T.L.J.; Sijbesma, R.P. Probing force with mechanobase-induced chemiluminescence. Angew. Chem. Int. Ed. 2016, 55, 1445–1449. [Google Scholar] [CrossRef]
- Piermattei, A.; Karthikeyan, S.; Sijbesma, R.P. Activating catalysts with mechanical force. Nat. Chem. 2009, 1, 133–137. [Google Scholar] [CrossRef]
- Larsen, M.B.; Boydston, A.J. “Flex-activated” mechanophores: Using polymer mechanochemistry to direct bond bending activation. J. Am. Chem. Soc. 2013, 135, 8189–8192. [Google Scholar] [CrossRef]
- Diesendruck, C.E.; Steinberg, B.D.; Sugai, N.; Silberstein, M.N.; Sottos, N.R.; White, S.R.; Braun, P.V.; Moore, J.S. Proton-coupled mechanochemical transduction: A mechanogenerated acid. J. Am. Chem. Soc. 2012, 134, 12446–12449. [Google Scholar] [CrossRef]
- Lenhardt, J.M.; Ong, M.T.; Choe, R.; Evenhuis, C.R.; Martinez, T.J.; Craig, S.L. Trapping a diradical transition state by mechanochemical polymer extension. Science 2010, 329, 1057–1060. [Google Scholar] [CrossRef]
- Lenhardt, J.M.; Black, A.L.; Craig, S.L. gem-Dichlorocyclopropanes as abundant and efficient mechanophores in polybutadiene copolymers under mechanical stress. J. Am. Chem. Soc. 2009, 131, 10818–10819. [Google Scholar] [CrossRef]
- Kean, Z.S.; Craig, S.L. Mechanochemical remodeling of synthetic polymers. Polymer 2012, 53, 1035–1048. [Google Scholar] [CrossRef]
- Howard, J.L.; Cao, Q.; Browne, D.L. Mechanochemistry as an emerging tool for molecular synthesis: What can it offer? Chem. Sci. 2018, 9, 3080–3094. [Google Scholar] [CrossRef]
- Li, J.; Nagamani, C.; Moore, J.S. Polymer mechanochemistry: From destructive to productive. Acc. Chem. Res. 2015, 48, 2181–2190. [Google Scholar] [CrossRef]
- French, A.S. Mechanotransduction. Annu. Rev. Physiol. 1992, 54, 135–152. [Google Scholar] [CrossRef]
- Huang, H.; Kamm, R.D.; Lee, R.T. Cell mechanics and mechanotransduction: Pathways, probes, and physiology. Am. J. Physiol. Cell Physiol. 2004, 287, C1–C11. [Google Scholar] [CrossRef]
- Goodman, M.B.; Lumpkin, E.A.; Ricci, A.; Tracey, W.D.; Kernan, M.; Nicolson, T. Molecules and mechanisms of mechanotransduction. J. Neurosci. 2004, 24, 9220–9222. [Google Scholar] [CrossRef]
- Comrie, J.E.; Huck, W.T.S. Exploring actuation and mechanotransduction properties of polymer brushes. Macromol. Rapid Commun. 2008, 29, 539–546. [Google Scholar] [CrossRef]
- Crawley, J.T.B.; de Groot, R.; Xiang, Y.; Luken, B.M.; Lane, D.A. Unraveling the scissile bond: How ADAMTS13 recognizes and cleaves von Willebrand factor. Blood 2011, 118, 3212–3221. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, P.G.; Walker, R.G. Molecular basis of mechanosensory transduction. Nature 2001, 413, 194. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.-H.; Lukacs, V.; de Nooij, J.C.; Zaytseva, D.; Criddle, C.R.; Francisco, A.; Jessell, T.M.; Wilkinson, K.A.; Patapoutian, A. Piezo2 is the principal mechanotransduction channel for proprioception. Nat. Neurosci. 2015, 18, 1756. [Google Scholar] [CrossRef] [PubMed]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Mahla, R.S. Stem cells applications in regenerative medicine and disease therapeutics. Int. J. Cell Biol. 2016, 2016, 24. [Google Scholar] [CrossRef]
- Rief, M.; Grubmüller, H. Force spectroscopy of single biomolecules. Chem. Phys. Chem. 2002, 3, 255–261. [Google Scholar] [CrossRef]
- Yao, M.; Goult, B.T.; Klapholz, B.; Hu, X.; Toseland, C.P.; Guo, Y.; Cong, P.; Sheetz, M.P.; Yan, J. The mechanical response of talin. Nat. Commun. 2016, 7, 11966. [Google Scholar] [CrossRef]
- Eckels, E.C.; Tapia-Rojo, R.; Rivas-Pardo, J.A.; Fernández, J.M. The work of titin protein folding as a major driver in muscle contraction. Annu. Rev. Physiol. 2018, 80, 327–351. [Google Scholar] [CrossRef]
- Holst, J.; Watson, S.; Lord, M.S.; Eamegdool, S.S.; Bax, D.V.; Nivison-Smith, L.B.; Kondyurin, A.; Ma, L.; Oberhauser, A.F.; Weiss, A.S.; et al. Substrate elasticity provides mechanical signals for the expansion of hemopoietic stem and progenitor cells. Nat. Biotechnol. 2010, 28. [Google Scholar] [CrossRef]
- Yeo, G.C.; Baldock, C.; Tuukkanen, A.; Roessle, M.; Dyksterhuis, L.B.; Wise, S.G.; Matthews, J.; Mithieux, S.M.; Weiss, A.S. Tropoelastin bridge region positions the cell-interactive C terminus and contributes to elastic fiber assembly. Proc. Natl. Acad. Sci. USA 2012, 109, 2878–2883. [Google Scholar] [CrossRef]
- Qin, G.; Hu, X.; Cebe, P.; Kaplan, D.L. Mechanism of resilin elasticity. Nat. Commun. 2012, 3, 1003. [Google Scholar] [CrossRef]
- Groote, R.; Szyja, B.M.; Pidko, E.A.; Hensen, E.J.M.; Sijbesma, R.P. Unfolding and mechanochemical scission of supramolecular polymers containing a metal–ligand coordination bond. Macromolecules 2011, 44, 9187–9195. [Google Scholar] [CrossRef]
- Wang, J.; Kouznetsova, T.B.; Niu, Z.; Ong, M.T.; Klukovich, H.M.; Rheingold, A.L.; Martinez, T.J.; Craig, S.L. Inducing and quantifying forbidden reactivity with single-molecule polymer mechanochemistry. Nat. Chem. 2015, 7, 323. [Google Scholar] [CrossRef]
- Kryger, M.J.; Munaretto, A.M.; Moore, J.S. Structure–mechanochemical activity relationships for cyclobutane mechanophores. J. Am. Chem. Soc. 2011, 133, 18992–18998. [Google Scholar] [CrossRef]
- Kean, Z.S.; Black Ramirez, A.L.; Yan, Y.; Craig, S.L. Bicyclo[3.2.0]heptane mechanophores for the non-scissile and photochemically reversible generation of reactive bis-enones. J. Am. Chem. Soc. 2012, 134, 12939–12942. [Google Scholar] [CrossRef]
- Lavalle, P.; Boulmedais, F.; Schaaf, P.; Jierry, L. Soft-mechanochemistry: Mechanochemistry inspired by nature. Langmuir 2016, 32, 7265–7276. [Google Scholar] [CrossRef]
- Polacheck, W.J.; Chen, C.S. Measuring cell-generated forces: A guide to the available tools. Nat. Methods 2016, 13, 415. [Google Scholar] [CrossRef]
- Jiang, G.; Giannone, G.; Critchley, D.R.; Fukumoto, E.; Sheetz, M.P. Two-piconewton slip bond between fibronectin and the cytoskeleton depends on talin. Nature 2003, 424, 334. [Google Scholar] [CrossRef]
- Finer, J.T.; Simmons, R.M.; Spudich, J.A. Single myosin molecule mechanics: Piconewton forces and nanometre steps. Nature 1994, 368, 113. [Google Scholar] [CrossRef] [PubMed]
- Vogel, V. Mechanotransduction involving multimodular proteins: Converting force into biochemical signals. Annu. Rev. Biophys. 2006, 35, 459–488. [Google Scholar] [CrossRef]
- Jasti, J.; Furukawa, H.; Gonzales, E.B.; Gouaux, E. Structure of acid-sensing ion channel 1 at 1.9 Å resolution and low pH. Nature 2007, 449, 316. [Google Scholar] [CrossRef]
- Chalfie, M. Neurosensory mechanotransduction. Nat. Rev. Mol. Cell Biol. 2009, 10, 44. [Google Scholar] [CrossRef]
- Davila, J.; Chassepot, A.; Longo, J.; Boulmedais, F.; Reisch, A.; Frisch, B.; Meyer, F.; Voegel, J.-C.; Mésini, P.J.; Senger, B.; et al. Cyto-mechanoresponsive polyelectrolyte multilayer films. J. Am. Chem. Soc. 2012, 134, 83–86. [Google Scholar] [CrossRef]
- Bacharouche, J.; Badique, F.; Fahs, A.; Spanedda, M.V.; Geissler, A.; Malval, J.-P.; Vallat, M.-F.; Anselme, K.; Francius, G.; Frisch, B.; et al. Biomimetic cryptic site surfaces for reversible chemo- and cyto-mechanoresponsive substrates. ACS Nano 2013, 7, 3457–3465. [Google Scholar] [CrossRef]
- Mertz, D.; Vogt, C.; Hemmerlé, J.; Mutterer, J.; Ball, V.; Voegel, J.-C.; Schaaf, P.; Lavalle, P. Mechanotransductive surfaces for reversible biocatalysis activation. Nat. Mater. 2009, 8, 731. [Google Scholar] [CrossRef] [PubMed]
- Longo, J.; Yao, C.; Rios, C.; Chau, N.T.T.; Boulmedais, F.; Hemmerlé, J.; Lavalle, P.; Schiller, S.M.; Schaaf, P.; Jierry, L. Reversible biomechano-responsive surface based on green fluorescent protein genetically modified with unnatural amino acids. Chem. Commun. 2015, 51, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Rios, C.; Longo, J.; Zahouani, S.; Garnier, T.; Vogt, C.; Reisch, A.; Senger, B.; Boulmedais, F.; Hemmerlé, J.; Benmlih, K.; et al. A new biomimetic route to engineer enzymatically active mechano-responsive materials. Chem. Commun. 2015, 51, 5622–5625. [Google Scholar] [CrossRef]
- Van der Hoff, B.M.E.; Glynn, P.A.R. The rate of degradation by ultrasonation of polystyrene in solution. J. Macromol. Sci. A 1974, 8, 429–449. [Google Scholar] [CrossRef]
- Van der Hoff, B.M.E.; Gall, C.E. A method for following changes in molecular weight distributions of polymers on degradation: Development and comparison with ultrasonic degradation experiments. J. Macromol. Sci. A 1977, 11, 1739–1758. [Google Scholar] [CrossRef]
- Xue, F.; Zhu, C.; Liu, F.; Wang, S.; Liu, H.; Li, C. Effects of high-intensity ultrasound treatment on functional properties of plum (Pruni domesticae semen) seed protein isolate. J. Sci. Food Agric. 2018. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Bravo-Rodriguez, K.; Phillips, C.; Seidel, R.W.; Wieberneit, F.; Stoll, R.; Doltsinis, N.L.; Sanchez-Garcia, E.; Sander, W. Conformation and dynamics of a cyclic disulfide-bridged peptide: Effects of temperature and solvent. J. Phys. Chem. B 2013, 117, 3560–3570. [Google Scholar] [CrossRef] [PubMed]
- Tauchman, J.; Císařová, I.; Štěpnička, P. Chiral phosphanylferrocenecarboxamides with amino acid pendant groups as ligands for Cu-mediated asymmetric conjugate additions of diethylzinc to chalcones— Structural characterisation of precursors to the Cu catalyst. Eur. J. Org. Chem. 2010, 2010, 4276–4287. [Google Scholar] [CrossRef]
- Alvarez, S.G.; Alvarez, M.T. A practical procedure for the synthesis of alkyl azides at ambient temperature in dimethyl sulfoxide in high purity and yield. Synthesis 1997, 1997, 413–414. [Google Scholar] [CrossRef]
- Chen, Y.; Guan, Z. Bioinspired modular synthesis of elastin-mimic polymers to probe the mechanism of elastin elasticity. J. Am. Chem. Soc. 2010, 132, 4577–4579. [Google Scholar] [CrossRef]
- Roy, S.; Eastman, A.; Gribble, G.W. Synthesis of bisindolylmaleimides related to GF109203x and their efficient conversion to the bioactive indolocarbazoles. Org. Biomol. Chem. 2006, 4, 3228–3234. [Google Scholar] [CrossRef]
- Chun, J.; Lee, H.S.; Jung, I.G.; Lee, S.W.; Kim, H.J.; Son, S.U. Cu2O: A versatile reagent for base-free direct synthesis of NHC-copper complexes and decoration of 3D-MOF with coordinatively unsaturated NHC-copper species. Organometallics 2010, 29, 1518–1521. [Google Scholar] [CrossRef]
- Michael, P.; Biewend, M.; Binder, W.H. Mechanochemical activation of fluorogenic CuAAC “click” reactions for stress-sensing applications. Macromol. Rapid Commun. 2018, 1800376. [Google Scholar] [CrossRef]
- Meneses, C.; Nicoll, S.L.; Trembleau, L. Multigram-scale synthesis of short peptides via a simplified repetitive solution-phase procedure. J. Org. Chem. 2010, 75, 564–569. [Google Scholar] [CrossRef]
- Cinzia, C.; Anna, B. Synthesis of α-N-linked glycopeptides. Eur. J. Org. Chem. 2011, 2011, 3911–3919. [Google Scholar] [CrossRef]
- Michael, P.; Sheidaee Mehr, S.K.; Binder, W.H. Synthesis and characterization of polymer linked copper(I) bis(N-heterocyclic carbene) mechanocatalysts. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 3893–3907. [Google Scholar] [CrossRef]
- Binder, W.H.; Sachsenhofer, R. ‘Click’ chemistry in polymer and material science: An update. Macromol. Rapid Commun. 2008, 29, 952–981. [Google Scholar] [CrossRef]
- Díez-González, S.; Nolan, S.P. [(NHC)2Cu]X complexes as efficient catalysts for azide-alkyne click chemistry at low catalyst loadings. Angew. Chem. Int. Ed. 2008, 47, 8881–8884. [Google Scholar] [CrossRef] [PubMed]
- Huisgen, R. Kinetics and mechanism of 1,3-dipolar cycloadditions. Angew. Chem. Int. Ed. Engl. 1963, 2, 633–645. [Google Scholar] [CrossRef]

| Entry | Catalyst | Ultrasound 2 | t (h) | T (°C) | Conversion 3 (%) |
|---|---|---|---|---|---|
| 1 | 3 | on | 42.5 | rt | 3.8 |
| 2 | 4 | on | 42.5 | rt | 3.4 |
| 3 | 4 | off | 42.5 | rt | 0 |
| 4 | 4 | off | 42.5 | 60 | 1.9 |
| 5 | 7 | on | 42.5 | rt | 9.9 |
| 6 | 8 | on | 42.5 | rt | 4.8 |
| 7 | 11 | on | 42.5 | rt | 6.9 |
| 8 | 12 | on | 42.5 | rt | 3.4 |
| 9 | 13 | on | 42.5 | rt | 3.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Funtan, S.; Michael, P.; Binder, W.H. Synthesis and Mechanochemical Activity of Peptide-Based Cu(I) Bis(N-heterocyclic carbene) Complexes. Biomimetics 2019, 4, 24. https://doi.org/10.3390/biomimetics4010024
Funtan S, Michael P, Binder WH. Synthesis and Mechanochemical Activity of Peptide-Based Cu(I) Bis(N-heterocyclic carbene) Complexes. Biomimetics. 2019; 4(1):24. https://doi.org/10.3390/biomimetics4010024
Chicago/Turabian StyleFuntan, Sebastian, Philipp Michael, and Wolfgang H. Binder. 2019. "Synthesis and Mechanochemical Activity of Peptide-Based Cu(I) Bis(N-heterocyclic carbene) Complexes" Biomimetics 4, no. 1: 24. https://doi.org/10.3390/biomimetics4010024
APA StyleFuntan, S., Michael, P., & Binder, W. H. (2019). Synthesis and Mechanochemical Activity of Peptide-Based Cu(I) Bis(N-heterocyclic carbene) Complexes. Biomimetics, 4(1), 24. https://doi.org/10.3390/biomimetics4010024