Comparing Different Registration and Visualization Methods for Navigated Common Femoral Arterial Access—A Phantom Model Study Using Mixed Reality
Abstract
1. Introduction
2. Materials and Methods
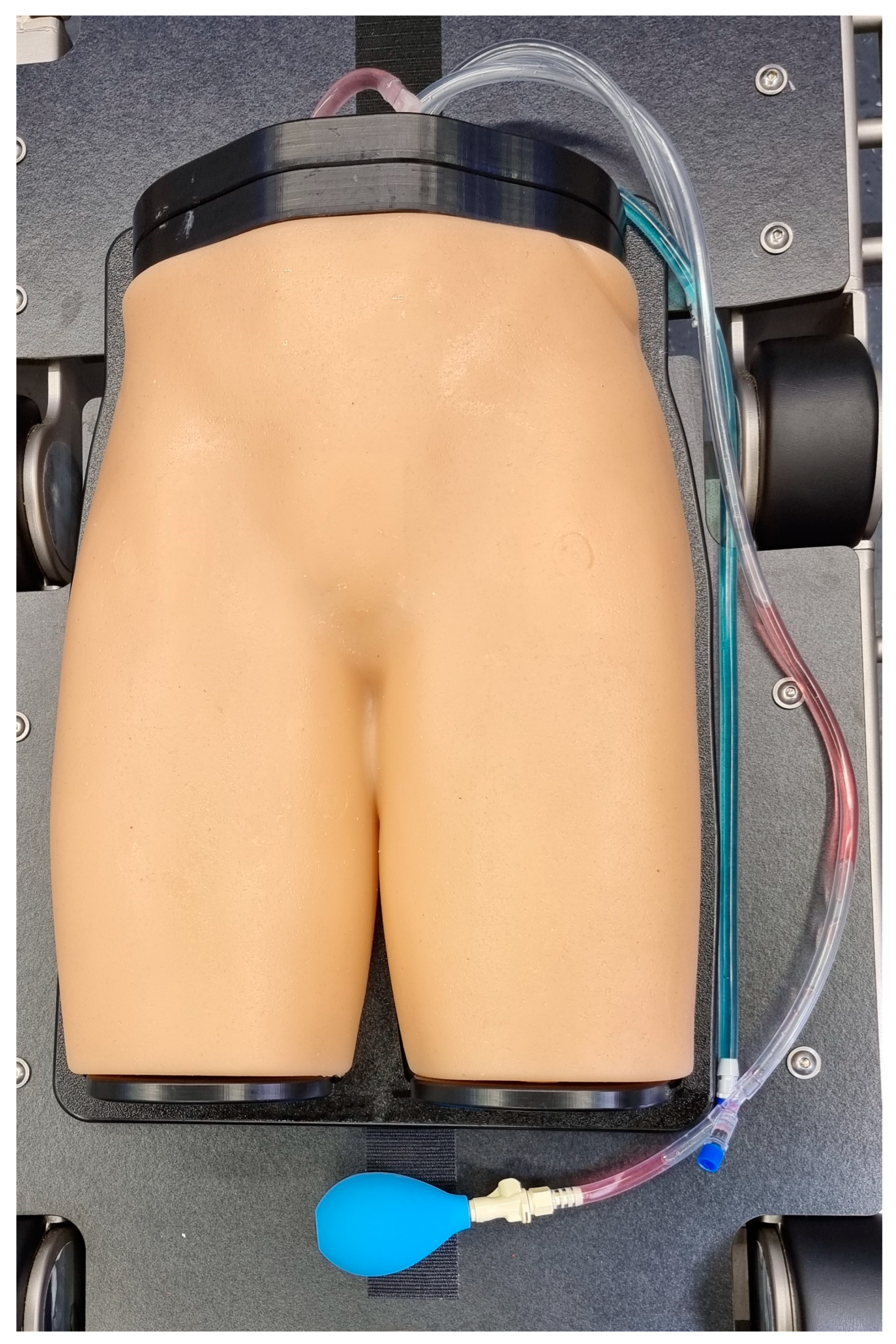
2.1. Phantom Model
2.2. Head-Mounted Display
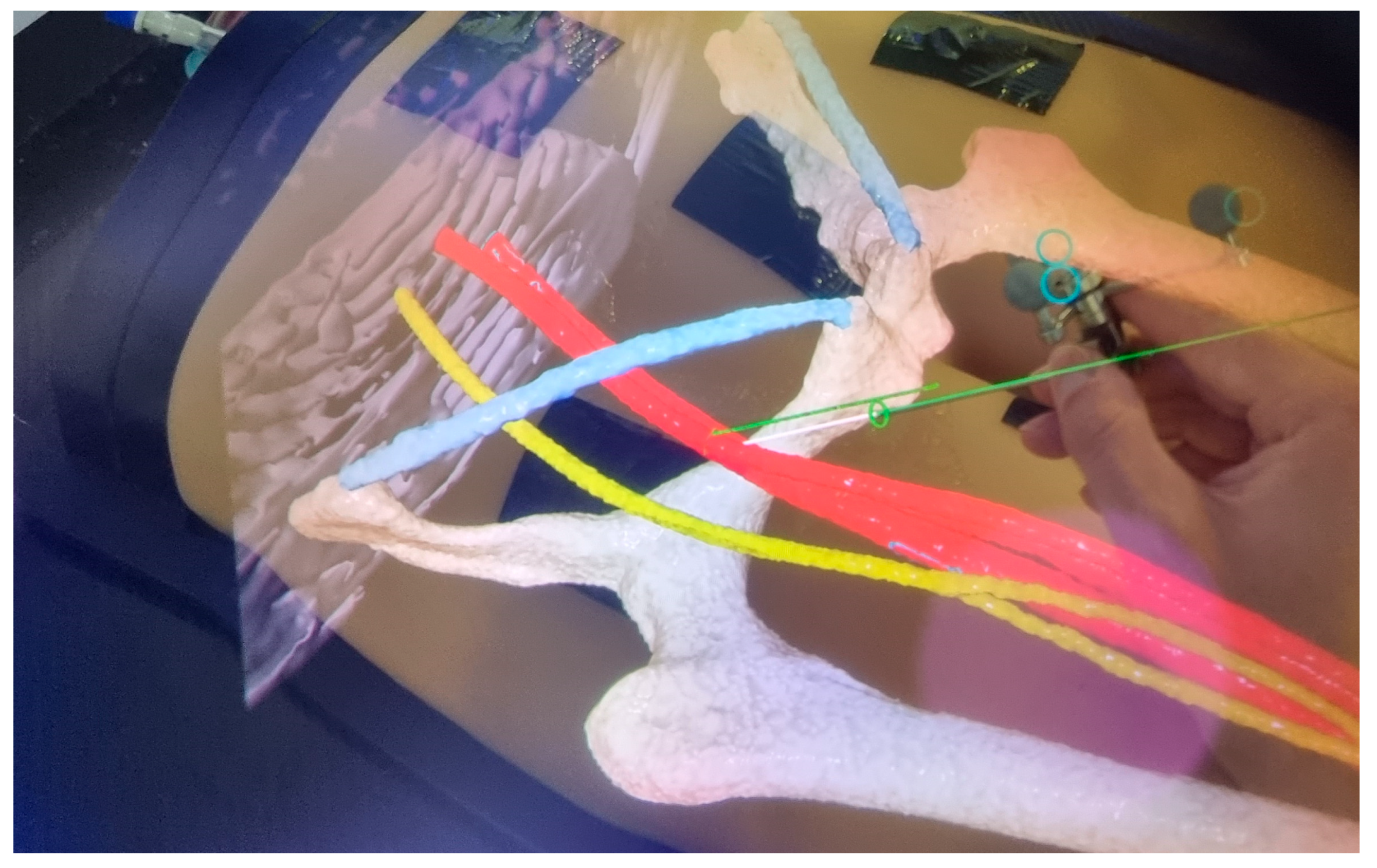
2.3. Workflows of Navigated CFA Access Techniques
2.4. System Usability of Registration and Visualization Methods
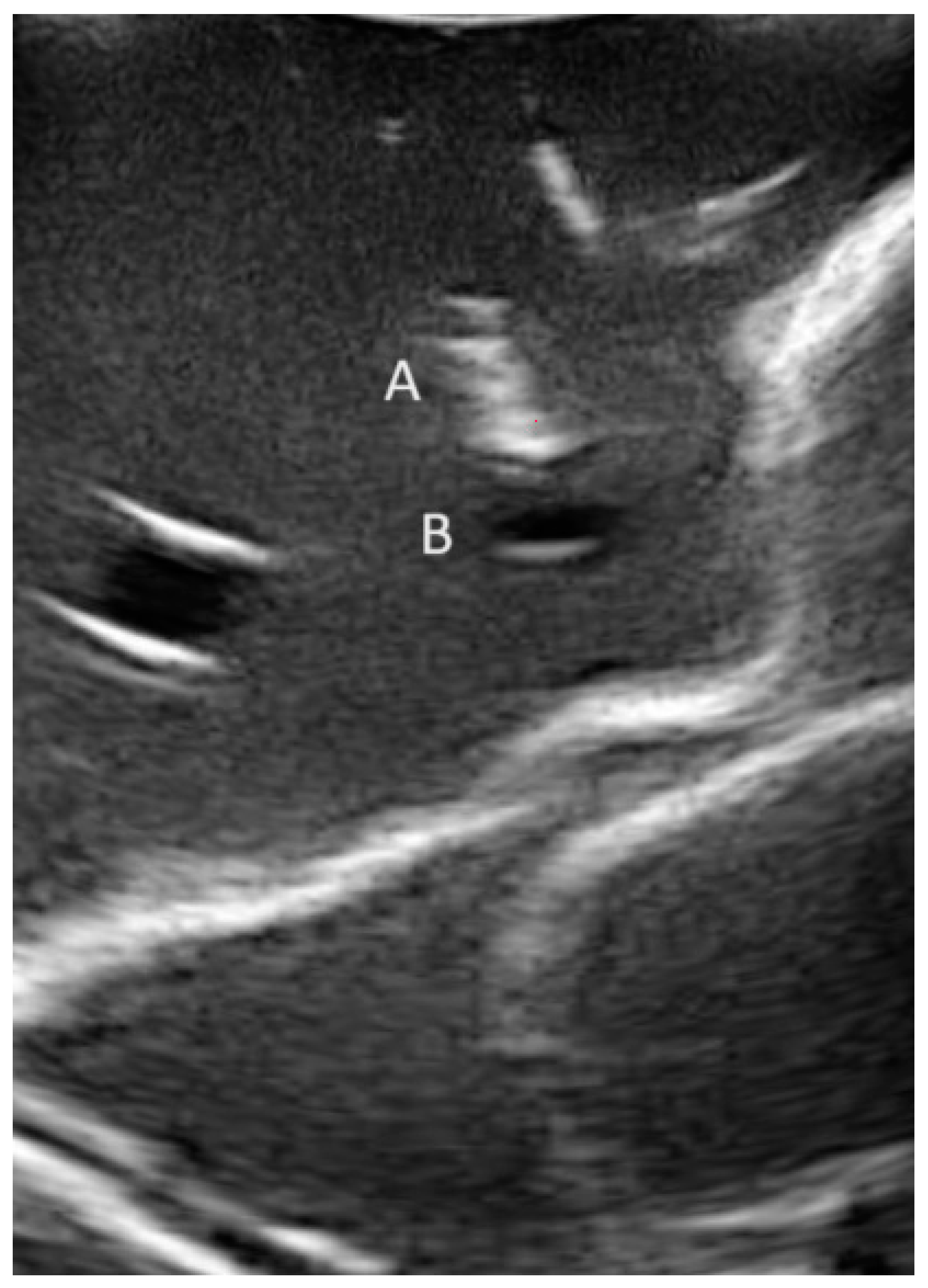
2.5. Definitions
2.5.1. Technical Success
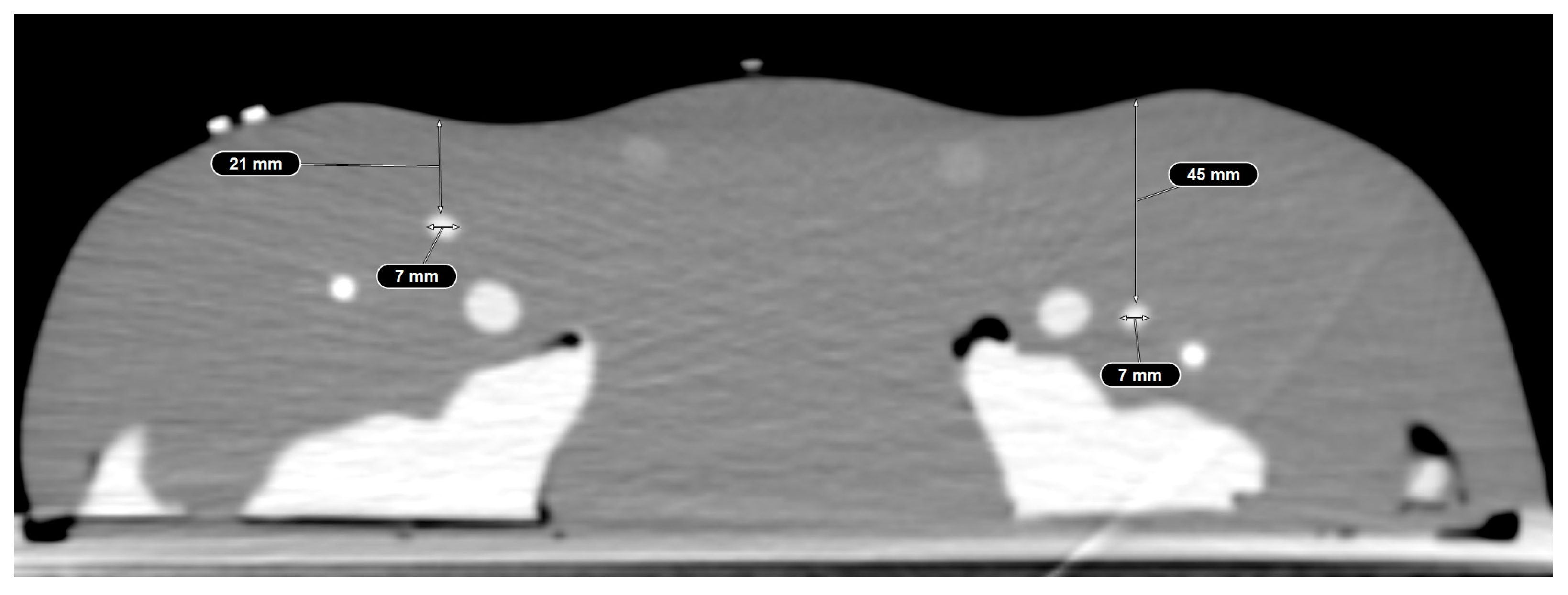
2.5.2. Positional Errors
2.6. Statistical Analysis
3. Results
3.1. Technical Success
3.2. Endpoints (1–4) in the Mixed Effects Model
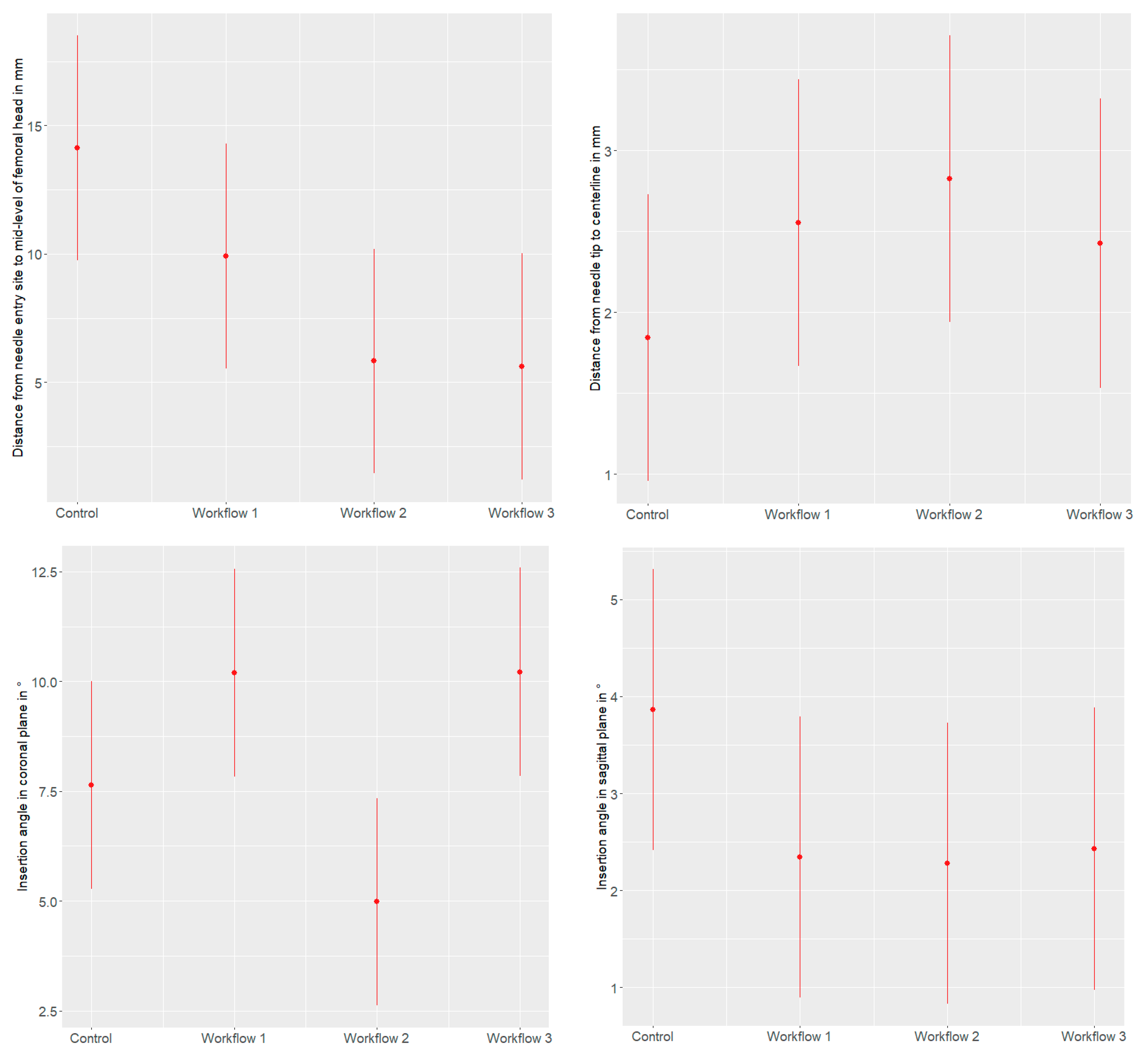
3.2.1. Distance from the Needle Entry Site to the Mid-Level of the Femoral Head
3.2.2. Distance of the Centerline to the Needle Tip in the Axial Plane
3.2.3. Insertion Angle in Coronal Plane
3.2.4. Insertion Angle in Sagittal Plane
3.3. System Usability
3.3.1. Registration
3.3.2. Visualization
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MxR | Mixed reality |
| HMD | Head-mounted display |
| CFA | Common femoral artery |
| CTA | Computed tomography angiography |
| SUS | System usability scale |
| CI | Confidence interval |
References
- Hatzl, J.; Bockler, D.; Meisenbacher, K.; Barb, A.; Hartmann, N.; Henning, D.; Uhl, C. Mixed Reality in Vascular Surgery—A Scoping Review. Zentralbl Chir. 2022, 147, 439–446. [Google Scholar] [CrossRef]
- Andress, S.; Johnson, A.; Unberath, M.; Winkler, A.F.; Yu, K.; Fotouhi, J.; Weidert, S.; Osgood, G.; Navab, N. On-the-fly augmented reality for orthopedic surgery using a multimodal fiducial. J. Med. Imaging 2018, 5, 021209. [Google Scholar] [CrossRef]
- Meulstee, J.W.; Nijsink, J.; Schreurs, R.; Verhamme, L.M.; Xi, T.; Delye, H.H.K.; Borstlap, W.A.; Maal, T.J.J. Toward Holographic-Guided Surgery. Surg. Innov. 2019, 26, 86–94. [Google Scholar] [CrossRef]
- Gregory, T.M.; Gregory, J.; Sledge, J.; Allard, R.; Mir, O. Surgery guided by mixed reality: Presentation of a proof of concept. Acta Orthop. 2018, 89, 480–483. [Google Scholar] [CrossRef]
- Cai, E.Z.; Gao, Y.; Ngiam, K.Y.; Lim, T.C. Mixed Reality Intraoperative Navigation in Craniomaxillofacial Surgery. Plast. Reconstr. Surg. 2021, 148, 686e–688e. [Google Scholar] [CrossRef]
- Jayender, J.; Xavier, B.; King, F.; Hosny, A.; Black, D.; Pieper, S.; Tavakkoli, A. A Novel Mixed Reality Navigation System for Laparoscopy Surgery. Med. Image Comput. Comput. Assist. Interv. 2018, 11073, 72–80. [Google Scholar] [CrossRef]
- Groves, L.; Li, N.; Peters, T.M.; Chen, E.C.S. Towards a First-Person Perspective Mixed Reality Guidance System for Needle Interventions. J. Imaging 2022, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, M.; Sugimoto, M.; Haruta, H.; Umezawa, A.; Kurokawa, Y. Intraoperative holography navigation using a mixed-reality wearable computer during laparoscopic cholecystectomy. Surgery 2022, 171, 1006–1013. [Google Scholar] [CrossRef]
- Porpiglia, F.; Checcucci, E.; Amparore, D.; Peretti, D.; Piramide, F.; De Cillis, S.; Piana, A.; Niculescu, G.; Verri, P.; Manfredi, M.; et al. Percutaneous Kidney Puncture with Three-dimensional Mixed-reality Hologram Guidance: From Preoperative Planning to Intraoperative Navigation. Eur. Urol. 2022, 81, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Youssef, S.; McDonnell, J.M.; Wilson, K.V.; Turley, L.; Cunniffe, G.; Morris, S.; Darwish, S.; Butler, J.S. Accuracy of augmented reality-assisted pedicle screw placement: A systematic review. Eur. Spine J. 2024, 33, 974–984. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hu, H.; Zhang, Y.; Wu, X.; Zeng, X.; Yang, J.; Fang, C. Efficacy of Augmented Reality Combined with Indocyanine Green Fluorescence Imaging Guided Laparoscopic Segmentectomy for Hepatocellular Carcinoma. J. Am. Coll. Surg. 2023, 238, 321–330. [Google Scholar] [CrossRef]
- Eves, J.; Sudarsanam, A.; Shalhoub, J.; Amiras, D. Augmented Reality in Vascular and Endovascular Surgery: Scoping Review. JMIR Serious Games 2022, 10, e34501. [Google Scholar] [CrossRef]
- Hatzl, J.; Henning, D.; Hartmann, N.; Bockler, D.; Uhl, C. A New Method for Common Femoral Arterial Access Using a Mixed Reality-Assisted Technique on a Phantom Model. J. Endovasc. Ther. 2023, 15266028231208640. [Google Scholar] [CrossRef]
- Uhl, C.; Hatzl, J.; Meisenbacher, K.; Zimmer, L.; Hartmann, N.; Bockler, D. Mixed-Reality-Assisted Puncture of the Common Femoral Artery in a Phantom Model. J. Imaging 2022, 8, 47. [Google Scholar] [CrossRef]
- Tuna Katircibasi, M.; Gunes, H.; Cagri Aykan, A.; Aksu, E.; Ozgul, S. Comparison of Ultrasound Guidance and Conventional Method for Common Femoral Artery Cannulation: A Prospective Study of 939 Patients. Acta Cardiol. Sin. 2018, 34, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Hahl, T.; Karvonen, R.; Uurto, I.; Protto, S.; Suominen, V. The Safety and Effectiveness of the Prostar XL Closure Device Compared to Open Groin Cutdown for Endovascular Aneurysm Repair. Vasc. Endovasc. Surg. 2023, 57, 848–855. [Google Scholar] [CrossRef]
- Hakeem, A.; Saqib, S.U.; Zafar, H. Return to Work in Patients with Unilateral Inguinal Hernia Surgery: A Comparative Study Between Laparoscopic Transabdominal Preperitoneal Approach and Lichtenstein Tension-Free Mesh Repair. Cureus 2023, 15, e39202. [Google Scholar] [CrossRef] [PubMed]
- Garabet, W.; Arnautovic, A.; Meurer, L.; Mulorz, J.; Rembe, J.D.; Duran, M.; Suss, J.D.; Schelzig, H.; Wagenhauser, M.U. Analysis of Determinants for Suture-mediated Closure Device Failure during EVAR Procedures. Vasc. Endovasc. Surg. 2024, 58, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Ko, Y.G.; Park, Y.; Shim, C.Y.; Hong, G.R.; Lee, S.H.; Lee, S.; Jung, H.W.; Hong, S.J.; Ahn, C.M.; et al. Risk Factors for Closure Failure following Percutaneous Transfemoral Transcatheter Aortic Valve Implantation. Ann. Vasc. Surg. 2020, 66, 406–414. [Google Scholar] [CrossRef]
- Chen, I.M.; Lee, T.H.; Chen, P.L.; Shih, C.C.; Chang, H.H. Factors in ProGlide(R) Vascular Closure Failure in Sheath Arteriotomies Greater than 16 French. Eur. J. Vasc. Endovasc. Surg. 2019, 58, 615–622. [Google Scholar] [CrossRef]
- Rijkee, M.P.; Statius van Eps, R.G.; Wever, J.J.; van Overhagen, H.; van Dijk, L.C.; Knippenberg, B. Predictors of Failure of Closure in Percutaneous EVAR Using the Prostar XL Percutaneous Vascular Surgery Device. Eur. J. Vasc. Endovasc. Surg. 2015, 49, 45–49. [Google Scholar] [CrossRef]
- Gabriel, M.; Pawlaczyk, K.; Waliszewski, K.; Krasinski, Z.; Majewski, W. Location of femoral artery puncture site and the risk of postcatheterization pseudoaneurysm formation. Int. J. Cardiol. 2007, 120, 167–171. [Google Scholar] [CrossRef]
- Frank, J.J.; Kamalakannan, D.; Kodenchery, M.; Savoy-Moore, R.T.; Rosman, H. Retroperitoneal hematoma in patients undergoing cardiac catheterization. J. Interv. Cardiol. 2010, 23, 569–574. [Google Scholar] [CrossRef]
- Fukuda, K.; Okazaki, S.; Shiozaki, M.; Okai, I.; Nishino, A.; Tamura, H.; Inoue, K.; Sumiyoshi, M.; Daida, H.; Minamino, T. Ultrasound-guided puncture reduces bleeding-associated complications, regardless of calcified plaque, after endovascular treatment of femoropopliteal lesions, especially using the antegrade procedure: A single-center study. PLoS ONE 2021, 16, e0248416. [Google Scholar] [CrossRef]
- Kent, K.C.; Moscucci, M.; Mansour, K.A.; DiMattia, S.; Gallagher, S.; Kuntz, R.; Skillman, J.J. Retroperitoneal hematoma after cardiac catheterization: Prevalence, risk factors, and optimal management. J. Vasc. Surg. 1994, 20, 905–910, discussion 910-903. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Iida, O.; Takahara, M.; Soga, Y.; Suzuki, K.; Tanigawa, T.; Ito, M.; Uematsu, M. Effect of perioperative complications after endovascular therapy in patients with peripheral artery disease due to femoropopliteal lesions. J. Vasc. Surg. 2015, 61, 1272–1277. [Google Scholar] [CrossRef]
- Spiliopoulos, S.; Karnabatidis, D.; Katsanos, K.; Diamantopoulos, A.; Ali, T.; Kitrou, P.; Cannavale, A.; Krokidis, M. Day-Case Treatment of Peripheral Arterial Disease: Results from a Multi-Center European Study. Cardiovasc. Interv. Radiol. 2016, 39, 1684–1691. [Google Scholar] [CrossRef]
- Perez-Pachon, L.; Sharma, P.; Brech, H.; Gregory, J.; Lowe, T.; Poyade, M.; Groning, F. Effect of marker position and size on the registration accuracy of HoloLens in a non-clinical setting with implications for high-precision surgical tasks. Int. J. Comput. Assist. Radiol. Surg. 2021, 16, 955–966. [Google Scholar] [CrossRef] [PubMed]
- Frisk, H.; Lindqvist, E.; Persson, O.; Weinzierl, J.; Bruetzel, L.K.; Cewe, P.; Burstrom, G.; Edstrom, E.; Elmi-Terander, A. Feasibility and Accuracy of Thoracolumbar Pedicle Screw Placement Using an Augmented Reality Head Mounted Device. Sensors 2022, 22, 522. [Google Scholar] [CrossRef] [PubMed]
- Molina, C.A.; Theodore, N.; Ahmed, A.K.; Westbroek, E.M.; Mirovsky, Y.; Harel, R.; Orru, E.; Khan, M.; Witham, T.; Sciubba, D.M. Augmented reality-assisted pedicle screw insertion: A cadaveric proof-of-concept study. J. Neurosurg. Spine 2019, 31, 139–146. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 11 February 2024).
- Brooke, J. SUS: A quick and dirty usability scale. Usability Eval. Ind. 1995, 189, 4–7. [Google Scholar]
- Condino, S.; Piazza, R.; Carbone, M.; Bath, J.; Troisi, N.; Ferrari, M.; Berchiolli, R. Bioengineering, augmented reality, and robotic surgery in vascular surgery: A literature review. Front. Surg. 2022, 9, 966118. [Google Scholar] [CrossRef] [PubMed]
- Morillas Perez, J.; Mecho Meca, S.; Caballero Galindo, G.; Miguel Perez-Llano, J. Validation of the effectiveness of augmented reality-assisted vascular puncture: An experimental model. J. Vasc. Access 2023, 11297298231156006. [Google Scholar] [CrossRef] [PubMed]


| Registration | Visualization | |
|---|---|---|
| Control | Conventional sonography | |
| Workflow 1 | Sonography-assisted (prototype) 1 | HMD 3 |
| Workflow 2 | Paired point 2 | HMD 3 |
| Workflow 3 | Sonography-assisted (prototype) 1 | Monitor 4 |
| Expert | Non-Expert | p-Value 1 | |
|---|---|---|---|
| Technical success (N = 158) [n/N (%)] Overall | 78/79 (98.7%) | 66/79 (83.5%) | |
| Right: 39/39 (100%) | Right: 35/39 (89.7%) | 0.002 | |
| Left: 39/40 (97.5%) | Left: 31/40 (77.5%) | ||
| Control | 20/20 (100%) | 19/20 (95.5%) | |
| Right: 10/10 | Right: 10/10 (100%) | 1.0 | |
| Left: 10/10 | Left: 9/10 (90%) | ||
| Navigated | 58/59 (98.3%) | 47/59 (79.7%) | |
| Right: 29/29 (100%) | Right: 25/29 (86.2%) | 0.003 | |
| Left: 29/30 (96.7%) | Left: 22/30 (73.3%) | ||
| Workflow 1 | 20/20 (100%) | 15/20 (75%) | |
| Right: 10/10 (100%) | Right: 9/10 (90%) | ||
| Left: 10/10 (100%) | Left: 6/10 (60%) | ||
| Workflow 2 | 20/20 (100%) | 16/20 (80%) | |
| Right: 10/10 (100%) | Right: 8/10 (80%) | ||
| Left: 10/10 (100%) | Left: 8/10 (80%) | ||
| Workflow 3 | 18/19 (94.7%) | 16/19 (84.2%) | |
| Right: 9/9 (100%) | Right: 8/9 (88.9%) | ||
| Left: 9/10 (90%) | Left: 8/10 (80%) |
| Distance from the Needle Entry Site to the Mid-Level of the Femoral Head (mm) | Distance of the Centerline to the Needle Tip in the Axial Plane (mm) | |||||
|---|---|---|---|---|---|---|
| Predictors | Estimates | CI | p | Estimates | CI | p |
| Intercept | 13.15 | [8.8]–[17.5] | <0.001 | 1.85 | [1.0]–[2.7] | <0.001 |
| Left side | 0.98 | [−2.2]–[4.2] | 0.550 | −0.01 | [−0.7]–[0.7] | 0.976 |
| Workflow 1 | −4.22 | [−8.7]–[0.3] | 0.068 | 0.71 | [−0.3]–[1.7] | 0.171 |
| Workflow 2 | −8.30 | [−12.8]–[−3.8] | <0.001 | 0.98 | [−0.04]–[2.0] | 0.059 |
| Workflow 3 | −8.52 | [−13.1]–[−3.9] | <0.001 | 0.59 | [−0.5]–[1.6] | 0.265 |
| Non-expert operator | 4.43 | [0.2]–[8.7] | 0.040 | 0.63 | [−0.1]–[1.4] | 0.091 |
| Random Effects | ||||||
| σ2 | 104.89 | 5.32 | ||||
| τ00operateur | 4.82 | 0.00 | ||||
| ICC | 0.04 | |||||
| Noperateur | 10 | 10 | ||||
| Observations | 158 | 158 | ||||
| Marginal R2/Conditional R2 | 0.137/0.175 | 0.041/NA | ||||
| Insertion angle in coronal plane (°) | Insertion angle in sagittal plane (°) | |||||
| Predictors | Estimates | CI | p | Estimates | CI | p |
| Intercept | 7.01 | [4.7]–[9.4] | <0.001 | 4.79 | [3.3]–[6.2] | <0.001 |
| Left side | 0.62 | [−0.9]–[2.1] | 0.413 | −0.92 | [−1.8]–[−0.01] | 0.047 |
| Workflow 1 | 2.56 | [0.5]–[4.7] | 0.017 | −1.52 | [−2.8]–[−0.2] | 0.021 |
| Workflow 2 | −2.65 | [−4.8]–[−0.6] | 0.014 | −1.59 | [−2.9]–[−0.3] | 0.016 |
| Workflow 3 | 2.58 | [0.5]–[4.7] | 0.018 | −1.44 | [−2.7]–[−0.1] | 0.031 |
| Non-expert operator | 0.51 | [−2.1]–[3.1] | 0.700 | −0.24 | [−1.8]–[1.4] | 0.767 |
| Random Effects | ||||||
| σ2 | 22.56 | 8.45 | ||||
| τ00operateur | 2.90 | 1.10 | ||||
| ICC | 0.11 | 0.11 | ||||
| Noperateur | 10 | 10 | ||||
| Observations | 158 | 158 | ||||
| Marginal R2/Conditional R2 | 0.161/0.257 | 0.066/0.173 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hatzl, J.; Henning, D.; Böckler, D.; Hartmann, N.; Meisenbacher, K.; Uhl, C. Comparing Different Registration and Visualization Methods for Navigated Common Femoral Arterial Access—A Phantom Model Study Using Mixed Reality. J. Imaging 2024, 10, 76. https://doi.org/10.3390/jimaging10040076
Hatzl J, Henning D, Böckler D, Hartmann N, Meisenbacher K, Uhl C. Comparing Different Registration and Visualization Methods for Navigated Common Femoral Arterial Access—A Phantom Model Study Using Mixed Reality. Journal of Imaging. 2024; 10(4):76. https://doi.org/10.3390/jimaging10040076
Chicago/Turabian StyleHatzl, Johannes, Daniel Henning, Dittmar Böckler, Niklas Hartmann, Katrin Meisenbacher, and Christian Uhl. 2024. "Comparing Different Registration and Visualization Methods for Navigated Common Femoral Arterial Access—A Phantom Model Study Using Mixed Reality" Journal of Imaging 10, no. 4: 76. https://doi.org/10.3390/jimaging10040076
APA StyleHatzl, J., Henning, D., Böckler, D., Hartmann, N., Meisenbacher, K., & Uhl, C. (2024). Comparing Different Registration and Visualization Methods for Navigated Common Femoral Arterial Access—A Phantom Model Study Using Mixed Reality. Journal of Imaging, 10(4), 76. https://doi.org/10.3390/jimaging10040076





