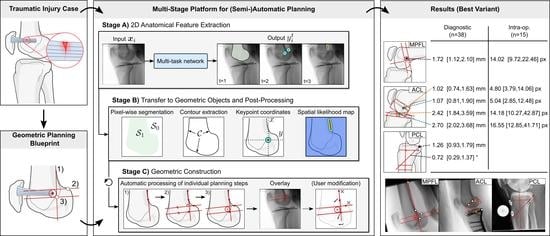
Multi-Stage Platform for (Semi-)Automatic Planning in Reconstructive Orthopedic Surgery
Abstract
:1. Introduction
- Most planning methods rely on the combination of salient proxy structures. This indirect description implies a greater number of error sources, which translates to higher observer-variability if the planning is done manually;
- Certain surgical planning can have a high level of geometric complexity. Sufficiently precise manual execution is only possible with tailored software tools or otherwise requires great amounts of time and labor;
- The ability to register pre-operative planning and intra-operative live data is highly complex due to the variable configuration of the joint and arbitrary relation between patient, table, and imaging system;
- Ad-hoc modifications of the surgical plan are essential to compensate for motion during the intervention;
- Manual interaction with a computer-assisted planning system is undesirable due to the surgery’s sterile setting. At the same time, the planning system should offer granular controls to correct each construction step with real-time visualization.
2. Related Work and Contribution
2.1. Image-Based Surgical Planning in Orthopedics and Traumatology
2.2. Multi-Task Learning and Task Weighting
2.3. Contribution
- This work establishes a multi-stage workflow that covers all necessary steps for image-based surgical planning on 2D X-ray images. The workflow is designed to mimic the clinically-established manual planning process, enabling granular control over each anatomical feature contributing to the planning geometry;
- We evaluate the model for three trauma-surgical planning applications on both diagnostic as well as intra-operative X-ray images of the knee joint. The numeric results match clinical requirements and encourage further clinical evaluation;
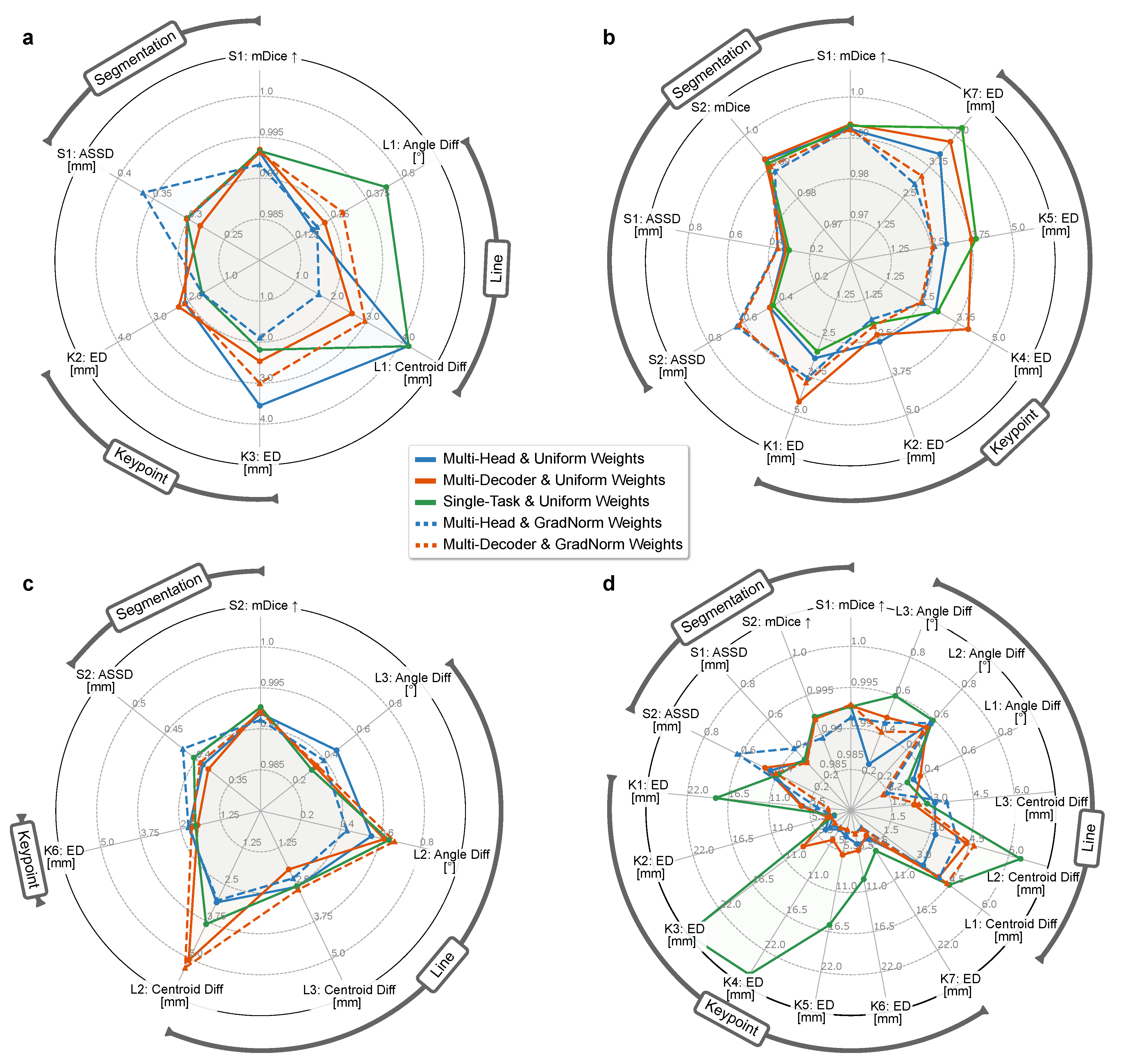
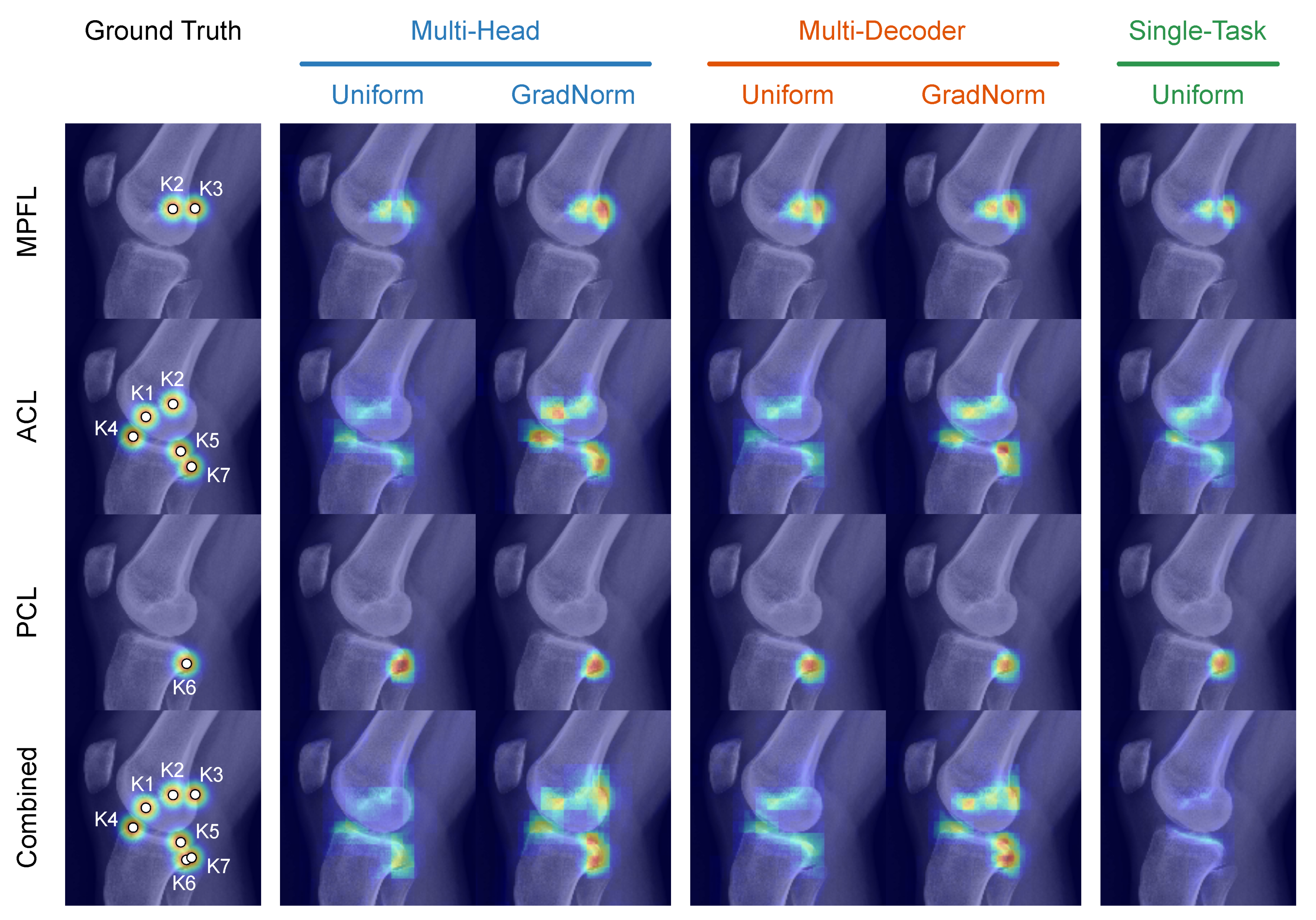
- We empirically show that the detection of anatomical landmarks benefits from a MTL setting. We confirm that explicit task weighting significantly reduces the landmark localization error, and illustrate that a multi-head network topology achieves similar performance to task-specific decoders, which are computationally much more expensive.
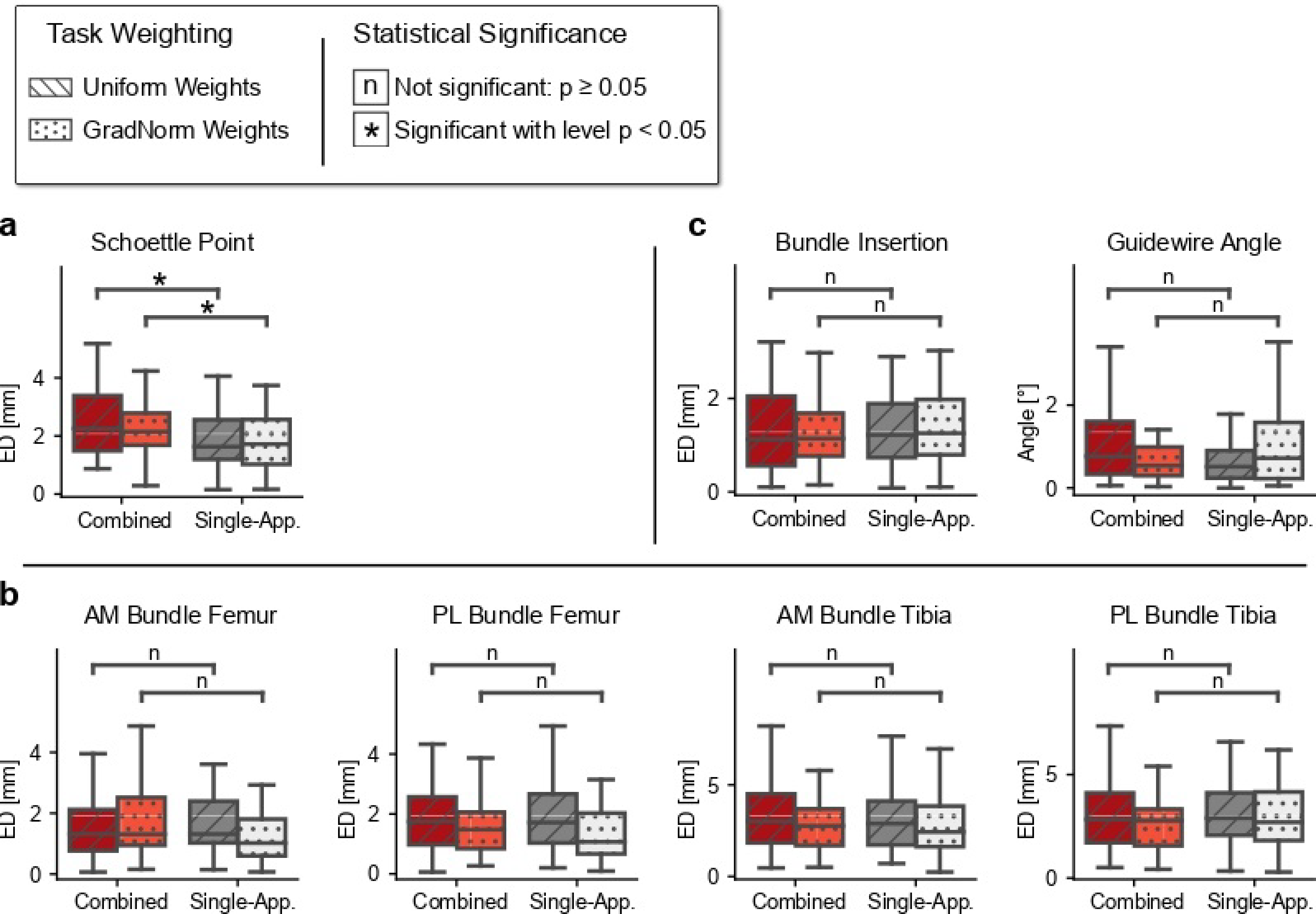
- Our study demonstrates that sharing tasks across anatomically related applications does not significantly improve performance compared to the single-application variant.
2.4. Article Structure
3. Materials and Methods
3.1. (Semi-)Automatic Workflow for 2D Surgical Planning
3.1.1. Multi-Stage Planning Algorithm
- Semantically coherent regions. Segmentation of connected regions that share certain characteristics, mostly bones and tools;
- Anatomical keypoints. Point-like landmarks that pinpoint features of interest on the bone surface;
- Elongated structures. Straight and curved lines that describe edges, ridges, or that refer to indirect features, such as anatomical axes.
3.1.2. Stage A) MTL for Joint Extraction of Anatomical Features
- Uniform (constant). All tasks are weighted uniformly: .
- Balanced relative learning rates (dynamic). Gradient normalization by Chen et al. [68] to ensure balanced training rates of all tasks, i.e., approximately equally-sized update steps for each task: .
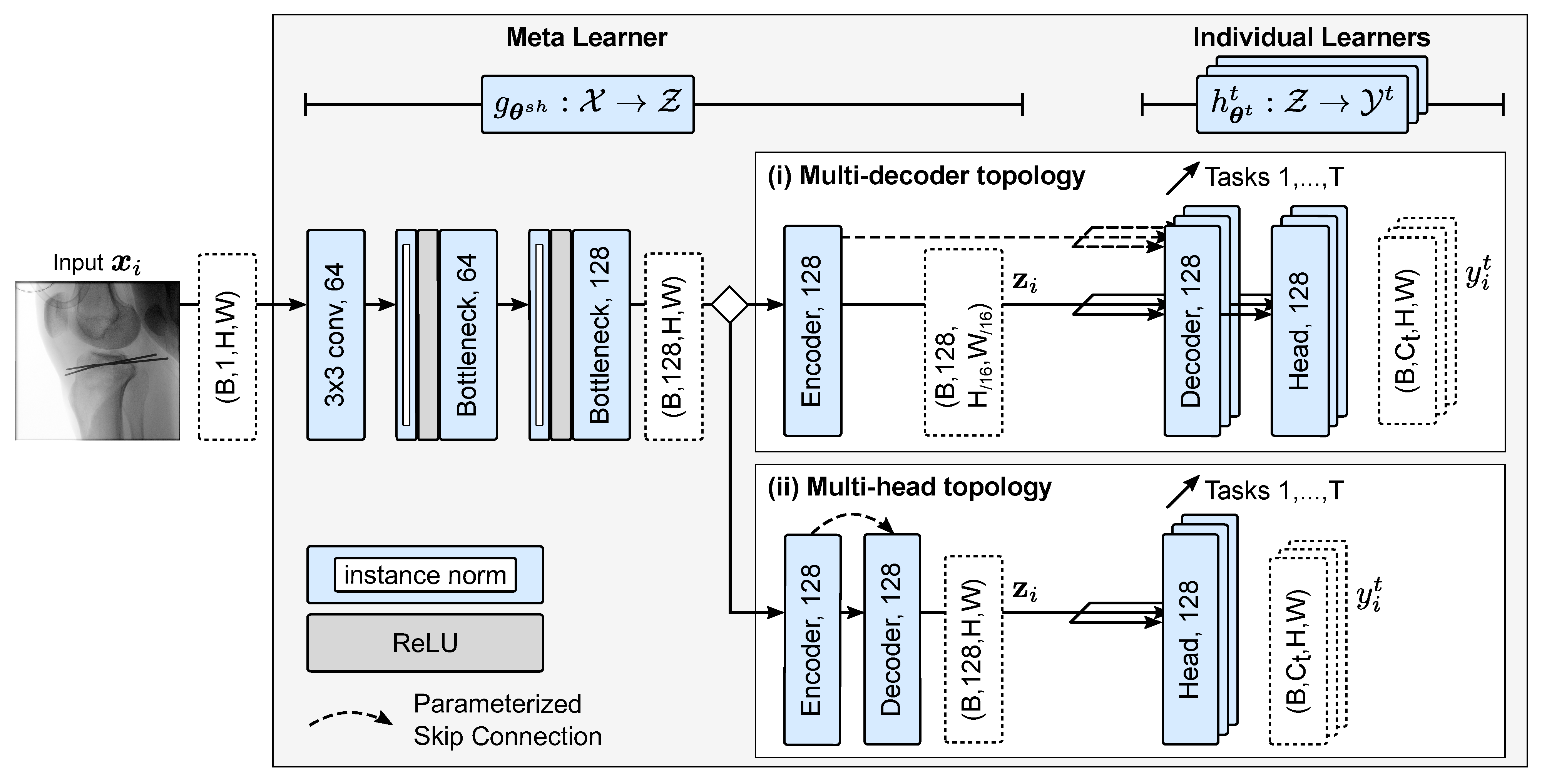
- Single-task baseline. For comparison, a STL baseline of independent encoder-decoder structures is optimized. Here, no parameters are shared;
- Multi-head topology. Both the encoder and decoder parameters are shared between the tasks in this variant. The output of the decoder is fed into task-specific prediction heads, which involve significantly less dedicated parameters and, thus, require a multi-purpose feature decoding. We argue that such a constrained decoder might benefit learning for highly similar tasks;
- Multi-decoder topology. After feature extraction in a shared feature encoder, the latent representation is used as input for the task-specific decoders and prediction heads. In other words, an abstract representation has to be found that serves the reconstruction for different kinds of tasks.
3.1.3. Stage B) Extraction of Geometric Objects and Post-Processing
3.1.4. Stage C) Geometric Construction of Individual Planning Steps
3.2. Multi-Task Network Architecture
3.3. Dataset, Ground Truth, and Augmentation Protocol
3.3.1. Cohort 1: Diagnostic X-ray Images
3.3.2. Cohort 2: Intra-Operative X-ray Images
3.3.3. Augmentation and Ground Truth
3.4. Training Policy and Implementation Details
3.5. Evaluation Protocol
4. Results
4.1. Research Questions
- Rq (1).
- How does the MTL network topology and task weighting strategy affect anatomical feature extraction?
- Rq (2).
- Does sharing tasks across anatomically related applications improve the feature extraction and target positioning compared to the single-application variant?
- Rq (3).
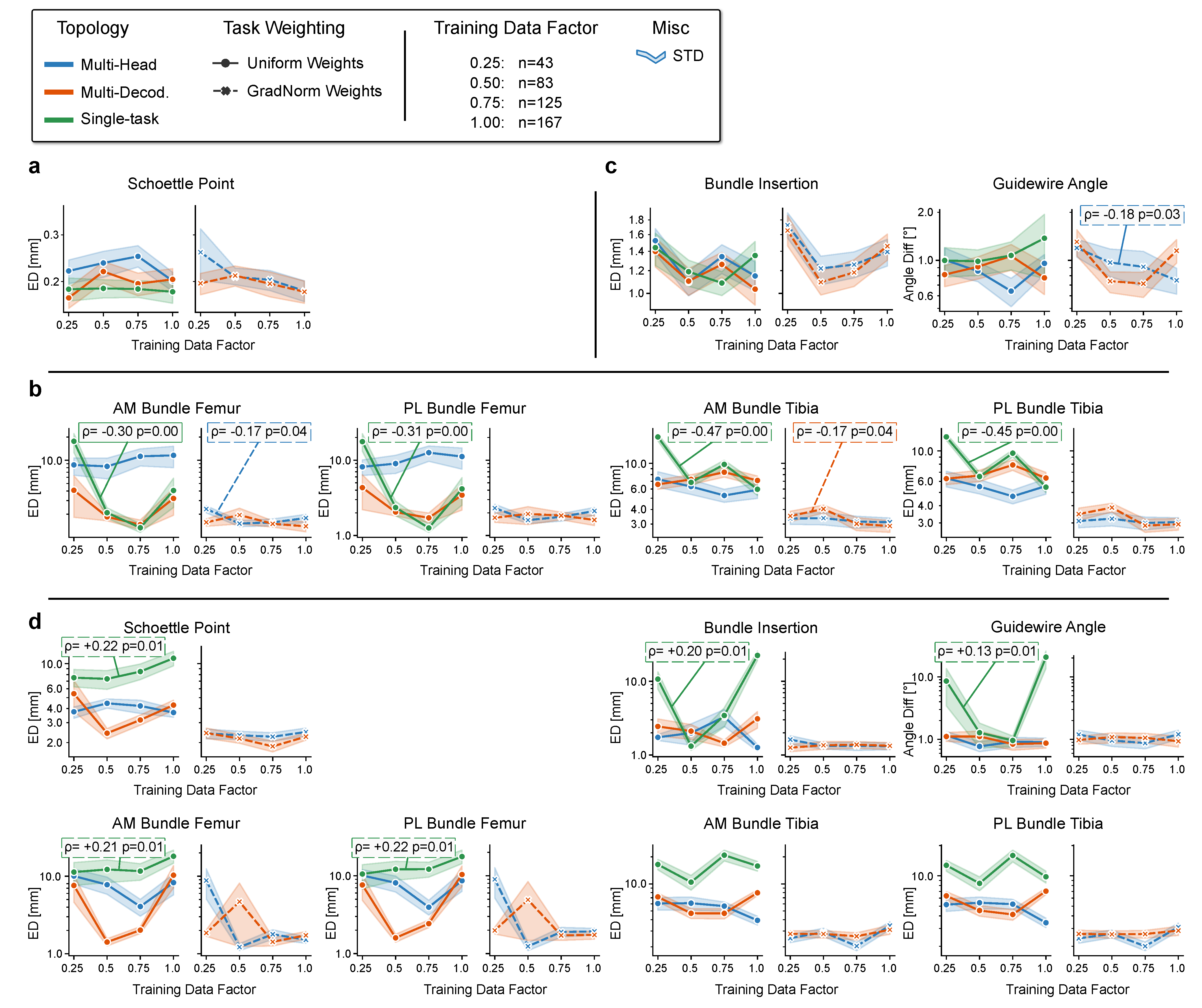
- How does the number of training data affect the planning accuracy?
- Rq (4).
- Can the performance on highly-standardized diagnostics images be applied to more complex imaging data in the intra-operative environment?
4.2. Rq (1) Network Topology and Task Weighting
4.3. Rq (2) Combining Tasks across Multiple Surgical Applications
4.4. Rq (3) Effect of the Number of Training Data
4.5. Rq (4) Application to Intra-Operative Data
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MPFL | Medial Patellofemoral Ligament |
| ACL | Anterior Cruciate Ligament |
| PCL | Posterior Cruciate Ligament |
| AM | Anteromedial |
| PL | Posterolateral |
| OR | Operating room |
| MTL | Multi-task learning |
| STL | Single-task learning |
| GradNorm | Gradient normalization |
| ASSD | Average symmetric surface distance |
| ED | Euclidean distance |
Appendix A. Pre-Operative Planning Examples
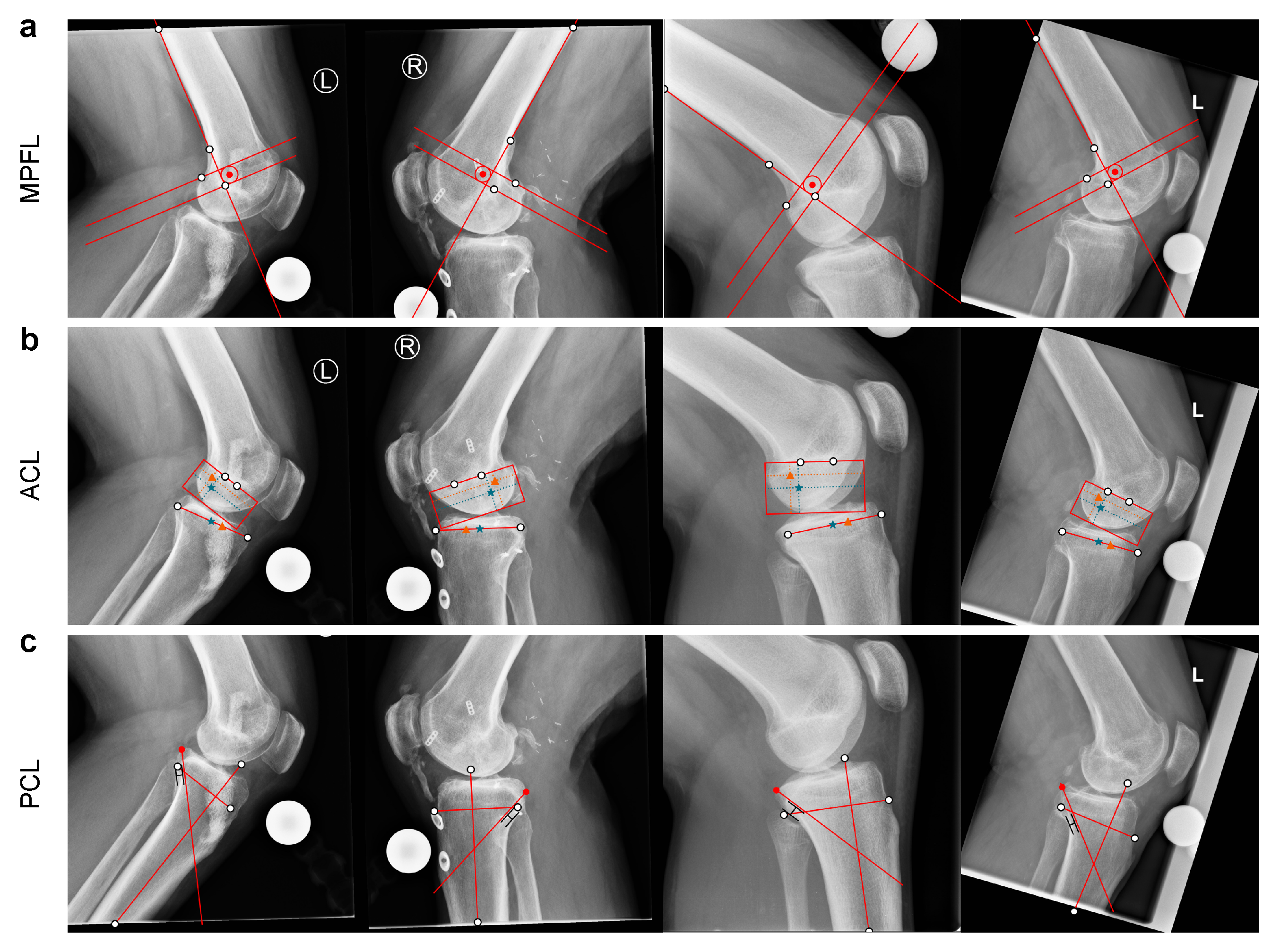
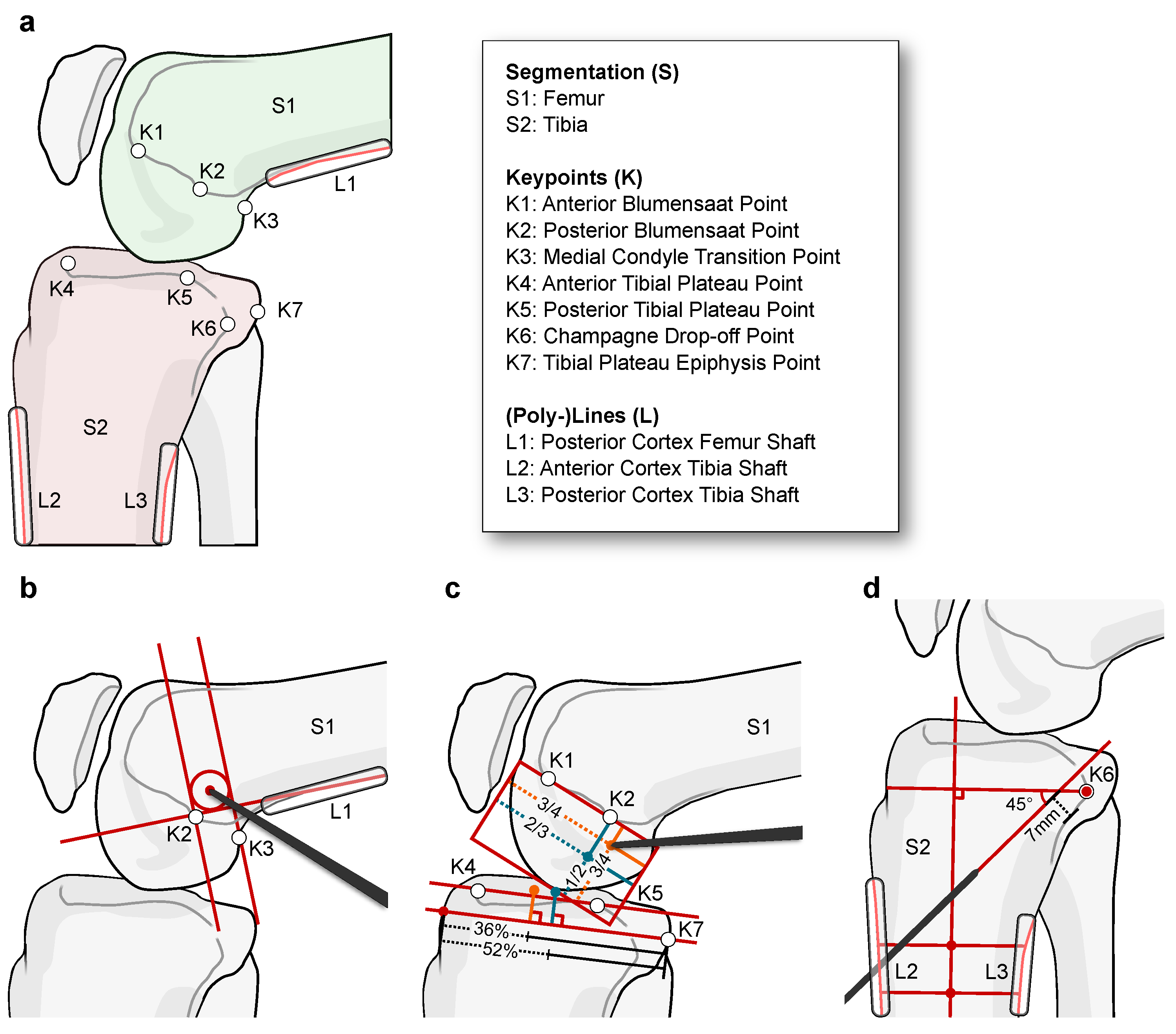
Appendix B. Planning Geometry
Appendix B.1. MPFL Reconstruction

Appendix B.2. ACL Reconstruction
Appendix B.2.1. Femoral Bundle Attachments
Appendix B.2.2. Tibial Bundle Attachments
Appendix B.3. PCL Reconstruction
Appendix C. Neural Network Topology
| Block | Input | Operation | Cin | Cout | Input Size | Output Size | |
|---|---|---|---|---|---|---|---|
| Pre | P1 | – | 3 × 3 conv, p = 1 | 1 | 64 | ||
| P2 | P1 | Bottleneck | 64 | 128 | |||
| P3 | P2 | Bottleneck | 128 | 128 | |||
| Encoder | E1 | P3 | Bottleneck | 128 | 128 | ||
| max pool, s = 2 | 128 | 128 | |||||
| E2 | E1 | Bottleneck | 128 | 128 | |||
| max pool, s = 2 | 128 | 128 | |||||
| E3 | E2 | Bottleneck | 128 | 128 | |||
| max pool, s = 2 | 128 | 128 | |||||
| E4 | E3 | Bottleneck | 128 | 128 | |||
| max pool, s = 2 | 128 | 128 | |||||
| Skip Con. | S1 | P3 | Bottleneck | 128 | 128 | ||
| S2 | E1 | Bottleneck | 128 | 128 | |||
| S3 | E2 | Bottleneck | 128 | 128 | |||
| S4 | E3 | Bottleneck | 128 | 128 | |||
| Decoder | D1 | E4 | Bottleneck | 128 | 128 | ||
| ×2 NN up-sampling | 128 | 128 | |||||
| D2 | D1⊕S1 | Bottleneck | 128 | 128 | |||
| ×2 NN up-sampling | 128 | 128 | |||||
| D3 | D2⊕S2 | Bottleneck | 128 | 128 | |||
| ×2 NN up-sampling | 128 | 128 | |||||
| D4 | D3⊕S3 | Bottleneck | 128 | 128 | |||
| ×2 NN up-sampling | 128 | 128 | |||||
| Heads | H1 (t = 1) | D4⊕S4 | Head | 128 | C1 | ||
| H2 (t = 2) | D4⊕S4 | Head | 128 | C2 | |||
| H3 (t = 3) | D4⊕S4 | Head | 128 | C3 |

References
- Hernandez, A.J.; Favaro, E.; Almeida, A.; Bonavides, A.; Demange, M.K.; Camanho, G.L. Reconstruction of the Medial Patellofemoral Ligament in Skeletally Immature Patients. Tech. Knee Surg. 2009, 8, 42–46. [Google Scholar] [CrossRef]
- Deie, M.; Ochi, M.; Sumen, Y.; Yasumoto, M.; Kobayashi, K.; Kimura, H. Reconstruction of the medial patellofemoral ligament for the treatment of habitual or recurrent dislocation of the patella in children. J. Bone Joint Surg. Br. 2003, 85, 887–890. [Google Scholar] [CrossRef] [Green Version]
- Diederichs, G.; Issever, A.S.; Scheffler, S. MR imaging of patellar instability: Injury patterns and assessment of risk factors. Radiographics 2010, 30, 961–981. [Google Scholar] [CrossRef] [PubMed]
- Longo, U.G.; Viganò, M.; Candela, V.; de Girolamo, L.; Cella, E.; Thiebat, G.; Salvatore, G.; Ciccozzi, M.; Denaro, V. Epidemiology of Posterior Cruciate Ligament Reconstructions in Italy: A 15-Year Study. J. Clin. Med. 2021, 10, 499. [Google Scholar] [CrossRef]
- Rünow, A. The Dislocating Patella. Acta Orthop. Scand. 1983, 54, 1–53. [Google Scholar] [CrossRef] [Green Version]
- Stefancin, J.J.; Parker, R.D. First-time traumatic patellar dislocation: A systematic review. Clin. Orthop. Relat. Res. 2007, 455, 93–101. [Google Scholar] [CrossRef] [Green Version]
- Dornacher, D.; Lippacher, S.; Nelitz, M.; Reichel, H.; Ignatius, A.; Dürselen, L.; Seitz, A.M. Impact of five different medial patellofemoral ligament-reconstruction strategies and three different graft pre-tensioning states on the mean patellofemoral contact pressure: A biomechanical study on human cadaver knees. J. Exp. Orthop. 2018, 5, 25. [Google Scholar] [CrossRef]
- Arendt, E.A.; Fithian, D.C.; Cohen, E. Current concepts of lateral patella dislocation. Clin. Sports Med. 2002, 21, 499–519. [Google Scholar] [CrossRef]
- Askenberger, M.; Ekström, W.; Finnbogason, T.; Janarv, P.M. Occult Intra-articular Knee Injuries in Children With Hemarthrosis. Am. J. Sports Med. 2014, 42, 1600–1606. [Google Scholar] [CrossRef]
- Fithian, D.C.; Paxton, E.W.; Stone, M.L.; Silva, P.; Davis, D.K.; Elias, D.A.; White, L.M. Epidemiology and natural history of acute patellar dislocation. Am. J. Sports Med. 2004, 32, 1114–1121. [Google Scholar] [CrossRef]
- Mäenpää, H.; Lehto, M.U.K. Patellar dislocation. The long-term results of nonoperative management in 100 patients. Am. J. Sports Med. 1997, 25, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Panni, A.S.; Cerciello, S.; Vasso, M. Patellofemoral instability: Surgical treatment of soft tissues. Joints 2013, 1, 34–39. [Google Scholar] [PubMed]
- Petri, M.; Ettinger, M.; Stuebig, T.; Brand, S.; Krettek, C.; Jagodzinski, M.; Omar, M. Current Concepts for Patellar Dislocation. Arch. Trauma Res. 2015, 4, e29301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amis, A.A.; Firer, P.; Mountney, J.; Senavongse, W.; Thomas, N.P. Anatomy and biomechanics of the medial patellofemoral ligament. Knee 2003, 10, 215–220. [Google Scholar] [CrossRef]
- Dragoo, J.L.; Nguyen, M.; Gatewood, C.T.; Taunton, J.D.; Young, S. Medial Patellofemoral Ligament Repair Versus Reconstruction for Recurrent Patellar Instability: Two-Year Results of an Algorithm-Based Approach. Orthop. J. Sports Med. 2017, 5, 2325967116689465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camp, C.L.; Krych, A.J.; Dahm, D.L.; Levy, B.A.; Stuart, M.J. Medial patellofemoral ligament repair for recurrent patellar dislocation. Am. J. Sports Med. 2010, 38, 2248–2254. [Google Scholar] [CrossRef]
- Buckens, C.F.M.; Saris, D.B.F. Reconstruction of the medial patellofemoral ligament for treatment of patellofemoral instability: A systematic review. Am. J. Sports Med. 2010, 38, 181–188. [Google Scholar] [CrossRef]
- Nelitz, M.; Williams, S.R.M. Kombinierte Trochleaplastik und Rekonstruktion des medialen patellofemoralen Ligaments zur Behandlung der patellofemoralen Instabilität. Oper. Orthop. Traumatol. 2015, 27, 495–504. [Google Scholar] [CrossRef]
- Shah, J.N.; Howard, J.S.; Flanigan, D.C.; Brophy, R.H.; Carey, J.L.; Lattermann, C. A systematic review of complications and failures associated with medial patellofemoral ligament reconstruction for recurrent patellar dislocation. Am. J. Sports Med. 2012, 40, 1916–1923. [Google Scholar] [CrossRef] [Green Version]
- Vainionpää, S.; Laasonen, E.; Silvennoinen, T.; Vasenius, J.; Rokkanen, P. Acute dislocation of the patella. A prospective review of operative treatment. J. Bone Joint Surg. Br. 1990, 72, 366–369. [Google Scholar] [CrossRef]
- Schöttle, P.B.; Schmeling, A.; Rosenstiel, N.; Weiler, A. Radiographic landmarks for femoral tunnel placement in medial patellofemoral ligament reconstruction. Am. J. Sports Med. 2007, 35, 801–804. [Google Scholar] [CrossRef]
- Chahla, J.; Nitri, M.; Civitarese, D.; Dean, C.S.; Moulton, S.G.; LaPrade, R.F. Anatomic Double-Bundle Posterior Cruciate Ligament Reconstruction. Arthrosc. Tech. 2016, 5, e149–e156. [Google Scholar] [CrossRef] [PubMed]
- Johannsen, A.M.; Anderson, C.J.; Wijdicks, C.A.; Engebretsen, L.; LaPrade, R.F. Radiographic landmarks for tunnel positioning in posterior cruciate ligament reconstructions. Am. J. Sports Med. 2013, 41, 35–42. [Google Scholar] [CrossRef] [PubMed]
- LaPrade, R.F.; Cinque, M.E.; Dornan, G.J.; DePhillipo, N.N.; Geeslin, A.G.; Moatshe, G.; Chahla, J. Double-Bundle Posterior Cruciate Ligament Reconstruction in 100 Patients at a Mean 3 Years’ Follow-up: Outcomes Were Comparable to Anterior Cruciate Ligament Reconstructions. Am. J. Sports Med. 2018, 46, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Spiridonov, S.I.; Slinkard, N.J.; LaPrade, R.F. Isolated and combined grade-III posterior cruciate ligament tears treated with double-bundle reconstruction with use of endoscopically placed femoral tunnels and grafts: Operative technique and clinical outcomes. J. Bone Joint Surg. Am. 2011, 93, 1773–1780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weimann, A.; Wolfert, A.; Zantop, T.; Eggers, A.K.; Raschke, M.; Petersen, W. Reducing the “killer turn” in posterior cruciate ligament reconstruction by fixation level and smoothing the tibial aperture. Arthroscopy 2007, 23, 1104–1111. [Google Scholar] [CrossRef]
- Jackson, D.W.; Proctor, C.S.; Simon, T.M. Arthroscopic assisted PCL reconstruction: A technical note on potential neurovascular injury related to drill bit configuration. Arthroscopy 1993, 9, 224–227. [Google Scholar] [CrossRef]
- Montgomery, S.R.; Johnson, J.S.; McAllister, D.R.; Petrigliano, F.A. Surgical management of PCL injuries: Indications, techniques, and outcomes. Curr. Rev. Musculoskelet. Med. 2013, 6, 115–123. [Google Scholar] [CrossRef] [Green Version]
- Nicodeme, J.D.; Löcherbach, C.; Jolles, B.M. Tibial tunnel placement in posterior cruciate ligament reconstruction: A systematic review. Knee Surg. Sports Traumatol. Arthrosc. 2014, 22, 1556–1562. [Google Scholar] [CrossRef]
- Yao, J.; Wen, C.Y.; Zhang, M.; Cheung, J.T.-M.; Yan, C.; Chiu, K.-Y.; Lu, W.W.; Fan, Y. Effect of tibial drill-guide angle on the mechanical environment at bone tunnel aperture after anatomic single-bundle anterior cruciate ligament reconstruction. Int. Orthop. 2014, 38, 973–981. [Google Scholar] [CrossRef] [Green Version]
- Bertollo, N.; Walsh, W.R. Drilling of Bone: Practicality, Limitations and Complications Associated with Surgical Drill-Bits. In Biomechanics in Applications; Klika, V., Ed.; IntechOpen: Rijeka, Croatia, 2011; Chapter 3. [Google Scholar] [CrossRef] [Green Version]
- Bernard, M.; Hertel, P.; Hornung, H.; Cierpinski, T. Femoral insertion of the ACL. Radiographic quadrant method. J. Knee Surg. 1997, 10, 14–21; discussion 21–22. [Google Scholar]
- Amis, A.A.; Jakob, R.P. Anterior cruciate ligament graft positioning, tensioning and twisting. Knee Surg. Sports Traumatol. Arthrosc. 1998, 6 (Suppl. 1), S2–S12. [Google Scholar] [CrossRef]
- Stäubli, H.U.; Rauschning, W. Tibial attachment area of the anterior cruciate ligament in the extended knee position. Anatomy and cryosections in vitro complemented by magnetic resonance arthrography in vivo. Knee Surg. Sports Traumatol. Arthrosc. 1994, 2, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Linte, C.A.; Moore, J.T.; Chen, E.C.; Peters, T.M. Image-Guided Procedures. In Bioengineering for Surgery; Elsevier: Amsterdam, The Netherlands, 2016; pp. 59–90. [Google Scholar] [CrossRef]
- Kubicek, J.; Tomanec, F.; Cerny, M.; Vilimek, D.; Kalova, M.; Oczka, D. Recent Trends, Technical Concepts and Components of Computer-Assisted Orthopedic Surgery Systems: A Comprehensive Review. Sensors 2019, 19, 5199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joskowicz, L.; Hazan, E.J. Computer Aided Orthopaedic Surgery: Incremental shift or paradigm change? Med. Image Anal. 2016, 33, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Yang, J.; Chen, X.; Sun, Y.; Li, X. Medical Robotics in Bone Fracture Reduction Surgery: A Review. Sensors 2019, 19, 3593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Innocenti, B.; Bori, E. Robotics in orthopaedic surgery: Why, what and how? Arch. Orthop. Trauma Surg. 2021, 141, 2035–2042. [Google Scholar] [CrossRef]
- Jinnah, A.H.; Luo, T.D.; Plate, J.F.; Jinnah, R.H. General Concepts in Robotics in Orthopedics. In Robotics in Knee and Hip Arthroplasty; Lonner, J.H., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 27–35. [Google Scholar] [CrossRef]
- Karthik, K.; Colegate-Stone, T.; Dasgupta, P.; Tavakkolizadeh, A.; Sinha, J. Robotic surgery in trauma and orthopaedics. Bone Joint J. 2015, 97, 292–299. [Google Scholar] [CrossRef]
- Stübig, T.; Windhagen, H.; Krettek, C.; Ettinger, M. Computer-Assisted Orthopedic and Trauma Surgery. Dtsch. Arztebl. Int. 2020, 117, 793–800. [Google Scholar] [CrossRef]
- Zhao, J.X.; Li, C.; Ren, H.; Hao, M.; Zhang, L.C.; Tang, P.F. Evolution and Current Applications of Robot-Assisted Fracture Reduction: A Comprehensive Review. Ann. Biomed. Eng. 2020, 48, 203–224. [Google Scholar] [CrossRef]
- Ewurum, C.H.; Guo, Y.; Pagnha, S.; Feng, Z.; Luo, X. Surgical Navigation in Orthopedics: Workflow and System Review. In Intelligent Orthopaedics: Artificial Intelligence and Smart Image-Guided Technology for Orthopaedics; Zheng, G., Tian, W., Zhuang, X., Eds.; Springer: Singapore, 2018; pp. 47–63. [Google Scholar] [CrossRef]
- Székely, G.; Nolte, L.P. Image guidance in orthopaedics and traumatology: A historical perspective. Med. Image Anal. 2016, 33, 79–83. [Google Scholar] [CrossRef]
- Zheng, G.; Nolte, L.P. Computer-Aided Orthopaedic Surgery: State-of-the-Art and Future Perspectives. In Intelligent Orthopaedics: Artificial Intelligence and Smart Image-guided Technology for Orthopaedics; Springer: Singapore, 2018; pp. 1–20. [Google Scholar] [CrossRef]
- Wang, M.; Li, D.; Shang, X.; Wang, J. A review of computer-assisted orthopaedic surgery systems. Int. J. Med. Robot. 2020, 16, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Casari, F.A.; Navab, N.; Hruby, L.A.; Kriechling, P.; Nakamura, R.; Tori, R.; de Lourdes Dos Santos Nunes, F.; Queiroz, M.C.; Fürnstahl, P.; Farshad, M. Augmented Reality in Orthopedic Surgery Is Emerging from Proof of Concept Towards Clinical Studies: A Literature Review Explaining the Technology and Current State of the Art. Curr. Rev. Musculoskelet. Med. 2021, 14, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Essert, C.; Joskowicz, L. Chapter 32—Image-based surgery planning. In Handbook of Medical Image Computing and Computer Assisted Intervention; Zhou, S.K., Rueckert, D., Fichtinger, G., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 795–815. [Google Scholar] [CrossRef]
- Kordon, F.; Maier, A.; Swartman, B.; Privalov, M.; El Barbari, J.S.; Kunze, H. Contour-Based Bone Axis Detection for X-ray Guided Surgery on the Knee. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); Martel, A.L., Abolmaesumi, P., Stoyanov, D., Mateus, D., Zuluaga, M.A., Zhou, S.K., Racoceanu, D., Joskowicz, L., Eds.; Springer: Cham, Switzerland, 2020; Volume 12266, pp. 671–680. [Google Scholar] [CrossRef]
- Bier, B.; Unberath, M.; Zaech, J.N.; Fotouhi, J.; Armand, M.; Osgood, G.; Navab, N.; Maier, A. X-ray-transform Invariant Anatomical Landmark Detection for Pelvic Trauma Surgery. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); Frangi, A.F., Schnabel, J.A., Davatzikos, C., Alberola-López, C., Fichtinger, G., Eds.; Springer International Publishing: Cham, Switzerland, 2018; Volume 11073, pp. 55–63. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.M.; Kim, H.P.; Jeon, K.; Lee, S.H.; Seo, J.K. Automatic 3D cephalometric annotation system using shadowed 2D image-based machine learning. Phys. Med. Biol. 2019, 64, 055002. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.E.; Yu, C.H.; Chang, C.J.; Wu, H.K.; Yu, T.H.; Tseng, C.S. C-Arm Image-Based Surgical Path Planning Method for Distal Locking of Intramedullary Nails. Appl. Bionics Biomech. 2018, 2018, 4530386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kordon, F.; Fischer, P.; Privalov, M.; Swartman, B.; Schnetzke, M.; Franke, J.; Lasowski, R.; Maier, A.; Kunze, H. Multi-task Localization and Segmentation for X-ray Guided Planning in Knee Surgery. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); Shen, D., Liu, T., Peters, T.M., Staib, L.H., Essert, C., Zhou, S., Yap, P.-T., Khan, A., Eds.; Springer: Cham, Switzerland, 2019; Volume 11769, pp. 622–630. [Google Scholar] [CrossRef] [Green Version]
- Kordon, F.; Maier, A.; Swartman, B.; Privalov, M.; El Barbari, J.S.; Kunze, H. Automatic Path Planning for Safe Guide Pin Insertion in PCL Reconstruction Surgery. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); de Bruijne, M., Cattin, P.C., Cotin, S., Padoy, N., Speidel, S., Zheng, Y., Essert, C., Eds.; Springer International Publishing: Cham, Switzerland, 2021; Volume 12904, pp. 560–570. [Google Scholar] [CrossRef]
- Swartman, B.; Frere, D.; Wei, W.; Schnetzke, M.; Beisemann, N.; Keil, H.; Franke, J.; Grützner, P.; Vetter, S. 2D projection-based software application for mobile C-arms optimises wire placement in the proximal femur—An experimental study. Injury 2017, 48, 2068–2073. [Google Scholar] [CrossRef]
- Kausch, L.; Scherer, M.; Thomas, S.; Klein, A.; Isensee, F.; Maier-Hein, K. Automatic image-based pedicle screw planning. In Medical Imaging 2021: Image-Guided Procedures, Robotic Interventions, and Modeling; Linte, C.A., Siewerdsen, J.H., Eds.; SPIE: Bellingham, WA, USA, 2021; Volume 11598, pp. 406–414. [Google Scholar] [CrossRef]
- Joskowicz, L. Future Perspectives on Statistical Shape Models in Computer-Aided Orthopedic Surgery: Beyond Statistical Shape Models and on to Big Data. In Computer Assisted Orthopaedic Surgery for Hip and Knee: Current State of the Art in Clinical Application and Basic Research; Springer: Singapore, 2018; pp. 199–206. [Google Scholar] [CrossRef]
- Kamiya, N. Deep Learning Technique for Musculoskeletal Analysis. In Deep Learning in Medical Image Analysis: Challenges and Applications; Springer International Publishing: Cham, Switzerland, 2020; pp. 165–176. [Google Scholar] [CrossRef]
- Benos, L.; Stanev, D.; Spyrou, L.; Moustakas, K.; Tsaopoulos, D.E. A Review on Finite Element Modeling and Simulation of the Anterior Cruciate Ligament Reconstruction. Front. Bioeng. Biotechnol. 2020, 8, 967. [Google Scholar] [CrossRef]
- Colombet, P.; Robinson, J.; Christel, P.; Franceschi, J.P.; Djian, P.; Bellier, G.; Sbihi, A. Morphology of anterior cruciate ligament attachments for anatomic reconstruction: A cadaveric dissection and radiographic study. Arthroscopy 2006, 22, 984–992. [Google Scholar] [CrossRef]
- Caruana, R. Multitask Learning. In Learning to Learn; Springer: Boston, MA, USA, 1998; pp. 95–133. [Google Scholar] [CrossRef]
- Ruder, S. An Overview of Multi-Task Learning in Deep Neural Networks. arXiv 2017, arXiv:1706.05098. [Google Scholar]
- Thung, K.H.; Wee, C.Y. A brief review on multi-task learning. Multimed. Tools Appl. 2018, 77, 29705–29725. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Q. An overview of multi-task learning. Natl. Sci. Rev. 2018, 5, 30–43. [Google Scholar] [CrossRef] [Green Version]
- Baxter, J. A Model of Inductive Bias Learning. J. Artif. Int. Res. 2000, 12, 149–198. [Google Scholar] [CrossRef]
- Lin, B.; Ye, F.; Zhang, Y. A Closer Look at Loss Weighting in Multi-Task Learning. arXiv 2021, arXiv:2111.10603. [Google Scholar]
- Chen, Z.; Badrinarayanan, V.; Lee, C.Y.; Rabinovich, A. GradNorm: Gradient Normalization for Adaptive Loss Balancing in Deep Multitask Networks. In Proceedings of the 35th International Conference on Machine Learning, Stockholm, Sweden, 10–15 July 2018; Dy, J., Krause, A., Eds.; PMLR: Stockholm, Sweden, 2018; 80, pp. 794–803. [Google Scholar]
- Liu, S.; Johns, E.; Davison, A.J. End-To-End Multi-Task Learning With Attention. In Proceedings of the 2019 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Long Beach, CA, USA, 15–20 June 2019; pp. 1871–1880. [Google Scholar] [CrossRef] [Green Version]
- Kendall, A.; Gal, Y.; Cipolla, R. Multi-Task Learning Using Uncertainty to Weigh Losses for Scene Geometry and Semantics. In Proceedings of the 2018 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Salt Lake City, UT, USA, 18–23 June 2018. [Google Scholar]
- Sener, O.; Koltun, V. Multi-Task Learning as Multi-Objective Optimization. In Advances in Neural Information Processing Systems; Bengio, S., Wallach, H., Larochelle, H., Grauman, K., Cesa-Bianchi, N., Garnett, R., Eds.; Curran Associates, Inc.: Red Hook, NY, USA, 2018; Volume 31. [Google Scholar]
- Yu, T.; Kumar, S.; Gupta, A.; Levine, S.; Hausman, K.; Finn, C. Gradient Surgery for Multi-Task Learning. In Advances in Neural Information Processing Systems; Larochelle, H., Ranzato, M., Hadsell, R., Balcan, M.F., Lin, H., Eds.; Curran Associates, Inc.: Red Hook, NY, USA, 2020; Volume 33, pp. 5824–5836. [Google Scholar]
- Wang, Z.; Tsvetkov, Y.; Firat, O.; Cao, Y. Gradient Vaccine: Investigating and Improving Multi-task Optimization in Massively Multilingual Models. arXiv 2021, arXiv:2010.05874. [Google Scholar]
- Chen, Z.; Ngiam, J.; Huang, Y.; Luong, T.; Kretzschmar, H.; Chai, Y.; Anguelov, D. Just Pick a Sign: Optimizing Deep Multitask Models with Gradient Sign Dropout. In Advances in Neural Information Processing Systems; Larochelle, H., Ranzato, M., Hadsell, R., Balcan, M.F., Lin, H., Eds.; Curran Associates, Inc.: Red Hook, NY, USA, 2020; Volume 33, pp. 2039–2050. [Google Scholar]
- Liu, L.; Li, Y.; Kuang, Z.; Xue, J.H.; Chen, Y.; Yang, W.; Liao, Q.; Zhang, W. Towards Impartial Multi-task Learning. In Proceedings of the International Conference on Learning Representations, Virtual Event, 3–7 May 2021. [Google Scholar]
- Newell, A.; Yang, K.; Deng, J. Stacked Hourglass Networks for Human Pose Estimation. In Computer Vision—ECCV 2016; Leibe, B., Matas, J., Sebe, N., Welling, M., Eds.; Springer: Cham, Switzerland, 2016; pp. 483–499. [Google Scholar]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Identity Mappings in Deep Residual Networks. In Computer Vision—ECCV 2016; Leibe, B., Matas, J., Sebe, N., Welling, M., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 630–645. [Google Scholar]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Deep Residual Learning for Image Recognition. In Proceedings of the 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Las Vegas, NV, USA, 27–30 June 2016; pp. 770–778. [Google Scholar] [CrossRef] [Green Version]
- Greff, K.; Srivastava, R.K.; Schmidhuber, J. Highway and Residual Networks learn Unrolled Iterative Estimation. In Proceedings of the 5th International Conference on Learning Representations, ICLR, Toulon, France, 24–26 April 2017. [Google Scholar]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); Springer: Berlin/Heidelberg, Germany, 2015; Volume 9351, pp. 234–241. [Google Scholar]
- Wada, K. Labelme: Image Polygonal Annotation with Python. 2016. Available online: https://github.com/wkentaro/labelme (accessed on 28 February 2022).
- Kordon, F.; Lasowski, R.; Swartman, B.; Franke, J.; Fischer, P.; Kunze, H. Improved X-ray Bone Segmentation by Normalization and Augmentation Strategies. In Bildverarbeitung für Die Medizin 2019; Handels, H., Deserno, T.M., Maier, A., Maier-Hein, K.H., Palm, C., Tolxdorff, T., Eds.; Informatik Aktuell, Springer Fachmedien Wiesbaden: Wiesbaden, Germany, 2019; pp. 104–109. [Google Scholar] [CrossRef]
- Sutskever, I.; Martens, J.; Dahl, G.; Hinton, G. On the Importance of Initialization and Momentum in Deep Learning. In Proceedings of the 30th International Conference on International Conference on Machine Learning, Atlanta, GA, USA, 17–19 June 2013. [Google Scholar]
- Smith, L.N. Cyclical learning rates for training neural networks. In Proceedings of the 2017 IEEE Winter Conference on Applications of Computer Vision (WACV), Santa Rosa, CA, USA, 24–31 March 2017; pp. 464–472. [Google Scholar]
- Akkus, Z.; Galimzianova, A.; Hoogi, A.; Rubin, D.L.; Erickson, B.J. Deep Learning for Brain MRI Segmentation: State of the Art and Future Directions. J. Digit. Imaging 2017, 30, 449–459. [Google Scholar] [CrossRef] [Green Version]
- Hu, M.K. Visual pattern recognition by moment invariants. IEEE Trans. Inf. Theory 1962, 8, 179–187. [Google Scholar] [CrossRef] [Green Version]
- Ziegler, C.G.; Fulkerson, J.P.; Edgar, C. Radiographic Reference Points Are Inaccurate with and without a True Lateral Radiograph: The Importance of Anatomy in Medial Patellofemoral Ligament Reconstruction. Am. J. Sports Med. 2016, 44, 133–142. [Google Scholar] [CrossRef]
- Sanchis-Alfonso, V.; Ramírez-Fuentes, C.; Montesinos-Berry, E.; Elía, I.; Martí-Bonmatí, L. Radiographic Location Does Not Ensure a Precise Anatomic Location of the Femoral Fixation Site in Medial Patellofemoral Ligament Reconstruction. Orthop. J. Sports Med. 2017, 5, 2325967117739252. [Google Scholar] [CrossRef]
- Kausch, L.; Thomas, S.; Kunze, H.; Privalov, M.; Vetter, S.; Franke, J.; Mahnken, A.H.; Maier-Hein, L.; Maier-Hein, K. Toward automatic C-arm positioning for standard projections in orthopedic surgery. Int. J. Comput. Assist. Radiol. Surg. 2020, 15, 1095–1105. [Google Scholar] [CrossRef]
- Kausch, L.; Thomas, S.; Kunze, H.; Norajitra, T.; Klein, A.; El Barbari, J.S.; Privalov, M.; Vetter, S.; Mahnken, A.; Maier-Hein, L.; et al. C-Arm Positioning for Spinal Standard Projections in Different Intra-operative Settings. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); de Bruijne, M., Cattin, P.C., Cotin, S., Padoy, N., Speidel, S., Zheng, Y., Essert, C., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 352–362. [Google Scholar]
- Cho, W.J.; Kim, J.M.; Kim, D.E.; Lee, J.G.; Park, J.W.; Han, Y.H.; Seo, H.G. Accuracy of the femoral tunnel position in robot-assisted anterior cruciate ligament reconstruction using a magnetic resonance imaging-based navigation system: A preliminary report. Int. J. Med. Robot. Comput. Assist. Surg. 2018, 14, e1933. [Google Scholar] [CrossRef]
- Morita, K.; Kobashi, S.; Kashiwa, K.; Nakayama, H.; Kambara, S.; Morimoto, M.; Yoshiya, S.; Aikawa, S. Computer-aided Surgical Planning of Anterior Cruciate Ligament Reconstruction in MR Images. Procedia Comput. Sci. 2015, 60, 1659–1667. [Google Scholar] [CrossRef] [Green Version]
- Morita, K. Computer-aided Diagnosis Systems Based on Medical Image Registration. Ph.D. Thesis, University of Hyogo, Kobe, Japan, 2019. [Google Scholar]
- Raposo, C.; Barreto, J.P.; Sousa, C.; Ribeiro, L.; Melo, R.; Oliveira, J.P.; Marques, P.; Fonseca, F.; Barrett, D. Video-based computer navigation in knee arthroscopy for patient-specific ACL reconstruction. Int. J. Comput. Assist. Radiol. Surg. 2019, 14, 1529–1539. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Lim, S.; Kim, Y.; Lee, D.; Park, S.; Park, S.; Wang, J.H. 3D preoperative surgical planning software for anterior cruciate ligament reconstruction. In Proceedings of the International Conference on Control, Automation and Systems (ICCAS), Gwangju, Korea, 20–23 October 2013; pp. 344–346. [Google Scholar] [CrossRef]
- Le Huynh, M.; Kim, Y.H. A computer-aided and robot-assisted surgical system for reconstruction of anterior cruciate ligament. Int. J. Precis. Eng. Manuf. 2013, 14, 49–54. [Google Scholar] [CrossRef]
- Kitamura, G.; Albers, M.B.V.; Lesniak, B.P.; Rabuck, S.J.; Musahl, V.; Andrews, C.L.; Ghodadra, A.; Fu, F. 3-Dimensional Printed Models May Be a Useful Tool When Planning Revision Anterior Cruciate Ligament Reconstruction. Arthrosc. Sports Med. Rehab. 2019, 1, e41–e46. [Google Scholar] [CrossRef] [Green Version]



| Anatomical Structure | Spatial Representation |
|---|---|
| Semantically coherent regions | Pixel-wise and multi-label segmentation. The multi-label aspect allows for overlap-aware segmentation, e.g., in areas between bones or metal implants. |
| Anatomical keypoints | Individual heatmaps/activation maps. For that purpose, a multivariate Gaussian distribution with its mean at the keypoint coordinate and a predefined standard deviation is sampled. |
| Elongated structures | Line-symmetric heatmap/activation map. The distance to the line segment or the axis of interest is evaluated and transformed using a Gaussian function. |
| Multi-Head | Multi-Decoder | Single-Task | |||
|---|---|---|---|---|---|
| Planning | Uniform | GradNorm | Uniform | GradNorm | Uniform |
| MPFL (n = 2) | |||||
| ACL (n = 5) | |||||
| PCL (n = 1) | |||||
| Comb. (n = 7) | |||||
| Multi-Head | Multi-Decoder | ||||
|---|---|---|---|---|---|
| Planning | Metric [px] | Median, | Cnt. | Median, | Cnt. |
| MPFL | Schoettle Pt. | 15 | 15 | ||
| ACL | AM Femur | 12 | 12 | ||
| PL Femur | 12 | 12 | |||
| AM Tibia | 12 | 12 | |||
| PL Tibia | 12 | 12 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kordon, F.; Maier, A.; Swartman, B.; Privalov, M.; El Barbari, J.S.; Kunze, H. Multi-Stage Platform for (Semi-)Automatic Planning in Reconstructive Orthopedic Surgery. J. Imaging 2022, 8, 108. https://doi.org/10.3390/jimaging8040108
Kordon F, Maier A, Swartman B, Privalov M, El Barbari JS, Kunze H. Multi-Stage Platform for (Semi-)Automatic Planning in Reconstructive Orthopedic Surgery. Journal of Imaging. 2022; 8(4):108. https://doi.org/10.3390/jimaging8040108
Chicago/Turabian StyleKordon, Florian, Andreas Maier, Benedict Swartman, Maxim Privalov, Jan Siad El Barbari, and Holger Kunze. 2022. "Multi-Stage Platform for (Semi-)Automatic Planning in Reconstructive Orthopedic Surgery" Journal of Imaging 8, no. 4: 108. https://doi.org/10.3390/jimaging8040108
APA StyleKordon, F., Maier, A., Swartman, B., Privalov, M., El Barbari, J. S., & Kunze, H. (2022). Multi-Stage Platform for (Semi-)Automatic Planning in Reconstructive Orthopedic Surgery. Journal of Imaging, 8(4), 108. https://doi.org/10.3390/jimaging8040108