Advances in Biomaterials for Tissue Regeneration: From Scaffold Design to CAP-Enabled Interfaces and AI-Driven Optimization
Abstract
1. Introduction
- (1)
- Compare natural and synthetic biomaterials, including hybrid systems;
- (2)
- Outline advanced fabrication strategies (electrospinning and 3D/4D printing) and architectural parameters;
- (3)
- Highlight surface engineering, with emphasis on CAP;
- (4)
- Discuss cell–material crosstalk and immunomodulation;
- (5)
- Explore the role of AI in predictive design;
- (6)
- Address safety considerations and translational challenges.
2. Materials and Methods
3. Results and Discussion
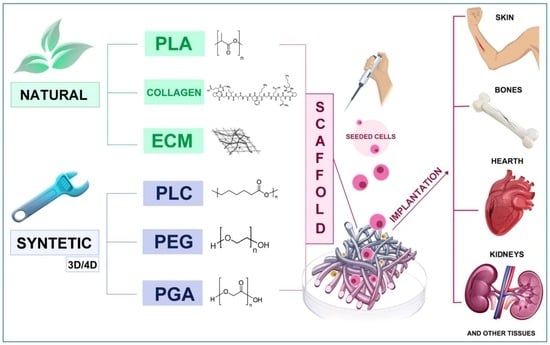
3.1. Tissue Engineering and Scaffold Categories
3.2. Cells Involved in Tissue Engineering
| Study | Type of Cells | Experimental Model | Main Findings |
|---|---|---|---|
| Lanza et al. [19] | Renal cells cloned from bovine fetuses | In vitro cultivation and in vivo reimplantation | Cells self-organized into structures capable of waste product elimination |
| Ross et al. [20] | Murine embryonic stem cells | Seeding onto rat kidney extracellular matrix | Cells proliferated and differentiated |
| Goto et al. [21] | Epithelial cells and fibroblasts | Amniotic membrane used as basement membrane | Formation of a multilayered epithelium without the need for added growth factors |
| Biniazan et al. [25] | Adipose tissue stem cells | In vitro cultivation | Formation of new blood vessels |
| Zhao et al. [26] | Adipose tissue stem cells from kidney | In vivo cultivation (rabbit model) | Formation of ureter structure: no signs of inflammation are observed |
3.3. Biomaterials for TE: Natural, Synthetic, and Hybrid Strategies
| Category | Subcategory | Examples |
|---|---|---|
| Natural | Biopolymers | Polylactic acid (PLA) and polyhydroxyalkanoates (PHAs) |
| Polysaccharides | Hyaluronic acid (HA), alginate, cellulose, and chitosan | |
| Polypeptides and proteins | Collagen, gelatine, and fibroin | |
| Synthetic | PCL | Polycaprolactone |
| PEG | Polyethylene glycol | |
| PGA | Polyglycolic acid | |
| PLGA | Polylactic glycolic acid |
| Feature | Natural | Synthetic | Hybrid |
|---|---|---|---|
| Bioactivity | High | Low | High |
| Mechanical strength | Low–moderate | High | Tunable |
| Degradation control | Limited | Precise | Tunable |
| Reproducibility | Low | High | Moderate |
| Clinical scalability | Moderate | High | Emerging |
3.3.1. Natural Biomaterials
- Bio polyesters, such as PLA, PHAs and their derivatives;
- Polysaccharides, such as HA, alginate, cellulose and chitosan.;
- Polypeptides and proteins, such as collagen, gelatin, fibroin, polyglutamic acids and antimicrobial peptides (AMPs) which contribute to ECM-like biofunctionality.
3.3.2. Collagen and Cellulose
3.3.3. Decellularized Extracellular Matrix
3.3.4. Synthetic Biomaterials
3.3.5. Hybrid Composites
3.4. Advanced Fabrication: From Electrospinning to 3D/4D Printing
3.5. Surface Engineering for Bioactivity and Hemocompatibility
3.6. Cold Atmospheric Plasma in Biomaterial Surface Modification
- CAP treatment significantly improves wettability, thereby enhancing protein adsorption and cell adhesion. For instance, in electrospun poly(vinyl alcohol)/poly(L-lactic acid) (PVA/PLLA) nanofibrous scaffolds, CAP reduces the water contact angle (WCA) from approximately 110° to 50°, promoting ECM protein adsorption and improving fibroblast and osteoblast attachment. This effect is accompanied by increased alkaline phosphatase (ALP) activity and accelerated calcium deposition in mesenchymal stem cells (MSCs), indicating enhanced osteogenic differentiation [72,73].
- In addition to supporting adhesion, CAP modulates cell proliferation and differentiation. In PCL/PLGA scaffolds loaded with β carotene, CAP enhances MSC adhesion, proliferation, and mineralization, while upregulating osteogenic markers such as RUNX2, SOX9, and osteonectin [74]. Similar effects have been observed in human calvaria osteoblasts (HCOs), with increased expression of ALP, COL1A1, RUNX2, and BMP2, suggesting activation of maturation [75].
- CAP is also effective in improving the performance of additively manufactured scaffolds. In 3D-printed PLA constructs, it increases nanoscale roughness (from approximately 1.2 nm to over 27 nm) while enhancing surface chemistry and hydrophilicity, resulting in improved osteoblast attachment, spreading, and proliferation [76].
- Beyond bone applications, CAP demonstrates versatility across different tissues. For example, electrospun PCL scaffolds loaded with TGF β1-releasing microspheres exhibit enhanced hydrophilicity, increased vitronectin adsorption, and deeper cell infiltration following CAP treatment. In 3D-cultures, these scaffolds promote glycosaminoglycan (GAG) deposition and type II collagen expression, supporting cartilage tissue maturation [77].
- The biological effects of CAP are primarily driven by ROS/RNS generation, UV radiation, localized electric fields, and reactive radicals. These factors increase surface energy, introduce polar functional groups (hydroxyl, carbonyl, and carboxyl), and modify surface topography. As a result, wettability improves, facilitating selective adsorption of ECM proteins such as fibronectin and vitronectin, activation of integrin-mediated adhesion, and downstream signaling pathways regulating migration, proliferation, and lineage commitment [78,79].
| Biomaterial/System | Type of CAP Treatment | Physicochemical Modifications | Biological Response | Main Application | |
|---|---|---|---|---|---|
| PVA/PLLA | Atmospheric cold plasma; low temperature | WCA reduction from 110° to 50°; increased surface energy | Enhanced fibroblast and osteoblast adhesion; ↑ ALP; ↑ Ca2+ deposition; ↑ ECM protein adsorption | Bone/soft tissue regeneration | [72] |
| PCL/PLGA + β-carotene | Surface activation via CAP | Introduction of polar groups (C–O and C=O); improved wettability | ↑ MSC adhesion, proliferation, and mineralization; ↑ osteogenic markers (RUNX2, SOX9, and osteonectin) | Bone regeneration | [74] |
| Human calvaria osteoblasts (HCOs) | Direct CAP exposure | Controlled ROS generation; sub-nm topography modification | ↑ ALP, COL1A1, RUNX2, and BMP2; ↑ mineralization | Osteoinduction/bone models | [75] |
| PLA | CAP with high reactive species density | Roughness increases from 1.2 to 27.6 nm; ↑ –OH/–COOH; ↑ hydrophilicity | Improved osteoblast adhesion, spreading, and proliferation; better 3D colonization | Bone regeneration | [76] |
| PCL + TGF-β1 microspheres | CAP pre-treatment | ↑ Vitronectin adsorption; ↑ hydrophilicity; improved cell infiltration | ↑ GAG; ↑ collagen II; enhanced cartilage maturation | Cartilage regeneration | [77] |
| PLA/CaCO3 composites | CAP interfacial enhancement | Improved polymer–mineral bonding; ↑ hydrophilicity; ↑ mechanical stability | ↑ MSC proliferation; improved biocompatibility | Bioactive composites/bone | [80,81] |
| PCL | CAP surface functionalization | Carbonyl/carboxyl introduction; ↑ surface energy | ↑ fibroblast, MSC, and chondrocyte adhesion and proliferation | Soft tissue, cartilage, and nerve | [78,79] |
3.7. Artificial Intelligence as a Driving Force in Next-Generation Biomaterial Design
3.8. Clinical Translation of Next-Generation Biomaterials
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAP | cold atmospheric plasma |
| AI | artificial intelligence |
| TE | tissue engineering |
| TR | tissue regeneration |
| ECM | extracellular matrix |
| ESCs | embryonic stem cells |
| iPSCs | induced pluripotent stem cells |
| ADMSCs | adipose-derived mesenchymal stem cells |
| ADSCs | adipose-derived stem cells |
| PLA | polylactic acid |
| PHAs | polyhydroxyalkanoates |
| HA | hyaluronic acid |
| PCL | polycaprolactone |
| PEG | polyethylene glycol |
| PGA | polyglycolic acid |
| PLGA | polylactic glycolic acid |
| AMPs | antimicrobial peptides |
| miRNAs | microRNAs |
| MBVs | matrix-bound nanovesicles |
| SCs | stem/progenitor cells |
| FGF | fibroblast growth factor |
| VEGF | vascular endothelial growth factor |
| PDGF | platelet-derived growth factor |
| TGF-β | transforming growth factor-beta |
| FAK | focal adhesion kinase |
| Src | proto-oncogene tyrosine-protein kinase |
| PVA/PLLA | poly(vinyl alcohol)/poly(L-lactic acid) |
| ALP | alkaline phosphatase |
| MSCs | mesenchymal stem cells |
| HCOs | human calvaria osteoblasts |
| GAG | glycosaminoglycan |
| ROS/RNS | reactive oxygen and nitrogen species |
| MACI | matrix-induced autologous chondrocyte implantation |
References
- Naik, R.R.; Singamaneni, S. Introduction: Bioinspired and Biomimetic Materials. Chem. Rev. 2017, 117, 12581–12583. [Google Scholar] [CrossRef]
- Tajurahim, N.A.N.; Mahmood, S.; Ngadiman, N.H.A.; Sing, S.L. Biomaterials for tissue engineering scaffolds: Balancing efficiency and eco-friendliness through life cycle assessment. Clean. Environ. Syst. 2025, 16, 100253. [Google Scholar] [CrossRef]
- Tabata, Y. Biomaterial technology for tissue engineering applications. J. R. Soc. Interface 2009, 6, S311–S324. [Google Scholar] [CrossRef]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, N.; Bagheri, S. The influence of topography on tissue engineering perspective. Mater. Sci. Eng. C 2016, 61, 906–921. [Google Scholar] [CrossRef] [PubMed]
- Norman, J.J.; Desai, T.A. Methods for Fabrication of Nanoscale Topography for Tissue Engineering Scaffolds. Ann. Biomed. Eng. 2006, 34, 89–101. [Google Scholar] [CrossRef]
- Burra, P.; Arcidiacono, D.; Bizzaro, D.; Chioato, T.; Di Liddo, R.; Banerjee, A.; Cappon, A.; Bo, P.; Conconi, M.T.; Parnigotto, P.P.; et al. Systemic administration of a novel human umbilical cord mesenchymal stem cells population accelerates the resolution of acute liver injury. BMC Gastroenterol. 2012, 12, 88. [Google Scholar] [CrossRef]
- Kitahara, S.; Nakagawa, K.; Sah, R.L.; Wada, Y.; Ogawa, T.; Moriya, H.; Masuda, K. In Vivo Maturation of Scaffold-free Engineered Articular Cartilage on Hydroxyapatite. Tissue Eng. Part A 2008, 14, 1905–1913. [Google Scholar] [CrossRef] [PubMed]
- Dahl, S.L.M.; Koh, J.; Prabhakar, V.; Niklason, L.E. Decellularized Native and Engineered Arterial Scaffolds for Transplantation. Cell Transplant. 2003, 12, 659–666. [Google Scholar] [CrossRef]
- Petersen, T.H.; Calle, E.A.; Colehour, M.B.; Niklason, L.E. Matrix Composition and Mechanics of Decellularized Lung Scaffolds. Cells Tissues Organs 2012, 195, 222–231. [Google Scholar] [CrossRef]
- Bakhshandeh, B.; Zarrintaj, P.; Oftadeh, M.O.; Keramati, F.; Fouladiha, H.; Sohrabi-Jahromi, S.; Ziraksaz, Z. Tissue engineering; strategies, tissues, and biomaterials. Biotechnol. Genet. Eng. Rev. 2017, 33, 144–172. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.M.; Haugh, M.G.; O’Brien, F.J. The effect of mean pore size on cell attachment, proliferation and migration in collagen–glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials 2010, 31, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Mukasheva, F.; Adilova, L.; Dyussenbinov, A.; Yernaimanova, B.; Abilev, M.; Akilbekova, D. Optimizing scaffold pore size for tissue engineering: Insights across various tissue types. Front. Bioeng. Biotechnol. 2024, 12, 1444986. [Google Scholar] [CrossRef] [PubMed]
- Giuseppe Longo, U.; Loppini, M.; Berton, A.; La Verde, L.; Khan, W.S.; Denaro, V. Stem Cells From Umbilical Cord and Placenta for Musculoskeletal Tissue Engineering. Curr. Stem Cell Res. Ther. 2012, 7, 272–281. [Google Scholar] [CrossRef]
- Petsche Connell, J.; Camci-Unal, G.; Khademhosseini, A.; Jacot, J.G. Amniotic Fluid-Derived Stem Cells for Cardiovascular Tissue Engineering Applications. Tissue Eng. Part B Rev. 2013, 19, 368–379. [Google Scholar] [CrossRef]
- Jin, J.; Wang, J.; Huang, J.; Huang, F.; Fu, J.; Yang, X.; Miao, Z. Transplantation of human placenta-derived mesenchymal stem cells in a silk fibroin/hydroxyapatite scaffold improves bone repair in rabbits. J. Biosci. Bioeng. 2014, 118, 593–598. [Google Scholar] [CrossRef]
- Gaggi, G.; Di Credico, A.; Izzicupo, P.; Sancilio, S.; Di Mauro, M.; Iannetti, G.; Dolci, S.; Amabile, G.; Di Baldassarre, A.; Ghinassi, B. Decellularized Extracellular Matrices and Cardiac Differentiation: Study on Human Amniotic Fluid-Stem Cells. Int. J. Mol. Sci. 2020, 21, 6317. [Google Scholar] [CrossRef]
- Gaggi, G.; Izzicupo, P.; Di Credico, A.; Sancilio, S.; Di Baldassarre, A.; Ghinassi, B. Spare Parts from Discarded Materials: Fetal Annexes in Regenerative Medicine. Int. J. Mol. Sci. 2019, 20, 1573. [Google Scholar] [CrossRef]
- Lanza, R.P.; Chung, H.Y.; Yoo, J.J.; Wettstein, P.J.; Blackwell, C.; Borson, N.; Hofmeister, E.; Schuch, G.; Soker, S.; Moraes, C.T.; et al. Generation of histocompatible tissues using nuclear transplantation. Nat. Biotechnol. 2002, 20, 689–696. [Google Scholar] [CrossRef]
- Ross, E.A.; Williams, M.J.; Hamazaki, T.; Terada, N.; Clapp, W.L.; Adin, C.; Ellison, G.W.; Jorgensen, M.; Batich, C.D. Embryonic Stem Cells Proliferate and Differentiate when Seeded into Kidney Scaffolds. J. Am. Soc. Nephrol. 2009, 20, 2338–2347. [Google Scholar] [CrossRef]
- Goto, Y.; Noguchi, Y.; Nomura, A.; Sakamoto, T.; Ishii, Y.; Bitoh, S.; Picton, C.; Fujita, Y.; Watanabe, T.; Hasegawa, S.; et al. In Vitro Reconstitution of the Tracheal Epithelium. Am. J. Respir. Cell Mol. Biol. 1999, 20, 312–318. [Google Scholar] [CrossRef]
- Gaharwar, A.K.; Singh, I.; Khademhosseini, A. Engineered biomaterials for in situ tissue regeneration. Nat. Rev. Mater. 2020, 5, 686–705. [Google Scholar] [CrossRef]
- Percival, K.M.; Paul, V.; Husseini, G.A. Recent Advancements in Bone Tissue Engineering: Integrating Smart Scaffold Technologies and Bio-Responsive Systems for Enhanced Regeneration. Int. J. Mol. Sci. 2024, 25, 6012. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.; Pham, K.T.; Prince, R.C.; Leiser-Miller, L.B.; Prasad, M.S.; Wang, X.; Nordberg, R.C.; Bielajew, B.J.; Hu, J.C.; Yamaga, K.; et al. Superstable lipid vacuoles endow cartilage with its shape and biomechanics. Science 2025, 387, eads9960. [Google Scholar] [CrossRef]
- Biniazan, F.; Stoian, A.; Haykal, S. Adipose-Derived Stem Cells: Angiogenetic Potential and Utility in Tissue Engineering. Int. J. Mol. Sci. 2024, 25, 2356. [Google Scholar] [CrossRef]
- Zhao, Z.; Yu, H.; Fan, C.; Kong, Q.; Liu, D.; Meng, L. Differentiate into urothelium and smooth muscle cells from adipose tissue-derived stem cells for ureter reconstruction in a rabbit model. Am. J. Transl. Res. 2016, 8, 3757–3768. [Google Scholar] [PubMed]
- Heljak, M.K.; Chlanda, A.; Swieszkowski, W.; Bil, M. Multiscale analysis of viscoelastic properties, topography and internal structure of a biodegradable thermo-responsive shape memory polyurethane. Polymer 2020, 191, 122273. [Google Scholar] [CrossRef]
- Vert, M.; Doi, Y.; Hellwich, K.-H.; Hess, M.; Hodge, P.; Kubisa, P.; Rinaudo, M.; Schué, F. Terminology for biorelated polymers and applications (IUPAC Recommendations 2012). Pure Appl. Chem. 2012, 84, 377–410. [Google Scholar] [CrossRef]
- Kim, H.S.; Kumbar, S.G.; Nukavarapu, S.P. Biomaterial-directed cell behavior for tissue engineering. Curr. Opin. Biomed. Eng. 2021, 17, 100260. [Google Scholar] [CrossRef]
- Igwe, J.; Amini, A.; Mikael, P.; Laurencin, C.; Nukavarapu, S. Nanostructured Scaffolds for Bone Tissue Engineering; Springer: Berlin/Heidelberg, Germany, 2011; pp. 169–192. [Google Scholar]
- Seeherman, H.; Wozney, J.; Li, R. Bone Morphogenetic Protein Delivery Systems. Spine 2002, 27, S16–S23. [Google Scholar] [CrossRef]
- Jaidev, L.; Chatterjee, K. Surface functionalization of 3D printed polymer scaffolds to augment stem cell response. Mater. Des. 2019, 161, 44–54. [Google Scholar] [CrossRef]
- Ouberai, M.M.; Xu, K.; Welland, M.E. Effect of the interplay between protein and surface on the properties of adsorbed protein layers. Biomaterials 2014, 35, 6157–6163. [Google Scholar] [CrossRef]
- Samir, A.; Ashour, F.H.; Hakim, A.A.A.; Bassyouni, M. Recent advances in biodegradable polymers for sustainable applications. npj Mater. Degrad. 2022, 6, 68. [Google Scholar] [CrossRef]
- Mary, S.M.S.; Raja, S.; Kalkura, S.N.; Babu, S.M. Tunable physicochemical and biocompatibility properties of PCL/HAp/BN composite films through γ-irradiation. Polym. Bull. 2025, 82, 10319–10340. [Google Scholar] [CrossRef]
- Liu, S.; Yu, J.-M.; Gan, Y.-C.; Qiu, X.-Z.; Gao, Z.-C.; Wang, H.; Chen, S.-X.; Xiong, Y.; Liu, G.-H.; Lin, S.-E.; et al. Biomimetic natural biomaterials for tissue engineering and regenerative medicine: New biosynthesis methods, recent advances, and emerging applications. Mil. Med. Res. 2023, 10, 16. [Google Scholar] [CrossRef]
- Ullah, S.; Chen, X. Fabrication, applications and challenges of natural biomaterials in tissue engineering. Appl. Mater. Today 2020, 20, 100656. [Google Scholar] [CrossRef]
- Costa, A.; Naranjo, J.D.; Londono, R.; Badylak, S.F. Biologic Scaffolds. Cold Spring Harb. Perspect. Med. 2017, 7, a025676. [Google Scholar] [CrossRef]
- Huang, G.; Li, F.; Zhao, X.; Ma, Y.; Li, Y.; Lin, M.; Jin, G.; Lu, T.J.; Genin, G.M.; Xu, F. Functional and Biomimetic Materials for Engineering of the Three-Dimensional Cell Microenvironment. Chem. Rev. 2017, 117, 12764–12850. [Google Scholar] [CrossRef] [PubMed]
- Tidu, A.; Ghoubay-Benallaoua, D.; Teulon, C.; Asnacios, S.; Grieve, K.; Portier, F.; Schanne-Klein, M.-C.; Borderie, V.; Mosser, G. Highly concentrated collagen solutions leading to transparent scaffolds of controlled three-dimensional organizations for corneal epithelial cell colonization. Biomater. Sci. 2018, 6, 1492–1502. [Google Scholar] [CrossRef]
- Akiyama, S.K. Integrins in cell adhesion and signaling. Hum. Cell 1996, 9, 181–186. [Google Scholar] [PubMed]
- Zhou, C.; Shi, Q.; Guo, W.; Terrell, L.; Qureshi, A.T.; Hayes, D.J.; Wu, Q. Electrospun Bio-Nanocomposite Scaffolds for Bone Tissue Engineering by Cellulose Nanocrystals Reinforcing Maleic Anhydride Grafted PLA. ACS Appl. Mater. Interfaces 2013, 5, 3847–3854. [Google Scholar] [CrossRef]
- Lin, J.; Li, C.; Zhao, Y.; Hu, J.; Zhang, L.-M. Co-electrospun Nanofibrous Membranes of Collagen and Zein for Wound Healing. ACS Appl. Mater. Interfaces 2012, 4, 1050–1057. [Google Scholar] [CrossRef]
- Crapo, P.M.; Gilbert, T.W.; Badylak, S.F. An overview of tissue and whole organ decellularization processes. Biomaterials 2011, 32, 3233–3243. [Google Scholar] [CrossRef]
- Swinehart, I.T.; Badylak, S.F. Extracellular matrix bioscaffolds in tissue remodeling and morphogenesis. Dev. Dyn. 2016, 245, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Kasper, J.C.; Friess, W. The freezing step in lyophilization: Physico-chemical fundamentals, freezing methods and consequences on process performance and quality attributes of biopharmaceuticals. Eur. J. Pharm. Biopharm. 2011, 78, 248–263. [Google Scholar] [CrossRef]
- Badylak, S.F. Xenogeneic extracellular matrix as a scaffold for tissue reconstruction. Transpl. Immunol. 2004, 12, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Valentin, J.E.; Badylak, J.S.; McCabe, G.P.; Badylak, S.F. Extracellular Matrix Bioscaffolds for Orthopaedic Applications. J. Bone Jt. Surg. 2006, 88, 2673–2686. [Google Scholar] [CrossRef]
- Gilbert, T.W.; Stolz, D.B.; Biancaniello, F.; Simmons-Byrd, A.; Badylak, S.F. Production and characterization of ECM powder: Implications for tissue engineering applications. Biomaterials 2005, 26, 1431–1435. [Google Scholar] [CrossRef]
- Freytes, D.O.; Martin, J.; Velankar, S.S.; Lee, A.S.; Badylak, S.F. Preparation and rheological characterization of a gel form of the porcine urinary bladder matrix. Biomaterials 2008, 29, 1630–1637. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, R.; Mao, X.; Li, X.; Li, T.; Liang, F.; He, J.; Wen, L.; Wang, W.; Li, X.; et al. Preparation of PLCL/ECM nerve conduits by electrostatic spinning technique and evaluation in vitro and in vivo. J. Neural Eng. 2024, 21, 026028. [Google Scholar] [CrossRef] [PubMed]
- Romero, D.J.; Hussey, G.; Capella-Monsonís, H. Immune Response to Extracellular Matrix Bioscaffolds: A Comprehensive Review. Biologics 2025, 5, 28. [Google Scholar] [CrossRef]
- Mayorca-Guiliani, A.E.; Leeming, D.J.; Henriksen, K.; Mortensen, J.H.; Nielsen, S.H.; Anstee, Q.M.; Sanyal, A.J.; Karsdal, M.A.; Schuppan, D. ECM formation and degradation during fibrosis, repair, and regeneration. npj Metab. Health Dis. 2025, 3, 25. [Google Scholar] [CrossRef]
- Naba, A. Mechanisms of assembly and remodelling of the extracellular matrix. Nat. Rev. Mol. Cell Biol. 2024, 25, 865–885. [Google Scholar] [CrossRef]
- Moeendarbary, E.; Harris, A.R. Cell mechanics: Principles, practices, and prospects. Wiley Interdiscip. Rev. Syst. Biol. Med. 2014, 6, 371–388. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.P.; Appel, E.A.; Ashby, P.D.; Baker, B.M.; Franco, E.; Gu, L.; Haynes, K.; Joshi, N.S.; Kloxin, A.M.; Kouwer, P.H.J.; et al. The living interface between synthetic biology and biomaterial design. Nat. Mater. 2022, 21, 390–397. [Google Scholar] [CrossRef]
- Roam, J.L.; Yan, Y.; Nguyen, P.K.; Kinstlinger, I.S.; Leuchter, M.K.; Hunter, D.A.; Wood, M.D.; Elbert, D.L. A modular, plasmin-sensitive, clickable poly(ethylene glycol)-heparin-laminin microsphere system for establishing growth factor gradients in nerve guidance conduits. Biomaterials 2015, 72, 112–124. [Google Scholar] [CrossRef]
- Annabi, N.; Fathi, A.; Mithieux, S.M.; Martens, P.; Weiss, A.S.; Dehghani, F. The effect of elastin on chondrocyte adhesion and proliferation on poly (ɛ-caprolactone)/elastin composites. Biomaterials 2011, 32, 1517–1525. [Google Scholar] [CrossRef]
- Joshi, M.K.; Tiwari, A.P.; Pant, H.R.; Shrestha, B.K.; Kim, H.J.; Park, C.H.; Kim, C.S. In Situ Generation of Cellulose Nanocrystals in Polycaprolactone Nanofibers: Effects on Crystallinity, Mechanical Strength, Biocompatibility, and Biomimetic Mineralization. ACS Appl. Mater. Interfaces 2015, 7, 19672–19683. [Google Scholar] [CrossRef] [PubMed]
- Crago, M.; Tan, R.; Hung, J.; Wise, S.G.; Akhavan, B.; Bilek, M.; Dehghani, F.; Talebian, S.; Naficy, S. Durable plasma-mediated zwitterionic grafting on polymeric surfaces for implantable medical devices. Commun. Mater. 2024, 5, 24. [Google Scholar] [CrossRef]
- Mededovic, M.; Zhong, X.; Kohn, D.H.; Lahann, J. Localized Gene Delivery and Enhanced Cell–Cell Communication via Bio-orthogonal Polymer Coatings. Regen. Eng. Transl. Med. 2025, 11, 1038–1051. [Google Scholar] [CrossRef]
- Zhou, W.; Qiao, Z.; Zare, E.N.; Huang, J.; Zheng, X.; Sun, X.; Shao, M.; Wang, H.; Wang, X.; Chen, D.; et al. 4D-Printed Dynamic Materials in Biomedical Applications: Chemistry, Challenges, and Their Future Perspectives in the Clinical Sector. J. Med. Chem. 2020, 63, 8003–8024. [Google Scholar] [CrossRef]
- Miao, S.; Zhu, W.; Castro, N.J.; Leng, J.; Zhang, L.G. Four-Dimensional Printing Hierarchy Scaffolds with Highly Biocompatible Smart Polymers for Tissue Engineering Applications. Tissue Eng. Part C Methods 2016, 22, 952–963. [Google Scholar] [CrossRef] [PubMed]
- Zarek, M.; Mansour, N.; Shapira, S.; Cohn, D. 4D Printing of Shape Memory-Based Personalized Endoluminal Medical Devices. Macromol. Rapid Commun. 2017, 38, 1600628. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Zainol, I.; Chowdhury, S.R.; Fauzi, M. Development of various composition multicomponent chitosan/fish collagen/glycerin 3D porous scaffolds: Effect on morphology, mechanical strength, biostability and cytocompatibility. Int. J. Biol. Macromol. 2018, 111, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Caliari, S.R.; Harley, B.A. The effect of anisotropic collagen-GAG scaffolds and growth factor supplementation on tendon cell recruitment, alignment, and metabolic activity. Biomaterials 2011, 32, 5330–5340. [Google Scholar] [CrossRef]
- Minardi, S.; Taraballi, F.; Wang, X.; Cabrera, F.J.; Van Eps, J.L.; Robbins, A.B.; Sandri, M.; Moreno, M.R.; Weiner, B.K.; Tasciotti, E. Biomimetic collagen/elastin meshes for ventral hernia repair in a rat model. Acta Biomater. 2017, 50, 165–177. [Google Scholar] [CrossRef]
- Shi, W.; Sun, M.; Hu, X.; Ren, B.; Cheng, J.; Li, C.; Duan, X.; Fu, X.; Zhang, J.; Chen, H.; et al. Structurally and Functionally Optimized Silk-Fibroin–Gelatin Scaffold Using 3D Printing to Repair Cartilage Injury In Vitro and In Vivo. Adv. Mater. 2017, 29, 1701089. [Google Scholar] [CrossRef]
- Hu, T.; Fang, J.; Shen, Y.; Li, M.; Wang, B.; Xu, Z.; Hu, W. Advances of naturally derived biomedical polymers in tissue engineering. Front. Chem. 2024, 12, 1469183. [Google Scholar] [CrossRef]
- Tabares, F.L.; Junkar, I. Cold Plasma Systems and Their Application in Surface Treatments for Medicine. Molecules 2021, 26, 1903. [Google Scholar] [CrossRef]
- Kumar, P. Plasma surface modification of biomaterials: Developing plasma-treated implants and prosthetics with improved biocompatibility. Int. J. Phys. Appl. 2025, 7, 266–275. [Google Scholar] [CrossRef]
- Yang, Q.; Zhao, W.; Yang, L.; Fan, Y.; Shao, C.; Wang, T.; Zhang, F. The Cold Atmospheric Plasma Inhibits Cancer Proliferation Through Reducing Glutathione Synthesis. Molecules 2025, 30, 2808. [Google Scholar] [CrossRef]
- Rifathin, A.; Wijaya, A.M.; Roziafanto, A.N.; Laksmono, J.A.; Nugraha, A.F.; Chalid, M. Cold Plasma-Induced Surface Modification of Microfibrillated Cellulose Using Lauric Acid to Enhance Compatibility in Polymer Composites. Indones. J. Chem. 2025, 25, 1167–1178. [Google Scholar] [CrossRef]
- Moradi, Y.; Ghiassadin, A.; Bakhshi, H.; Irani, S.; Atyabi, S.M.; Dadgar, N. Cold Atmosphere Plasma Modification on Beta-Carotene-Loaded Nanofibers to Enhance Osteogenic Differentiation. Fibers Polym. 2022, 23, 18–27. [Google Scholar] [CrossRef]
- Eggers, B.; Wagenheim, A.-M.; Jung, S.; Kleinheinz, J.; Nokhbehsaim, M.; Kramer, F.-J.; Sielker, S. Effect of Cold Atmospheric Plasma (CAP) on Osteogenic Differentiation Potential of Human Osteoblasts. Int. J. Mol. Sci. 2022, 23, 2503. [Google Scholar] [CrossRef]
- Wang, M.; Favi, P.; Cheng, X.; Golshan, N.H.; Ziemer, K.S.; Keidar, M.; Webster, T.J. Cold atmospheric plasma (CAP) surface nanomodified 3D printed polylactic acid (PLA) scaffolds for bone regeneration. Acta Biomater. 2016, 46, 256–265. [Google Scholar] [CrossRef]
- Zhu, W.; Castro, N.J.; Cheng, X.; Keidar, M.; Zhang, L.G. Cold Atmospheric Plasma Modified Electrospun Scaffolds with Embedded Microspheres for Improved Cartilage Regeneration. PLoS ONE 2015, 10, e0134729. [Google Scholar] [CrossRef]
- Zhai, S.-Y.; Kong, M.G.; Xia, Y.-M. Cold Atmospheric Plasma Ameliorates Skin Diseases Involving Reactive Oxygen/Nitrogen Species-Mediated Functions. Front. Immunol. 2022, 13, 868386. [Google Scholar] [CrossRef] [PubMed]
- Chiper, A.S.; Borcia, G. Stable Surface Modification by Cold Atmospheric-Pressure Plasma: Comparative Study on Cellulose-Based and Synthetic Polymers. Polymers 2023, 15, 4172. [Google Scholar] [CrossRef]
- Busco, G.; Robert, E.; Chettouh-Hammas, N.; Pouvesle, J.-M.; Grillon, C. The emerging potential of cold atmospheric plasma in skin biology. Free Radic. Biol. Med. 2020, 161, 290–304. [Google Scholar] [CrossRef] [PubMed]
- Luan, P.; Oehrlein, G.S. Interaction of long-lived reactive species from cold atmospheric pressure plasma with polymers: Chemical modification by ozone and reactive oxygen-nitrogen species. J. Vac. Sci. Technol. A 2019, 37, 051303. [Google Scholar] [CrossRef]
- Gokcekuyu, Y.; Ekinci, F.; Guzel, M.S.; Acici, K.; Aydin, S.; Asuroglu, T. Artificial Intelligence in Biomaterials: A Comprehensive Review. Appl. Sci. 2024, 14, 6590. [Google Scholar] [CrossRef]
- Bagherpour, R.; Bagherpour, G.; Mohammadi, P. Application of Artificial Intelligence in Tissue Engineering. Tissue Eng. Part B Rev. 2025, 31, 31–43. [Google Scholar] [CrossRef]
- Ercan, U.K.; Özdemir, G.D.; Özdemir, M.A.; Güren, O. Plasma medicine: The era of artificial intelligence. Plasma Process. Polym. 2023, 20, e2300066. [Google Scholar] [CrossRef]
- Jeznach, O.; Tabakoglu, S.; Zaszczyńska, A.; Sajkiewicz, P. Review on machine learning application in tissue engineering: What has been done so far? Application areas, challenges, and perspectives. J. Mater. Sci. 2024, 59, 21222–21250. [Google Scholar] [CrossRef]
- Lin, L.; Wang, Q.; Hou, Z.; Keidar, M. Low-temperature plasma adaptation in the course of machine learning controls of plasma medicine. Phys. Plasmas 2025, 32, 093509. [Google Scholar] [CrossRef]
- Wu, Y.; Ding, X.; Wang, Y.; Ouyang, D. Harnessing the power of machine learning into tissue engineering: Current progress and future prospects. Burn. Trauma 2024, 12, tkae053. [Google Scholar] [CrossRef]
| Tissue | Pore Size (µm) | Function |
|---|---|---|
| Skin | 100–200 [65] | Promotes fibroblast migration and neovascularization |
| Bone | 200–500 [13] | Enhances osteoblast infiltration and bone formation |
| Cartilage | 250–350 [68] | Supports chondrocyte distribution and ECM deposition |
| Tendon | 20–50 [66] | Guides directional cell growth |
| Heart (Myocardium) | 50–100 [13] | Improves cardiomyocyte viability and oxygen diffusion |
| Endothelium | 30–40 [67] | Limits cellular infiltration during inflammation |
| Study | Description | Applications |
|---|---|---|
| Percival et al. [23] | Intelligent scaffolds that dynamically respond to biological signals, optimizing the environment for bone growth. | Bone reconstruction; treatment of fractures and bone diseases |
| Hu et al. [69] | Self-healing hydrogels mimicking the natural extracellular matrix, capable of regenerating after damage. | Implantable medical devices |
| Ramos et al. [24] | Discovery of lipo-chondrocytes, cells with both adipose and cartilage characteristics. | Cartilage transplants; tissue integration |
| Crago et al. [60] | Plasma-mediated zwitterionic grafting on polymer surfaces, improving biocompatibility and thrombogenic resistance. | Long-term implantable devices |
| Gaharwar et al. [22] | Plasma treatment introduces reactive functional groups for anchoring bioactive molecules without altering mechanical properties. | Smart implants; promotion of cell adhesion, proliferation, and differentiation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Del Gaudio, L.; Lattanzio, S.; Di Pietro, R.; Sancilio, S. Advances in Biomaterials for Tissue Regeneration: From Scaffold Design to CAP-Enabled Interfaces and AI-Driven Optimization. Biomimetics 2026, 11, 330. https://doi.org/10.3390/biomimetics11050330
Del Gaudio L, Lattanzio S, Di Pietro R, Sancilio S. Advances in Biomaterials for Tissue Regeneration: From Scaffold Design to CAP-Enabled Interfaces and AI-Driven Optimization. Biomimetics. 2026; 11(5):330. https://doi.org/10.3390/biomimetics11050330
Chicago/Turabian StyleDel Gaudio, Laura, Stefano Lattanzio, Roberta Di Pietro, and Silvia Sancilio. 2026. "Advances in Biomaterials for Tissue Regeneration: From Scaffold Design to CAP-Enabled Interfaces and AI-Driven Optimization" Biomimetics 11, no. 5: 330. https://doi.org/10.3390/biomimetics11050330
APA StyleDel Gaudio, L., Lattanzio, S., Di Pietro, R., & Sancilio, S. (2026). Advances in Biomaterials for Tissue Regeneration: From Scaffold Design to CAP-Enabled Interfaces and AI-Driven Optimization. Biomimetics, 11(5), 330. https://doi.org/10.3390/biomimetics11050330