HF_IDS_Cam: Fast Video Capture with ImageJ for Real-Time Analysis
Abstract
:1. Introduction
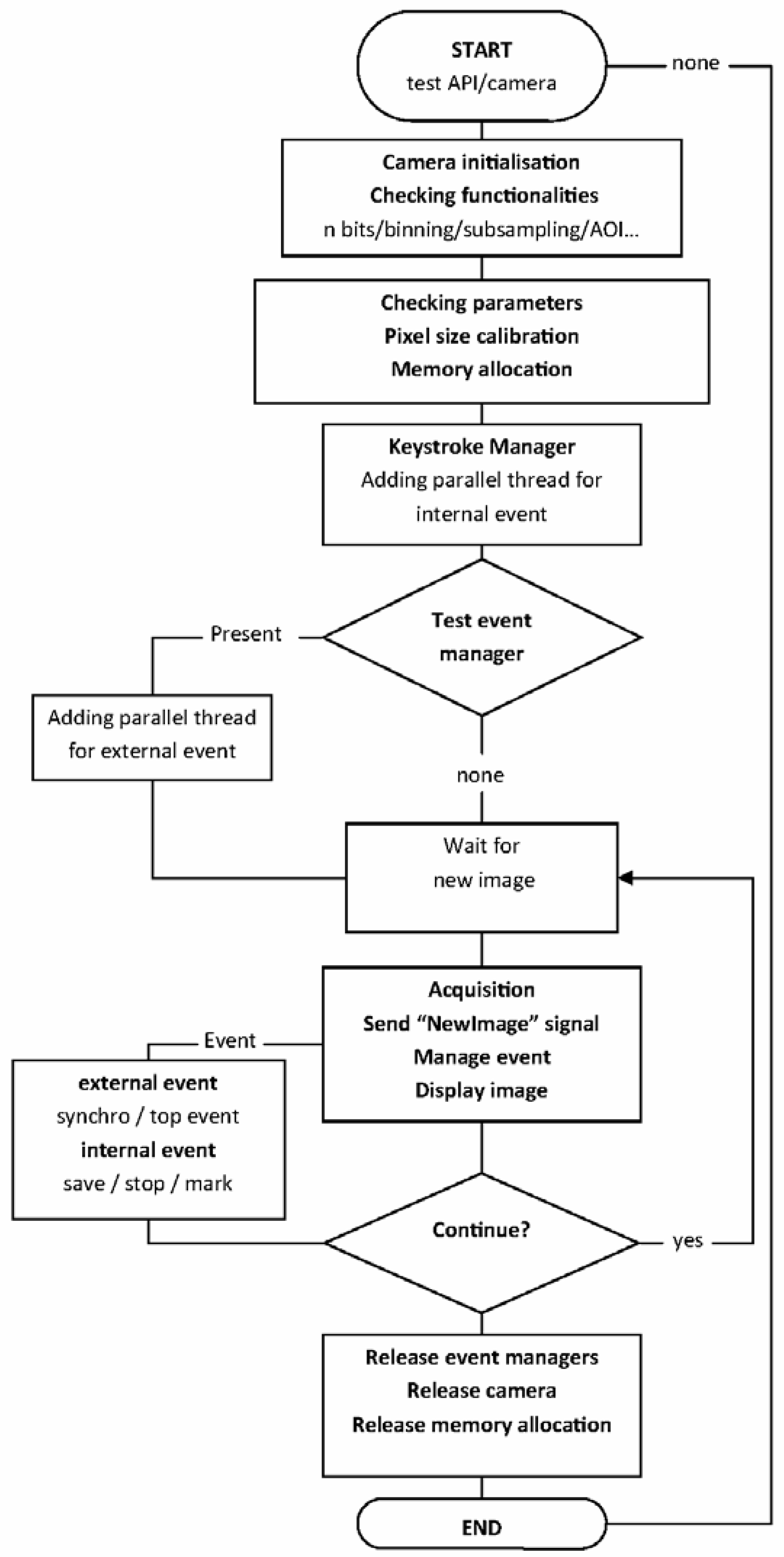
2. Materials and Methods
3. Results
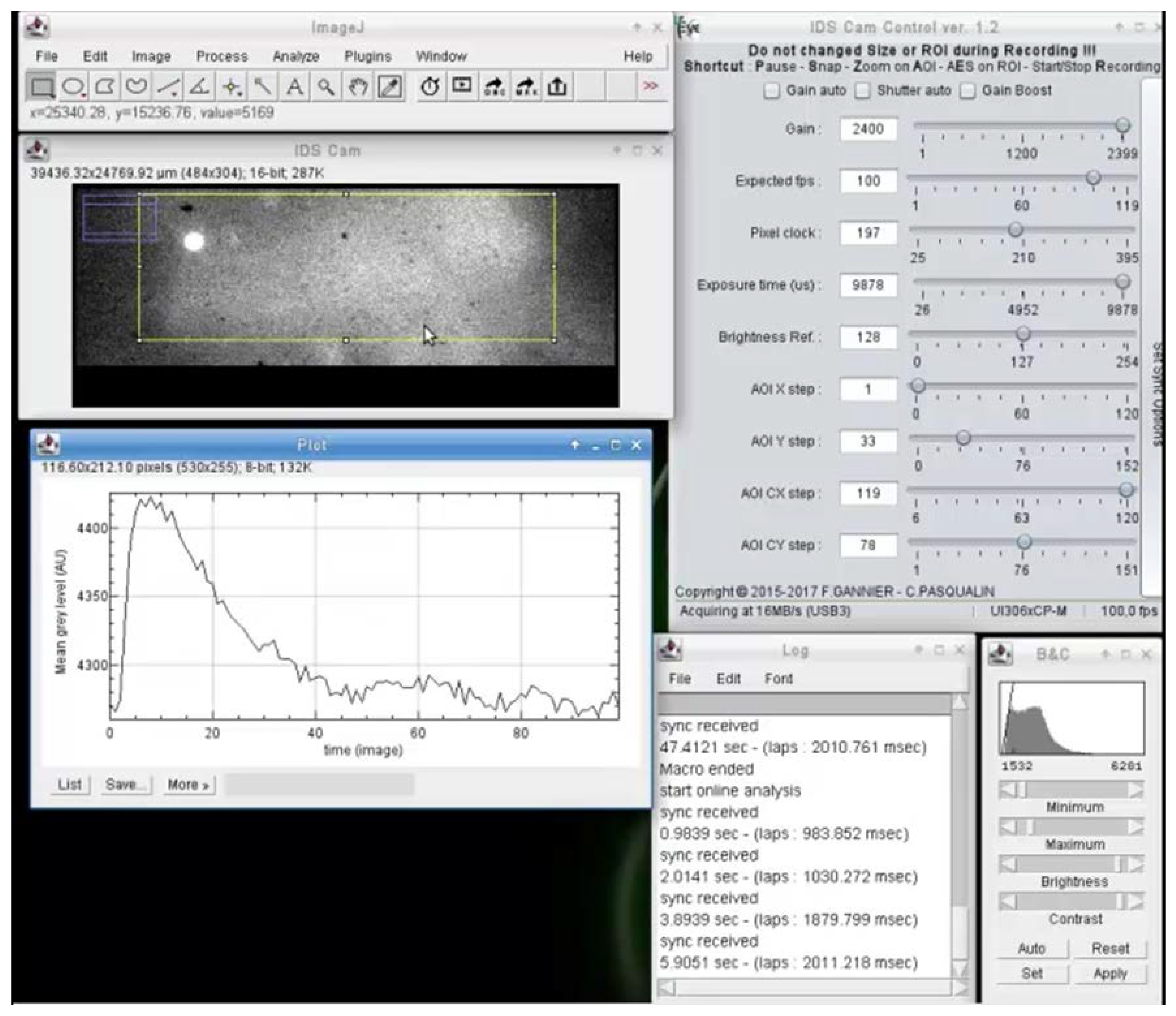
3.1. Image Acquisition Features and Outputs
3.2. Real-Time Video Analysis
3.3. Synchronizations
4. Limitations and Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Edelstein, A.D.; Tsuchida, M.A.; Amodaj, N.; Pinkard, H.; Vale, R.D.; Stuurman, N. Advanced methods of microscope control using μManager software. J. Biol. Methods 2014, 1, e10. [Google Scholar] [CrossRef] [PubMed]
- Pasqualin, C.; Gannier, F.; Yu, A.; Malécot, C.O.; Bredeloux, P.; Maupoil, V. SarcOptiM for ImageJ: high-frequency online sarcomere length computing on stimulated cardiomyocytes. Am. J. Physiol.-Cell Physiol. 2016, 311, C277–C283. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasqualin, C.; Gannier, F.; Bredeloux, P.; Maupoil, V. HF_IDS_Cam: Fast Video Capture with ImageJ for Real-Time Analysis. J. Imaging 2018, 4, 44. https://doi.org/10.3390/jimaging4020044
Pasqualin C, Gannier F, Bredeloux P, Maupoil V. HF_IDS_Cam: Fast Video Capture with ImageJ for Real-Time Analysis. Journal of Imaging. 2018; 4(2):44. https://doi.org/10.3390/jimaging4020044
Chicago/Turabian StylePasqualin, Côme, François Gannier, Pierre Bredeloux, and Véronique Maupoil. 2018. "HF_IDS_Cam: Fast Video Capture with ImageJ for Real-Time Analysis" Journal of Imaging 4, no. 2: 44. https://doi.org/10.3390/jimaging4020044
APA StylePasqualin, C., Gannier, F., Bredeloux, P., & Maupoil, V. (2018). HF_IDS_Cam: Fast Video Capture with ImageJ for Real-Time Analysis. Journal of Imaging, 4(2), 44. https://doi.org/10.3390/jimaging4020044




