Extraction of Antioxidant Phenolics from Agri-Food Waste Biomass Using a Newly Designed Glycerol-Based Natural Low-Transition Temperature Mixture: A Comparison with Conventional Eco-Friendly Solvents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Low Transition Temperature Mixture (LTTM) Synthesis
2.3. Agri-Food Wastes
2.4. Extraction Procedure
2.5. The Determination of Yields in Total Polyphenols (YTP), Total Chlorogenates (YTCg), and Total Flavonoids (YTFn)
2.6. Antioxidant Activity Assays
2.7. Statistical Analysis
3. Results and Discussion
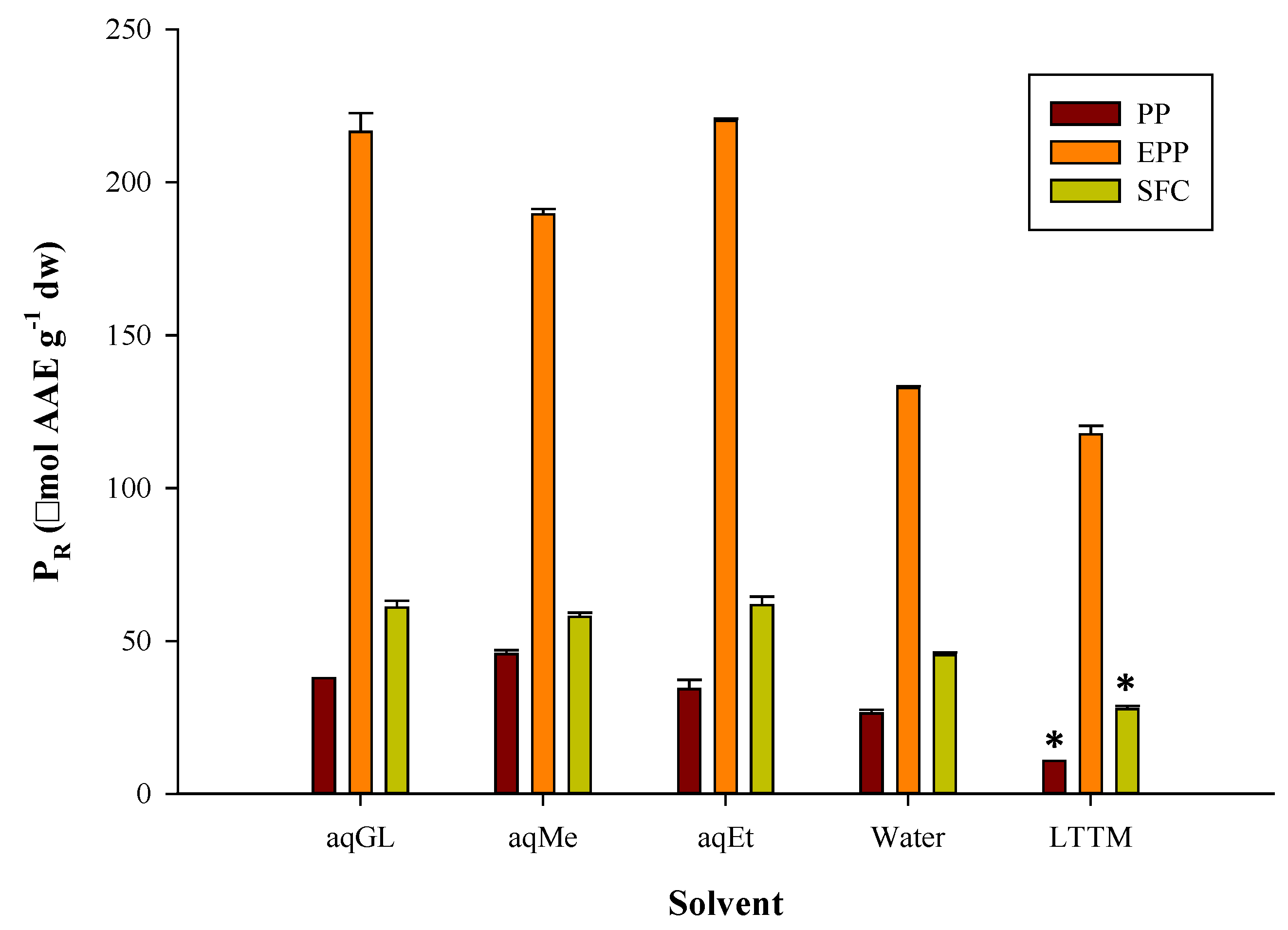
3.1. Extraction Efficiency
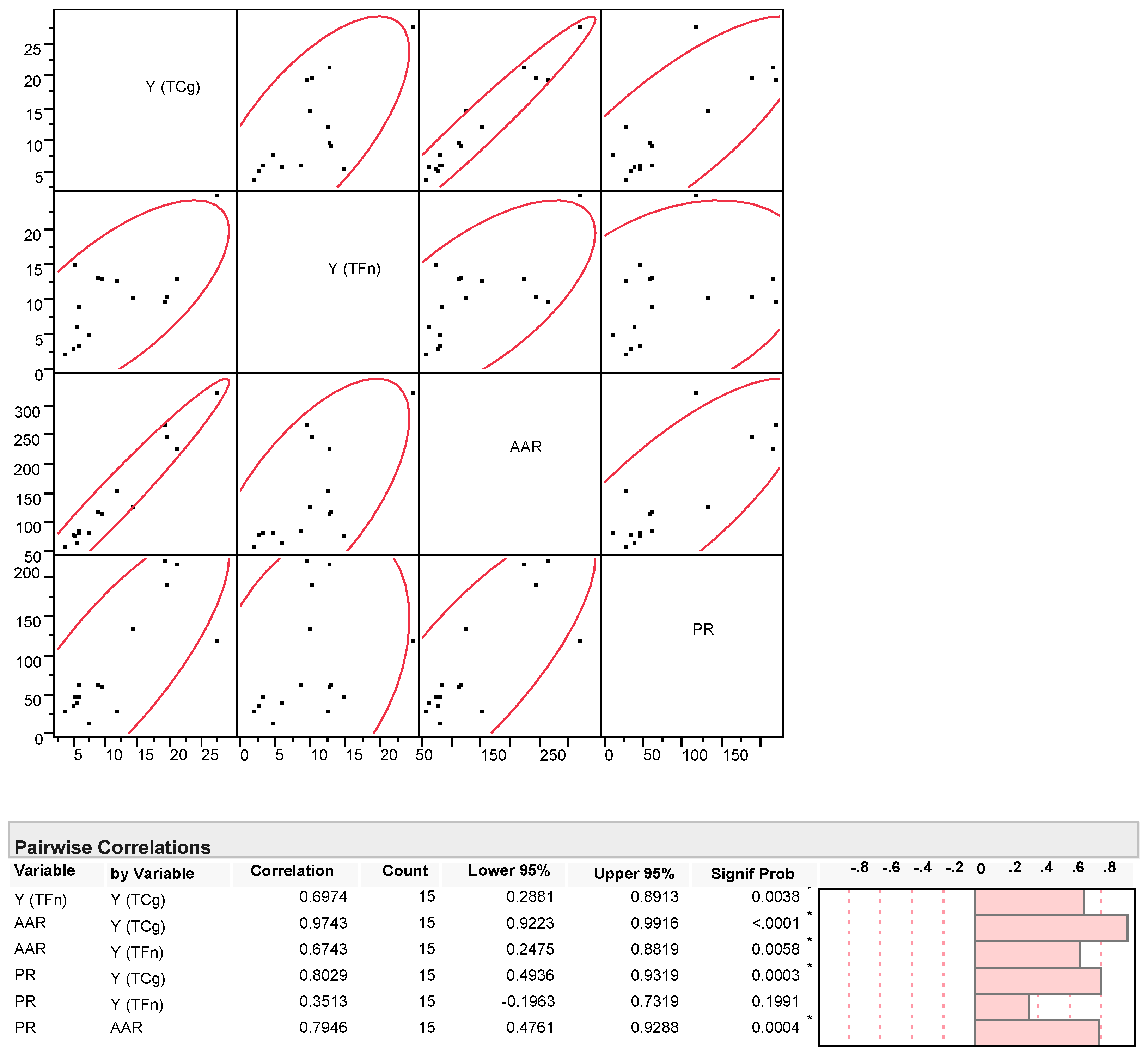
3.2. Polyphenolic Profile and Antioxidant Activity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Nomenclature
| AAR | antiradical activity (μmol·DPPH·g−1) |
| CTFn | total flavonoid concentration (mg·RtE·L−1) |
| PR | reducing power (μmol·AAE·g−1) |
| RL/S | liquid-to-solid ratio (mL·g−1) |
| YTCg | yield in total chlorogenates (mg·CGAE·g−1) |
| YTFn | yield in total flavonoids (mg·RtE·g−1) |
| YTP | yield in total polyphenols (mg·g−1) |
Abbreviations
| AAE | ascorbic acid equivalents |
| DPPH• | 2,2-diphenyl-picrylhydrazyl radical |
| dw | dry weight |
| EPP | eggplant peels |
| HBA | hydrogen bond acceptor |
| HBD | hydrogen bond donor |
| LTTM | low-transition temperature mixture |
| PP | potato peels |
| SFC | spent filter coffee |
| TPTZ | 2,4,6-tripyridyl-s-triazine |
References
- Mirabella, N.; Castellani, V.; Sala, S. Current options for the valorization of food manufacturing waste: A review. J. Clean. Prod. 2014, 65, 28–41. [Google Scholar] [CrossRef]
- Makris, D.P.; Boskou, D. Plant-derived antioxidants as food additives. In Plants as a Source of Natural Antioxidants; Dubei, N.K., Ed.; CABI Publishers: Oxfordshire, UK, 2014; pp. 169–190. [Google Scholar]
- Babbar, N.; Oberoi, H.S.; Sandhu, S.K. Therapeutic and nutraceutical potential of bioactive compounds extracted from fruit residues. Crit. Rev. Food Sci. Nutr. 2015, 55, 319–337. [Google Scholar] [CrossRef] [PubMed]
- Peralbo-Molina, Á.; de Castro, M.D.L. Potential of residues from the Mediterranean agriculture and agrifood industry. Trends Food Sci. Technol. 2013, 32, 16–24. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Namieśnik, J. Ionic liquids and deep eutectic mixtures: Sustainable solvents for extraction processes. Chem. Sus. Chem. 2014, 7, 1784–1800. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; van Spronsen, J.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N. Chlorogenic acids and other cinnamates–nature, occurrence, dietary burden, absorption and metabolism. J. Sci. Food Agric. 2000, 80, 1033–1043. [Google Scholar] [CrossRef]
- Shahidi, F.; Chandrasekara, A. Hydroxycinnamates and their in vitro and in vivo antioxidant activities. Phytochem. Rev. 2010, 9, 147–170. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Mouratoglou, E.; Malliou, V.; Makris, D.P. Novel glycerol-based natural eutectic mixtures and their efficiency in the ultrasound-assisted extraction of antioxidant polyphenols from agri-food waste biomass. Waste Biomass Valoriz. 2016. [Google Scholar] [CrossRef]
- Michail, A.; Sigala, P.; Grigorakis, S.; Makris, D.P. Kinetics of ultrasound-assisted polyphenol extraction from spent filter coffee using aqueous glycerol. Chem. Eng. Commun. 2015, 203, 407–413. [Google Scholar] [CrossRef]
- Philippi, K.; Tsamandouras, N.; Grigorakis, S.; Makris, D.P. Ultrasound-assisted green extraction of eggplant peel (Solanum melongena) polyphenols using aqueous mixtures of glycerol and ethanol: Optimisation and kinetics. Environ. Proc. 2016, 3. [Google Scholar] [CrossRef]
- Paleologou, I.; Vasiliou, A.; Grigorakis, S.; Makris, D.P. Optimisation of a green ultrasound-assisted extraction process for potato peel (Solanum tuberosum) polyphenols using bio-solvents and response surface methodology. Biomass Convers. Biorefin. 2016, 9. [Google Scholar] [CrossRef]
- Apostolakis, A.; Grigorakis, S.; Makris, D.P. Optimisation and comparative kinetics study of polyphenol extraction from olive leaves (Olea europaea) using heated water/glycerol mixtures. Sep. Purif. Technol. 2014, 128, 89–95. [Google Scholar] [CrossRef]
- Makris, D.P. Kinetics of ultrasound-assisted flavonoid extraction from agri-food solid wastes using water/glycerol mixtures. Resources 2016, 5. [Google Scholar] [CrossRef]
- Karakashov, B.; Grigorakis, S.; Loupassaki, S.; Mourtzinos, I.; Makris, D. Optimisation of organic solvent-free polyphenol extraction from Hypericum triquetrifolium Turra using Box–Behnken experimental design and kinetics. Int. J. Ind. Chem. 2015, 2, 85–92. [Google Scholar] [CrossRef]
- Shehata, E.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Extraction optimisation using water/glycerol for the efficient recovery of polyphenolic antioxidants from two Artemisia species. Sep. Purif. Technol. 2015, 149, 462–469. [Google Scholar] [CrossRef]
- Abbott, A.P.; Harris, R.C.; Ryder, K.S.; D’Agostino, C.; Gladden, L.F.; Mantle, M.D. Glycerol eutectics as sustainable solvent systems. Green Chem. 2011, 13, 82–90. [Google Scholar] [CrossRef]
- Bakirtzi, C.; Triantafyllidou, K.; Makris, D.P. Novel lactic acid-based natural deep eutectic solvents: Efficiency in the ultrasound-assisted extraction of antioxidant polyphenols from common native Greek medicinal plants. J. Appl. Res. Med. Aromat. Plants 2016, 3. [Google Scholar] [CrossRef]
- Blidi, S.; Bikaki, M.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. A comparative evaluation of bio-solvents for the efficient extraction of polyphenolic phytochemicals: Apple waste peels as a case study. Waste Biomass Valoriz. 2015, 6, 1125–1133. [Google Scholar] [CrossRef]
- Pandey, A.; Rai, R.; Pal, M.; Pandey, S. How polar are choline chloride-based deep eutectic solvents? Phys. Chem. Chem. Phys. 2014, 16, 1559–1568. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Row, K.H. Recent developments in deep eutectic solvents in chemical sciences. Monatsh. Chemie Chem. Mon. 2013, 144, 1427–1454. [Google Scholar] [CrossRef]
- Bi, W.; Tian, M.; Row, K.H. Evaluation of alcohol-based deep eutectic solvent in extraction and determination of flavonoids with response surface methodology optimization. J. Chromatogr. A 2013, 1285, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Wu, L. Effect of chlorogenic acid on antioxidant activity of Flos Lonicerae extracts. J. Zhejiang Univ. Sci. B 2007, 8, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Babova, O.; Occhipinti, A.; Maffei, M.E. Chemical partitioning and antioxidant capacity of green coffee (Coffea arabica and Coffea canephora) of different geographical origin. Phytochemistry 2016, 123, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Michalska, A.; Wojdyło, A.; Bogucka, B. The influence of nitrogen and potassium fertilisation on the content of polyphenolic Compounds and Antioxidant capacity of coloured potato. J. Food Compos. Anal. 2016, 47, 69–75. [Google Scholar] [CrossRef]
- Budryn, G.; Nebesny, E. Antioxidant properties of Arabica and Robusta coffee extracts prepared under different conditions. Dtsch. Lebensm.Rundsch. 2008, 104, 69–78. [Google Scholar]
- Andjelković, M.; Van Camp, J.; De Meulenaer, B.; Depaemelaere, G.; Socaciu, C.; Verloo, M.; Verhe, R. Iron-chelation properties of phenolic acids bearing catechol and galloyl groups. Food Chem. 2006, 98, 23–31. [Google Scholar] [CrossRef]
- Bassil, D.; Makris, D.P.; Kefalas, P. Oxidation of caffeic acid in the presence of L-cysteine: Isolation of 2-S-cysteinylcaffeic acid and evaluation of its antioxidant properties. Food Res. Int. 2005, 38, 395–402. [Google Scholar] [CrossRef]
- Amorati, R.; Pedulli, G.F.; Cabrini, L.; Zambonin, L.; Landi, L. Solvent and pH effects on the antioxidant activity of caffeic and other phenolic acids. J. Agric. Food Chem. 2006, 54, 2932–2937. [Google Scholar] [CrossRef] [PubMed]
- Kono, Y.; Kobayashi, K.; Tagawa, S.; Adachi, K.; Ueda, A.; Sawa, Y.; Shibata, H. Antioxidant activity of polyphenolics in diets: Rate constants of reactions of chlorogenic acid and caffeic acid with reactive species of oxygen and nitrogen. Biochim. Biophys. Acta Gen. Subj. 1997, 1335, 335–342. [Google Scholar] [CrossRef]
- Foti, M.C.; Daquino, C.; Geraci, C. Electron-transfer reaction of cinnamic acids and their methyl esters with the DPPH radical in alcoholic solutions. J. Org. Chem. 2004, 69, 2309–2314. [Google Scholar] [CrossRef] [PubMed]

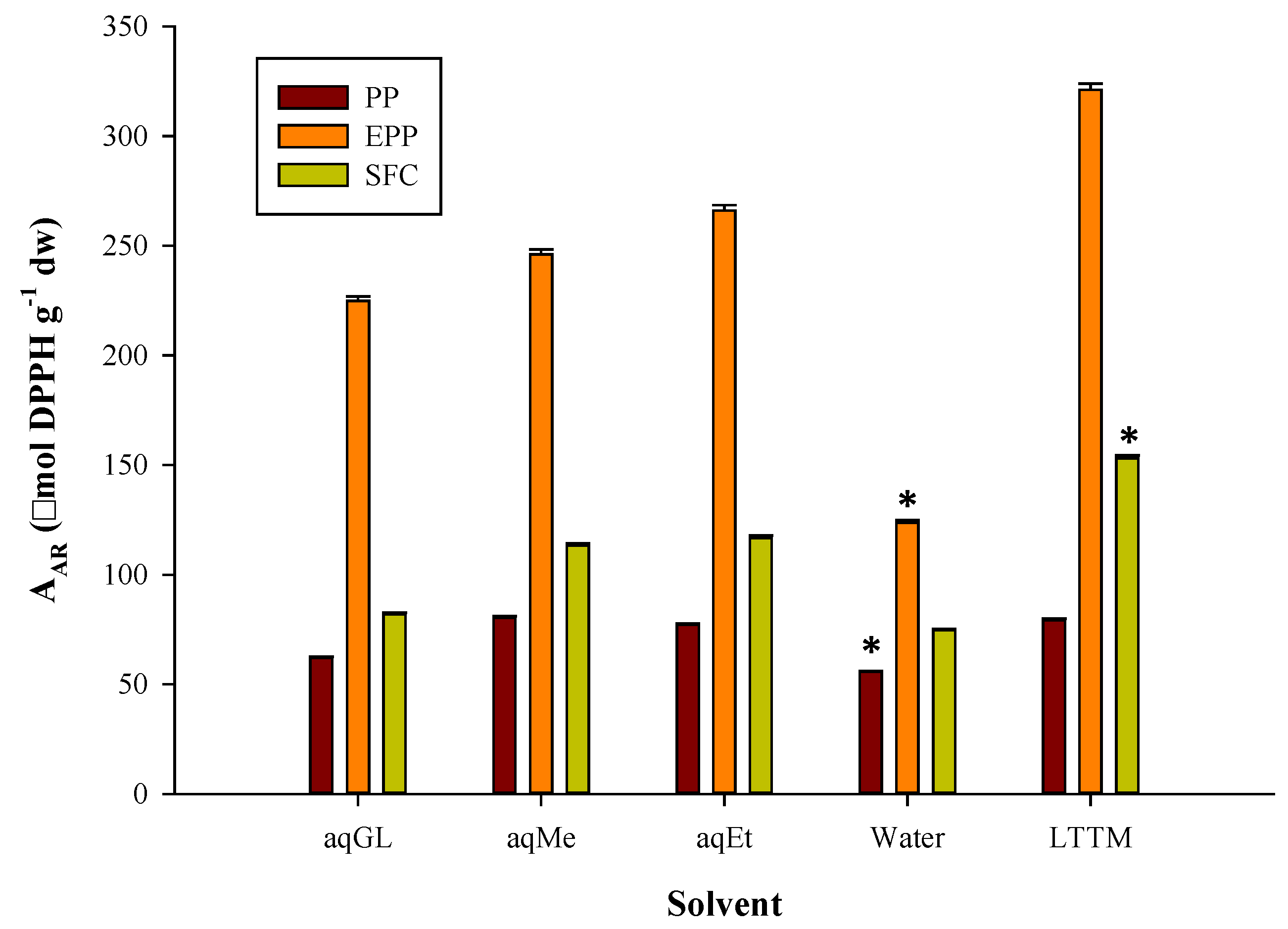
| Solvent | Waste | ||
|---|---|---|---|
| PPs | EPPs | SFC | |
| 80% (w/v) aq glycerol | 5.49 ± 0.29 | 21.09 ± 0.73 | 5.89 ± 0.19 |
| 50% (v/v) aq methanol | 5.74 ± 0.22 | 19.67 ± 0.28 | 9.24 ± 0.20 |
| 50% (v/v) aq ethanol | 5.05 ± 0.14 | 19.33 ± 0.31 | 8.79 ± 0.08 |
| Water | 3.53 ± 0.14 a | 14.30 ± 0.28 a | 5.29 ± 0.15 |
| LTTM | 7.38 ± 0.55 a | 27.63 ± 0.31 a | 11.74 ± 0.22 a |
| Solvent | Waste | ||
|---|---|---|---|
| PPs | EPPs | SFC | |
| 80% (w/v) aq glycerol | 6.02 ± 0.09 a | 12.85 ± 0.27 | 8.70 ± 0.13 a |
| 50% (v/v) aq methanol | 3.14 ± 0.09 | 10.13 ± 0.14 | 12.78 ± 0.25 |
| 50% (v/v) aq ethanol | 2.68 ± 0.13 | 9.60 ± 0.14 | 12.94 ± 0.05 |
| Water | 1.98 ± 0.20 | 9.94 ± 0.60 | 14.82 ± 0.98 |
| LTTM | 4.66 ± 0.05 | 24.68 ± 0.80 a | 12.48 ± 0.05 |
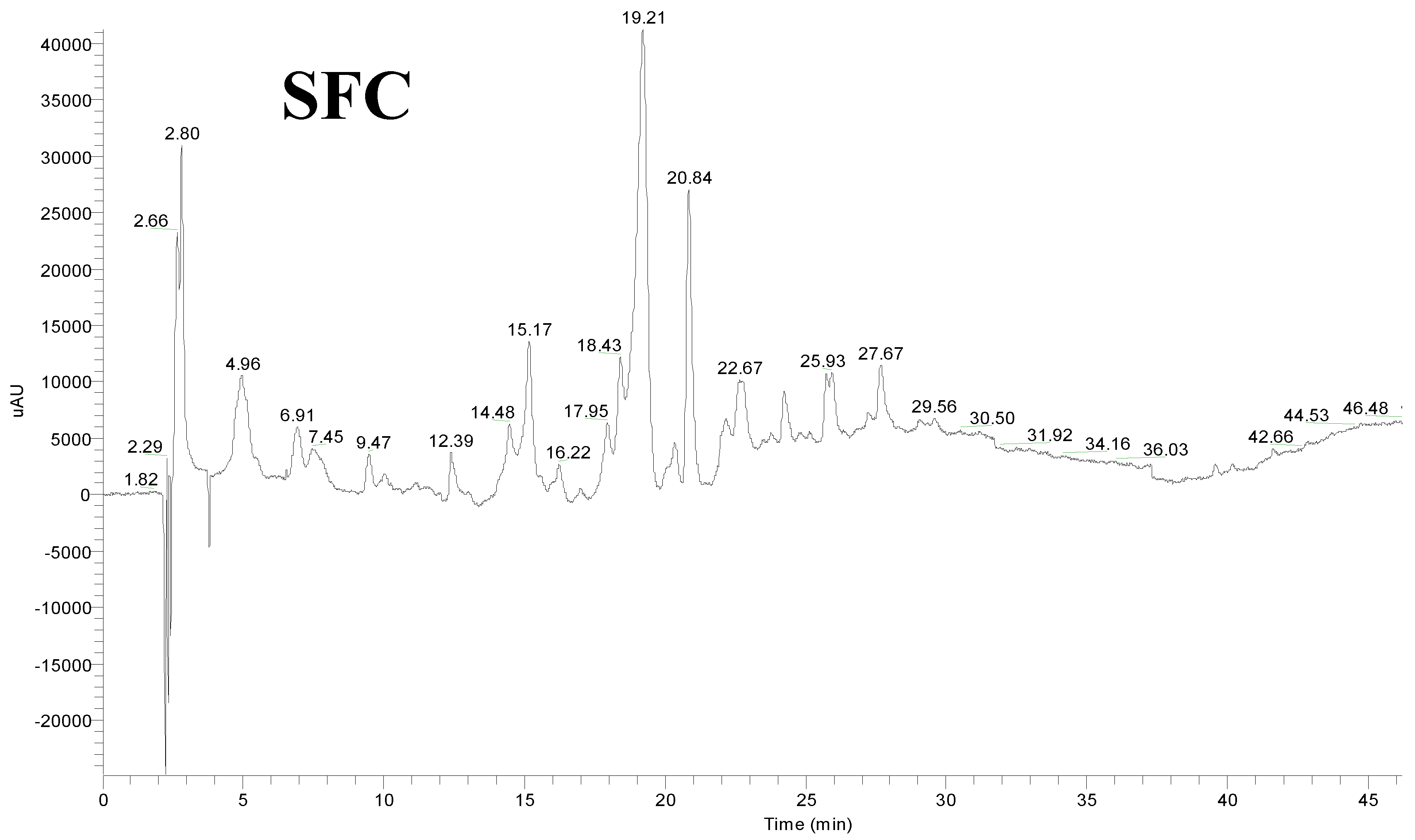
| No | Rt (min) | UV-Vis | [M + H]+ (m/z) | Fragment Ions (m/z) | Tentative Identity | Waste | ||
|---|---|---|---|---|---|---|---|---|
| PPs | EPPs | SFC | ||||||
| 1 | 12.87 | 244, 292, 316 | 251 | - | p-Coumaric acid derivative | ○ | ● | ○ |
| 2 | 14.48 | 242, 328 | 355 | 163 | Caffeoylquinic acid | ○ | ○ | ● |
| 3 | 15.17 | 248, 320 | 355 | 163 | Caffeoylquinic acid | ○ | ○ | ● |
| 4 | 15.25 | 246, 328 | 355 | 377 [M + Na]+, 163 | Caffeoylquinic acid | ○ | ● | ○ |
| 5 | 15.90 | 244, 292, 316 | 339 | 195 | p-Coumaroylquinic acid | ○ | ● | ○ |
| 6 | 17.95 | 246, 318 | 339 | 195 | p-Coumaroylquinic acid | ○ | ○ | ● |
| 7 | 18.07 | 246, 292, 318 | 472 | 220 | N1-(hydro)caffeoyl-N8-(hydro) caffeoylspermidine | ○ | ● | ○ |
| 8 | 18.43 | 240, 316 | 339 | 195 | p-Coumaroylquinic acid | ○ | ○ | ● |
| 9 | 19.43 | 246, 328 | 355 | 377 [M + Na]+, 163 | Caffeoylquinic acid | ● | ● | ● |
| 10 | 20.84 | 246, 320 | 339 | 195 | p-Coumaroylquinic acid | ○ | ○ | ● |
| 11 | 20.93 | 246, 328 | 695 | 717 [M + Na]+ | Caffeic acid derivative | ○ | ● | ○ |
| 12 | 21.01 | 242, 342 | 595 | 289 | Kaempferol rutinoside | ● | ● | ○ |
| 13 | 22.67 | 242, 320 | 339 | 195 | p-Coumaroylquinic acid | ○ | ○ | ● |
| 14 | 25.48 | 242, 320 | 613 | Caffeoylspermine | ● | ○ | ○ | |
| 15 | 25.93 | 246, 320 | 517 | di-Caffeoylquinic acid | ○ | ○ | ● | |
| 16 | 27.67 | 252, 350 | 611 | 303 | Quercetin rutinoside | ○ | ○ | ● |
| 17 | 28.93 | 238, 322 | 517 | di-Caffeoylquinic acid | ● | ● | ● | |
| 18 | 29.56 | 242, 320 | 517 | 539 [M + Na]+ | di-Caffeoylquinic acid | ○ | ○ | ● |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manousaki, A.; Jancheva, M.; Grigorakis, S.; Makris, D.P. Extraction of Antioxidant Phenolics from Agri-Food Waste Biomass Using a Newly Designed Glycerol-Based Natural Low-Transition Temperature Mixture: A Comparison with Conventional Eco-Friendly Solvents. Recycling 2016, 1, 194-204. https://doi.org/10.3390/recycling1010194
Manousaki A, Jancheva M, Grigorakis S, Makris DP. Extraction of Antioxidant Phenolics from Agri-Food Waste Biomass Using a Newly Designed Glycerol-Based Natural Low-Transition Temperature Mixture: A Comparison with Conventional Eco-Friendly Solvents. Recycling. 2016; 1(1):194-204. https://doi.org/10.3390/recycling1010194
Chicago/Turabian StyleManousaki, Areti, Magdalena Jancheva, Spyros Grigorakis, and Dimitris P. Makris. 2016. "Extraction of Antioxidant Phenolics from Agri-Food Waste Biomass Using a Newly Designed Glycerol-Based Natural Low-Transition Temperature Mixture: A Comparison with Conventional Eco-Friendly Solvents" Recycling 1, no. 1: 194-204. https://doi.org/10.3390/recycling1010194








