Navigating Battery Choices in IoT: An Extensive Survey of Technologies and Their Applications
Abstract
:1. Introduction
- Analysis of a range of battery technologies for suitability in IoT applications.
- Development of a battery–application compatibility matrix for data-driven critical analysis.
2. IoT Applications
2.1. Healthcare
Wearable
2.2. Smart Cities
2.3. Smart Home
2.4. Smart Farm
2.5. Smart Industry
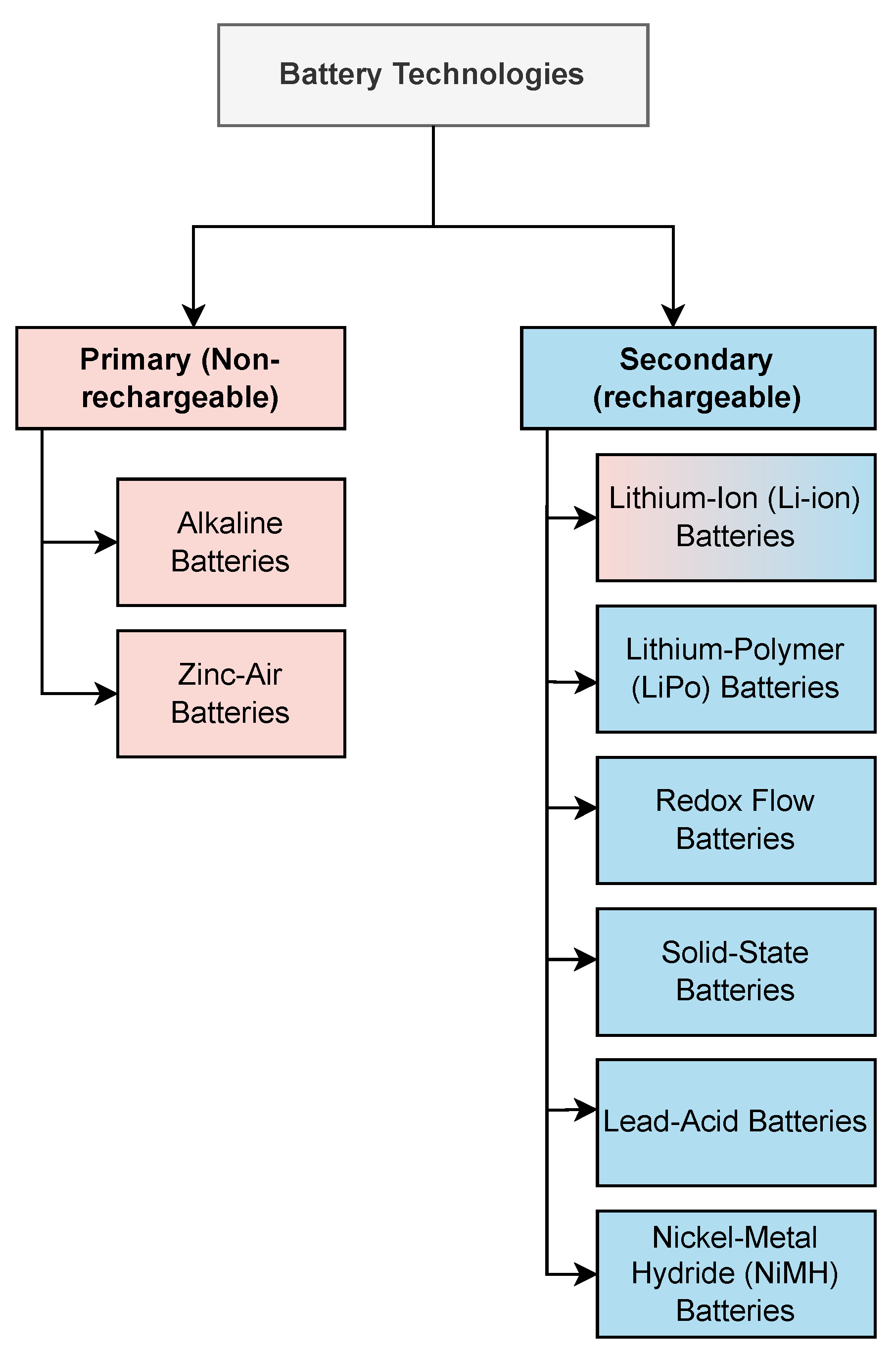
3. Batteries for IoT Applications
3.1. Lead-Acid Batteries
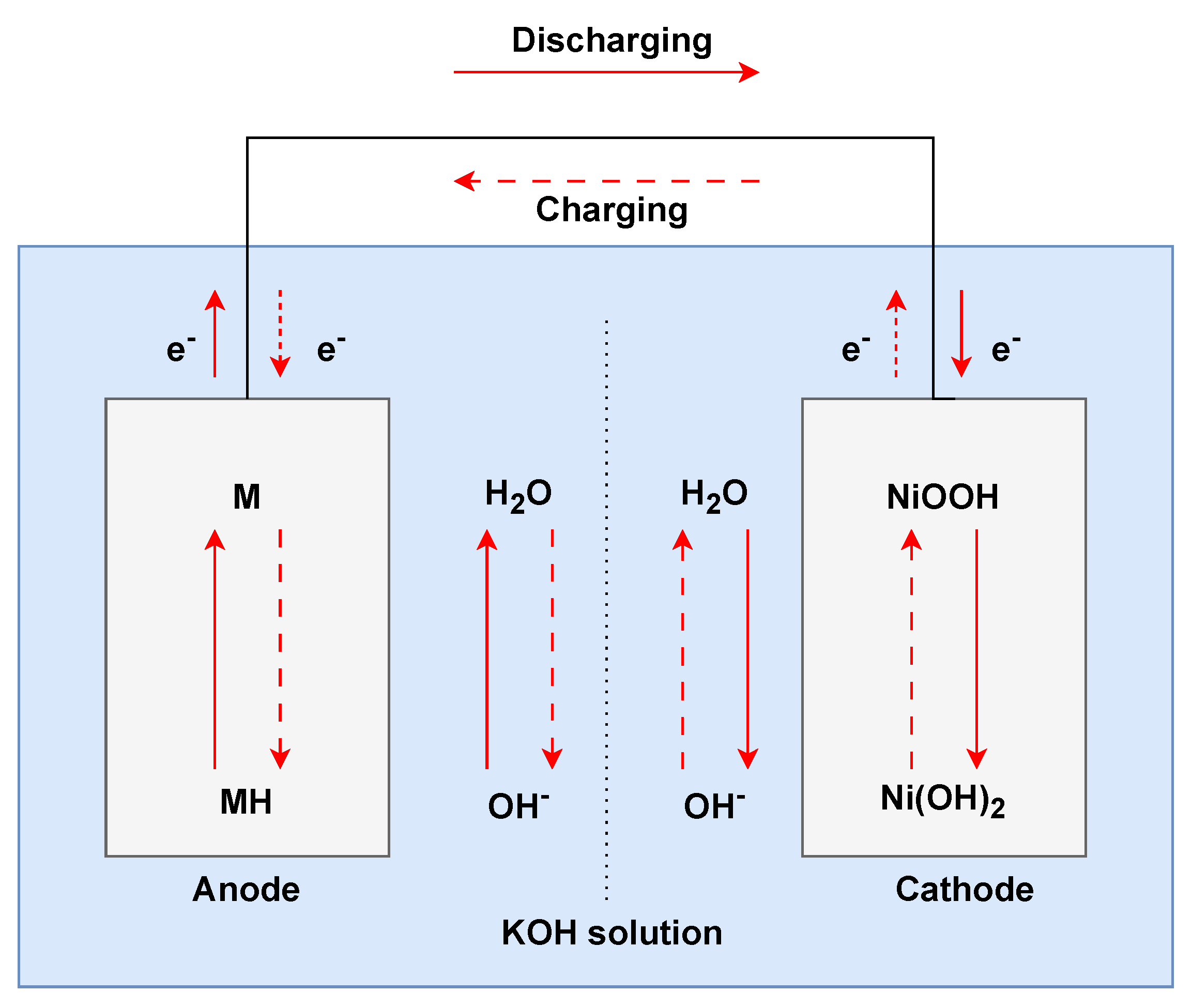
3.2. Nickel–Metal Hydride (NiMH) Batteries
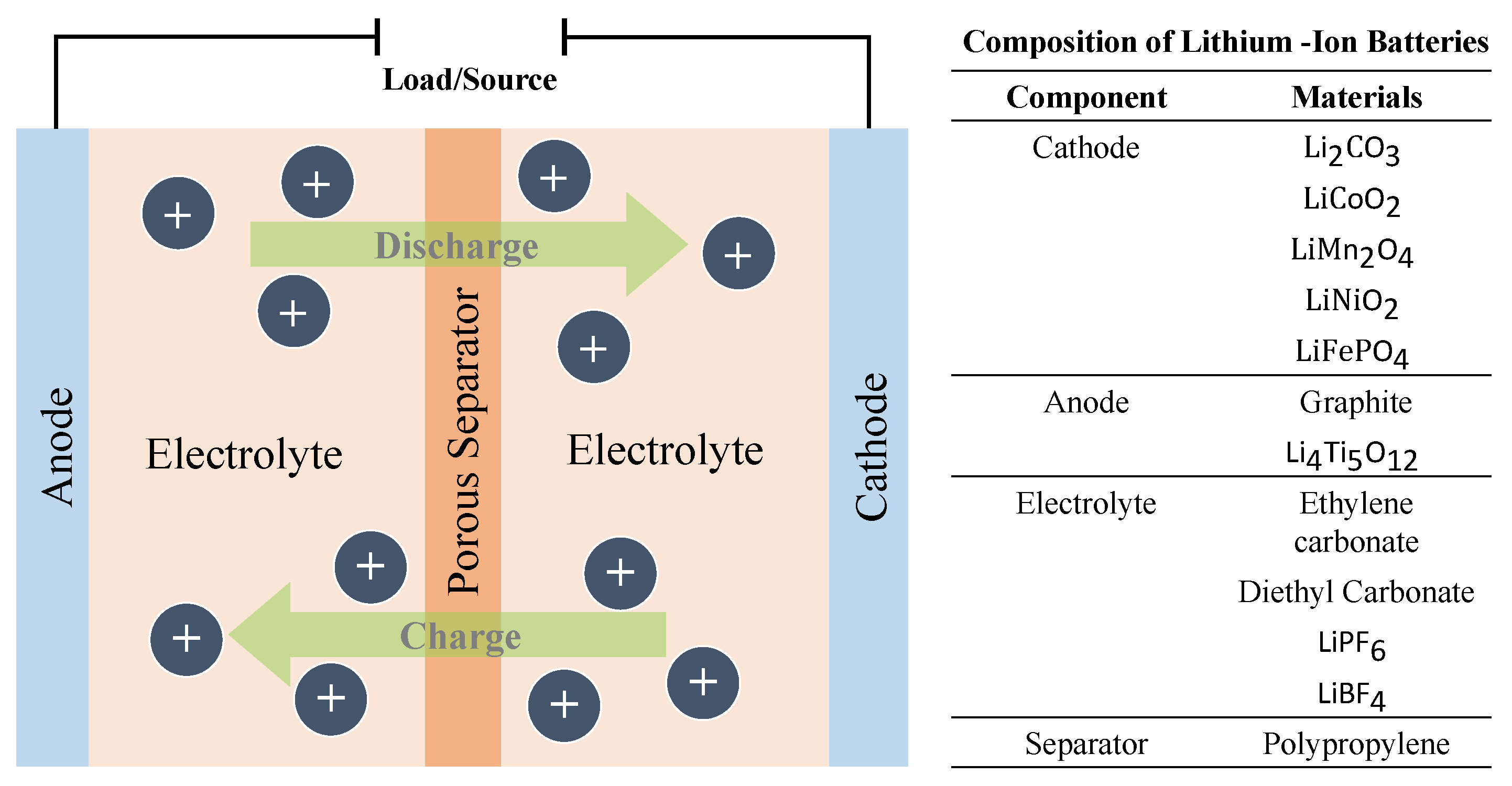
3.3. Lithium–Ion (Li–Ion) Batteries
3.3.1. Lithium Cobalt Oxide (LCO) Batteries
3.3.2. Lithium Manganese Oxide (LMO) Batteries
3.3.3. Lithium Nickel Manganese Cobalt Oxide
3.3.4. Lithium Iron Phosphate (LiFePO) Batteries
3.3.5. Lithium–Polymer (LiPo) Batteries
3.4. Solid-State Batteries
3.5. Alkaline Batteries
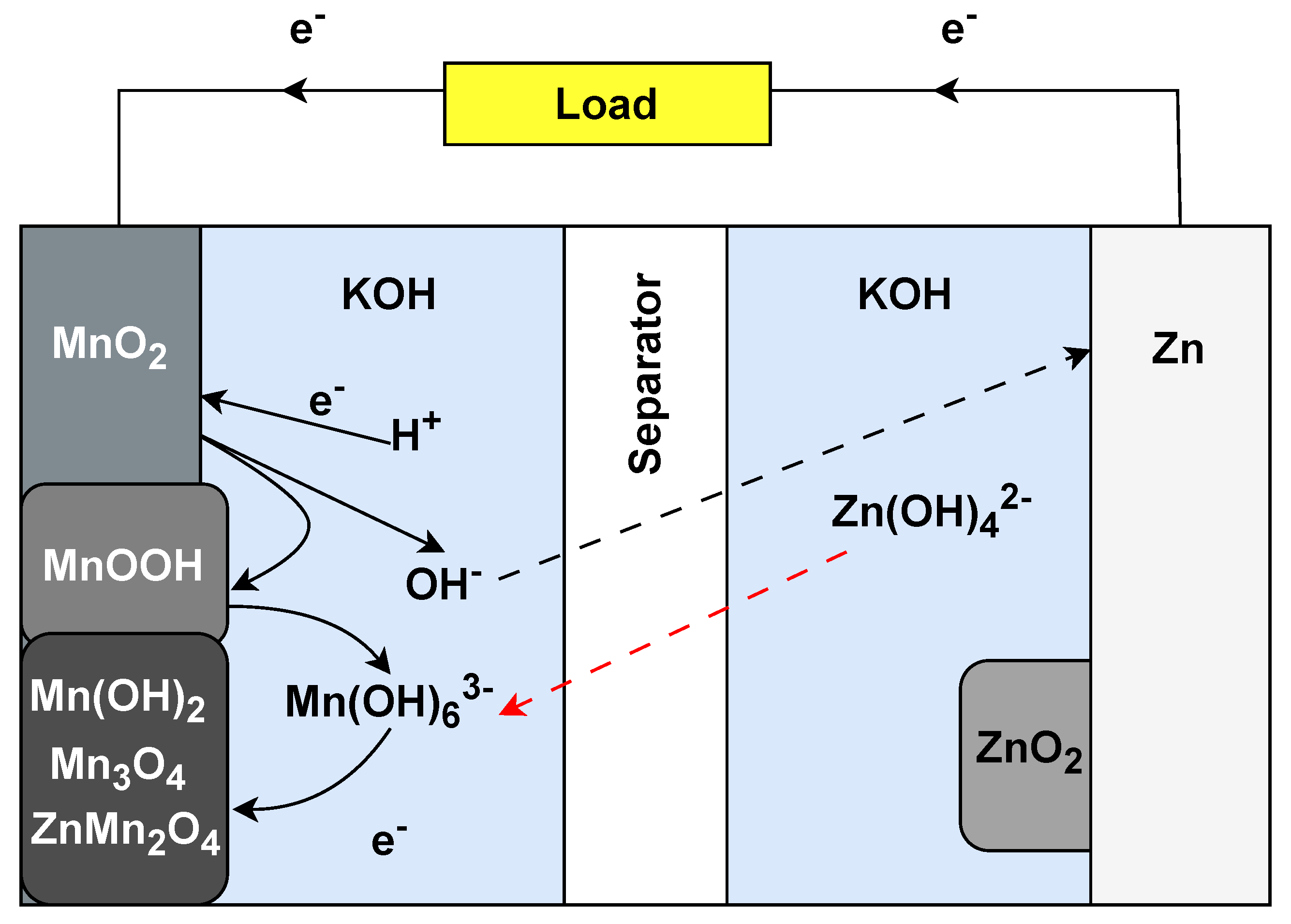
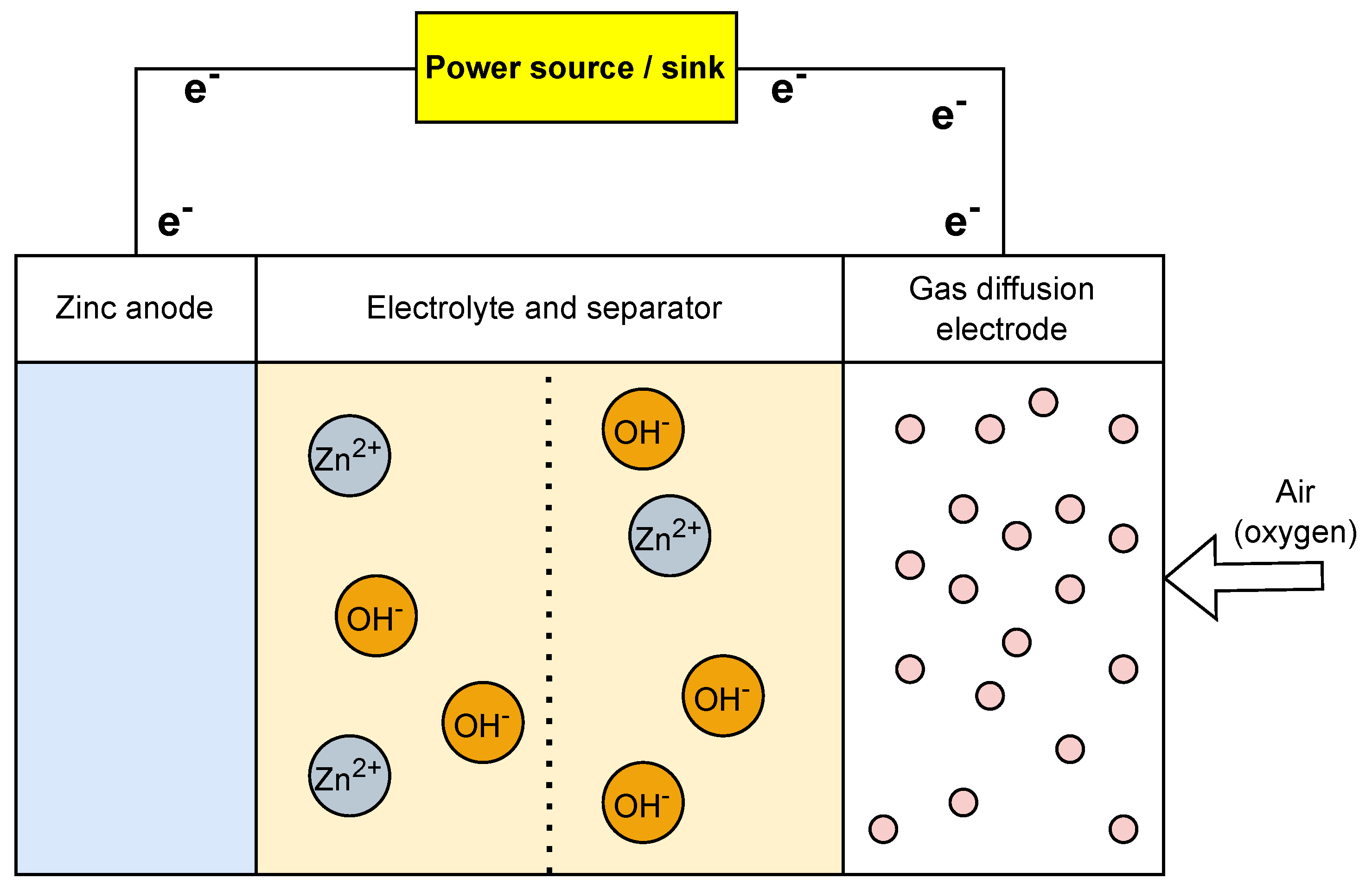
3.6. Zinc–Air Batteries
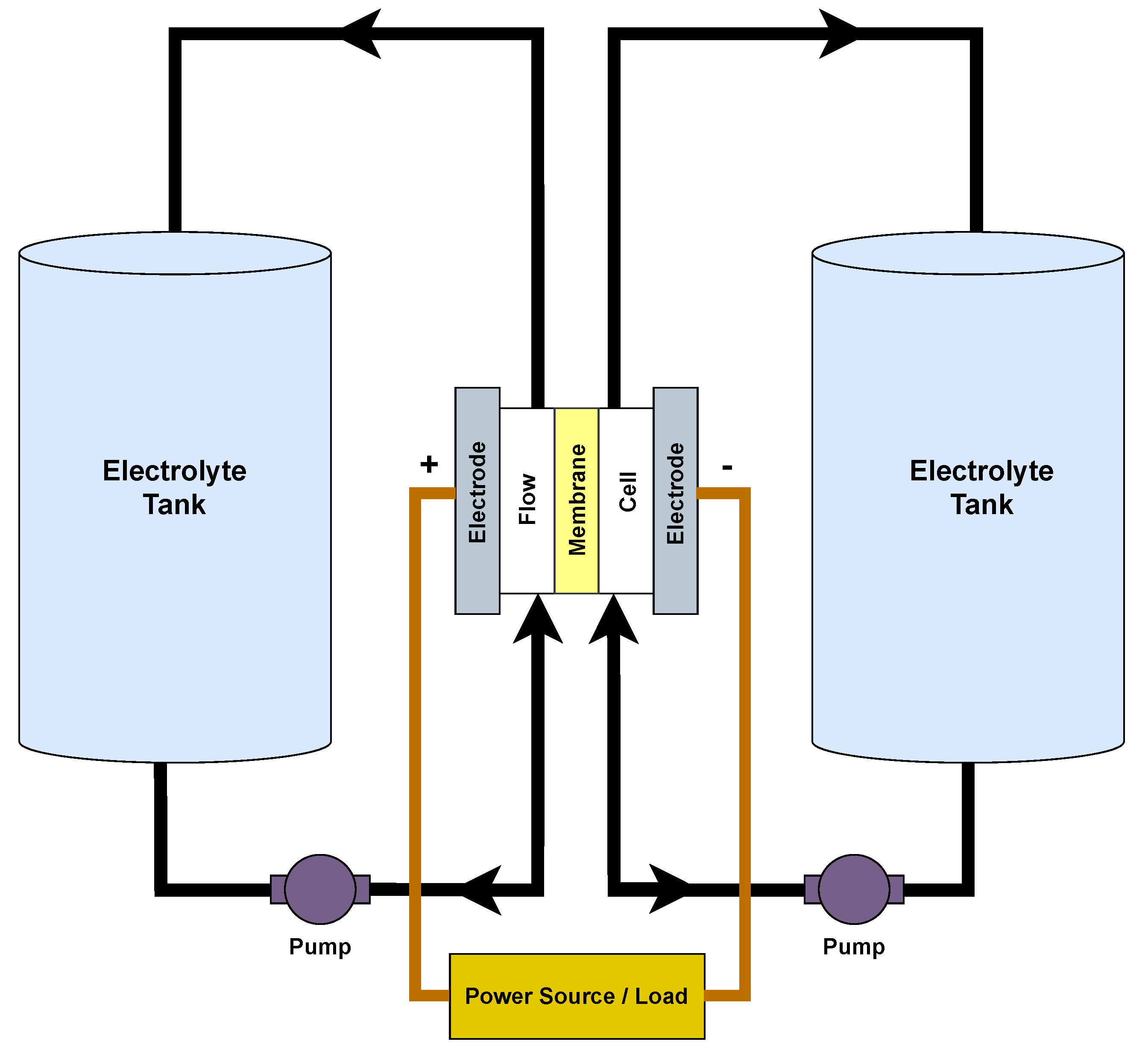
3.7. Flow Batteries
3.8. Supercapacitors
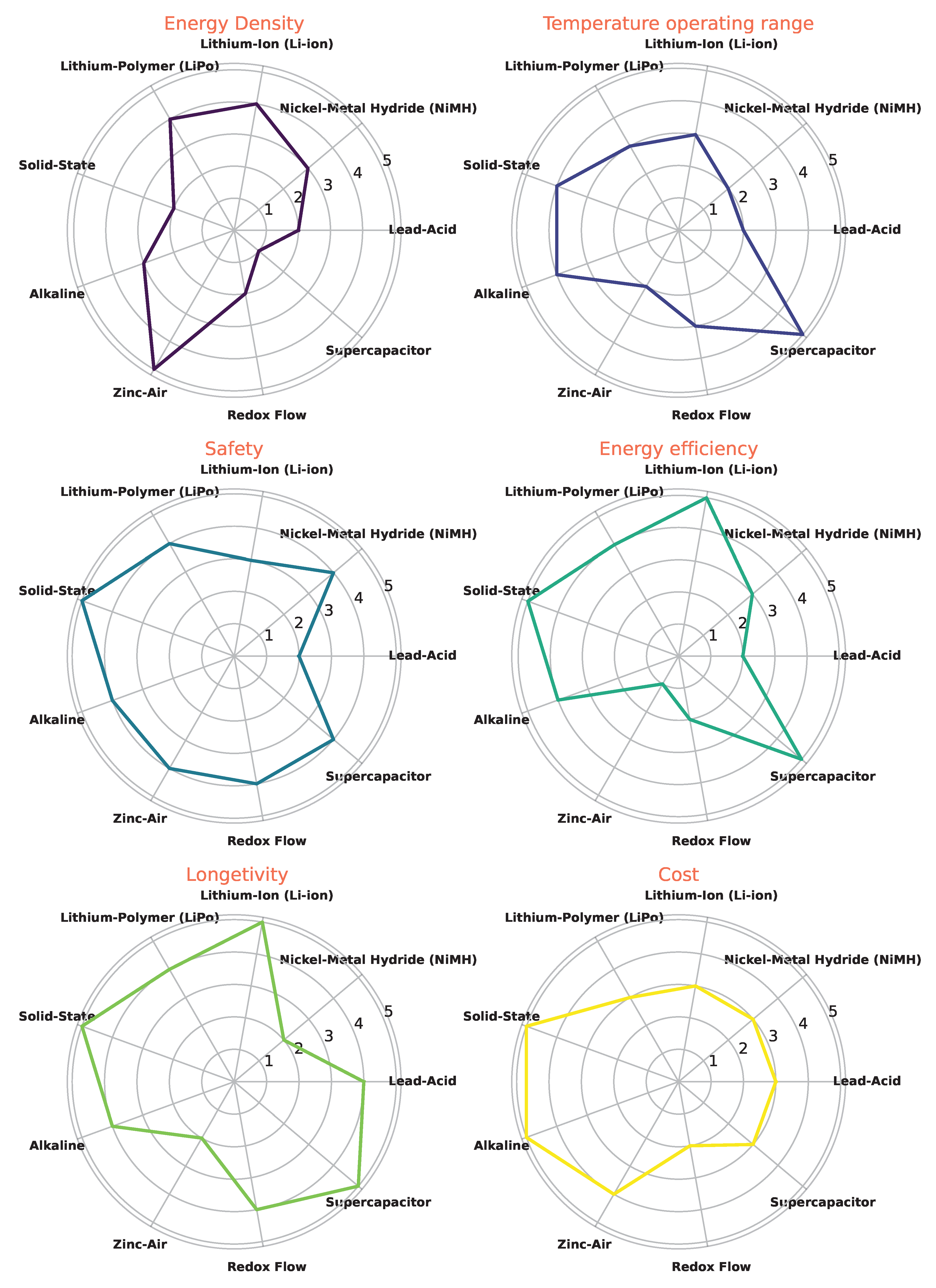
4. Battery Evaluation
- Energy density;
- Temperature range;
- Longevity;
- Nominal cell voltage;
- Safety;
- Cost;
- Energy efficiency.
4.1. Energy and Power Densities
4.1.1. Gravimetric Energy Density
4.1.2. Volumetric Energy Density
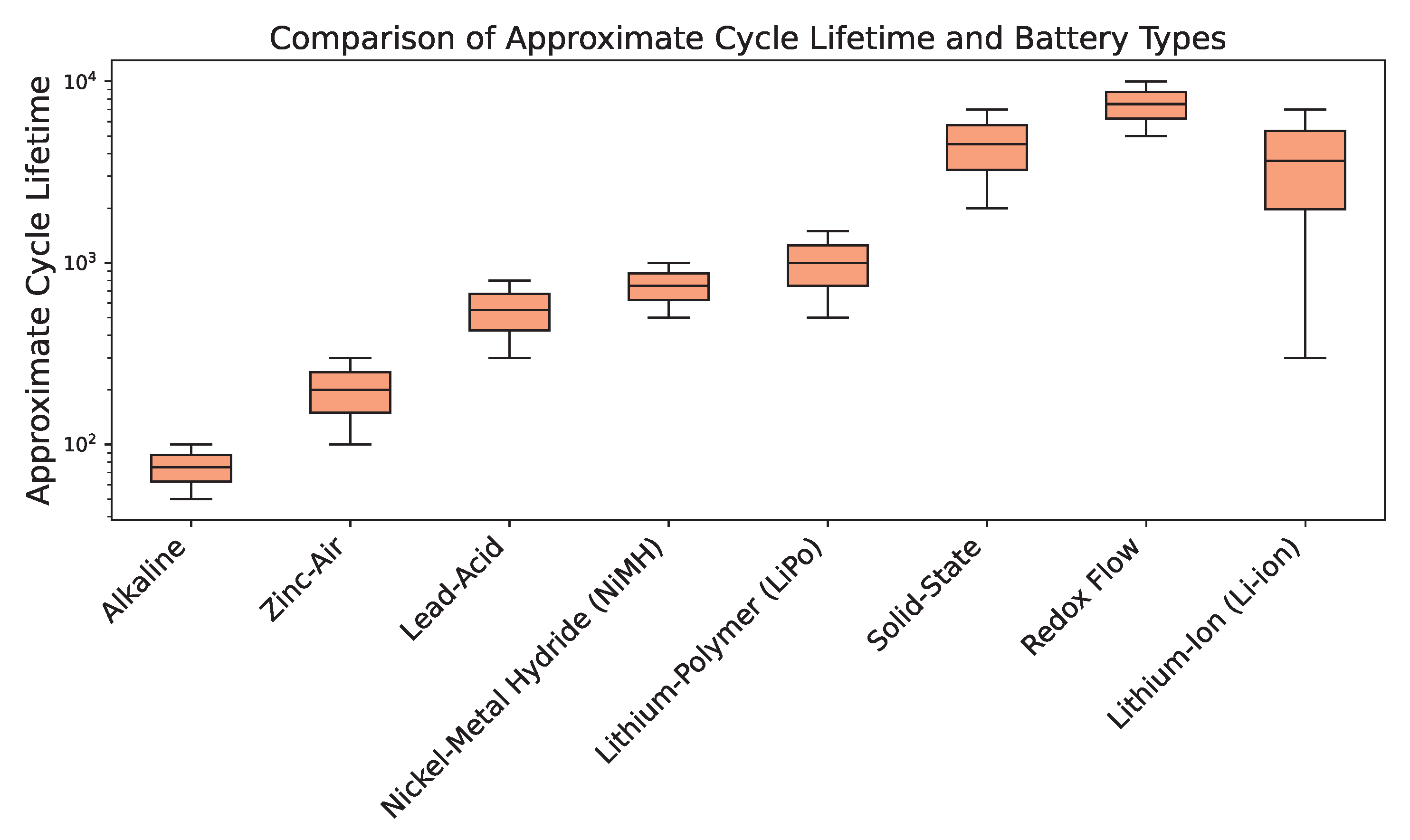
4.2. Longevity
4.3. Nominal Cell Voltage
4.4. Cost
4.5. Operating Temperature
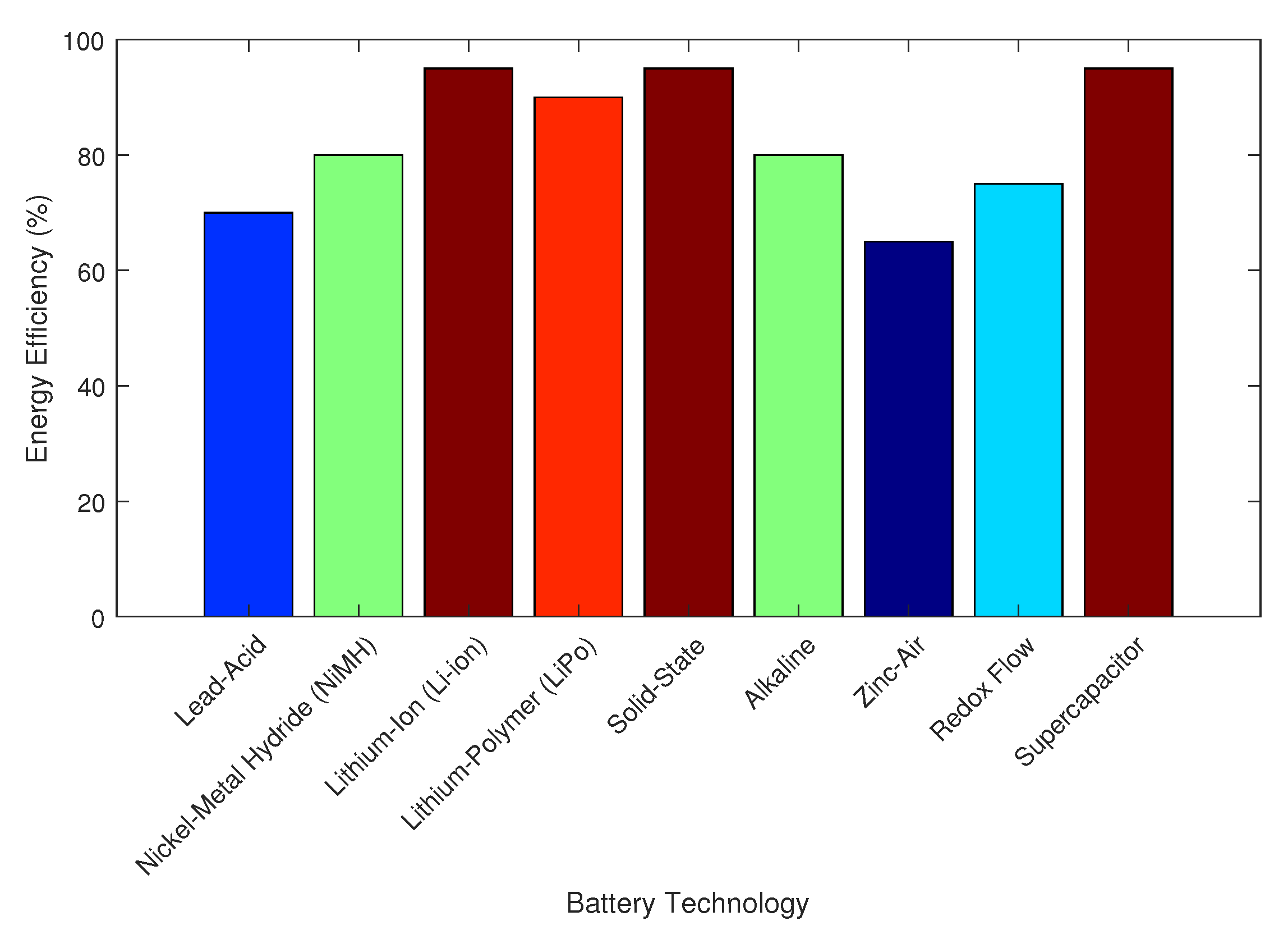
4.6. Energy Efficiency
4.7. Safety
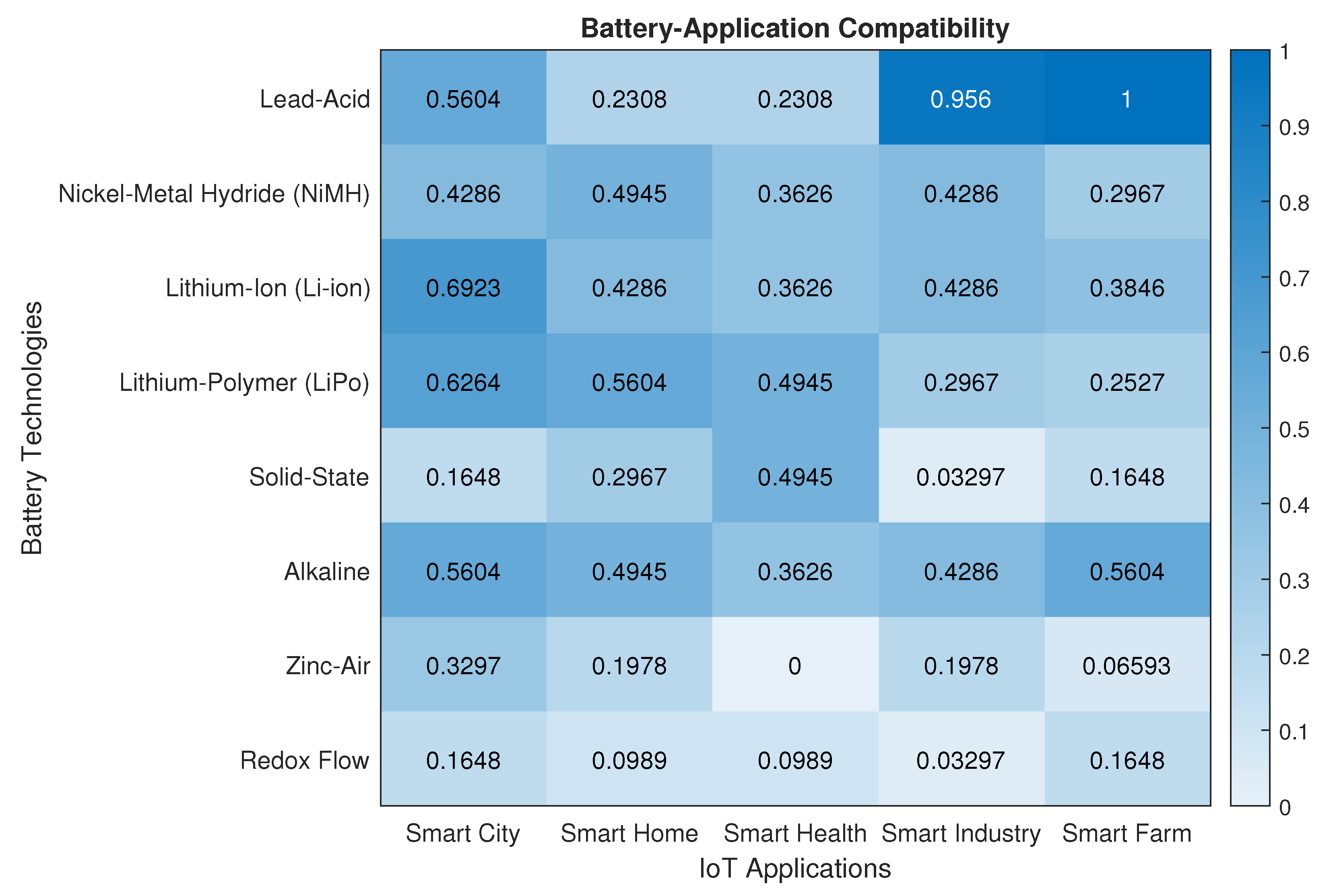
5. Requirements for IoT Applications
6. Conclusions
Funding
Conflicts of Interest
References
- Lohiya, R.; Thakkar, A. Application Domains, Evaluation Data Sets, and Research Challenges of IoT: A Systematic Review. IEEE Internet Things J. 2021, 8, 8774–8798. [Google Scholar] [CrossRef]
- Al-kahtani, M.S.; Khan, F.; Taekeun, W. Application of Internet of Things and Sensors in Healthcare. Sensors 2022, 22, 5738. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, Y.; Yang, J.; Miao, Y.; Li, W. Big Health Application System based on Health Internet of Things and Big Data. IEEE Access 2017, 5, 7885–7897. [Google Scholar] [CrossRef]
- Baker, S.B.; Xiang, W.; Atkinson, I. Internet of Things for Smart Healthcare: Technologies, Challenges, and Opportunities. IEEE Access 2017, 5, 26521–26544. [Google Scholar] [CrossRef]
- Wu, F.; Rüdiger, C.; Redouté, J.M.; Yuce, M.R. WE-Safe: A wearable IoT sensor node for safety applications via LoRa. In Proceedings of the 2018 IEEE 4th World Forum on Internet of Things (WF-IoT), Singapore, 5–8 February 2018; pp. 144–148. [Google Scholar] [CrossRef]
- Xu, J.; Gu, B.; Tian, G. Review of agricultural IoT technology. Artif. Intell. Agric. 2022, 6, 10–22. [Google Scholar] [CrossRef]
- Dhanaraju, M.; Chenniappan, P.; Ramalingam, K.; Pazhanivelan, S.; Kaliaperumal, R. Smart Farming: Internet of Things (IoT)-Based Sustainable Agriculture. Agriculture 2022, 12, 1745. [Google Scholar] [CrossRef]
- Miao, Y.; Hynan, P.; von Jouanne, A.; Yokochi, A. Current Li-Ion Battery Technologies in Electric Vehicles and Opportunities for Advancements. Energies 2019, 12, 1074. [Google Scholar] [CrossRef]
- Rangarajan, S.S.; Sunddararaj, S.P.; Sudhakar, A.; Shiva, C.K.; Subramaniam, U.; Collins, E.R.; Senjyu, T. Lithium-Ion Batteries—The Crux of Electric Vehicles with Opportunities and Challenges. Clean Technol. 2022, 4, 908–930. [Google Scholar] [CrossRef]
- Chen, W.; Liang, J.; Yang, Z.; Li, G. A Review of Lithium-Ion Battery for Electric Vehicle Applications and Beyond. Energy Procedia 2019, 158, 4363–4368. [Google Scholar] [CrossRef]
- Armenta-Déu, C.; Boucheix, B. Evaluation of Lithium-Ion Battery Performance under Variable Climatic Conditions: Influence on the Driving Range of Electric Vehicles. Future Transp. 2023, 3, 535–551. [Google Scholar] [CrossRef]
- Homssi, B.A.; Al-Hourani, A.; Magowe, K.; Delaney, J.; Tom, N.; Ying, J.; Wolf, H.; Maselli, S.; Kandeepan, S.; Wang, K.; et al. A Framework for the Design and Deployment of Large-Scale LPWAN Networks for Smart Cities Applications. IEEE Internet Things Mag. 2021, 4, 53–59. [Google Scholar] [CrossRef]
- Winter, M.; Brodd, R.J. What Are Batteries, Fuel Cells, and Supercapacitors? Chem. Rev. 2004, 104, 4245–4270. [Google Scholar] [CrossRef]
- Abdulmalek, S.; Nasir, A.; Jabbar, W.A.; Almuhaya, M.A.M.; Bairagi, A.K.; Khan, M.A.M.; Kee, S.H. IoT-Based Healthcare-Monitoring System towards Improving Quality of Life: A Review. Healthcare 2022, 10, 1993. [Google Scholar] [CrossRef]
- Singh, R.P.; Javaid, M.; Haleem, A.; Suman, R. Internet of things (IoT) applications to fight against COVID-19 pandemic. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 521–524. [Google Scholar] [CrossRef]
- Wu, T.; Wu, F.; Qiu, C.; Redouté, J.M.; Yuce, M.R. A Rigid-Flex Wearable Health Monitoring Sensor Patch for IoT-Connected Healthcare Applications. IEEE Internet Things J. 2020, 7, 6932–6945. [Google Scholar] [CrossRef]
- Farrokhi, A.; Farahbakhsh, R.; Rezazadeh, J.; Minerva, R. Application of Internet of Things and artificial intelligence for smart fitness: A survey. Comput. Netw. 2021, 189, 107859. [Google Scholar] [CrossRef]
- Passos, J.; Lopes, S.I.; Clemente, F.M.; Moreira, P.M.; Rico-González, M.; Bezerra, P.; Rodrigues, L.P. Wearables and Internet of Things (IoT) technologies for fitness assessment: A systematic review. Sensors 2021, 21, 5418. [Google Scholar] [CrossRef]
- Lingg, E.; Leone, G.; Spaulding, K.; B’Far, R. Cardea: Cloud based employee health and wellness integrated wellness application with a wearable device and the HCM data store. In Proceedings of the 2014 IEEE World Forum on Internet of Things (WF-IoT), Seoul, Republic of Korea, 6–8 March 2014; pp. 265–270. [Google Scholar] [CrossRef]
- Nagaraj, P.; Muneeswaran, V.; Sudar, K.M.; Ali, R.S.; Someshwara, A.L.; Kumar, T.S. Internet of Things Based Smart Hospital Saline Monitoring System. In Proceedings of the 2021 5th International Conference on Computer, Communication and Signal Processing (ICCCSP), Chennai, India, 24–25 May 2021; pp. 53–58. [Google Scholar] [CrossRef]
- Rathnayaka, A.; Gendy, M.E.G.; Wu, F.; Mamun, M.A.A.; Curtis, S.J.; Bingham, G.; Peleg, A.Y.; Stewardson, A.J.; Yuce, M.R. An Autonomous IoT-Based Contact Tracing Platform in a COVID-19 Patient Ward. IEEE Internet Things J. 2023, 10, 8706–8717. [Google Scholar] [CrossRef]
- Siriwardhana, Y.; De Alwis, C.; Gür, G.; Ylianttila, M.; Liyanage, M. The Fight against the COVID-19 Pandemic with 5G Technologies. IEEE Eng. Manag. Rev. 2020, 48, 72–84. [Google Scholar] [CrossRef]
- Hasan, K.; Pour Ebrahim, M.; Yuce, M.R. Real-Time People Counting Using IR-UWB Radar. In Body Area Networks. Smart IoT and Big Data for Intelligent Health Management; Ur Rehman, M., Zoha, A., Eds.; Springer: Cham, Switzerland, 2022; pp. 63–70. [Google Scholar]
- Alsamhi, S.H.; Ma, O.; Ansari, M.S.; Almalki, F.A. Survey on Collaborative Smart Drones and Internet of Things for Improving Smartness of Smart Cities. IEEE Access 2019, 7, 128125–128152. [Google Scholar] [CrossRef]
- Anagnostopoulos, T.; Zaslavsky, A.; Kolomvatsos, K.; Medvedev, A.; Amirian, P.; Morley, J.; Hadjieftymiades, S. Challenges and Opportunities of Waste Management in IoT-Enabled Smart Cities: A Survey. IEEE Trans. Sustain. Comput. 2017, 2, 275–289. [Google Scholar] [CrossRef]
- Kwon, K.; Lee, S.; Kim, S. AI-Based Home Energy Management System Considering Energy Efficiency and Resident Satisfaction. IEEE Internet Things J. 2022, 9, 1608–1621. [Google Scholar] [CrossRef]
- Kelly, S.D.T.; Suryadevara, N.K.; Mukhopadhyay, S.C. Towards the Implementation of IoT for Environmental Condition Monitoring in Homes. IEEE Sens. J. 2013, 13, 3846–3853. [Google Scholar] [CrossRef]
- Al-Ali, A.; Zualkernan, I.A.; Rashid, M.; Gupta, R.; Alikarar, M. A smart home energy management system using IoT and big data analytics approach. IEEE Trans. Consum. Electron. 2017, 63, 426–434. [Google Scholar] [CrossRef]
- Ramson, S.R.J.; León-Salas, W.D.; Brecheisen, Z.; Foster, E.J.; Johnston, C.T.; Schulze, D.G.; Filley, T.; Rahimi, R.; Soto, M.J.C.V.; Bolivar, J.A.L.; et al. A Self-Powered, Real-Time, LoRaWAN IoT-Based Soil Health Monitoring System. IEEE Internet Things J. 2021, 8, 9278–9293. [Google Scholar] [CrossRef]
- Hu, W.J.; Fan, J.; Du, Y.X.; Li, B.S.; Xiong, N.; Bekkering, E. MDFC–ResNet: An Agricultural IoT System to Accurately Recognize Crop Diseases. IEEE Access 2020, 8, 115287–115298. [Google Scholar] [CrossRef]
- Sharma, A.; Jain, A.; Gupta, P.; Chowdary, V. Machine Learning Applications for Precision Agriculture: A Comprehensive Review. IEEE Access 2021, 9, 4843–4873. [Google Scholar] [CrossRef]
- Farooq, M.S.; Sohail, O.O.; Abid, A.; Rasheed, S. A Survey on the Role of IoT in Agriculture for the Implementation of Smart Livestock Environment. IEEE Access 2022, 10, 9483–9505. [Google Scholar] [CrossRef]
- Lopes, P.P.; Stamenkovic, V.R. Past, present, and future of lead–acid batteries. Science 2020, 369, 923–924. [Google Scholar] [CrossRef]
- Zito, R. Energy Storage: A New Approach; John Wiley & Sons: Hoboken, NJ, USA, 2010; Volume 26. [Google Scholar]
- Chang, S.; Young, K.; Nei, J.; Fierro, C. Reviews on the U.S. Patents Regarding Nickel/Metal Hydride Batteries. Batteries 2016, 2, 10. [Google Scholar] [CrossRef]
- Ruetschi, P.; Meli, F.; Desilvestro, J. Nickel-metal hydride batteries. The preferred batteries of the future? J. Power Sources 1995, 57, 85–91. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Park, K.S. The Li-Ion Rechargeable Battery: A Perspective. J. Am. Chem. Soc. 2013, 135, 1167–1176. [Google Scholar] [CrossRef]
- Nitta, N.; Wu, F.; Lee, J.T.; Yushin, G. Li-ion battery materials: Present and future. Mater. Today 2015, 18, 252–264. [Google Scholar] [CrossRef]
- Manthiram, A. A reflection on lithium-ion battery cathode chemistry. Nat. Commun. 2020, 11, 1550. [Google Scholar] [CrossRef]
- Deng, D. Li-ion batteries: Basics, progress, and challenges. Energy Sci. Eng. 2015, 3, 385–418. [Google Scholar] [CrossRef]
- Wang, K.; Wan, J.; Xiang, Y.; Zhu, J.; Leng, Q.; Wang, M.; Xu, L.; Yang, Y. Recent advances and historical developments of high voltage lithium cobalt oxide materials for rechargeable Li-ion batteries. J. Power Sources 2020, 460, 228062. [Google Scholar] [CrossRef]
- Thackeray, M.M. Manganese oxides for lithium batteries. Prog. Solid State Chem. 1997, 25, 1–71. [Google Scholar] [CrossRef]
- Dou, S. Review and prospect of layered lithium nickel manganese oxide as cathode materials for Li-ion batteries. J. Solid State Electrochem. 2013, 17, 911–926. [Google Scholar] [CrossRef]
- Fröhlich, K.; Legotin, E.; Bärhold, F.; Trifonova, A. New large-scale production route for synthesis of lithium nickel manganese cobalt oxide. J. Solid State Electrochem. 2017, 21, 3403–3410. [Google Scholar] [CrossRef]
- Satyavani, T.; Kumar, A.S.; Rao, P.S. Methods of synthesis and performance improvement of lithium iron phosphate for high rate Li-ion batteries: A review. Eng. Sci. Technol. Int. J. 2016, 19, 178–188. [Google Scholar] [CrossRef]
- Long, L.; Wang, S.; Xiao, M.; Meng, Y. Polymer electrolytes for lithium polymer batteries. J. Mater. Chem. A 2016, 4, 10038–10069. [Google Scholar] [CrossRef]
- Stephan, A.M.; Nahm, K. Review on composite polymer electrolytes for lithium batteries. Polymer 2006, 47, 5952–5964. [Google Scholar] [CrossRef]
- Zurbuchen, A.; Haeberlin, A.; Pfenniger, A.; Bereuter, L.; Schaerer, J.; Jutzi, F.; Huber, C.; Fuhrer, J.; Vogel, R. Towards Batteryless Cardiac Implantable Electronic Devices—The Swiss Way. IEEE Trans. Biomed. Circuits Syst. 2017, 11, 78–86. [Google Scholar] [CrossRef]
- Munshi, M.Z.A. Handbook of Solid State Batteries & Capacitors; World Scientific: Singapore, 1995. [Google Scholar]
- Julien, C.; Nazri, G.A. Solid State Batteries: Materials Design and Optimization; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; Volume 271. [Google Scholar]
- Hossain, E.; Faruque, H.M.R.; Sunny, M.S.H.; Mohammad, N.; Nawar, N. A comprehensive review on energy storage systems: Types, comparison, current scenario, applications, barriers, and potential solutions, policies, and future prospects. Energies 2020, 13, 3651. [Google Scholar] [CrossRef]
- Salkind, A.J.; Klein, M. Batteries, Alkaline Secondary Cells; Wiley: Hoboken, NJ, USA, 2000. [Google Scholar]
- Olabi, A.G.; Sayed, E.T.; Wilberforce, T.; Jamal, A.; Alami, A.H.; Elsaid, K.; Rahman, S.M.A.; Shah, S.K.; Abdelkareem, M.A. Metal-Air Batteries—A Review. Energies 2021, 14, 7373. [Google Scholar] [CrossRef]
- Wang, C.; Yu, Y.; Niu, J.; Liu, Y.; Bridges, D.; Liu, X.; Pooran, J.; Zhang, Y.; Hu, A. Recent progress of metal–air batteries—A mini review. Appl. Sci. 2019, 9, 2787. [Google Scholar] [CrossRef]
- Li, Y.; Dai, H. Recent advances in zinc–air batteries. Chem. Soc. Rev. 2014, 43, 5257–5275. [Google Scholar] [CrossRef]
- Yao, Y.; Lei, J.; Shi, Y.; Ai, F.; Lu, Y.C. Assessment methods and performance metrics for redox flow batteries. Nat. Energy 2021, 6, 582–588. [Google Scholar] [CrossRef]
- Berrueta, A.; Ursúa, A.; Martín, I.S.; Eftekhari, A.; Sanchis, P. Supercapacitors: Electrical Characteristics, Modeling, Applications, and Future Trends. IEEE Access 2019, 7, 50869–50896. [Google Scholar] [CrossRef]
- Şahin, M.E.; Blaabjerg, F.; Sangwongwanich, A. A Comprehensive Review on Supercapacitor Applications and Developments. Energies 2022, 15, 674. [Google Scholar] [CrossRef]
- Anzola, J.; Jiménez, A.; Tarazona, G. Self-sustainable power-collecting node in IoT. Internet Things 2019, 7, 100082. [Google Scholar] [CrossRef]
- Manupibul, U.; Tanthuwapathom, R.; Jarumethitanont, W.; Kaimuk, P.; Limroongreungrat, W.; Charoensuk, W. Integration of force and IMU sensors for developing low-cost portable gait measurement system in lower extremities. Sci. Rep. 2023, 13, 10653. [Google Scholar] [CrossRef]
- Kadechkar, A.; Riba, J.R.; Moreno-Eguilaz, M.; Pérez, J. SmartConnector: A Self-Powered IoT Solution to Ease Predictive Maintenance in Substations. IEEE Sens. J. 2020, 20, 11632–11641. [Google Scholar] [CrossRef]
- Chakraborty, S.; Arvind, P.; Poddar, S.; Acharya, A.K.; Kumar, S.D. Integration of IoT Based PLC for Smart Relaying of a PV-Fed Induction Motor Driven Conveyor Belt. In Proceedings of the Fifth International Conference on Microelectronics, Computing and Communication Systems, Ranchi, India, 2021; pp. 155–165. [Google Scholar]
- Elsts, A.; Fafoutis, X.; Woznowski, P.; Tonkin, E.; Oikonomou, G.; Piechocki, R.; Craddock, I. Enabling Healthcare in Smart Homes: The SPHERE IoT Network Infrastructure. IEEE Commun. Mag. 2018, 56, 164–170. [Google Scholar] [CrossRef]
- Lee, B.G.; Lee, S.M. Smart Wearable Hand Device for Sign Language Interpretation System With Sensors Fusion. IEEE Sens. J. 2018, 18, 1224–1232. [Google Scholar] [CrossRef]
- Dieffenderfer, J.P.; Goodell, H.; Bent, B.; Beppler, E.; Jayakumar, R.; Yokus, M.; Jur, J.S.; Bozkurt, A.; Peden, D. Wearable wireless sensors for chronic respiratory disease monitoring. In Proceedings of the 2015 IEEE 12th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Cambridge, MA, USA, 9–12 June 2015; pp. 1–6. [Google Scholar]
- Shin, J.H.; Kwon, J.; Kim, J.U.; Ryu, H.; Ok, J.; Joon Kwon, S.; Park, H.; Kim, T.I. Wearable EEG electronics for a Brain–AI Closed-Loop System to enhance autonomous machine decision-making. npj Flex. Electron. 2022, 6, 32. [Google Scholar] [CrossRef]
- Hina Fathima, A.; Palanisamy, K. Battery energy storage applications in wind integrated systems—A review. In Proceedings of the 2014 International Conference on Smart Electric Grid (ISEG), Guntur, India, 19–20 September 2014; pp. 1–8. [Google Scholar]
- Archibong, E.I.; Ozuomba, S.; Ekott, E. Internet of Things (IoT)-based, Solar Powered Street Light System with Anti-vandalisation Mechanism. In Proceedings of the 2020 International Conference in Mathematics, Computer Engineering and Computer Science (ICMCECS), Ayobo, Nigeria, 18–21 March 2020; pp. 1–6. [Google Scholar]
- Sharma, P.; Salkuti, S.R.; Kim, S.C. Advancements in energy storage technologies for smart grid development. Int. J. Elect. Comput. Syst. Eng. 2022, 12, 3421. [Google Scholar] [CrossRef]
- Aljafari, B.; Vasantharaj, S.; Indragandhi, V.; Vaibhav, R. Optimization of DC, AC, and Hybrid AC/DC Microgrid-Based IoT Systems: A Review. Energies 2022, 15, 6813. [Google Scholar] [CrossRef]
- Vaghasiya, J.V.; Mayorga-Martinez, C.C.; Vyskočil, J.; Sofer, Z.; Pumera, M. Integrated biomonitoring sensing with wearable asymmetric supercapacitors based on ti 3 C 2 MXene and 1T-phase WS 2 nanosheets. Adv. Funct. Mater. 2020, 30, 2003673. [Google Scholar] [CrossRef]
- Ram, S.K.; Das, B.B.; Mahapatra, K.; Mohanty, S.P.; Choppali, U. Energy perspectives in IoT driven smart villages and smart cities. IEEE Consum. Electron. Mag. 2020, 10, 19–28. [Google Scholar] [CrossRef]
- Shrestha, K.; Sharma, S.; Pradhan, G.B.; Bhatta, T.; Rana, S.S.; Lee, S.; Seonu, S.; Shin, Y.; Park, J.Y. A triboelectric driven rectification free self-charging supercapacitor for smart IoT applications. Nano Energy 2022, 102, 107713. [Google Scholar] [CrossRef]
- Mekonnen, Y.; Burton, L.; Sarwat, A.; Bhansali, S. IoT Sensor Network Approach for Smart Farming: An Application in Food, Energy and Water System. In Proceedings of the 2018 IEEE Global Humanitarian Technology Conference (GHTC), San Jose, CA, USA, 18–21 October 2018; pp. 1–5. [Google Scholar] [CrossRef]
- Lee, S.Y.; Hong, J.H.; Hsieh, C.H.; Liang, M.C.; Chang Chien, S.Y.; Lin, K.H. Low-Power Wireless ECG Acquisition and Classification System for Body Sensor Networks. IEEE J. Biomed. Health Inform. 2015, 19, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Rodriguez-Villegas, E. System-level design trade-offs for truly wearable wireless medical devices. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 1441–1444. [Google Scholar] [CrossRef]
- Jörke, P.; Falkenberg, R.; Wietfeld, C. Power Consumption Analysis of NB-IoT and eMTC in Challenging Smart City Environments. In Proceedings of the 2018 IEEE Globecom Workshops (GC Wkshps), Abu Dhabi, United Arab Emirates, 9–13 December 2018; pp. 1–6. [Google Scholar] [CrossRef]
- Ikpehai, A.; Adebisi, B.; Rabie, K.M.; Anoh, K.; Ande, R.E.; Hammoudeh, M.; Gacanin, H.; Mbanaso, U.M. Low-Power Wide Area Network Technologies for Internet-of-Things: A Comparative Review. IEEE Internet Things J. 2019, 6, 2225–2240. [Google Scholar] [CrossRef]
- Sarangi, S.; Naik, V.; Choudhury, S.B.; Jain, P.; Kosgi, V.; Sharma, R.; Bhatt, P.; Srinivasu, P. An Affordable IoT Edge Platform for Digital Farming in Developing Regions. In Proceedings of the 2019 11th International Conference on Communication Systems & Networks (COMSNETS), Bengaluru, India, 7–11 January 2019; pp. 556–558. [Google Scholar] [CrossRef]
- Kontogiannis, S. An Internet of Things-Based Low-Power Integrated Beekeeping Safety and Conditions Monitoring System. Inventions 2019, 4, 52. [Google Scholar] [CrossRef]
- Buchmann, I. Batteries in a Portable World: A Handbook on Rechargeable Batteries for Non-Engineers, 4th ed.; Cadex Electronics Inc.: Richmond, BC, Canada, 2023. [Google Scholar]
- Armand, M.; Tarascon, J.M. Building better batteries. Nature 2008, 451, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Linden, D.; Reddy, T. Handbook of Batteries; McGraw-Hill Handbooks; McGraw-Hill Education: New York, NY, USA, 2001. [Google Scholar]
- Mitcheson, P.D. Energy harvesting for human wearable and implantable bio-sensors. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 3432–3436. [Google Scholar] [CrossRef]
- May, G.J.; Davidson, A.; Monahov, B. Lead batteries for utility energy storage: A review. J. Energy Storage 2018, 15, 145–157. [Google Scholar] [CrossRef]
- Cano, Z.P.; Banham, D.; Ye, S.; Hintennach, A.; Lu, J.; Fowler, M.; Chen, Z. Batteries and fuel cells for emerging electric vehicle markets. Nat. Energy 2018, 3, 279–289. [Google Scholar] [CrossRef]
- Ding, Y.; Cano, Z.P.; Yu, A.; Lu, J.; Chen, Z. Automotive Li-ion batteries: Current status and future perspectives. Electrochem. Energy Rev. 2019, 2, 1–28. [Google Scholar] [CrossRef]
- Zhao, J.; Burke, A.F. Review on supercapacitors: Technologies and performance evaluation. J. Energy Chem. 2021, 59, 276–291. [Google Scholar] [CrossRef]
- Wang, W.; Luo, Q.; Li, B.; Wei, X.; Li, L.; Yang, Z. Recent Progress in Redox Flow Battery Research and Development. Adv. Funct. Mater. 2013, 23, 970–986. [Google Scholar] [CrossRef]
- Alotto, P.; Guarnieri, M.; Moro, F. Redox flow batteries for the storage of renewable energy: A review. Renew. Sustain. Energy Rev. 2014, 29, 325–335. [Google Scholar] [CrossRef]
- Leong, K.W.; Wang, Y.; Ni, M.; Pan, W.; Luo, S.; Leung, D.Y. Rechargeable Zn-air batteries: Recent trends and future perspectives. Renew. Sustain. Energy Rev. 2022, 154, 111771. [Google Scholar] [CrossRef]
- Powers, R. Batteries for low power electronics. Proc. IEEE 1995, 83, 687–693. [Google Scholar] [CrossRef]
- Collath, N.; Tepe, B.; Englberger, S.; Jossen, A.; Hesse, H. Aging aware operation of lithium-ion battery energy storage systems: A review. J. Energy Storage 2022, 55, 105634. [Google Scholar] [CrossRef]
- Dehghani-Sanij, A.; Tharumalingam, E.; Dusseault, M.; Fraser, R. Study of energy storage systems and environmental challenges of batteries. Renew. Sustain. Energy Rev. 2019, 104, 192–208. [Google Scholar] [CrossRef]
- Kebede, A.A.; Kalogiannis, T.; Van Mierlo, J.; Berecibar, M. A comprehensive review of stationary energy storage devices for large scale renewable energy sources grid integration. Renew. Sustain. Energy Rev. 2022, 159, 112213. [Google Scholar] [CrossRef]
- Luo, X.; Wang, J.; Dooner, M.; Clarke, J. Overview of current development in electrical energy storage technologies and the application potential in power system operation. Appl. Energy 2015, 137, 511–536. [Google Scholar] [CrossRef]
- Lu, Q.; Zou, X.; Bu, Y.; An, L.; Wang, Y.; Shao, Z. What matters in engineering next-generation rechargeable Zn-air batteries? Next Energy 2023, 1, 100025. [Google Scholar] [CrossRef]
- Alipour, M.; Ziebert, C.; Conte, F.V.; Kizilel, R. A Review on Temperature-Dependent Electrochemical Properties, Aging, and Performance of Lithium-Ion Cells. Batteries 2020, 6, 35. [Google Scholar] [CrossRef]
- Goikolea, E.; Mysyk, R. Chapter Four - Nanotechnology in Electrochemical Capacitors. In Emerging Nanotechnologies in Rechargeable Energy Storage Systems; Rodriguez-Martinez, L.M., Omar, N., Eds.; Micro and Nano Technologies; Elsevier: Boston, MA, USA, 2017; pp. 131–169. [Google Scholar] [CrossRef]
- Townsend, A.; Gouws, R. A Comparative Review of Lead-Acid, Lithium-Ion and Ultra-Capacitor Technologies and Their Degradation Mechanisms. Energies 2022, 15, 4930. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, W.; Yuan, X.; Tang, H.; Tang, Y.; Wang, M.; Zuo, J.; Song, Z.; Sun, J. Environmental impact analysis and process optimization of batteries based on life cycle assessment. J. Clean. Prod. 2018, 174, 1262–1273. [Google Scholar] [CrossRef]
- Porzio, J.; Scown, C.D. Life-Cycle Assessment Considerations for Batteries and Battery Materials. Adv. Energy Mater. 2021, 11, 2100771. [Google Scholar] [CrossRef]
- Troy, S.; Schreiber, A.; Reppert, T.; Gehrke, H.G.; Finsterbusch, M.; Uhlenbruck, S.; Stenzel, P. Life Cycle Assessment and resource analysis of all-solid-state batteries. Appl. Energy 2016, 169, 757–767. [Google Scholar] [CrossRef]
- Mandade, P.; Weil, M.; Baumann, M.; Wei, Z. Environmental life cycle assessment of emerging solid-state batteries: A review. Chem. Eng. J. Adv. 2023, 13, 100439. [Google Scholar] [CrossRef]
- Yang, S.Y.; Sencadas, V.; You, S.S.; Jia, N.Z.X.; Srinivasan, S.S.; Huang, H.W.; Ahmed, A.E.; Liang, J.Y.; Traverso, G. Powering Implantable and Ingestible Electronics. Adv. Funct. Mater. 2021, 31, 2009289. [Google Scholar] [CrossRef] [PubMed]
- Deutsches Institut für Normung e.V. IEC 60086-2:2015; International Standard: Primary Batteries—Part 2: Physical and Electrical Specifications (IEC 60086-2:2015). Beuth-Verlag: Berlin, Germany, 2016. [CrossRef]
- Morin, É.; Maman, M.; Guizzetti, R.; Duda, A. Comparison of the Device Lifetime in Wireless Networks for the Internet of Things. IEEE Access 2017, 5, 7097–7114. [Google Scholar] [CrossRef]
- Ates, H.C.; Nguyen, P.Q.; Gonzalez-Macia, L.; Morales-Narváez, E.; Güder, F.; Collins, J.J.; Dincer, C. End-to-end design of wearable sensors. Nat. Rev. Mater. 2022, 7, 887–907. [Google Scholar] [CrossRef]
- Alaa, M.; Zaidan, A.; Zaidan, B.; Talal, M.; Kiah, M. A review of smart home applications based on Internet of Things. J. Netw. Comput. Appl. 2017, 97, 48–65. [Google Scholar] [CrossRef]
- Sinha, B.B.; Dhanalakshmi, R. Recent advancements and challenges of Internet of Things in smart agriculture: A survey. Future Gener. Comput. Syst. 2022, 126, 169–184. [Google Scholar] [CrossRef]
- Cerchecci, M.; Luti, F.; Mecocci, A.; Parrino, S.; Peruzzi, G.; Pozzebon, A. A Low Power IoT Sensor Node Architecture for Waste Management Within Smart Cities Context. Sensors 2018, 18, 1282. [Google Scholar] [CrossRef]
- Sanislav, T.; Mois, G.D.; Zeadally, S.; Folea, S.C. Energy Harvesting Techniques for Internet of Things (IoT). IEEE Access 2021, 9, 39530–39549. [Google Scholar] [CrossRef]
- Han, W.; Anaya, D.V.; Wu, T.; Wu, F.; Yuce, M.R. Self-powered wearable sensors design considerations. J. Micromech. Microeng. 2022, 32, 083002. [Google Scholar] [CrossRef]
- Garg, N.; Garg, R. Energy harvesting in IoT devices: A survey. In Proceedings of the 2017 International Conference on Intelligent Sustainable Systems (ICISS), Palladam, India, 7–8 December 2017; pp. 127–131. [Google Scholar] [CrossRef]
- Sun, Y.; Li, Y.Z.; Yuan, M. Requirements, challenges, and novel ideas for wearables on power supply and energy harvesting. Nano Energy 2023, 115, 108715. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Bruce, P.G. Batteries for wearables. Natl. Sci. Rev. 2022, 10, nwac062. [Google Scholar] [CrossRef]
- Sheng, Z.; Mahapatra, C.; Zhu, C.; Leung, V.C.M. Recent Advances in Industrial Wireless Sensor Networks Toward Efficient Management in IoT. IEEE Access 2015, 3, 622–637. [Google Scholar] [CrossRef]
- Peng, S. Challenges and Prospects for Zinc-Air Batteries. In Zinc-Air Batteries: Fundamentals, Key Materials and Application; Springer Nature Singapore: Singapore, 2023; pp. 205–215. [Google Scholar] [CrossRef]




| Research Article | IoT Application | Battery Type | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | I | II | III | IV | V | VI | VII | VIII | IX | |
| [59] | • | • | ||||||||||||
| [60] | • | • | • | |||||||||||
| [61] | • | • | ||||||||||||
| [62] | • | • | ||||||||||||
| [63] | • | • | ||||||||||||
| [64] | • | • | ||||||||||||
| [65] | • | • | ||||||||||||
| [66] | • | • | ||||||||||||
| [67] | • | |||||||||||||
| [68] | • | • | ||||||||||||
| [69] | • | • | ||||||||||||
| [70] | • | • | ||||||||||||
| [71] | • | • | ||||||||||||
| [72] | • | • | ||||||||||||
| [73] | • | • | ||||||||||||
| [74] | • | • | ||||||||||||
| [75] | • | • | ||||||||||||
| [76] | • | • | • | |||||||||||
| [77] | • | • | ||||||||||||
| [78] | • | • | ||||||||||||
| [79] | • | • | ||||||||||||
| [80] | • | • | ||||||||||||
| Battery Type | Shelf Life (at Ideal Conditions) |
|---|---|
| Lead–Acid | 3–8 years |
| Nickel–Metal Hydride (NiMH) | 4–6 years |
| Lithium–Ion (Li–ion) (Non-Rechargable) | 5–10 years |
| Lithium–Ion (Li–ion) (Rechargable) | 5 year |
| Lithium–Polymer (LiPo) | 2–3 years |
| Solid-State | ≈33 years |
| Alkaline | 4–7 years |
| Zinc–Air | ≈2 years |
| Redox Flow | ≈20 years |
| Supercapacitor | Unlimited at discharged state |
| Battery Type | Nominal Cell Voltage (V) |
|---|---|
| Lead–Acid | 2.1 |
| Nickel–Metal Hydride (NiMH) | 1.2 |
| Lithium–Ion (Li–ion) | 3.6 |
| Lithium–Polymer (LiPo) | 3.7 |
| Solid-State | 3.7 |
| Alkaline | 1.5 |
| Zinc–Air | 1.4 |
| Redox Flow | 1.4 |
| Supercapacitor | 2.7 |
| Battery Types | Approximate per Wh Cost Range (USD) |
|---|---|
| Lithium–Ion | 0.9361 |
| Lead–Acid | 0.6975 |
| Nickel Metal Hydride | 0.8546 |
| Alkaline | 0.1 |
| Lithium–Polymer | 2.3095 |
| Solid-State Battery | 0.8 |
| Zinc–Air | 0.3095 |
| Redox Flow | 5.7 |
| Supercapacitors | 5 |
| Battery Type | Notes |
|---|---|
| Lead–Acid Batteries | Generally safe if maintained properly. Hazardous materials present. |
| Nickel–Metal Hydride (NiMH) Batteries | Known for good safety profile, but can experience thermal runaway if overcharged. |
| Lithium–Ion (Li–ion) Batteries | Known for potential safety risks such as fires and explosions, particularly in electric vehicles. |
| Lithium–Polymer (LiPo) Batteries | Similar safety concerns as Li–ion, with additional risk due to flexible casing. |
| Solid-State Batteries | Known for better safety due to solid electrolyte, reducing the risk of leakage and thermal runaway. |
| Alkaline Batteries | Generally safe, but may leak caustic potassium hydroxide if damaged or over-discharged. |
| Zinc–Air Batteries | Known for safety, but can suffer from drying out which can affect performance. |
| Flow Batteries | Known for good safety profile as they typically contain non-flammable electrolytes. |
| Supercapacitor | Generally safe, but may pose risks if subjected to over-voltage conditions. |
| Technology | Power Consumption | Energy Density Requirements | Range | Battery Life Expectancy | Typical Applications |
|---|---|---|---|---|---|
| BLE | Very Low | Low | Short (up to 10 m) | Medium | Wearables, Beacons |
| LoRaWAN | Low | Medium | Long | Long | Remote Sensors, Agriculture Monitoring |
| IoT over Cellular Network | Low | Medium | Long | Long | Smart Meters, Asset Trackers |
| WiFi | High | High | Short | Short | Smart Home, Industrial IoT |
| Energy Density | Temperature Operating Range | Energy Efficiency | Longevity | Cost | Safety | |
|---|---|---|---|---|---|---|
| Smart City | Important | Low | Important | Important | Crucial | Moderate |
| Smart Home | Important | Moderate | Important | Moderate | Crucial | Crucial |
| Smart Health | Moderate | Important | Important | Important | Crucial | Crucial |
| Smart Industry | Moderate | Low | Important | Important | Crucial | Low |
| Smart Farm | Low | Low | Important | Important | Crucial | Low |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, K.; Tom, N.; Yuce, M.R. Navigating Battery Choices in IoT: An Extensive Survey of Technologies and Their Applications. Batteries 2023, 9, 580. https://doi.org/10.3390/batteries9120580
Hasan K, Tom N, Yuce MR. Navigating Battery Choices in IoT: An Extensive Survey of Technologies and Their Applications. Batteries. 2023; 9(12):580. https://doi.org/10.3390/batteries9120580
Chicago/Turabian StyleHasan, Kareeb, Neil Tom, and Mehmet Rasit Yuce. 2023. "Navigating Battery Choices in IoT: An Extensive Survey of Technologies and Their Applications" Batteries 9, no. 12: 580. https://doi.org/10.3390/batteries9120580
APA StyleHasan, K., Tom, N., & Yuce, M. R. (2023). Navigating Battery Choices in IoT: An Extensive Survey of Technologies and Their Applications. Batteries, 9(12), 580. https://doi.org/10.3390/batteries9120580






