Fabrication of Mechanically Stable Superhydrophobic Aluminium Surface with Excellent Self-Cleaning and Anti-Fogging Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
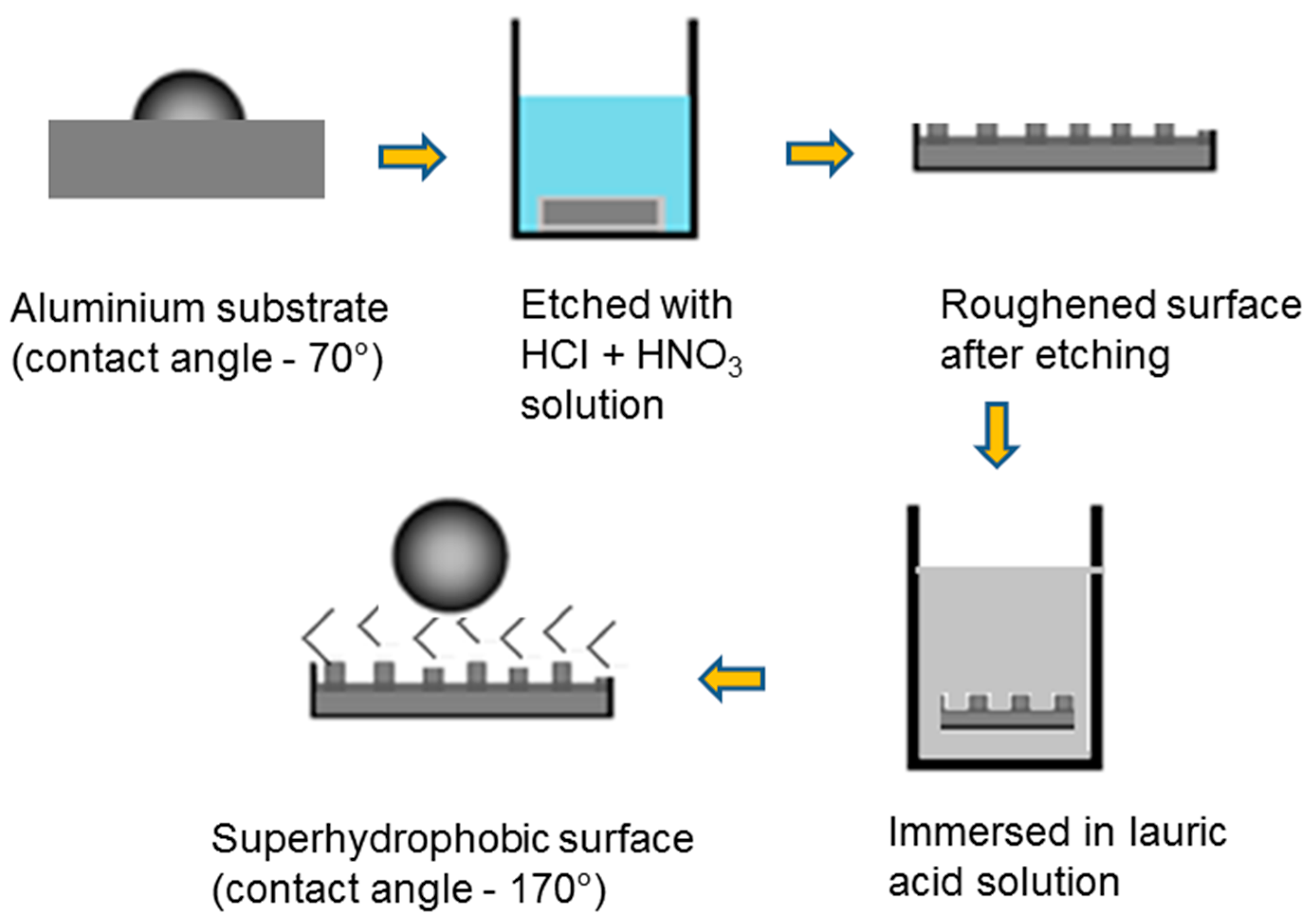
2.2. Synthesis of Superhydrophobic Coatings
2.3. Characterization of Superhydrophobic Coatings
3. Results and Discussion
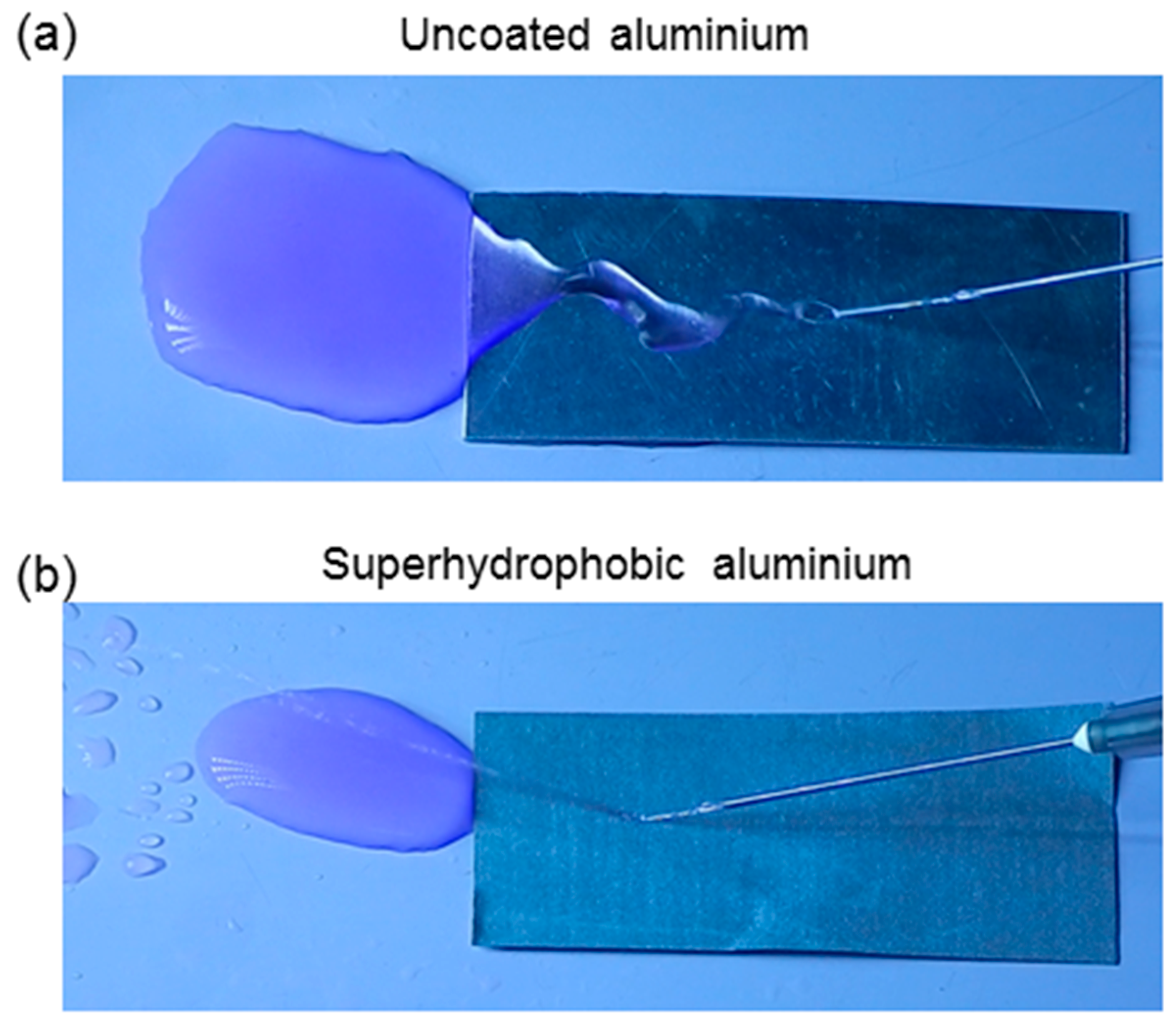
3.1. Surface Morphology and Wetting Properties
3.2. Wetting Stability of the Coatings under Perturbation Conditions
3.3. Self-Cleaning and Anti-Fogging Properties of the Coatings
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Guo, Z.; Liu, W. Highly transparent superhydrophobic thin film with low refractive index prepared by one-step coating of modified silica nanoparticles. Plant Sci. 2007, 172, 1103–1112. [Google Scholar] [CrossRef]
- Bixler, G.D.; Bhushan, B. Fluid drag reduction and efficient self-cleaning with rice leaf and butterfly wing bioinspired surfaces. Nanoscale 2014, 6, 76–96. [Google Scholar] [CrossRef] [PubMed]
- Blossey, R. Self-cleaning surfaces—virtual realities. Nature 2003, 2, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Gao, X.; Jiang, L. Application of superhydrophobic surface with high adhesive force in no lost transport of superparamagnetic microdroplet. J. Am. Chem. Soc. 2007, 129, 1478–1479. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Sato, O.; Taguchi, M.; Einaga, Y.; Murakami, T.; Fujishima, A. Self-cleaning particle coating with antireflection properties. Chem. Mater. 2005, 17, 696–700. [Google Scholar] [CrossRef]
- Cheng, Z.; Lai, H.; Zhang, N.; Sun, K.; Jiang, L. Magnetically Induced Reversible Transition between Cassie and Wenzel States of Superparamagnetic Microdroplets on Highly Hydrophobic Silicon Surface. J. Phys. Chem. C 2012, 116, 18796–18802. [Google Scholar] [CrossRef]
- Qu, M.; Zhao, G.; Cao, X.; Zhang, J. Biomimetic Fabrication of Lotus-Leaf-like Structured Polyaniline Film with Stable Superhydrophobic and Conductive Properties. Langmuir 2008, 24, 4185–4189. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Park, B.G.; Kim, D.H.; Ahn, D.J.; Park, Y.; Lee, S.H.; Lee, K.B. Nanostructure-Dependent Water-Droplet Adhesiveness Change in Superhydrophobic Anodic Aluminum Oxide Surfaces: From Highly Adhesive to Self-Cleanable. Langmuir 2010, 26, 1412–1415. [Google Scholar] [CrossRef] [PubMed]
- Desbief, S.; Grignard, B.; Detrembleur, C.; Rioboo, R.; Vaillant, A.; Seveno, D.; Voue, M.; de Coninck, J.; Jonas, A.M.; Jerome, C. Superhydrophobic Aluminum Surfaces by Deposition of Micelles of Fluorinated Block Copolymers. Langmuir 2010, 26, 2057–2067. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.J.; Guo, Y.B.; Fang, S.M. A facial approach to fabricate superhydrophobic aluminum surface. Surf. Interface Anal. 2010, 42, 1–6. [Google Scholar] [CrossRef]
- Qian, B.; Shen, Z. Fabrication of Superhydrophobic Surfaces by Dislocation-Selective Chemical Etching on Aluminum, Copper, and Zinc Substrates. Langmuir 2005, 21, 9007–9009. [Google Scholar] [CrossRef] [PubMed]
- Xiu, Y.; Zhu, L.; Hess, D.W.; Wong, C.P. Hierarchical silicon etched structures for controlled hydrophobicity/superhydrophobicity. Nano Lett. 2007, 7, 3388. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D.K.; Farzaneh, M.; Paynter, R.W. Superhydrophobic properties of ultrathin rf-sputtered Teflon films coated etched aluminum surfaces. Mater. Lett. 2008, 62, 1226. [Google Scholar] [CrossRef]
- Borras, A.; Barranco, A.; Gonzalez-Elipe, A.R. Reversible Superhydrophobic to Superhydrophilic Conversion of Ag@TiO2 Composite Nanofiber Surfaces. Langmuir 2008, 24, 8021. [Google Scholar] [CrossRef] [PubMed]
- Pastine, S.J.; Okawa, D.; Kessler, B.; Rolandi, M.; Llorente, M.; Zettl, A.; Frechet, J.M.J. A facile and patternable method for the surface modification of carbon nanotube forests using perfluoroarylazides. J. Am. Chem. Soc. 2008, 130, 4238. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.C.; McCarthy, T.J. (CH3)3SiCl/SiCl4 Azeotrope Grows Superhydrophobic Nanofilaments. Langmuir 2008, 24, 362–364. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Koratkar, N.; Ci, L.; Ajayan, P.M. Combined micro-/nanoscale surface roughness for enhanced hydrophobic stability in carbon nanotube arrays. Appl. Phys. Lett. 2007, 90, 143117. [Google Scholar] [CrossRef]
- Larmour, I.A.; Saunders, G.C.; Bell, S.E.J. Sheets of large superhydrophobic metal particles self-assembled on water by the cheerios effect. Angew. Chem. 2008, 47, 5043. [Google Scholar] [CrossRef] [PubMed]
- Ofir, Y.; Samanta, B.; Arumugam, P.; Rotello, V.M. Controlled fluorination of FePt nanoparticles: Hydrophobic to superhydrophobic surfaces. Adv. Mater. 2007, 19, 407. [Google Scholar] [CrossRef]
- Motornov, M.; Sheparovych, R.; Lupitskyy, R.; MacWilliams, E.; Minko, S. Responsive colloidal systems: Reversible aggregation and fabrication of superhydrophobic surfaces. J. Colloid Interface Sci. 2007, 310, 481. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Iardino, G.; Bertarelli, C.; Miozzo, L.; Papagni, A.; Zerbi, G. Modification of surface properties of electrospun polyamide nanofibers by means of a perfluorinated acridine. Appl. Surf. Sci. 2007, 253, 8360. [Google Scholar] [CrossRef]
- Menini, R.; Farzaneh, M. Production of superhydrophobic polymer fibers with embedded particles using the electrospinning technique. Polym. Int. 2008, 57, 77. [Google Scholar] [CrossRef]
- Rao, A.V.; Gurav, A.B.; Latthe, S.S.; Vhatkar, R.S.; Imai, H.; Kappenstein, C.; Wagh, P.B.; Gupta, S.C. Water repellent porous silica films by sol–gel dip coating method. J. Colloid Interface Sci. 2010, 352, 30. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.F.; Wang, J.N. A superhydrophobic coating on aluminium foil with an anti-corrosive property. New J. Chem. 2009, 33, 734. [Google Scholar] [CrossRef]
- Lee, J.A.; McCarthy, T.J. Polymer surface modification: Topography effects leading to extreme wettability behavior. Macromolecules 2007, 40, 3965. [Google Scholar] [CrossRef]
- Pozzato, A.; Dal, Z.S.; Fois, G.; Vendramin, D.; Mistura, G.; Belotti, M.; Chen, Y.; Natali, M. Superhydrophobic surfaces fabricated by nanoimprint lithography. Microelectron. Eng. 2006, 83, 884. [Google Scholar] [CrossRef]
- Davis, J.R. ASM Special Handbook: Aluminum and Aluminum Alloys; ASM International: Materials Park, OH, USA, 1993. [Google Scholar]
- Hatch, J.E. Aluminum-Properties and Physical Metallurgy; ASM International: Materials Park, OH, USA, 1984. [Google Scholar]
- Peng, S.; Bhushan, B. Mechanically durable superoleophobic aluminum surfaces with microstep and nanoreticula hierarchical structure for self-cleaning and anti-smudge properties. J. Colloid Interface Sci. 2016, 461, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Cakir, O. Chemical etching of aluminium. J. Mater. Process. Technol. 2008, 199, 337–340. [Google Scholar] [CrossRef]
- He, M.; Zhou, X.; Zeng, X.; Cui, D.; Zhang, Q.; Chen, J.; Li, H.; Wang, J.; Cao, Z.; Song, Y. Hierarchically structured porous aluminum surfaces for high-efficient removal of condensed water. Soft Matter 2012, 8, 6680–6683. [Google Scholar] [CrossRef]
- Ren, S.; Yang, S.; Zhao, Y. Nano-tribological study on a super-hydrophobic film formed on rough aluminum substrates. Acta Mech. Sin. 2004, 20, 159–164. [Google Scholar]
- Guo, Z.G.; Zhou, F.; Hao, J.C.; Liu, W.M. Stable Biomimetic Super-Hydrophobic Engineering Materials. J. Am. Chem. Soc. 2005, 127, 15670–15671. [Google Scholar] [CrossRef] [PubMed]
- Saleema, N.; Sarkar, D.K.; Paynter, R.W.; Chen, X.G. Superhydrophobic aluminum alloy surfaces by a novel one-step process. Appl. Mater. Interfaces 2010, 2, 2500–2502. [Google Scholar] [CrossRef] [PubMed]
- Saleema, N.; Sarkar, D.K.; Gallant, D.; Paynter, R.W.; Chen, X.G. Superhydrophobic aluminum alloy surfaces by a novel one-step process. ACS Appl. Mater. Interfaces 2011, 3, 4775–4781. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Li, W. A novel simple approach to preparation of superhydrophobic surfaces of aluminum alloys. Appl. Surf. Sci. 2011, 258, 1004–1007. [Google Scholar] [CrossRef]
- Fu, X.Y.; He, X.H. Fabrication of super-hydrophobic surfaces on aluminum alloy substrates. Appl. Surf. Sci. 2008, 255, 1776–1781. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, B.W.; Qu, M.N.; Zhang, J.Y.; He, D.Y. Fabrication of superhydrophobic surfaces on engineering material surfaces with stearic acid. Appl. Surf. Sci. 2008, 254, 2009–2012. [Google Scholar] [CrossRef]
- Shi-heng, Y.I.N.; Bin, Z.H.U.; Yun-chun, L.I.U.; Ji, Y.; Tong-chun, K. Fabrication of superhydrophobic aluminum plate by surface etching and fluorosilane modification. Chem. Res. Chin. Univ. 2012, 28, 903–906. [Google Scholar]
- Li, S.M.; Li, B.; Liu, J.H.; Yu, M. Corrosion resistance of superhydrophobic film on aluminum alloy surface fabricated by chemical etching and anodization. Chin. J. Inorg. Chem. 2012, 28, 1755–1762. [Google Scholar]
- Zhang, Y.; Ge, D.; Yang, S. Spray-coating of superhydrophobic aluminum alloys with enhanced mechanical robustness. J. Colloid Interface Sci. 2014, 423, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Latthe, S.S.; Sudhagar, P.; Ravidhas, C.; Christy, A.J.; Kirubakaran, D.D.; Venkatesh, R.; Devadoss, A.; Terashima, C.; Nakata, K.; Fujishima, A. Self-cleaning and superhydrophobic CuO coating by jet-nebulizer spray pyrolysis technique. Cryst. Eng. Commun. 2015, 17, 2624–2628. [Google Scholar] [CrossRef]
- Varshney, P.; Mohapatra, S.S.; Kumar, A. Superhydrophobic coatings for aluminium surfaces synthesized by chemical etching process. Int. J. Smart Nanomater. 2016, 7, 248–264. [Google Scholar] [CrossRef]
- Kibar, A.; Karabay, H.; Yi˘git, K.S.; Ucar, I.; Erbil, H.Y. Experimental investigation of inclined liquid water jet flow onto vertically located superhydrophobic surfaces. Exp. Fluids 2010, 49, 1135–1145. [Google Scholar] [CrossRef]
- Jung, Y.C.; Bhushan, B. Mechanically Durable Carbon Nanotube−Composite Hierarchical Structures with Superhydrophobicity, Self-Cleaning, and Low-Drag. ACS Nano 2009, 3, 4155–4163. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lopez, C.; Hirsa, A.; Koratkar, N. Impact dynamics and rebound of water droplets on superhydrophobic carbon nanotube arrays. Appl. Phys. Lett. 2007, 91, 023105. [Google Scholar] [CrossRef]
- Karapanagiotis, I.; Aifantis, K.E.; Konstantinidis, A. Capturing the evaporation process of water drops on sticky superhydrophobic polymer-nanoparticle surfaces. Mater. Lett. 2016, 164, 117–119. [Google Scholar] [CrossRef]
- Wang, N.; Xiong, D.; Deng, Y.; Shi, Y.; Wang, K. Mechanically Robust Superhydrophobic Steel Surface with Anti-Icing, UV-Durability, and Corrosion Resistance Properties. ACS Appl. Mater. Interfaces 2015, 7, 6260–6272. [Google Scholar] [CrossRef] [PubMed]
- Latthe, S.S.; Sudhagar, P.; Devadoss, A.; Kumar, A.M.; Liu, S.; Terashim, C.; Nakataa, K.; Fujishima, A. A mechanically bendable superhydrophobic steel surface with self-cleaning and corrosion-resistant properties. J. Mater. Chem. A 2015, 3, 14263. [Google Scholar] [CrossRef]
- Lomga, J.; Varshney, P.; Nanda, D.; Satapathy, M.; Mohapatra, S.S.; Kumar, A. Fabrication of durable and regenerable superhydrophobic coatings with excellent self-cleaning and anti-fogging properties for aluminium surfaces. J. Alloys Compd. 2017, 702, 161–170. [Google Scholar] [CrossRef]
- Sas, I.; Gorga, R.E.; Joines, J.A.; Thoney, K.A. Literature review on superhydrophobic self-cleaning surfaces produced by electrospinning. J. Polym. Sci. Part B Polym. Phys. 2012, 50, 824–845. [Google Scholar] [CrossRef]
- Mundo, R.D.; Agostino, R.; Palumbo, F. Long-Lasting Antifog Plasma Modification of Transparent Plastics. ACS Appl. Mater. Interface 2014, 6, 17059–17066. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, X.; Wu, H.; Wu, J. Facile fabrication of superhydrophobic nanostructures on aluminum foils with controlled-condensation and delayed-icing effects. Appl. Surf. Sci. 2012, 258, 8253–8257. [Google Scholar] [CrossRef]






© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varshney, P.; Mohapatra, S.S.; Kumar, A. Fabrication of Mechanically Stable Superhydrophobic Aluminium Surface with Excellent Self-Cleaning and Anti-Fogging Properties. Biomimetics 2017, 2, 2. https://doi.org/10.3390/biomimetics2010002
Varshney P, Mohapatra SS, Kumar A. Fabrication of Mechanically Stable Superhydrophobic Aluminium Surface with Excellent Self-Cleaning and Anti-Fogging Properties. Biomimetics. 2017; 2(1):2. https://doi.org/10.3390/biomimetics2010002
Chicago/Turabian StyleVarshney, Priya, Soumya S. Mohapatra, and Aditya Kumar. 2017. "Fabrication of Mechanically Stable Superhydrophobic Aluminium Surface with Excellent Self-Cleaning and Anti-Fogging Properties" Biomimetics 2, no. 1: 2. https://doi.org/10.3390/biomimetics2010002
APA StyleVarshney, P., Mohapatra, S. S., & Kumar, A. (2017). Fabrication of Mechanically Stable Superhydrophobic Aluminium Surface with Excellent Self-Cleaning and Anti-Fogging Properties. Biomimetics, 2(1), 2. https://doi.org/10.3390/biomimetics2010002




