Bacterial Treatment of Recycled Concrete Aggregate
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Concrete
2.1.2. Bacterial Strains
2.1.3. Bunt and Rovira Medium
2.2. Tests
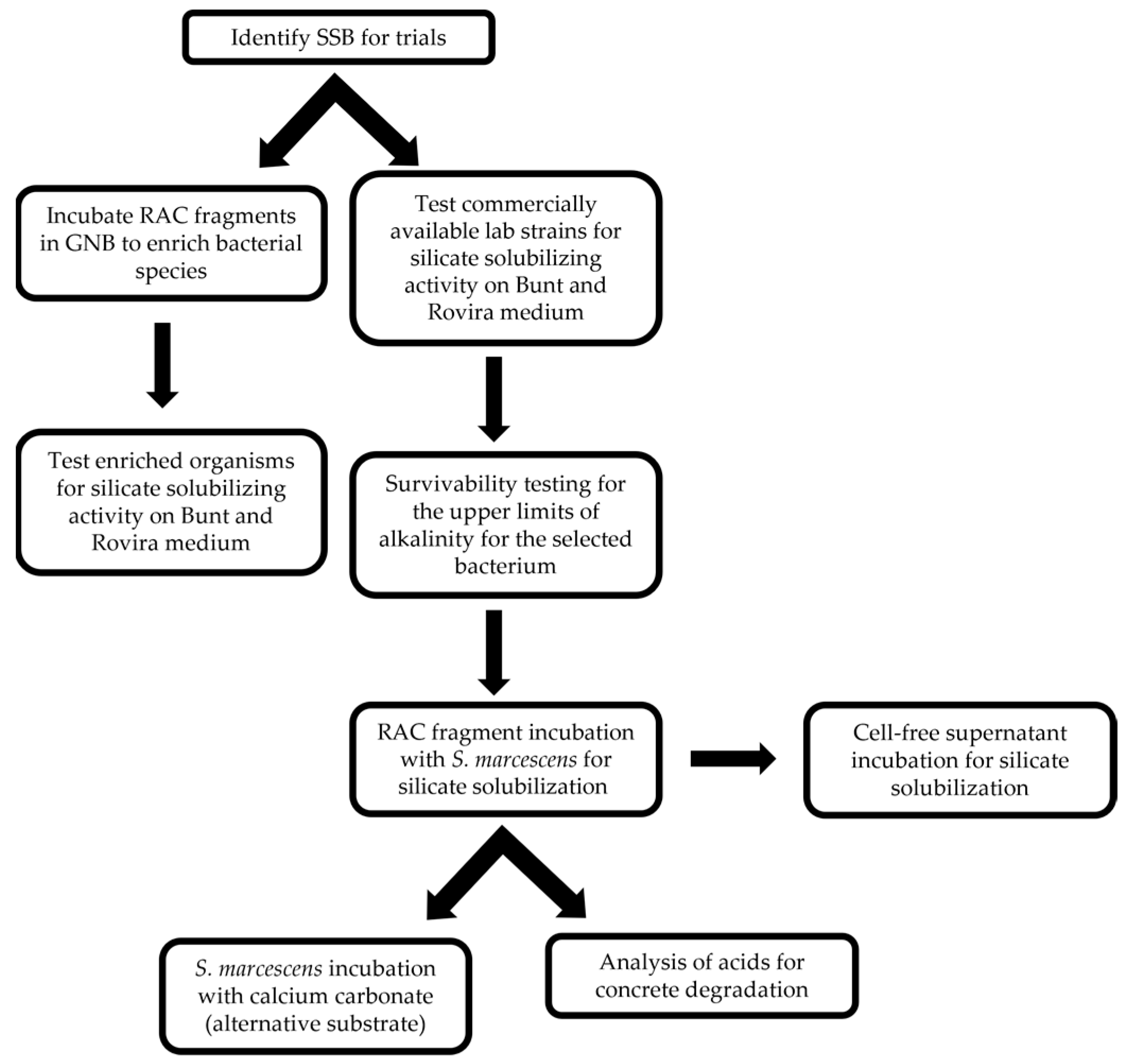
2.2.1. Identifying Silicate-Solubilizing Bacteria (SSB)
2.2.2. Selection of Bacteria
2.2.3. Identifying Favorable Environment
2.2.4. Silicate Solubilization Test
2.2.5. Supernatant-Only Trial
3. Results
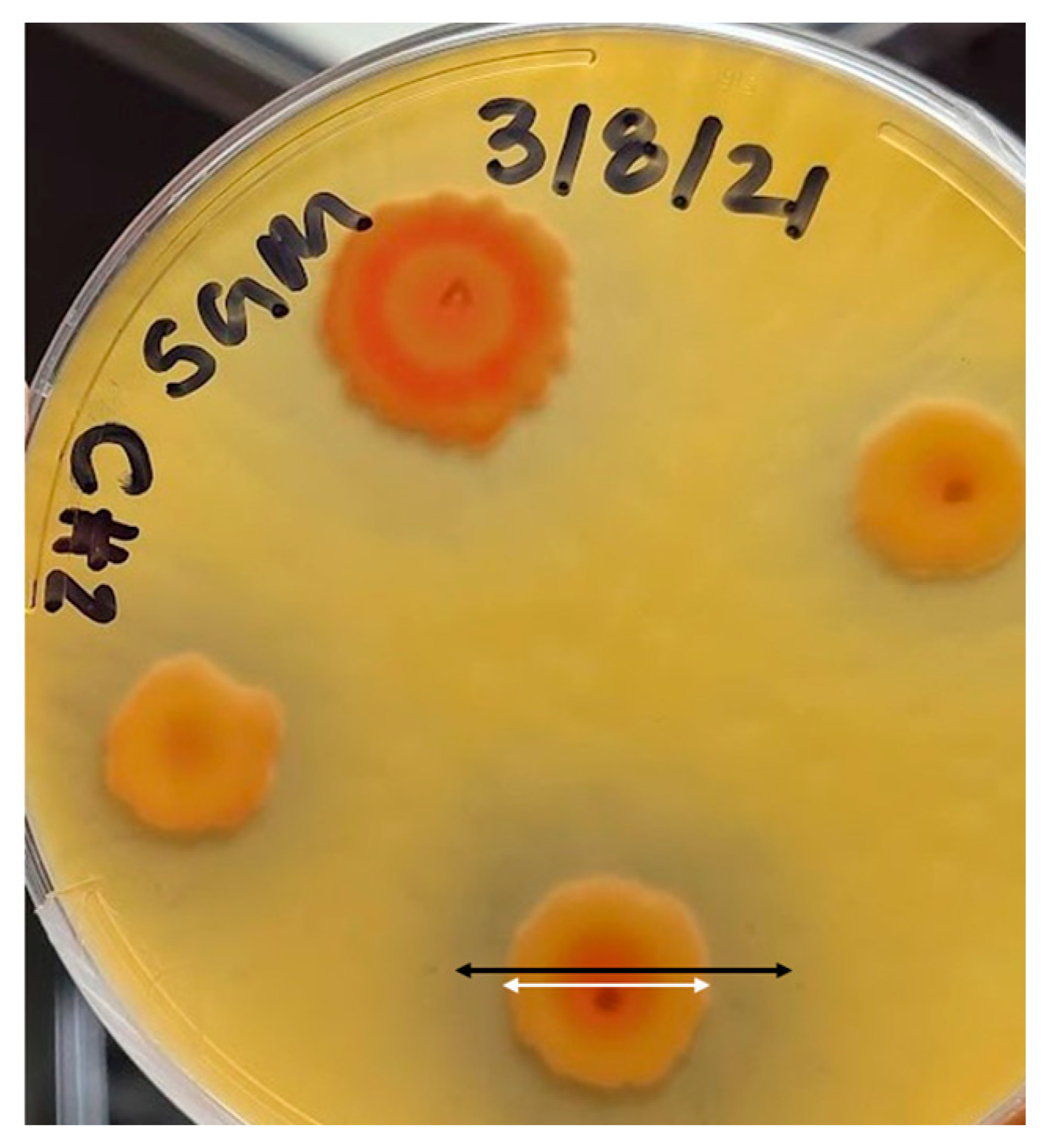
3.1. Identification of Bacteria
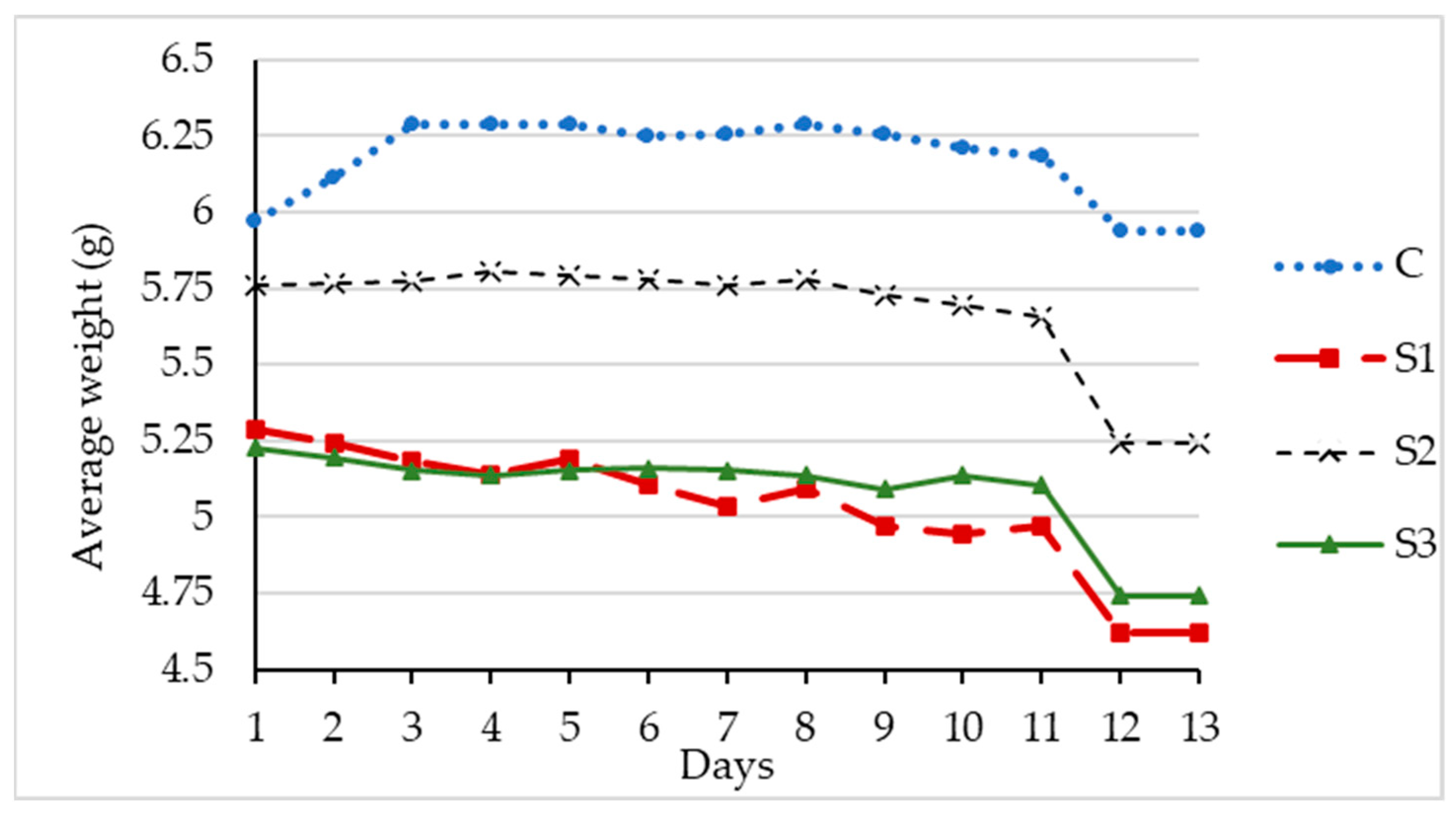
3.2. Favorable Environment for Bacteria
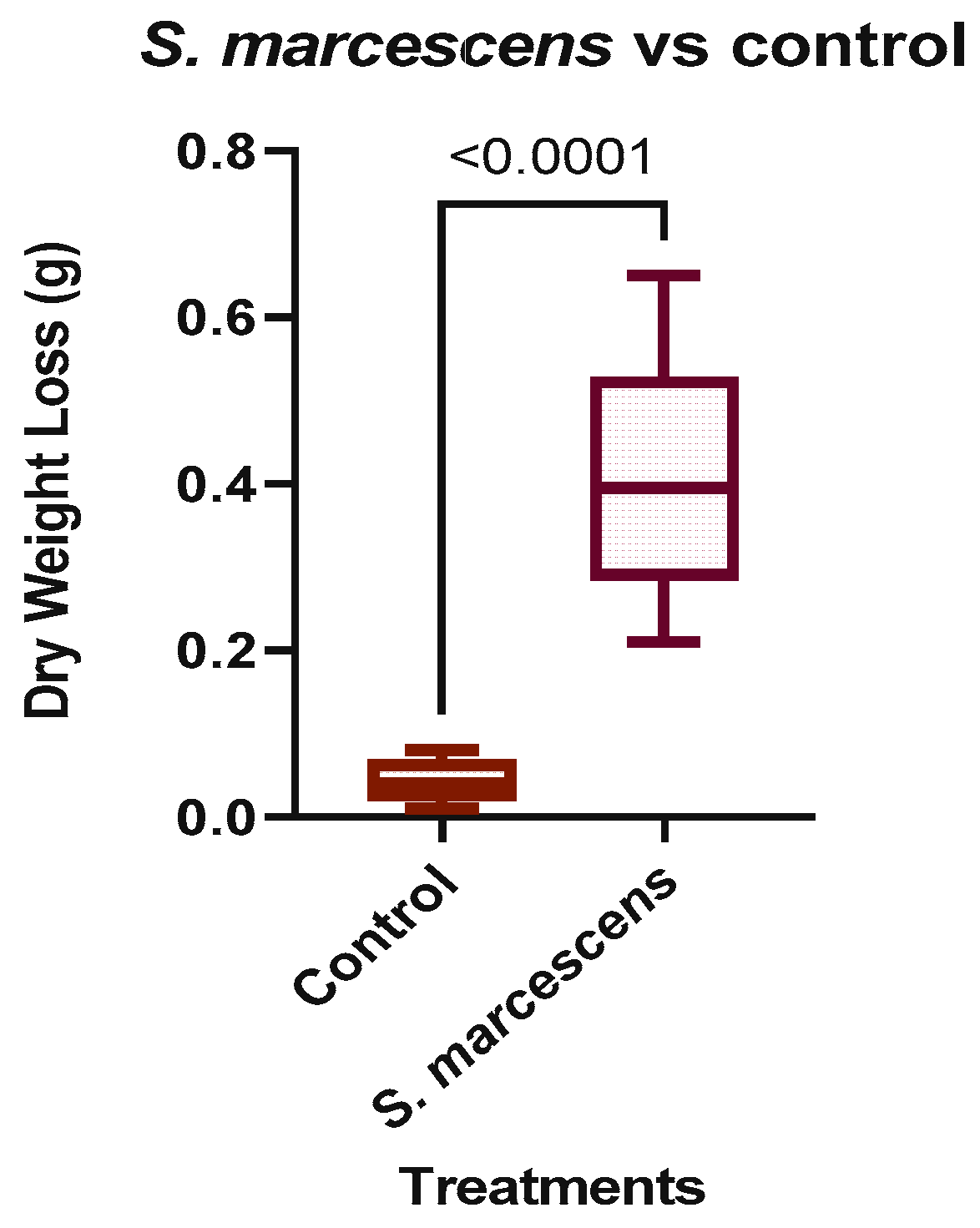
3.3. Silicate Solubilization
3.4. Testing Calcium Carbonate
3.5. Performance of Supernatant
4. Discussion
5. Conclusions
- S. marcescens bacterial strain can survive the alkaline environment in concrete and solubilize the silicates present in cement paste.
- SSB are capable of breaking down the CSH phase in cement paste by solubilizing the silicates.
- Cell-free supernatant did not significantly affect the RA, possibly due to the lower acid concentration in the supernatant.
- SSB treatment of RA to remove the adhered mortar is effective but a slow process by only reducing 10% in 14 days. Further investigation is required to speed up the process.
- The results of this research could serve as a basis for further investigation that could enhance the properties of RAC and the recycling of aggregate.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GCCA. Global Cement and Concrete Industy Announces Roadmap to Achieve Groundbreaking ‘Net Zero’ CO2 Emission by 2050. 2021. Available online: https://gccassociation.org/news/global-cement-and-concrete-industry-announces-roadmap-to-achieve-groundbreaking-net-zero-co2-emissions-by-2050/ (accessed on 30 September 2022).
- GAIN. Global Aggregates Information Network. Available online: https://www.gain.ie/ (accessed on 8 March 2023).
- Business, A. Growing Global Aggregates Sustainability. Aggreg. Bus. 2021. Available online: https://www.aggbusiness.com/feature/growing-global-aggregates-sustainably (accessed on 30 September 2022).
- Drew, L.J.; Langer, W.H.; Sachs, J.S. Environmentalism and Natural Aggregate Mining. Nat. Resour. Res. 2002, 11, 19–28. [Google Scholar] [CrossRef]
- Akhtar, A.; Sarmah, A.K. Construction and demolition waste generation and properties of recycled aggregate concrete: A global perspective. J. Clean. Prod. 2018, 186, 262–281. [Google Scholar] [CrossRef]
- Wang, R.; Yu, N.; Li, Y. Methods for improving the microstructure of recycled concrete aggregate: A review. Constr. Build. Mater. 2020, 242, 118164. [Google Scholar] [CrossRef]
- Revilla-Cuesta, V.; Skaf, M.; Faleschini, F.; Manso, J.M.; Ortega-López, V. Self-compacting concrete manufactured with recycled concrete aggregate: An overview. J. Clean. Prod. 2020, 262, 121362. [Google Scholar] [CrossRef]
- Tayeh, B.A.; Saffar, D.M.A.; Alyousef, R. The Utilization of Recycled Aggregate in High Performance Concrete: A Review. J. Mater. Res. Technol. 2020, 9, 8469–8481. [Google Scholar] [CrossRef]
- Cakir, O. Experimental analysis of properties of recycled coarse aggregate (RCA) concrete with mineral additives. Constr. Build. Mater. 2014, 68, 17–25. [Google Scholar] [CrossRef]
- Hansen, T.C. Recycling of Demolished Concrete Masonry; CRC Press: London, UK, 1992. [Google Scholar]
- Collins, R.J. The use of recycled aggregates in concrete. BRE Rep. 1994, 5, 49. [Google Scholar]
- Sagoe-Crentsil, K.K.; Brown, T.; Taylor, A.H. Performance of concrete made with commerically produced coarse recycled concrete aggregate. Cem. Concr. Res. 2001, 31, 707–712. [Google Scholar] [CrossRef]
- Sri Ravindrarajah, R.; Loo, Y.H.; Tam, C.T. Recycled Concrete as Fine and Coarse Aggregate in Concrete. Mag. Concr. Res. 1987, 39, 214–220. [Google Scholar] [CrossRef]
- Gomez-Soberon, J.M.V. Porosity of recycled concrete with substitution of recycled concrete agregate. Cem. Concr. Res. 2002, 32, 1301–1311. [Google Scholar] [CrossRef]
- Saravanakumar, P.; Abhiram, K.; Manoj, B. Properties of treated recycled aggregates and its influence on concrete strength characteristics. Constr. Build. Mater. 2016, 111, 611–617. [Google Scholar] [CrossRef]
- Tam, V.W.Y.; Tam, C.M.; Le, K.N. Removal of cement mortar remains from recycled aggregate using pre-soaking approaches. Resour. Conserv. Recycl. 2007, 50, 82–101. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.L.; Qian, X.; Chen, P.Y.; Xu, Y.; Guo, J.X. An environmentally friendly method to improve the quality of recycled concrete aggregates. Constr. Build. Mater. 2017, 144, 432–441. [Google Scholar] [CrossRef]
- Kazmi, S.M.S.; Munir, M.J.; Wu, Y.-F.; Patnaikuni, I.; Zhou, Y.; Xing, F. Effect of different aggregate treatment techniques on the freeze-thaw and sulfate resistance of recycled aggregate concrete. Cold Reg. Sci. Technol. 2020, 178, 103126. [Google Scholar] [CrossRef]
- Akbarnezhad, A.; Ong, K.C.G.; Zhang, M.H.; Tam, C.T.; Foo, T.W.J. Microwave-assisted beneficiation of recycled concrete aggregates. Constr. Build. Mater. 2011, 25, 3469–3479. [Google Scholar] [CrossRef]
- Larbi, J.A.; Heijnen, W.M.M.; Brouwer, J.P.; Mulder, E. Preliminary laboratory investigation of thermally treated recycled concrete aggregate for general use in concrete. In Waste Management Series; Woolley, G.R., Goumans, J.J.J.M., Wainwright, P.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2000; Volume 1, pp. 129–139. [Google Scholar]
- Kou, S.C.; Poon, C.S. Properties of concrete prepared with PVA-impregnated recycled concrete aggregates. Cem. Concr. Comp. 2010, 32, 649–654. [Google Scholar] [CrossRef]
- Lei, B.; Li, W.; Tang, Z.; Li, Z.; Tam, V.W.Y. Effects of environmental actions, recycled aggregate quality and modification treatments on durability performance of recycled concrete. J. Mater. Res. Technol. 2020, 9, 13375–13389. [Google Scholar] [CrossRef]
- Shi, C.J.; Li, Y.K.; Zhang, J.K.; Li, W.G.; Chong, L.L.; Xie, Z.B. Performance enhancement of recycled concrete aggregate—A review. J. Clean. Prod. 2016, 112, 466–472. [Google Scholar] [CrossRef]
- Santos, W.F.; Quattrone, M.; John, V.M.; Angulo, S.C. Roughness, wettability and water absorption of water repellent treated recycled aggregates. Constr. Build. Mater. 2017, 146, 502–513. [Google Scholar] [CrossRef]
- Spaeth, V.; Tegguer, A.D. Improvement of recycled concrete aggregate properties by polymer treatment. Int. J. Sustain. Built Environ. 2013, 2, 143–152. [Google Scholar]
- Giri, J.P.; Panda, M.; Sahoo, U.C. Performance of Bituminous Mixes Containing Treated Recycled Concrete Aggregates and Modified by Waste Polyethylene. J. Mater. Civ. Eng. 2018, 30, 04018184. [Google Scholar] [CrossRef]
- Kareem, A.I.; Nikraz, H.; Asadi, H. Application of Double-Coated Recycled Concrete Aggregates for Hot-Mix Asphalt. J. Mater. Civ. Eng. 2019, 31, 04019036. [Google Scholar] [CrossRef]
- Ma, J.; Sun, D.; Pang, Q.; Sun, G.; Hu, M.; Lu, T. Potential of recycled concrete aggregate pretreated with waste cooking oil residue for hot mix asphalt. J. Clean. Prod. 2019, 221, 469–479. [Google Scholar] [CrossRef]
- Esen, Y.; Orhan, E. Investigation of the effect on the physical and mechanical properties of the dosage of additive in self-consolidating concrete. KSCE J. Civ. Eng. 2016, 20, 2849–2858. [Google Scholar] [CrossRef]
- Katz, A. Treatments for the Improvement of Recycled Aggregate. J. Mater. Civ. Eng. 2004, 16, 597–603. [Google Scholar] [CrossRef]
- Arora, S.; Singh, S.P. Flexural fatigue performance of concrete made with recycled concrete aggregates and ternary blended cements. J. Sustain. Cem. Based Mater. 2018, 7, 182–202. [Google Scholar] [CrossRef]
- Shi, C.; Wu, Z.; Cao, Z.; Ling, T.C.; Zheng, J. Performance of mortar prepared with recycled concrete aggregate enhanced by CO2 and pozzolan slurry. Cem. Concr. Compos. 2018, 86, 130–138. [Google Scholar] [CrossRef]
- Zhang, H.; Ji, T.; Liu, H.; Su, S. Modifying recycled aggregate concrete by aggregate surface treatment using sulphoaluminate cement and basalt powder. Constr. Build. Mater. 2018, 192, 526–537. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, S.; Lu, L.; Gong, C. Evaluation of pre-coated recycled aggregate for concrete and mortar. Constr. Build. Mater. 2013, 43, 191–196. [Google Scholar] [CrossRef]
- Lee, C.-H.; Du, J.-C.; Shen, D.-H. Evaluation of pre-coated recycled concrete aggregate for hot mix asphalt. Constr. Build. Mater. 2012, 28, 66–71. [Google Scholar] [CrossRef]
- Esen, Y.; Kurt, A. Effect of High Temperature in Concrete for Different Mineral Additives and Rates. KSCE J. Civ. Eng. 2018, 22, 1288–1294. [Google Scholar] [CrossRef]
- Luo, Z.; Li, W.; Tam, V.W.Y.; Xiao, J.; Shah, S.P. Current progress on nanotechnology application in recycled aggregate concrete. J. Sustain. Cem. Based Mater. 2019, 8, 79–96. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, Y.; Meng, T.; Shah, S.P. Surface Treatment on Recycled Coarse Aggregates with Nanomaterials. J. Mater. Civ. Eng. 2016, 28, 04015094. [Google Scholar] [CrossRef]
- Mukharjee, B.B.; Barai, S.V. Influence of incorporation of nano-silica and recycled aggregates on compressive strength and microstructure of concrete. Constr. Build. Mater. 2014, 71, 570–578. [Google Scholar] [CrossRef]
- Mukharjee, B.B.; Barai, S.V. Influence of Nano-Silica on the properties of recycled aggregate concrete. Constr. Build. Mater. 2014, 55, 29–37. [Google Scholar] [CrossRef]
- Li, W.; Luo, Z.; Long, C.; Wu, C.; Duan, W.H.; Shah, S.P. Effects of nanoparticle on the dynamic behaviors of recycled aggregate concrete under impact loading. Mater. Des. 2016, 112, 58–66. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, C.; Li, Y.; Pan, X.; Poon, C.-S.; Xie, Z. Performance Enhancement of Recycled Concrete Aggregates through Carbonation. J. Mater. Civ. Eng. 2015, 27, 04015029. [Google Scholar] [CrossRef]
- Zhang, H.R.; Zhao, Y.X.; Meng, T.; Shah, S.P. The modification effects of a nano-silica slurry on microstructure, strength, and strain development of recycled aggregate concrete applied in an enlarged structural test. Constr. Build. Mater. 2015, 95, 721–735. [Google Scholar] [CrossRef]
- Shaikh, F.; Chavda, V.; Minhaj, N.; Arel, H.S. Effect of mixing methods of nano silica on properties of recycled aggregate concrete. Struct. Concr. 2018, 19, 387–399. [Google Scholar] [CrossRef]
- Hosseini, P.; Booshehrian, A.; Delkash, M.; Ghavami, S.; Zanjani, M.K. Use of Nano-SiO2 to Improve Microstructure and Compressive Strength of Recycled Aggregate Concretes. In Nanotechnology in Construction 3; Springer: Berlin, Heidelberg, 2009; pp. 215–221. [Google Scholar] [CrossRef]
- Wang, Y.; Hughes, P.; Niu, H.; Fan, Y. A new method to improve the properties of recycled aggregate concrete: Composite addition of basalt fiber and nano-silica. J. Clean. Prod. 2019, 236, 117602. [Google Scholar] [CrossRef]
- Meng, R.; Tao, M.; Huang, M.; Xu, Q. Research on Composite Strengthening Nano-Technique of Recycled Aggregate. Appl. Mech. Mater. 2012, 357–360, 2211–2214. [Google Scholar] [CrossRef]
- Li, W.; Long, C.; Tam, V.W.Y.; Poon, C.-S.; Hui Duan, W. Effects of nano-particles on failure process and microstructural properties of recycled aggregate concrete. Constr. Build. Mater. 2017, 142, 42–50. [Google Scholar] [CrossRef]
- Gao, C.; Huang, L.; Yan, L.; Jin, R.; Chen, H. Mechanical properties of recycled aggregate concrete modified by nano-particles. Constr. Build. Mater. 2020, 241, 118030. [Google Scholar] [CrossRef]
- Ying, J.; Zhou, B.; Xiao, J. Pore structure and chloride diffusivity of recycled aggregate concrete with nano-SiO2 and nano-TiO2. Constr. Build. Mater. 2017, 150, 49–55. [Google Scholar] [CrossRef]
- Moro, C.; Francioso, V.; Velay-Lizancos, M. Nano-TiO2 effects on high temperature resistance of recycled mortars. J. Clean. Prod. 2020, 263, 121581. [Google Scholar] [CrossRef]
- Mohammed, R.; Aladdin, A.; Farhad, M. Effect of Nano-Alumina on Microstructure And Mechanical Properties of Recycled Concrete; Journal of Engineering and Sustainable Development, 2018, 2, 90–103. J. Eng. Sustain. Dev. 2018, 2, 90–103. [Google Scholar]
- Fernández Bertos, M.; Simons, S.J.R.; Hills, C.D.; Carey, P.J. A review of accelerated carbonation technology in the treatment of cement-based materials and sequestration of CO2. J. Hazard. Mater. 2004, 112, 193–205. [Google Scholar] [CrossRef]
- Thiery, M.; Villain, G.; Dangla, P.; Platret, G. Investigation of the carbonation front shape on cementitious materials: Effects of the chemical kinetics. Cem. Concr. Res. 2007, 37, 1047–1058. [Google Scholar] [CrossRef]
- Tam, V.W.Y.; Wattage, H.; Le, K.N.; Buteraa, A.; Soomro, M. Methods to improve microstructural properties of recycled concrete aggregate: A critical review. Constr. Build. Mater. 2021, 270, 121490. [Google Scholar] [CrossRef]
- Liang, C.; Pan, B.; Ma, Z.; He, Z.; Duan, Z. Utilization of CO2 curing to enhance the properties of recycled aggregate and prepared concrete: A review. Cem. Concr. Compos. 2020, 105, 103446. [Google Scholar] [CrossRef]
- Grabiec, A.M.; Klama, J.; Zawal, D.; Krupa, D. Modification of recycled concrete aggregate by calcium carbonate biodeposition. Constr. Build. Mater. 2012, 34, 145–150. [Google Scholar] [CrossRef]
- García-González, J.; Rodríguez-Robles, D.; Wang, J.; De Belie, N.; Morán-del Pozo, J.M.; Guerra-Romero, M.I.; Juan-Valdés, A. Quality improvement of mixed and ceramic recycled aggregates by biodeposition of calcium carbonate. Constr. Build. Mater. 2017, 154, 1015–1023. [Google Scholar] [CrossRef]
- Wu, C.-R.; Zhu, Y.-G.; Zhang, X.-T.; Kou, S.-C. Improving the properties of recycled concrete aggregate with bio-deposition approach. Cem. Concr. Compos. 2018, 94, 248–254. [Google Scholar] [CrossRef]
- Cheng, H.L.; Wang, C.Y. The influence of sodium silicate on the properties of recycled aggregates. Gypsum Cem. Build. 2005, 12, 12–14. [Google Scholar]
- Patra, P.; Natarajan, K.A. Role of mineral specific bacterial proteins in selective flocculation and flotation. Int. J. Miner. Process. 2008, 88, 53–58. [Google Scholar] [CrossRef]
- Santhiya, D.; Subramanian, S.; Natarajan, K.A.; Rao, K.H.; Forssberg, K.S.E. Biomodulation of galena and sphalerite surfaces using Thiobacillus thiooxidans. Int. J. Miner. Process. 2001, 62, 121–141. [Google Scholar] [CrossRef]
- Sharma, P.K.; Rao, K.H. Adhesion of Paenibacillus polymyxa on chalcopyrite and pyrite: Surface thermodynamics and extended DLVO theory. Colloid. Surf. B 2003, 29, 21–38. [Google Scholar] [CrossRef]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–643. [Google Scholar] [CrossRef]
- Lauwers, A.M.; Heinen, W. Biodegradation and Utilization of Silica and Quartz. Arch. Microbiol. 1974, 95, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Vasanthi, N.; Saleena, L.M.; Raj, S.A. Silica Solubilization Potential of Certain Bacterial Species in the Presence of Different Silicate Minerals. Silicon 2016, 10, 267–275. [Google Scholar] [CrossRef]
- Rashid, H.; Nawaz, H.; Bhatti, T.M. Bioleaching studies of bauxite ore using Aspergillus niger. J. Biol. Sci. 2001, 1, 501–504. [Google Scholar]
- Zhan, S.F.; Liu, J.J.; Chen, Y.; Sun, D.S. Single and Coorperative Bauxite Bioleaching by Silicate Bacteria. In Proceedings of the 2013 International Conference on Agricultural and Natural Resources Engineering (Icanre 2013), Singapore, 1–2 May 2013; Volume 5, p. 172. [Google Scholar] [CrossRef]
- Krylova, L.N.; Gusakov, M.S.; Adamov, E.V.; Vainshtein, M.B. Application of bacterial-chemical oxidation for processing of nickel-containing raw materials. Russ. J. Non-Ferr. Met. 2011, 52, 410–415. [Google Scholar] [CrossRef]
- Yang, Z.C.; Feng, Y.L.; Li, H.R.; Wang, W.D.; Teng, Q. Effect of biological pretreatment on flotation recovery of pyrolusite. Trans. Nonferrous Met. Soc. China 2014, 24, 1571–1577. [Google Scholar] [CrossRef]
- Vandevivere, P.; Welch, S.A.; Ullman, W.J.; Kirchman, D.L. Enhanced Dissolution of Silicate Minerals by Bacteria at near-Neutral Ph. Microb. Ecol. 1994, 27, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Santelli, C.M.; Welch, S.A.; Westrich, H.R.; Banfield, J.F. The effect of Fe-oxidizing bacteria on Fe-silicate mineral dissolution. Chem. Geol. 2001, 180, 99–115. [Google Scholar] [CrossRef]
- Banfield, J.F.; Barker, W.W.; Welch, S.A.; Taunton, A. Bilogical impact on mineral dissolution: Application of the lichen model to understanding mineral weathering in the rhizosphere. Proc. Natl. Acad. Sci. USA 1999, 96, 3404–3411. [Google Scholar]
- Teng, Q.; Feng, Y.L.; Li, H.R. Effects of silicate-bacteria pretreatment on desiliconization of magnesite by reverse flotation. Colloid. Surf. A 2018, 544, 60–67. [Google Scholar] [CrossRef]
- Brown, J.; Chen, C.; Fernández, M.; Carr, D. Urban endoliths: Incidental microbial communities occurring inside concrete. AIMS Microbiol. 2023, 2, 277–312. [Google Scholar] [CrossRef]
- Mohamed, E.; Farag, A.; Youssef, S. Phosphate Solubilization by Bacillus subtilis and Serratia marcescens Isolated from Tomato Plant Rhizosphere. J. Environ. Prot. 2018, 9, 266–277. [Google Scholar] [CrossRef]
- Solé, M.; Rius, N.; Lorén, J.G. Rapid extracellular acidification induced by glucose metabolism in non-proliferating cells of Serratia marcescens. Int. Microbiol. 2000, 3, 39–43. [Google Scholar]
- Bunt, J.S.; Rovira, A.D. Microbiological Studies of Some Subantarctic Soils. J. Soil Sci. 1955, 1, 119–128. [Google Scholar] [CrossRef]
- Pastore, G.; Kernchen, S.; Spohn, M. Microbial solubilization of silicon and phosphorus from bedrock in relation to abundance of phosphorus-solubilizing bacteria in temperate forest soils. Soil Biol. Biochem. 2020, 151, 108050. [Google Scholar] [CrossRef]
- Kang, S.-M.; Waqas, M.; Shahzad, R.; You, Y.-H.; Asaf, S.; Khan, M.A.; Lee, K.-E.; Joo, G.-J.; Kim, S.-J.; Lee, I.-J. Isolation and characterization of a novel silicate-solubilizing bacterial strain Burkholderia eburnea CS4-2 that promotes growth of japonica rice (Oryza sativa L. cv. Dongjin). Soil.Sci. Plant Nutr. 2017, 63, 233–241. [Google Scholar] [CrossRef]
- Chandrakala, C.; Voleti, S.R.; Bandeppa, S.; Sunil Kumar, N.; Latha, P.C. Silicate Solubilization and Plant Growth Promoting Potential of Rhizobium sp. Isolated from Rice Rhizosphere. Silicon 2019, 11, 2895–2906. [Google Scholar] [CrossRef]
- Hollmann, B.; Perkins, M.; Chauhan, V.M.; Aylott, J.W.; Hardie, K.R. Fluorescent nanosensors reveal dynamic pH gradients during biofilm formation. npj Biofilms Microbiomes 2021, 7, 50. [Google Scholar] [CrossRef]
- Shi, C.; Stegemann, J.A. Acid corrosion resistance of different cementing materials. Cem. Concr. Res. 2000, 30, 803–808. [Google Scholar] [CrossRef]
- Lee, K.-E.; Adhikari, A.; Kang, S.-M.; You, Y.-H.; Joo, G.-J.; Kim, J.-H.; Kim, S.-J.; Lee, I.-J. Isolation and Characterization of the High Silicate and Phosphate Solubilizing Novel Strain Enterobacter ludwigii GAK2 that Promotes Growth in Rice Plants. Agronomy 2019, 9, 144. Available online: https://www.mdpi.com/2073-4395/9/3/144 (accessed on 8 August 2021). [CrossRef]
- Grimont, F.; Grimont, P.A.D. The Genus Serratia. In The Prokaryotes: A Handbook on the Biology of Bacteria Volume 6: Proteobacteria: Gamma Subclass; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 219–244. [Google Scholar] [CrossRef]



| Ingredient | Amount Per Liter |
|---|---|
| Difco powdered nutrient broth | 8 g |
| α-D(+)-Glucose | 100 g |
| Bromothymol Blue (BTB) | 48 mg |
| Optional: Agar (for plates) | 15 g |
| Ingredient | Amount Per Liter |
|---|---|
| Peptone | 10 g |
| Glucose | 20 g |
| Magnesium chloride | 0.1 g |
| Ferric chloride | 0.01 g |
| Yeast extract | 1 g |
| Ammonium sulphate | 0.5 g |
| Disodium hydrogen phosphate | 0.4 g |
| Agar | 20 g |
| Insoluble magnesium trisilicate | 0.25% of the volume |
| Culture ID | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Starting Mass (g) | 5.28 | 5.76 | 5.76 | 5.23 | 4.31 | 5.28 | 5.76 | 5.23 | 4.31 | 8.05 |
| Final Mass (g) | 4.94 | 5.46 | 5.58 | 4.89 | 3.86 | 4.63 | 5.24 | 4.74 | 3.76 | 7.81 |
| Δ Mass (g) | 0.34 | 0.30 | 0.21 | 0.34 | 0.45 | 0.65 | 0.52 | 0.49 | 0.55 | 0.24 |
| Δ Mass (%) | 6.44 | 5.21 | 3.63 | 6.50 | 10.44 | 12.31 | 9.03 | 9.37 | 12.76 | 2.98 |
| Average Δ Mass (g) | 0.409 | |||||||||
| Average Δ Mass (%) | 7.87 |
| Culture ID | C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 |
|---|---|---|---|---|---|---|---|---|
| Starting Mass (g) | 5.10 | 5.59 | 4.79 | 4.55 | 4.65 | 4.34 | 5.96 | 5.96 |
| Final Mass (g) | 5.07 | 5.58 | 4.72 | 4.47 | 4.63 | 4.29 | 5.90 | 5.94 |
| Δ Mass (g) | 0.03 | 0.01 | 0.07 | 0.08 | 0.02 | 0.05 | 0.06 | 0.02 |
| Δ Mass (%) | 0.59 | 0.18 | 1.46 | 1.76 | 0.43 | 1.15 | 1.01 | 0.34 |
| Average Δ Mass (g) | 0.04 | |||||||
| Average Δ Mass (%) | 0.86 |
| Culture ID | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Starting Mass (g) | 6.69 | 5.01 | 6.80 | 4.45 | 4.70 | 7.63 |
| Final Mass (g) | 6.66 | 4.97 | 6.77 | 4.43 | 4.69 | 7.58 |
| Δ Mass (g) | 0.03 | 0.04 | 0.03 | 0.02 | 0.01 | 0.05 |
| Δ Mass (%) | 0.45 | 0.80 | 0.44 | 0.45 | 0.21 | 0.66 |
| Average Δ Mass (g) | 0.03 | |||||
| Average Δ Mass (%) | 0.50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rowell, A.; Ghebrab, T.; Jeter, R. Bacterial Treatment of Recycled Concrete Aggregate. Recycling 2023, 8, 68. https://doi.org/10.3390/recycling8050068
Rowell A, Ghebrab T, Jeter R. Bacterial Treatment of Recycled Concrete Aggregate. Recycling. 2023; 8(5):68. https://doi.org/10.3390/recycling8050068
Chicago/Turabian StyleRowell, Austen, Tewodros Ghebrab, and Randall Jeter. 2023. "Bacterial Treatment of Recycled Concrete Aggregate" Recycling 8, no. 5: 68. https://doi.org/10.3390/recycling8050068
APA StyleRowell, A., Ghebrab, T., & Jeter, R. (2023). Bacterial Treatment of Recycled Concrete Aggregate. Recycling, 8(5), 68. https://doi.org/10.3390/recycling8050068






