Contact Dermatitis of the Vulva
Abstract
:1. Introduction
2. Purpose and Methods
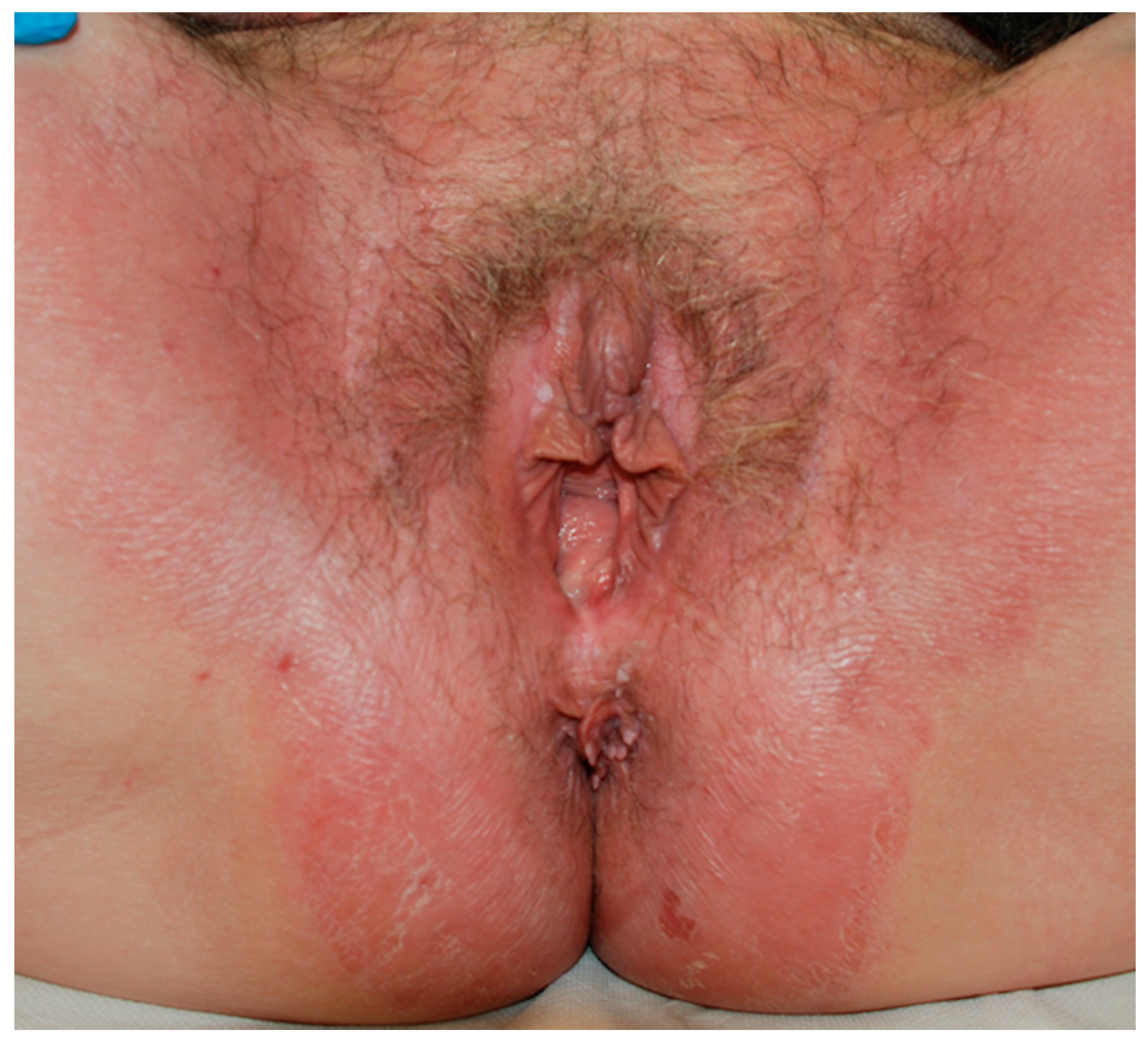
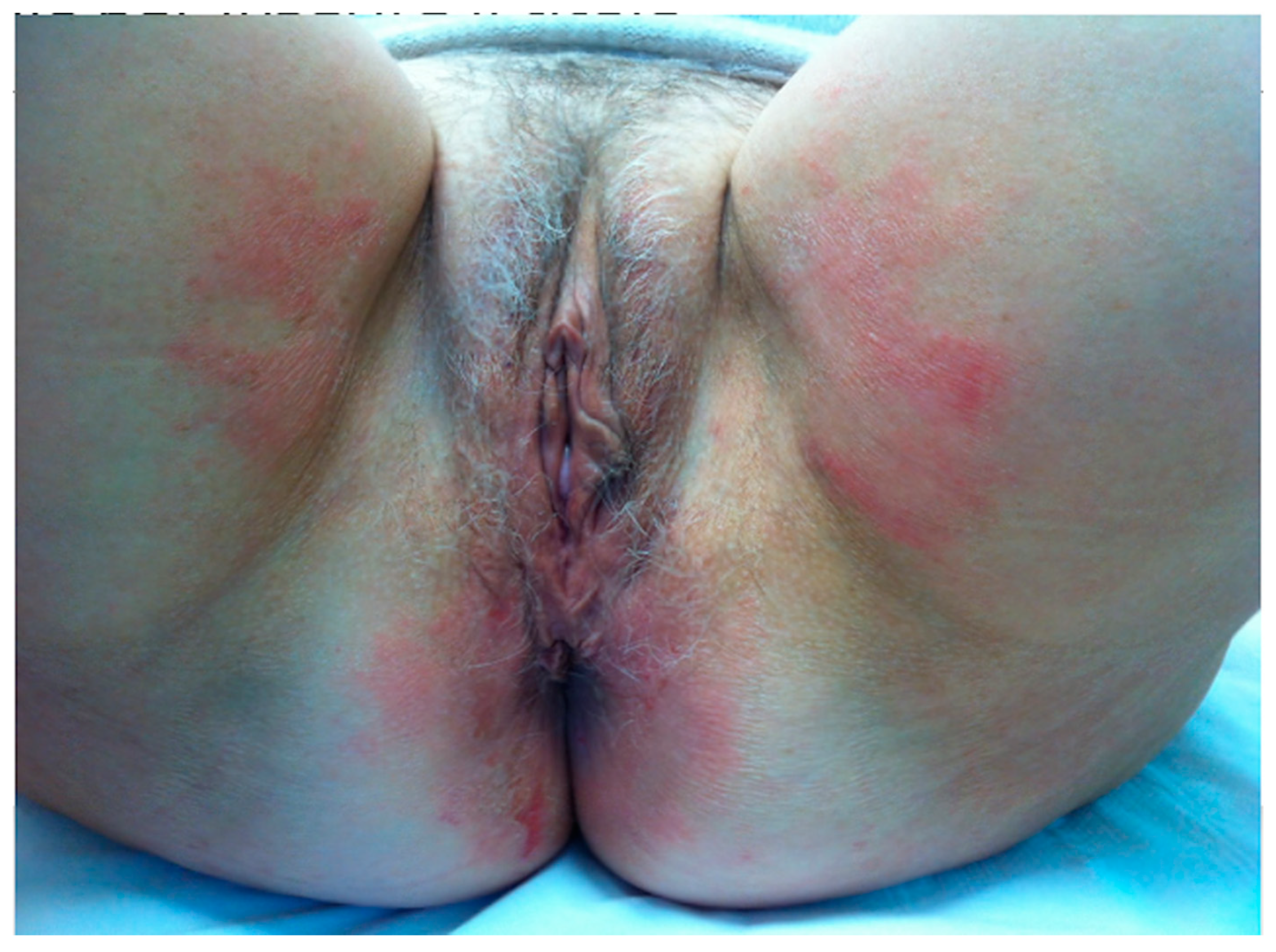
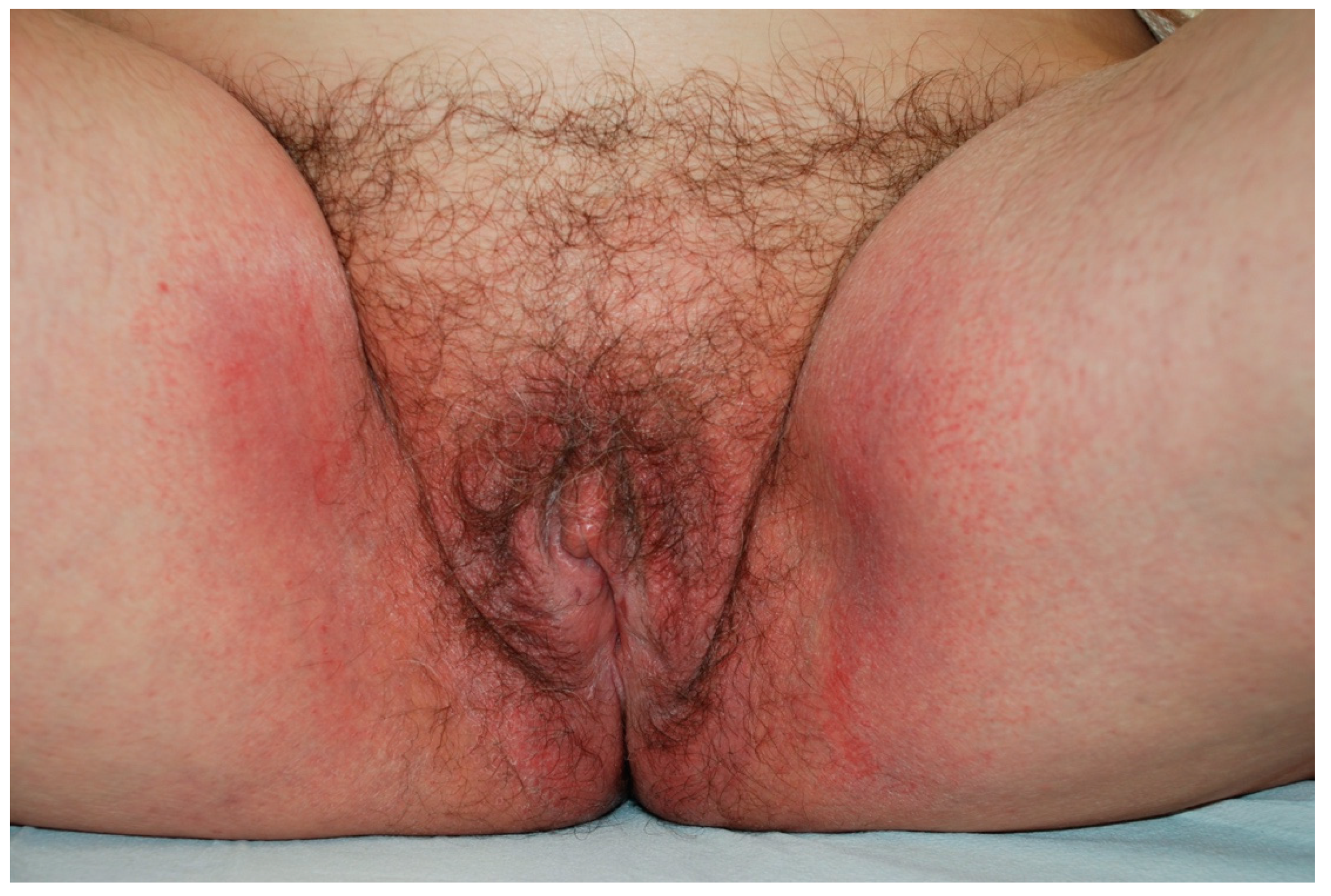
3. Irritant Contact Dermatitis
4. Allergic Contact Dermatitis
4.1. Sensitizers
4.1.1. Fragrances
4.1.2. Preservatives
4.1.3. Medicaments
4.1.4. Botanical Products
5. Conclusions
Funding
Conflicts of Interest
References
- Farage, M.A. Vulvar susceptibility to contact irritants and allergens: A review. Arch. Gynecol. Obstet. 2005, 272, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Bains, S.N.; Nash, P. Irritant Contact Dermatitis. Clin. Rev. Allergy Immunol. 2019, 56, 99–109. [Google Scholar] [CrossRef]
- Nosbaum, A.; Vocanson, M.; Rozieres, A.; Hennino, A.; Nicolas, J.F. Allergic and irritant contact dermatitis. Eur. J. Dermatol. EJD 2009, 19, 325–332. [Google Scholar] [CrossRef]
- Davis, M.D.P. Unusual patterns in contact dermatitis: Medicaments. Dermatol. Clin. 2009, 27, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Marren, P.; Wojnarowska, F.; Powell, S. Allergic contact dermatitis and vulvar dermatoses. Br. J. Dermatol. 1992, 126, 52–56. [Google Scholar] [CrossRef]
- Brenan, J.A.; Dennerstein, G.J.; Sfameni, S.F.; Drinkwater, P.; Marin, G.; Scurry, J.P. Evaluation of patch testing in patients with chronic vulvar symptoms. Australas. J. Dermatol. 1996, 37, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, P.C.; Rycroft, R.J.G.; White, I.R.; Ridley, C.M.; Neill, S.M.; McFadden, J.P. Contact sensitivity in women with anogenital dermatoses. Contact Dermat. 1997, 36, 174–175. [Google Scholar] [CrossRef]
- Lewis, F.M.; Harrington, C.I.; Gawkrodger, D.J. Contact sensitivity in pruritus vulvae: A common and manageable problem. Contact Dermat. 1994, 31, 264–265. [Google Scholar] [CrossRef]
- Lucke, T.W.; Fleming, C.J.; McHenry, P.; Lever, R. Patch testing in vulval dermatoses: How relevant is nickel? Contact Dermat. 1998, 38, 111–112. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.; Geier, J.; Elsner, P. Allergic contact dermatitis in patients with anogenital complaints. J. Reprod. Med. 2000, 45, 649–654. [Google Scholar]
- Crone, A.M.; Stewart, E.; Wojnarowska, F.; Powell, S.M. Aetiological factors in vulvar dermatitis. J. Eur. Acad. Dermatol. Venereol. JEADV 2000, 14, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Utaş, S.; Ferahbaş, A.; Yildiz, S. Patients with vulval pruritus: Patch test results. Contact Dermat. 2008, 58, 296–298. [Google Scholar] [CrossRef]
- Vermaat, H.; Van Meurs, T.; Rustemeyer, T.; Bruynzeel, D.P.; Kirtschig, G. Vulval allergic contact dermatitis due to peppermint oil in herbal tea. Contact Dermat. 2008, 58, 364–365. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, S.M.; Torgerson, R.R. Allergic contact dermatitis of the vulva. Dermat. Contact Atopic Occup. Drug 2013, 24, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Al-Niaimi, F.; Felton, S.; Williams, J. Patch testing for vulval symptoms: Our experience with 282 patients. Clin. Exp. Dermatol. 2014, 39, 439–442. [Google Scholar] [CrossRef]
- Trivedi, M.K.; Woodruff, C.M.; Kornik, R.; Botto, N. Patch Testing in Vulvar Allergic Contact Dermatitis. Dermat. Contact Atopic Occup. Drug 2018, 29, 95–96. [Google Scholar] [CrossRef] [PubMed]
- Nardelli, A.; Degreef, H.; Goossens, A. Contact allergic reactions of the vulva: A 14-year review. Dermat. Contact Atopic Occup. Drug 2004, 15, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Warshaw, E.M.; Furda, L.M.; Maibach, H.I.; Rietschel, R.L.; Fowler, J.F., Jr.; Belsito, D.V.; Zug, K.A.; DeLeo, V.A.; Marks, J.G., Jr.; Toby Mathias, C.G.; et al. Anogenital dermatitis in patients referred for patch testing: Retrospective analysis of cross-sectional data from the North American Contact Dermatitis Group, 1994–2004. Arch. Dermatol. 2008, 144, 749–755. [Google Scholar] [CrossRef]
- Haverhoek, E.; Reid, C.; Gordon, L.; Marshman, G.; Wood, J.; Selva-Nayagam, P. Prospective study of patch testing in patients with vulval pruritus. Australas. J. Dermatol. 2008, 49, 80–85. [Google Scholar] [CrossRef]
- Virgili, A.; Bacilieri, S.; Corazza, M. Evaluation of contact sensitization in vulvar lichen simplex chronicus. A proposal for a battery of selected allergens. J. Reprod. Med. 2003, 48, 33–36. [Google Scholar]
- Woodruff, C.M.; Trivedi, M.K.; Botto, N.; Kornik, R. Allergic Contact Dermatitis of the Vulva. Dermat. Contact Atopic Occup. Drug 2018, 29, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Yale, K.; Awosika, O.; Pardo, M.R.; Ehrlich, A. Genital Allergic Contact Dermatitis. Dermat. Contact Atopic Occup. Drug 2018, 29, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Bhate, K.; Landeck, L.; Gonzalez, E.; Neumann, K.; Schalock, P. Genital contact dermatitis: A retrospective analysis. Dermat. Contact Atopic Occup. Drug 2010, 21, 317–320. [Google Scholar] [CrossRef]
- Ljubojević, S.; Lipozencić, J.; Celić, D.; Turcić, P. Genital contact allergy. Acta Dermatovenerol. Croat. ADC 2009, 17, 285–288. [Google Scholar] [PubMed]
- Gilissen, L.; Schollaert, I.; Huygens, S.; Goossens, A. Iatrogenic allergic contact dermatitis in the (peri)anal and genital area. Contact Dermat. 2021, 84, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, A.; De Hertog, S.; Gladys, K.; Vanacker, H.; Goossens, A. ‘Dermatologically tested’ baby toilet tissues: A cause of allergic contact dermatitis in adults. Contact Dermat. 2007, 57, 97–99. [Google Scholar] [CrossRef] [PubMed]
- Tosti, A.; Voudouris, S.; Pazzaglia, M. Contact sensitization to 5-chloro-2-methyl-4-isothiazolin-3-one and 2-methyl-4-isothiazolin-3-one in children. Contact Dermat. 2003, 49, 215–216. [Google Scholar] [CrossRef] [PubMed]
- Fields, K.S.; Nelson, T.; Powell, U. Contact dermatitis caused by baby wipes. J. Am. Acad. Dermatol. 2006, 54, S230–S232. [Google Scholar] [CrossRef]
- Gardner, K.H.; Davis, M.D.P.; Richardson, D.M.; Pittelkow, M.R. The hazards of moist toilet paper: Allergy to the preservative methylchloroisothiazolinone/methylisothiazolinone. Arch. Dermatol. 2010, 146, 886–890. [Google Scholar] [CrossRef] [Green Version]
- Foote, C.A.; Brady, S.P.; Brady, K.L.; Clark, N.S.; Mercurio, M.G. Vulvar dermatitis from allergy to moist flushable wipes. J. Low. Genit. Tract Dis. 2014, 18, E16–E18. [Google Scholar] [CrossRef] [PubMed]
- Monroe, H.R.; Hu, J.C.; Chiu, M.W. Methylchloroisothiazolinone / methylisothiazolinone and moist wipe dermatitis. Dermatol. Online J. 2010, 16, 14. [Google Scholar] [PubMed]
- Arriaga-Gomez, E.; Arriaga-Gomez, E.; Kline, J.; Emanuel, E.; Neamonitaki, N.; Yangdon, T.; Zacheis, H.; Pasha, D.; Lim, J.; Bush, S.; et al. Repeated Vaginal Exposures to the Common Cosmetic and Household Preservative Methylisothiazolinone Induce Persistent, Mast Cell-Dependent Genital Pain in ND4 Mice. Int. J. Mol. Sci. 2019, 20, 5361. [Google Scholar] [CrossRef] [Green Version]
- Schlosser, B.J. Contact dermatitis of the vulva. Dermatol. Clin. 2010, 28, 697–706. [Google Scholar] [CrossRef]
- Margesson, L.J. Contact dermatitis of the vulva. Dermatol. Ther. 2004, 17, 20–27. [Google Scholar] [CrossRef]
- Corazza, M.; Scuderi, V.; Toni, G.; Forconi, R.; Zedde, P.; Borghi, A. Severe vulvovaginal allergic contact dermatitis due to clotrimazole contained in multiple topical products. Contact Dermat. 2020, 82, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, V.; Mehta, K.; Chauhan, P.; Abhinav, C. Allergic contact dermatitis due to clotrimazole with cross-reaction to miconazole. Indian J. Dermatol. Venereol. Leprol. 2015, 81, 80–82. [Google Scholar] [CrossRef]
- Wasilewski, C. Allergic contact dermatitis from nystatin. Arch. Dermatol. 1971, 104, 437. [Google Scholar] [CrossRef]
- Corazza, M.; Borghi, A.; Lauriola, M.M.; Virgili, A. Use of topical herbal remedies and cosmetics: A questionnaire-based investigation in dermatology out-patients. J. Eur. Acad. Dermatol. Venereol. JEADV 2009, 23, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Corazza, M.; Virgili, A.; Toni, G.; Minghetti, S.; Tiengo, S.; Borghi, A. Level of use and safety of botanical products for itching vulvar dermatoses. Are patch tests useful? Contact Dermat. 2016, 74, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, S.; Reeder, M. Botanicals in Dermatology: Essential Oils, Botanical Allergens, and Current Regulatory Practices. Dermat. Contact Atopic Occup. Drug 2016, 27, 317–324. [Google Scholar] [CrossRef]
- Kiken, D.A.; Cohen, D.E. Contact dermatitis to botanical extracts. Am. J. Contact Dermat. Off. J. Am. Contact Dermat. Soc. 2002, 13, 148–152. [Google Scholar]
- de Groot, A.C.; Schmidt, E. Essential Oils, Part IV: Contact Allergy. Dermat. Contact Atopic Occup. Drug 2016, 27, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.L.; Law, S.V.; Storrs, F.J. Prevalence of botanical extract allergy in patients with contact dermatitis. Dermat. Contact Atopic Occup. Drug 2004, 15, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Romita, P.; Foti, C.; Gullo, G.; Ambrogio, F.; DiMauro, D.; Calogiuri, G. Genital allergic contact dermatitis in response to contact to thyme in a sanitary pad. Contact Dermat. 2020, 82, 169–170. [Google Scholar] [CrossRef]
- Corazza, M.; Borghi, A.; Gallo, R.; Schena, D.; Pigatto, P.; Lauriola, M.M.; Guarneri, F.; Stingeni, L.; Vincenzi, C.; Foti, C.; et al. Topical botanically derived products: Use, skin reactions, and usefulness of patch tests. A multicentre Italian study. Contact Dermat. 2014, 70, 90–97. [Google Scholar] [CrossRef] [PubMed]

| Vulvar Irritants | |
|---|---|
| Strong Irritants | Weak Irritants |
| Imiquimod | Friction/rubbing |
| Trichloroacetic acid | Urine/feces/vaginal secretions/sweat/semen/saliva |
| Podophyllin | Sanitary napkins |
| 5-fluorouracil | Soaps/detergents |
| Antiseptic or scented wipes | |
| Sodium hypochlorite | Deodorants |
| Authors | Pathologies | N° Patients | % Positiveness | % Relevant p.t. | Allergens | |
|---|---|---|---|---|---|---|
| Doherty et al. | 1990 | Vulvar itching | 50 | 78 | - | nickel, fragrances, neomycin, local anesthetics |
| Marren et al. | 1992 | Vulvar dermatoses | 135 | 47 | 29 | nickel, fragrances, preservatives, ethylenediamine, topical medicaments |
| Brenan et al. | 1996 | Chronic vulvar symptoms | 700 | 42 | - | nickel, fragrances, ethylenediamine |
| Goldsmith et al. | 1997 | Anogenital dermatoses | 201 | 39 | 28 | antibiotics, local anesthetics, fragrances, corticosteroids |
| Lewis et al. | 1997 | Vulvar symptoms | 121 | 58.7 | 49 | local anesthetics, fragrances, neomycin |
| Lucke et al. | 1998 | Vulvar dermatoses | 55 | 65 | - | nickel, fragrances, medicaments, dyes |
| Bauer et al. | 2000 | Anogenital symptoms | 351 | 47 | 34.8 | nickel, fragrances, local anesthetics |
| Crone et al. | 2000 | Vulvar dermatoses | 38 | 47 | 28 | fragrances, preservatives, medicaments |
| Virgili et al. | 2003 | Vulvar lichen simplex chronicus | 61 | 47.5 | 26 | nickel, preservatives, fragrances, medicaments |
| Nardelli et al. | 2004 | Vulvar symptoms | 92 | 38 | 16 | medicaments |
| Utas et al. | 2008 | Vulvar itching | 50 | 52 | 16 | preservatives, fragrances, medicaments |
| Haverhoek et al. | 2008 | Vulvar pruritus | 43 | 81.4 | 44 | preservatives, fragrances, medicaments |
| Warshsoaw et al. | 2008 | Anogenital dermatoses | 570 | 44.1 | 27 | medicaments, corticosteroids |
| Vermaat et al. | 2008 | Anogenital dermatoses | 53 | 66 | 20 | fragrances, spices |
| O’Gorman et al. | 2013 | Vulvar itching | 90 | 69 | 39 | preservatives, fragrances, medicaments |
| Al-Niaimi at al. | 2014 | Vulvar symptoms | 282 | 54 | 49 | nickel, fragrances, neomycin |
| Trivedi et al. | 2018 | Vulvar itching | - | 64 | 54 | preservatives, fragrances |
| MEDICAMENTS | ||
|---|---|---|
| ANTIBIOTICS | Framycetin sulfate Neomycin Bacitracin Streptomycin sulfate Polymyxin | |
| ANTIFUNGALS | Terconazole Clotrimazole Miconazole Nystatin | |
| CORTICOSTEROIDS | Hydrocortisone-17-butyrate Clobetasol-17-propionate Budesonide 0.1% Tixocortol-21-pivalate | |
| ANESTHETICS | Esters (benzocaine, tetracaine, procaine) Amides (dibucaine, lidocaine, cinchocaine) | |
| BUFEXAMAE | ||
| CROTAMITON | ||
| ANTISEPTICS | Chlorhexidine Povidone-iodone Cetrimide Benzethonium chloride Mercuric chloride Gentian violet Phenvlmercurium salts Thiomerosal Potassium dichromate | |
| FRAGRANCES | Balsam of Peru Fragrances mix I and II Perfume mix Eugenol and Isoeugenol Cinnamic aldehyde Hydroxycitronellal Hydroxyisohexyl 3-cyclohexene carboldehyde |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corazza, M.; Toni, G.; Zedde, P.; Schettini, N.; Borghi, A. Contact Dermatitis of the Vulva. Allergies 2021, 1, 206-215. https://doi.org/10.3390/allergies1040019
Corazza M, Toni G, Zedde P, Schettini N, Borghi A. Contact Dermatitis of the Vulva. Allergies. 2021; 1(4):206-215. https://doi.org/10.3390/allergies1040019
Chicago/Turabian StyleCorazza, Monica, Giulia Toni, Pierantonia Zedde, Natale Schettini, and Alessandro Borghi. 2021. "Contact Dermatitis of the Vulva" Allergies 1, no. 4: 206-215. https://doi.org/10.3390/allergies1040019
APA StyleCorazza, M., Toni, G., Zedde, P., Schettini, N., & Borghi, A. (2021). Contact Dermatitis of the Vulva. Allergies, 1(4), 206-215. https://doi.org/10.3390/allergies1040019





