Secondary Zinc–Air Batteries: A View on Rechargeability Aspects
Abstract
1. Introduction
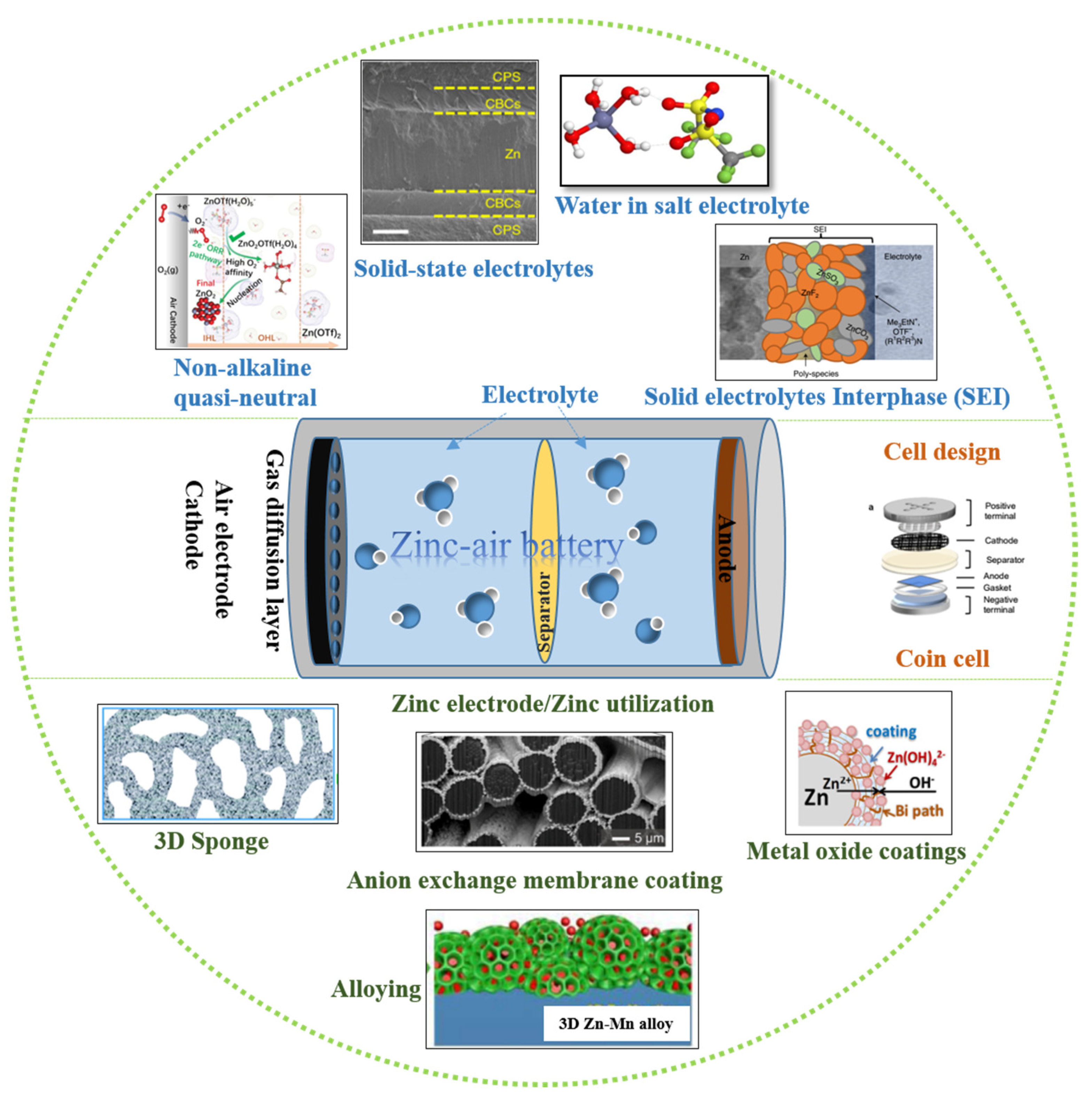
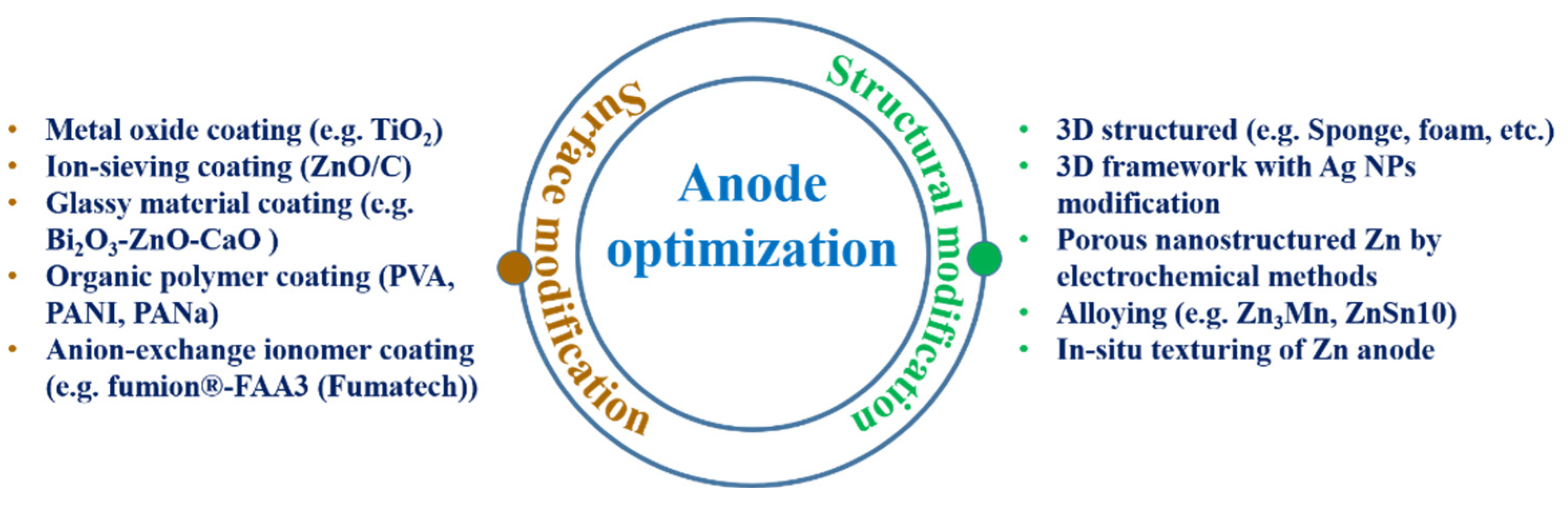
2. Anode Optimization as a Key to Zinc Utilization
2.1. Zinc Electrode-Structural and Surface Optimization
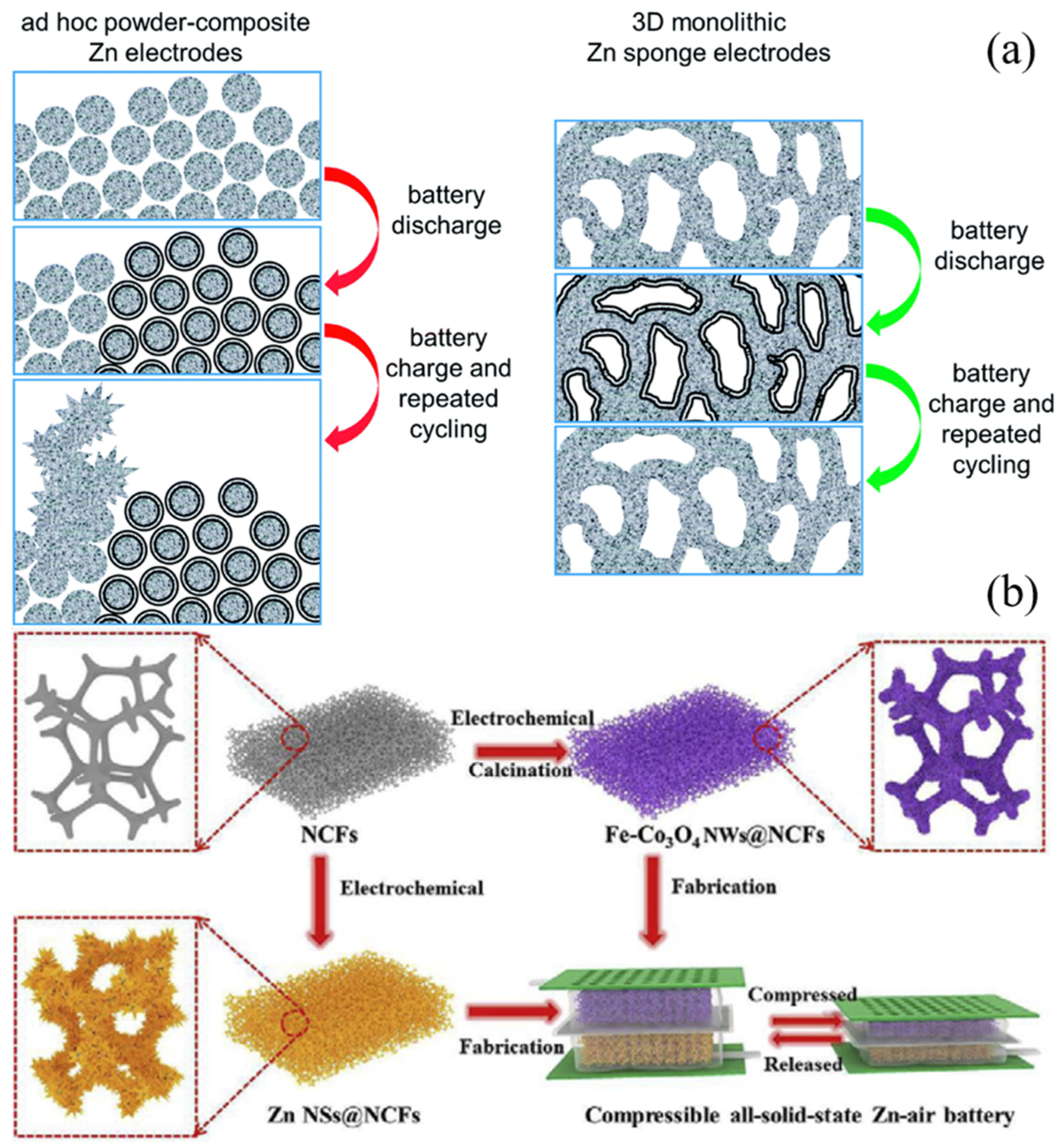
2.1.1. Structural Modification
2.1.2. Surface Modification
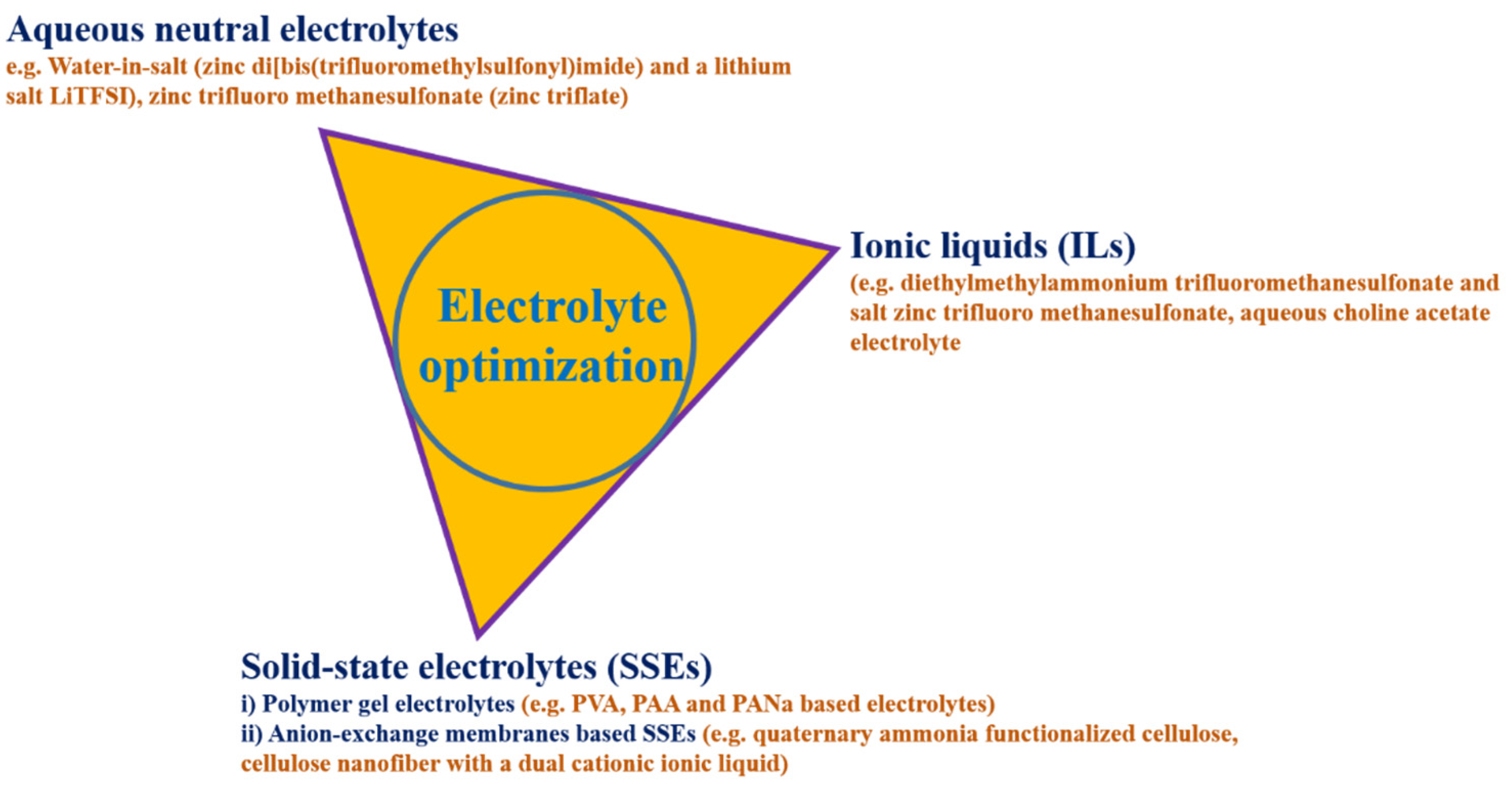
2.2. Electrolyte Optimization in Zinc–Air Secondary Cells
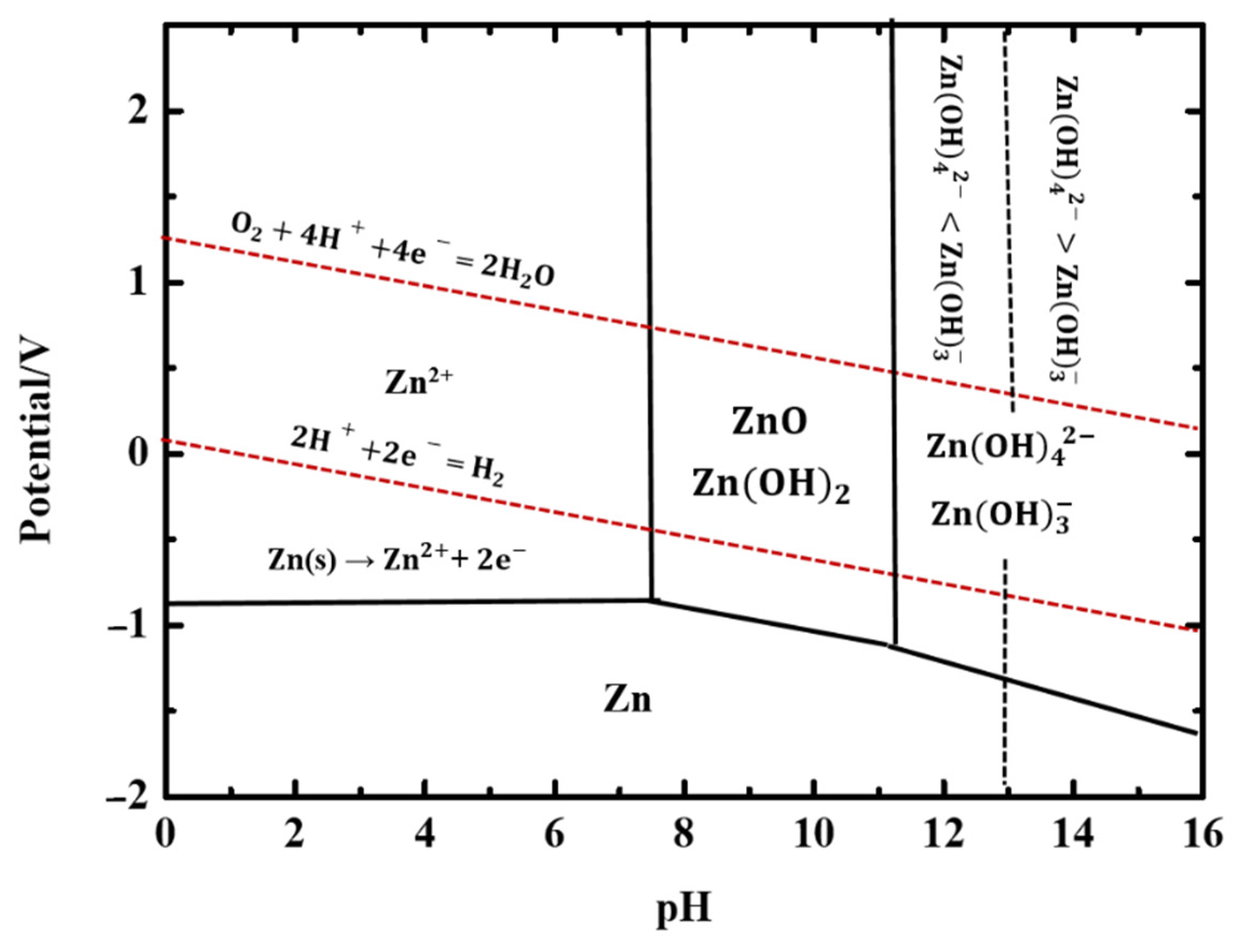
2.2.1. Aqueous Neutral Electrolytes
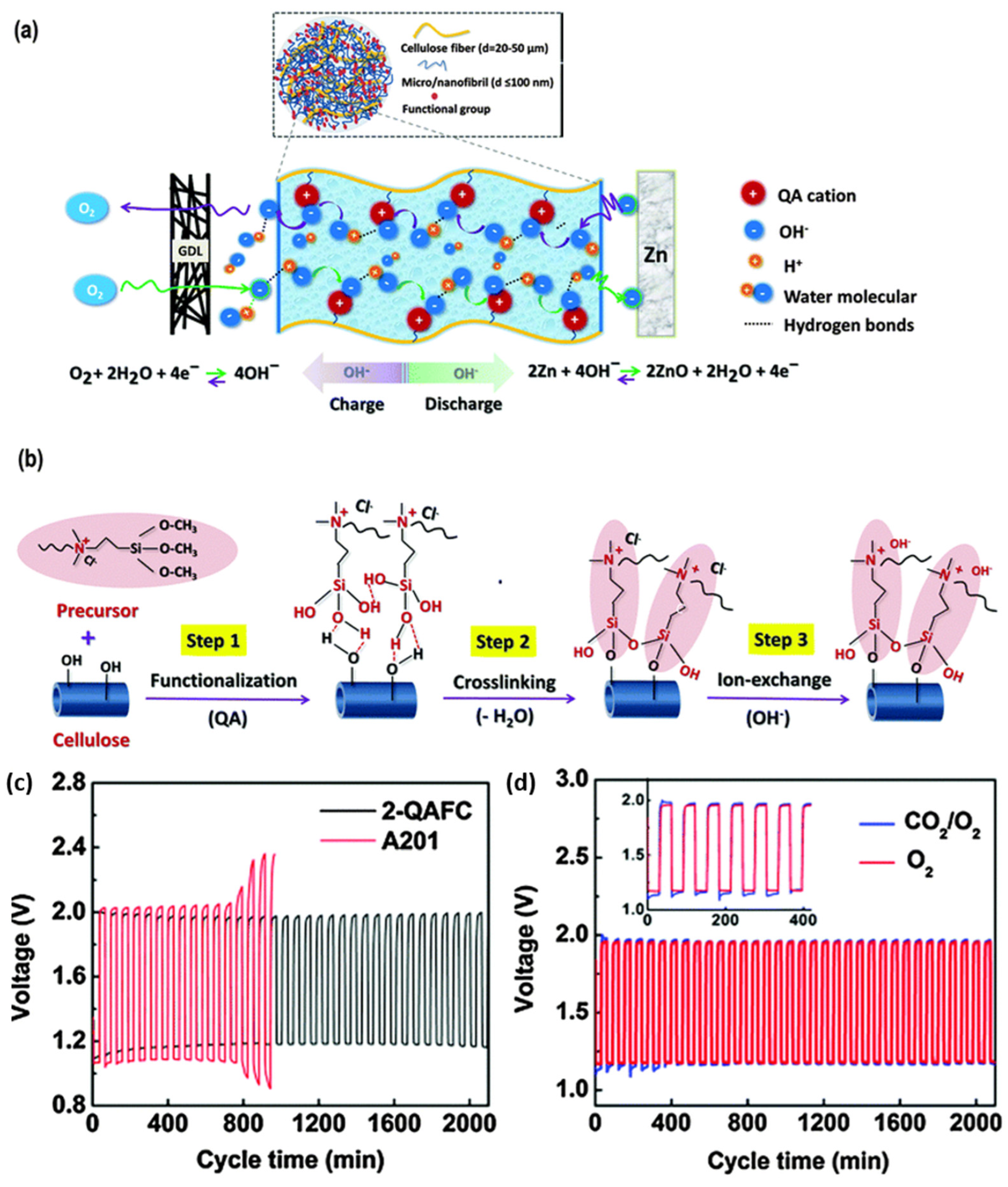
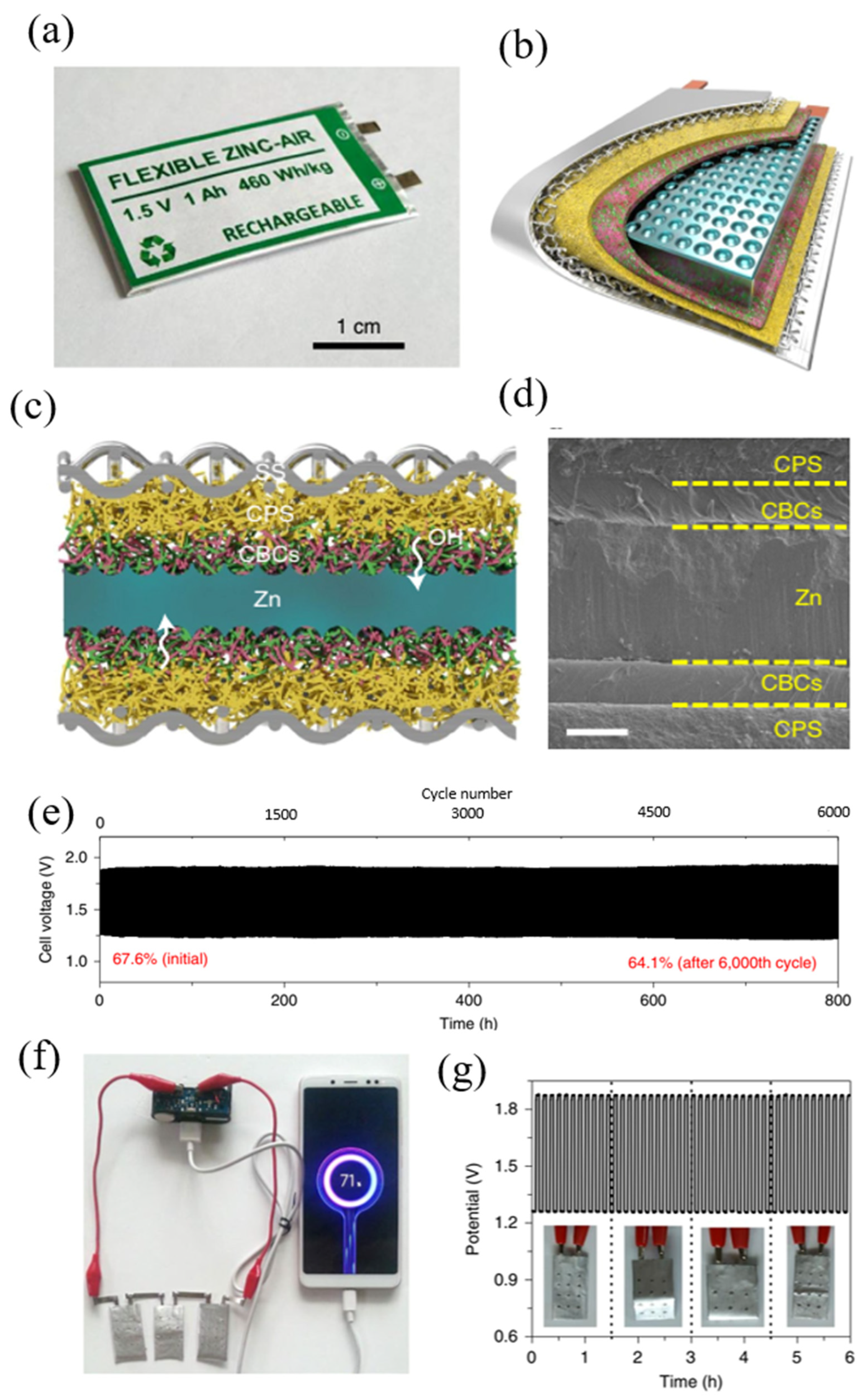
2.2.2. Solid State Electrolytes (SSEs)
2.2.3. Ionic Liquid Electrolytes
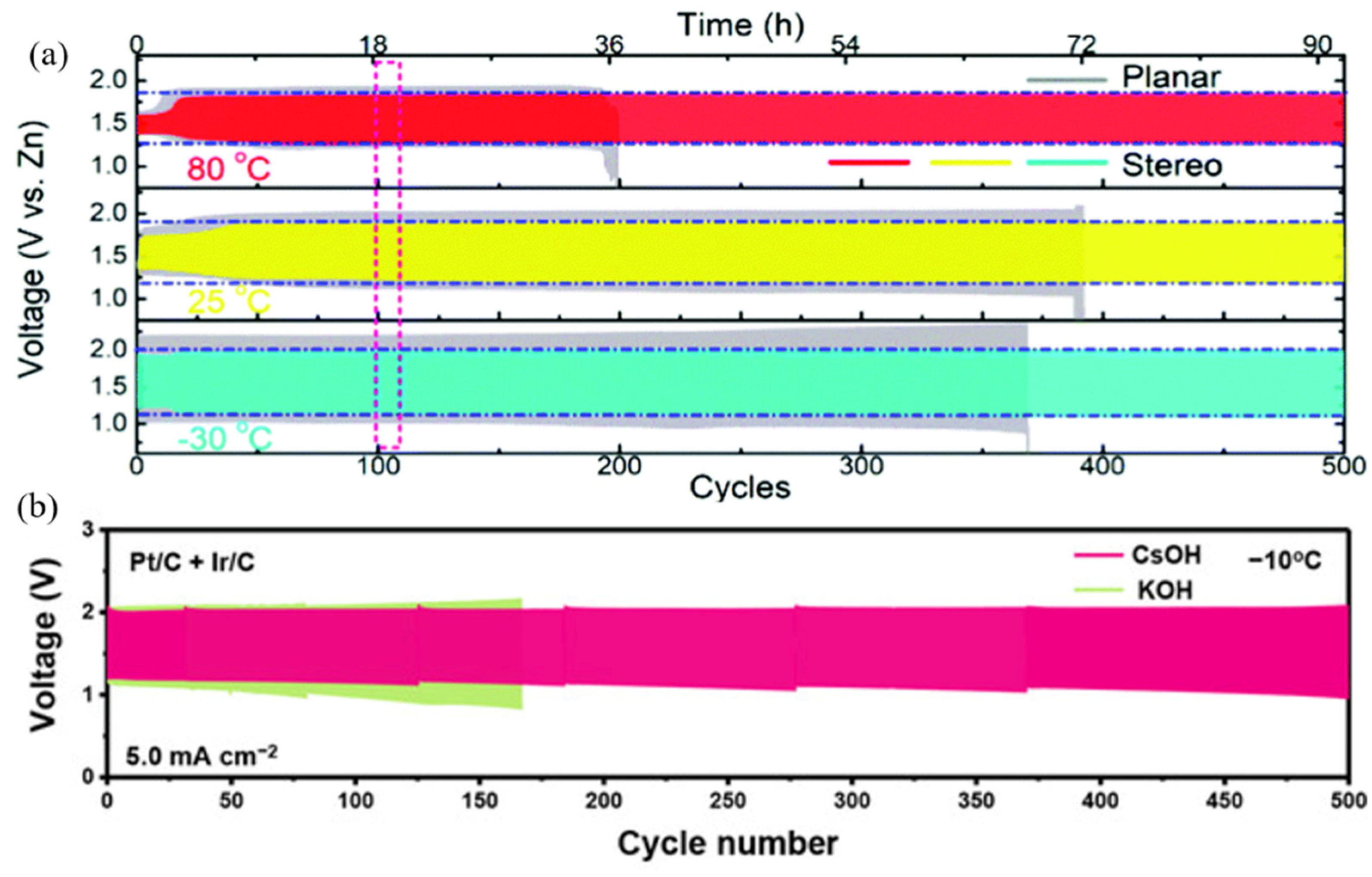
2.2.4. Wide Working Temperature
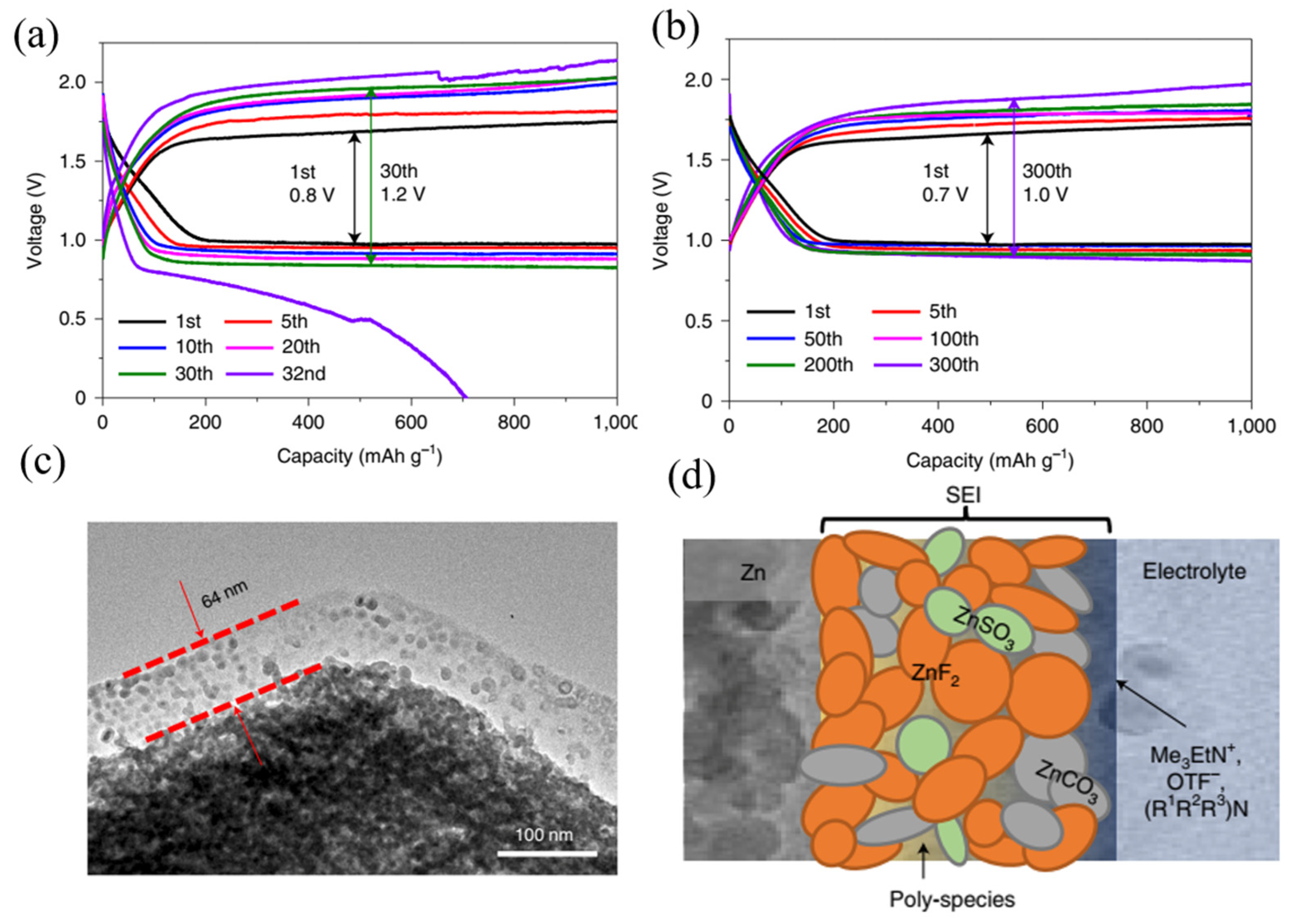
2.3. Solid Electrolyte Interphase (SEI) in Secondary Zinc Air Batteries
2.4. Cell Design for Secondary Zinc–Air Batteries
3. Conclusions and Future Perspectives
- Future theoretical investigations could be helpful to provide a framework for comprehension and interpretation of experimental outcome from modified zinc anode, additives and different electrolytes. Implementing simulation results in configuration processes will help researchers crafting rechargeable zinc–air batteries with maximum zinc utilization. Implementation of advance in situ/operando techniques to interpret passivation and corrosion mechanism of the zinc anode under different electrolytes will help to choose best configuration with maximum zinc utilization. Future research investigation requires to focus on multiple anode problems at one time. Along experimental investigations the aim should be to consider the detrimental processes including zinc anode passivation, HER and dendritic growth altogether in the charging/discharging of the batteries. These three issues of zinc anode are correlated to each other in real applications.
- Electrolytes portray a significant part in rechargeable zinc–air batteries and are responsible for ionic conductivity between the electrodes. In order to improve anode reversibility, researchers have supplemented traditional alkaline electrolytes with additives and polymer gel electrolytes. However, alkaline electrolytes are susceptible to atmospheric CO2, leading to carbonate production and cell degradation. Thus, we strongly encourage more research contributions on new electrolyte systems. For instance, quasi-neutral electrolytes could completely evade the alkaline-based parasitic side reactions restricting the long-term rechargeability in zinc–air batteries. The research on quasi-neutral electrolytes is still growing slowly with only few available publications, and their specific utilization in zinc–air batteries with detailed studies is still scarce. For further advancement, it is significant to develop a thorough understanding of the anode reversibility mechanism and oxygen redox chemistries in the quasi-neutral electrolytes. The quasi-neutral electrolytes with additives such as negatively charged organic moieties facilitate solid electrostatic interaction at the electrolyte anode interface and promote SEI formation, positively affecting anode utilization and rechargeability in zinc–air batteries. Thus, robust SEI formation should be in the criteria before designing an electrolyte system for the zinc–air batteries. Flexible solid-state zinc–air batteries have appeared as another encouraging prospect in the energy field due to parallel growth in interest in portable and wearable electronic appliances. However, the practical application of flexible zinc–air batteries is inhibited due to their poor electrochemical performance and short lifespan. To deal with those issues, the core of flexible zinc–air batteries development lies in designing SSEs with flexibility, mechanical robustness, high ion conductivity, and good water retention.
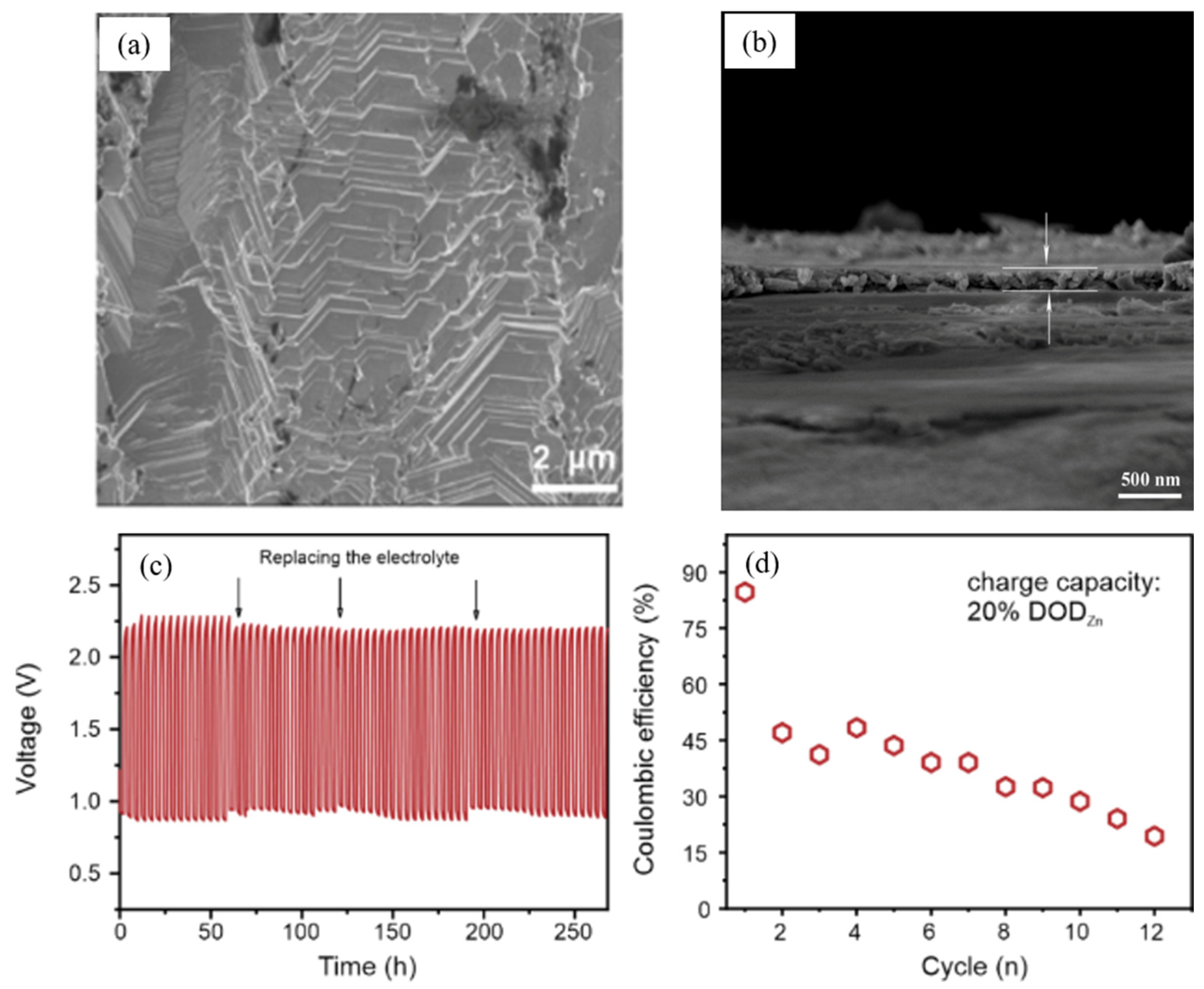
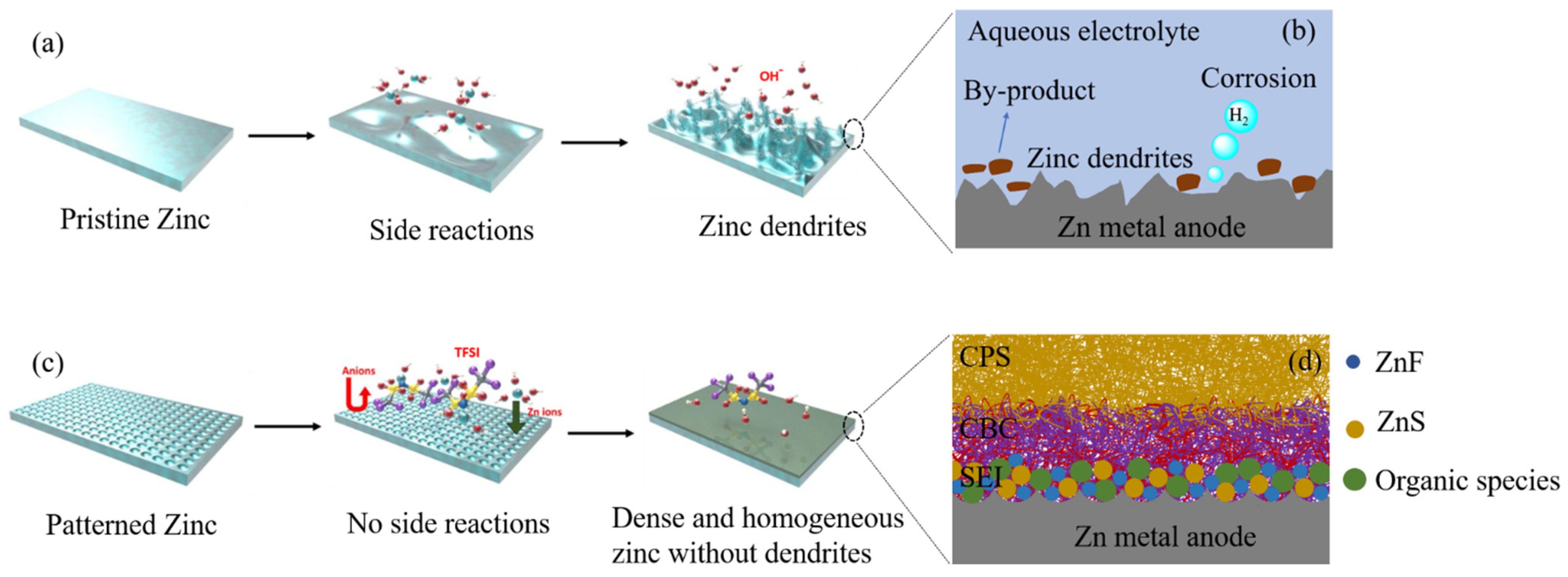
- The importance of SEI formation and its functionality in the betterment of battery performance is established for lithium-ion batteries. Because of its high redox potential, zinc is stable in both aqueous and organic electrolytes. In aqueous alkaline electrolytes, products are ionically non-conducting, which means the spontaneous generation of the SEI in zinc metal batteries is insignificant compared to the lithium-ion case. There are countable reports of SEI formation in zinc–air batteries due to additive addition in electrolytes of zinc anode structural variation. Patterning of anode surface along with addition of organic additives could lead to electrostatic interaction and development of robust and uniform SEI. Creation of robust and uniform SEI across zinc anode will lead‚ to uniform zinc stripping/plating, which leads to enhance reversibility of zinc anode ultimately enhancing rechargeability and cycle life of the batteries. However, an in-depth study of SEI in the zinc–air battery is still scarce, and composition of the SEI, their structure on the zinc anode and its effect on stripping/platting efficiency must be still an active research area.
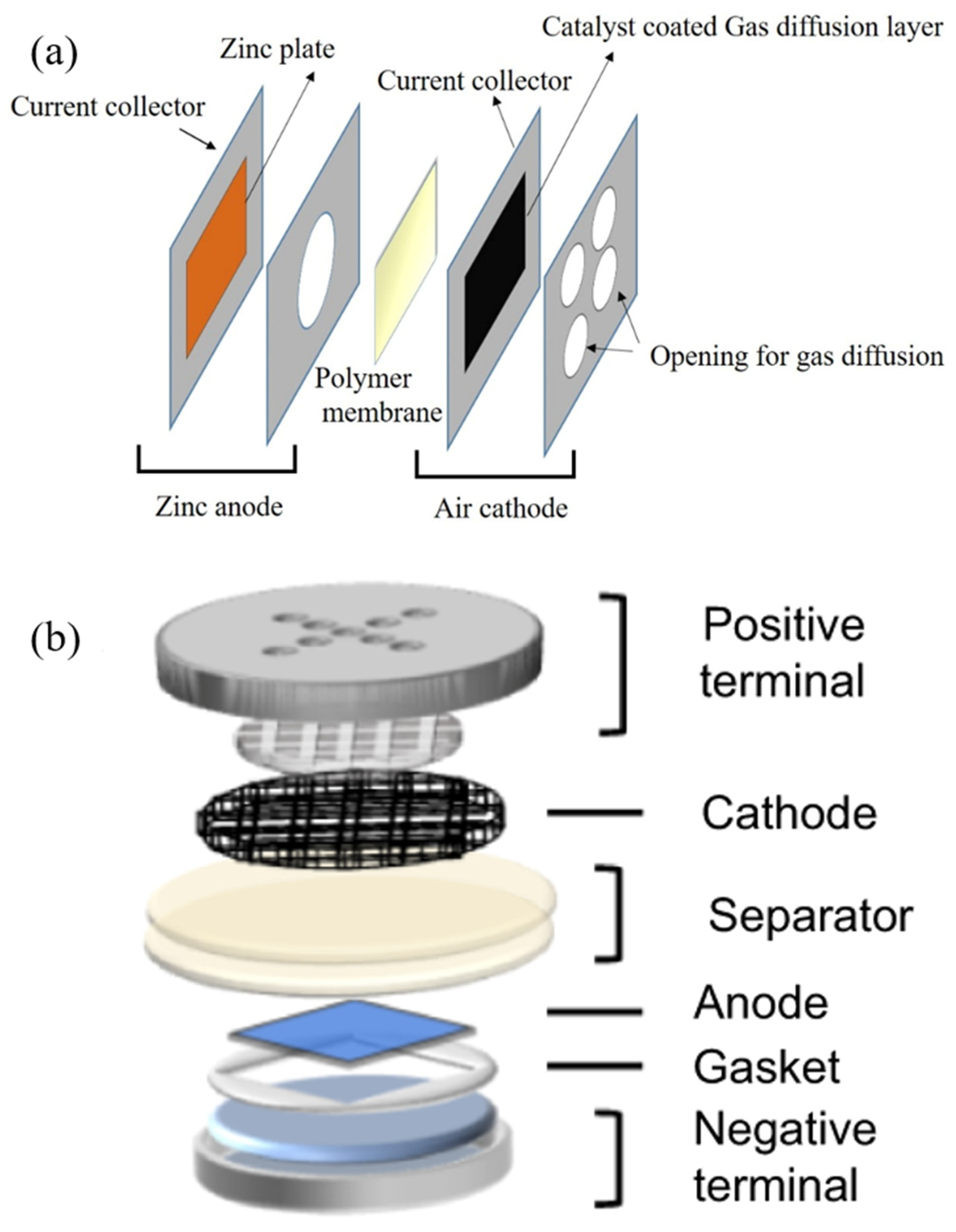
- Descriptor and cell design. To analyze the performance of zinc–air batteries, essential descriptors and metrics should be included in research reports. Discussing these significant descriptors in future research studies would assist in evaluating the practical relevancy of zinc anodes for zinc–air battery applications and permit credible cross-comparison of different research investigations. This is significant for the speedy transport of zinc–air battery technical knowledge from mostly lab-based research towards practical applications. The anode issues arising from battery configuration design or other physical components must be addressed. Currently only a few studies take account of experimental conditions and design of battery configurations. Only a minority of research reports consider the design of battery configurations with the optimization of anodes or electrolytes to get close to commercial batteries. We suggest employing coin cell-type designs because of their near-to-commercial configuration. Standard cell designs and experimental conditions enable easy comparison and interpretation of results.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, L.; Chang, Z.; Zhang, X.-B. Recent Progress on the Development of Metal-Air Batteries. Adv. Sustain. Syst. 2017, 1, 1700036. [Google Scholar] [CrossRef]
- Fu, J.; Cano, Z.P.; Park, M.G.; Yu, A.; Fowler, M.; Chen, Z. Electrically Rechargeable Zinc–Air Batteries: Progress, Challenges, and Perspectives. Adv. Mater. 2017, 29, 1604685. [Google Scholar] [CrossRef]
- Parker, J.F.; Nelson, E.S.; Wattendorf, M.D.; Chervin, C.N.; Long, J.W.; Rolison, D.R. Retaining the 3D Framework of Zinc Sponge Anodes upon Deep Discharge in Zn-Air Cells. ACS Appl. Mater. Interfaces 2014, 6, 19471–19476. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhang, Y.; Chen, P.; Wu, Y.; Yang, H.; Ding, H.; Zhang, Y.; Wang, Z.; Du, X.; Liu, N. Graphene Oxide-Modified Zinc Anode for Rechargeable Aqueous Batteries. Chem. Eng. Sci. 2019, 194, 142–147. [Google Scholar] [CrossRef]
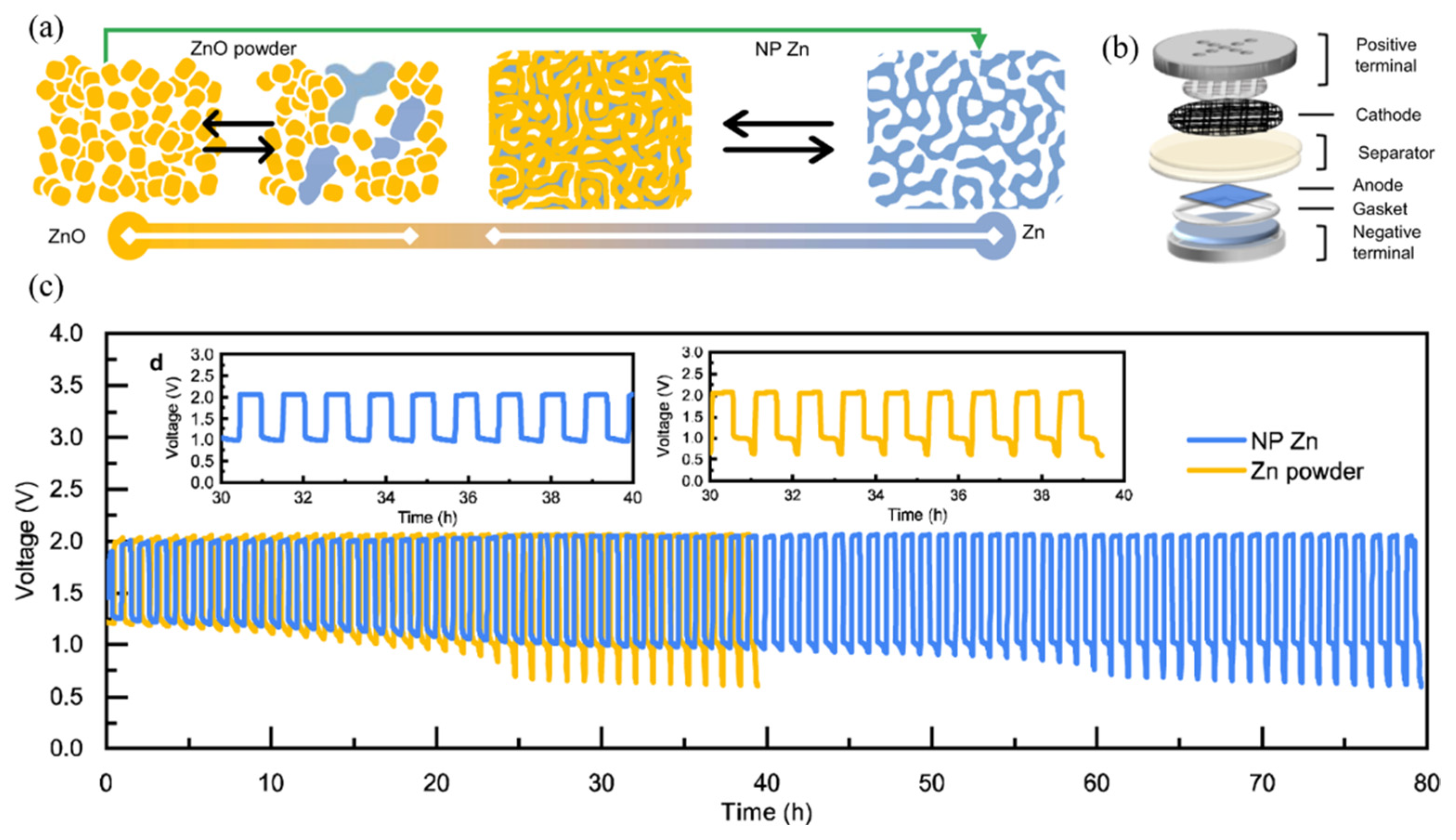
- Zhou, T.; Zhang, N.; Wu, C.; Xie, Y. Surface/Interface Nanoengineering for Rechargeable Zn-Air Batteries. Energy Environ. Sci. 2020, 13, 1132–1153. [Google Scholar] [CrossRef]
- Olabi, A.G.; Sayed, E.T.; Wilberforce, T.; Jamal, A.; Alami, A.H.; Elsaid, K.; Mohammod, S.; Rahman, A.; Shah, S.K. Metal-Air Batteries—A Review. Energies 2021, 14, 7373. [Google Scholar] [CrossRef]
- Ren, S.; Duan, X.; Liang, S.; Zhang, M.; Zheng, H. Bifunctional Electrocatalysts for Zn-Air Batteries: Recent Developments and Future Perspectives. J. Mater. Chem. A 2020, 8, 6144–6182. [Google Scholar] [CrossRef]
- Wang, H.F.; Xu, Q. Materials Design for Rechargeable Metal-Air Batteries. Matter 2019, 1, 565–595. [Google Scholar] [CrossRef]
- Xu, M.; Ivey, D.G.; Xie, Z.; Qu, W. Rechargeable Zn-Air Batteries: Progress in Electrolyte Development and Cell Configuration Advancement. J. Power Sources 2015, 283, 358–371. [Google Scholar] [CrossRef]
- Mainar, A.R.; Iruin, E.; Colmenares, L.C.; Kvasha, A.; de Meatza, I.; Bengoechea, M.; Leonet, O.; Boyano, I.; Zhang, Z.; Blazquez, J.A. An Overview of Progress in Electrolytes for Secondary Zinc-Air Batteries and Other Storage Systems Based on Zinc. J. Energy Storage 2018, 15, 304–328. [Google Scholar] [CrossRef]
- Prakoso, B.; Mahbub, M.A.A.; Yilmaz, M.; Khoiruddin; Wenten, I.G.; Handoko, A.D.; Sumboja, A. Recent Progress in Extending the Cycle-Life of Secondary Zn-Air Batteries. ChemNanoMat 2021, 7, 354–367. [Google Scholar] [CrossRef]
- Meng, F.L.; Liu, K.H.; Zhang, Y.; Shi, M.M.; Zhang, X.B.; Yan, J.M.; Jiang, Q. Recent Advances toward the Rational Design of Efficient Bifunctional Air Electrodes for Rechargeable Zn–Air Batteries. Small 2018, 14, 1703843. [Google Scholar] [CrossRef]
- Pan, J.; Tian, X.L.; Zaman, S.; Dong, Z.; Liu, H.; Park, H.S.; Xia, B.Y. Recent Progress on Transition Metal Oxides as Bifunctional Catalysts for Lithium-Air and Zinc-Air Batteries. Batter. Supercaps 2019, 2, 336–347. [Google Scholar] [CrossRef]
- Wang, X.; Peng, L.; Xu, N.; Wu, M.; Wang, Y.; Guo, J.; Sun, S.; Qiao, J. Cu/S-Occupation Bifunctional Oxygen Catalysts for Advanced Rechargeable Zinc-Air Batteries. ACS Appl. Mater. Interfaces 2020, 12, 52836–52844. [Google Scholar] [CrossRef]
- Zhou, C.; Chen, X.; Liu, S.; Han, Y.; Meng, H.; Jiang, Q.; Zhao, S.; Wei, F.; Sun, J.; Tan, T.; et al. Superdurable Bifunctional Oxygen Electrocatalyst for High-Performance Zinc-Air Batteries. J. Am. Chem. Soc. 2022, 144, 2694–2704. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, Q.; Tang, Y.; Zhang, L.; Li, Y. Zinc-Air Batteries: Are They Ready for Prime Time? Chem. Sci. 2019, 10, 8924–8929. [Google Scholar] [CrossRef]
- Deckenbach, D.; Schneider, J.J. A 3D Hierarchically Porous Nanoscale ZnO Anode for High-Energy Rechargeable Zinc-Air Batteries. J. Power Sources 2021, 488, 229393. [Google Scholar] [CrossRef]
- Mainar, A.R.; Leonet, O.; Bengoechea, M.; Boyano, I.; Meatza, I.D.; Kvasha, A.; Guerfi, A.; Blázquez, J.A. Alkaline Aqueous Electrolytes for Secondary Zinc–Air Batteries: An Overview. Int. J. Energy Res. 2016, 40, 1032–1049. [Google Scholar] [CrossRef]
- Pourzolfaghar, H.; Hosseini, S.; Zuki, F.M.; Alinejad, M.; Li, Y.Y. Recent Advancements to Mitigate Zinc Oxide Formation in Zinc-Air Batteries: A Technical Review. Mater. Today Commun. 2021, 29, 102954. [Google Scholar] [CrossRef]
- Arise, I.; Kawai, S.; Fukunaka, Y.; McLarnon, F.R. Numerical Calculation of Ionic Mass-Transfer Rates Accompanying Anodic Zinc Dissolution in Alkaline Solution. J. Electrochem. Soc. 2010, 157, A171–A178. [Google Scholar] [CrossRef]
- Horn, Q.C.; Shao-Horn, Y. Morphology and Spatial Distribution of ZnO Formed in Discharged Alkaline Zn/MnO2 AA Cells. J. Electrochem. Soc. 2003, 150, A652–A658. [Google Scholar] [CrossRef]
- Huot, J.Y.; Malservisi, M. High-Rate Capability of Zinc Anodes in Alkaline Primary Cells. J. Power Sources 2001, 96, 133–139. [Google Scholar] [CrossRef]
- Christensen, M.K.; Mathiesen, J.K.; Simonsen, S.B.; Norby, P. Transformation and Migration in Secondary Zinc-Air Batteries Studied by: In Situ Synchrotron X-Ray Diffraction and X-Ray Tomography. J. Mater. Chem. A 2019, 7, 6459–6466. [Google Scholar] [CrossRef]
- Mainar, A.R.; Colmenares, L.C.; Blázquez, J.A.; Urdampilleta, I. A Brief Overview of Secondary Zinc Anode Development: The Key of Improving Zinc-Based Energy Storage Systems. Int. J. Energy Res. 2018, 42, 903–918. [Google Scholar] [CrossRef]
- Zhao, Z.; Fan, X.; Ding, J.; Hu, W.; Zhong, C.; Lu, J. Challenges in Zinc Electrodes for Alkaline Zinc–Air Batteries: Obstacles to Commercialization. ACS Energy Lett. 2019, 4, 2259–2270. [Google Scholar] [CrossRef]
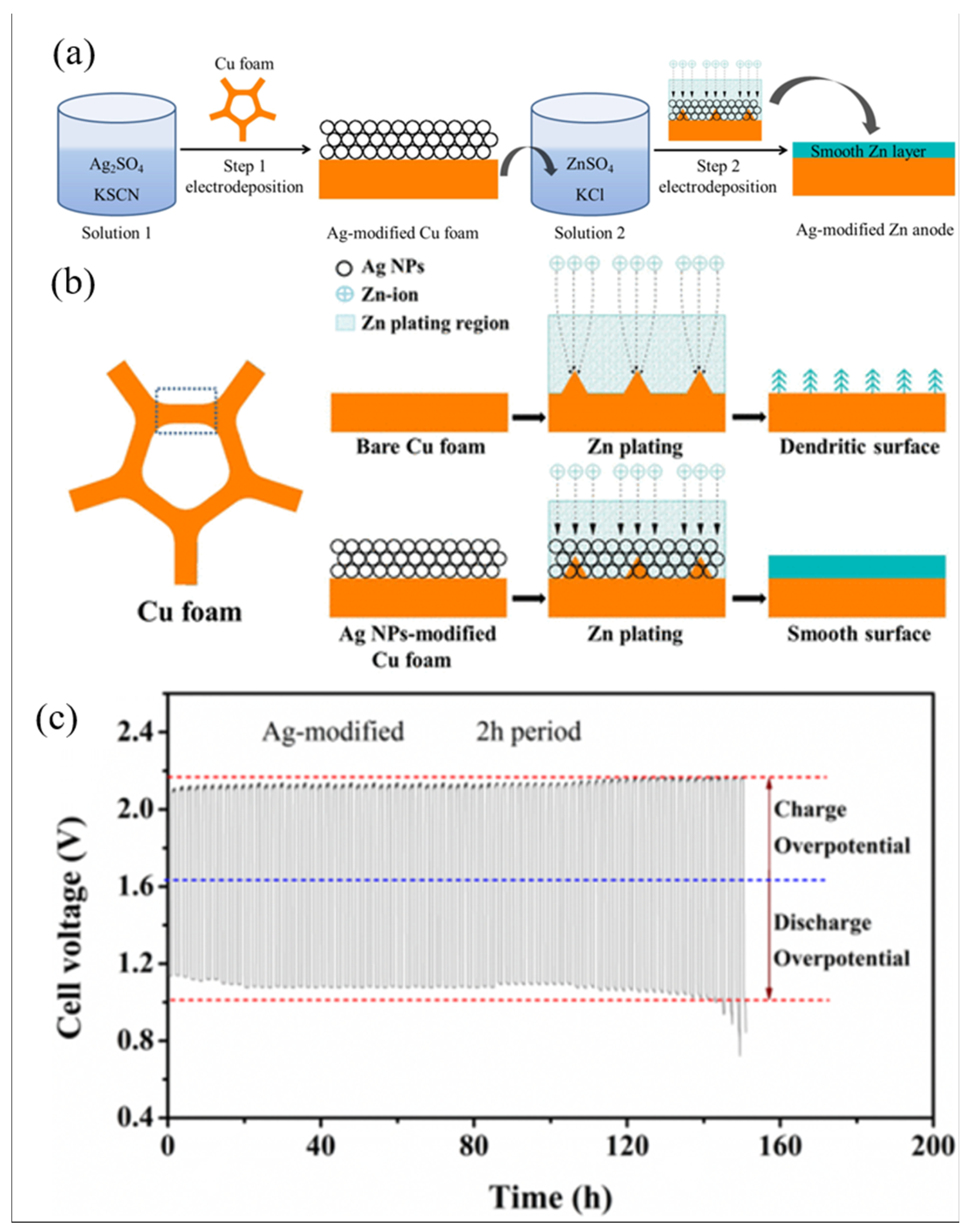
- Yu, J.; Chen, F.; Tang, Q.; Gebremariam, T.T.; Wang, J.; Gong, X.; Wang, X. Ag-Modified Cu Foams as Three-Dimensional Anodes for Rechargeable Zinc-Air Batteries. ACS Appl. Nano Mater. 2019, 2, 2679–2688. [Google Scholar] [CrossRef]
- Li, Y.; Lu, J. Metal-Air Batteries: Will They Be the Future Electrochemical Energy Storage Device of Choice? ACS Energy Lett. 2017, 2, 1370–1377. [Google Scholar] [CrossRef]
- Pan, Z.; Yang, J.; Zang, W.; Kou, Z.; Wang, C.; Ding, X.; Guan, C.; Xiong, T.; Chen, H.; Zhang, Q.; et al. All-Solid-State Sponge-like Squeezable Zinc-Air Battery. Energy Storage Mater. 2019, 23, 375–382. [Google Scholar] [CrossRef]
- Parker, J.F.; Chervin, C.N.; Nelson, E.S.; Rolison, D.R.; Long, J.W. Wiring Zinc in Three Dimensions Re-Writes Battery Performance—Dendrite-Free Cycling. Energy Environ. Sci. 2014, 7, 1117–1124. [Google Scholar] [CrossRef]
- Li, L.; Tsang, Y.C.A.; Xiao, D.; Zhu, G.; Zhi, C.; Chen, Q. Phase-Transition Tailored Nanoporous Zinc Metal Electrodes for Rechargeable Alkaline Zinc-Nickel Oxide Hydroxide and Zinc-Air Batteries. Nat. Commun. 2022, 13, 2870. [Google Scholar] [CrossRef]
- Tian, H.; Li, Z.; Feng, G.; Yang, Z.; Fox, D.; Wang, M.; Zhou, H.; Zhai, L.; Kushima, A.; Du, Y.; et al. Stable, High-Performance, Dendrite-Free, Seawater-Based Aqueous Batteries. Nat. Commun. 2021, 12, 237. [Google Scholar] [CrossRef]
- Peng, Y.; Lai, C.; Zhang, M.; Liu, X.; Yin, Y.; Li, Y.; Wu, Z. Zn–Sn Alloy Anode with Repressible Dendrite Grown and Meliorative Corrosion Resistance for Zn-Air Battery. J. Power Sources 2022, 526, 231173. [Google Scholar] [CrossRef]
- Durmus, Y.E.; Montiel Guerrero, S.S.; Tempel, H.; Hausen, F.; Kungl, H.; Eichel, R.A. Influence of Al Alloying on the Electrochemical Behavior of Zn Electrodes for Zn–Air Batteries With Neutral Sodium Chloride Electrolyte. Front. Chem. 2019, 7, 800. [Google Scholar] [CrossRef]
- Park, D.J.; Aremu, E.O.; Ryu, K.S. Bismuth Oxide as an Excellent Anode Additive for Inhibiting Dendrite Formation in Zinc-Air Secondary Batteries. Appl. Surf. Sci. 2018, 456, 507–514. [Google Scholar] [CrossRef]
- Jo, Y.N.; Prasanna, K.; Kang, S.H.; Ilango, P.R.; Kim, H.S.; Eom, S.W.; Lee, C.W. The Effects of Mechanical Alloying on the Self-Discharge and Corrosion Behavior in Zn-Air Batteries. J. Ind. Eng. Chem. 2017, 53, 247–252. [Google Scholar] [CrossRef]
- Aremu, E.O.; Park, D.J.; Ryu, K.S. The Effects of Anode Additives towards Suppressing Dendrite Growth and Hydrogen Gas Evolution Reaction in Zn-Air Secondary Batteries. Ionics 2019, 25, 4197–4207. [Google Scholar] [CrossRef]
- Chotipanich, J.; Arpornwichanop, A.; Yonezawa, T.; Kheawhom, S. Electronic and Ionic Conductivities Enhancement of Zinc Anode for Flexible Printed Zinc-Air Battery. Eng. J. 2018, 22, 47–57. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, J.; Lu, Q.; Hantusch, M.; Zhang, H.; Qu, Z.; Tang, H.; Dong, H.; Schmidt, O.G.; Holze, R.; et al. Highly Enhanced Reversibility of a Zn Anode by In-Situ Texturing. Energy Storage Mater. 2022, 47, 98–104. [Google Scholar] [CrossRef]
- Schmid, M.; Willert-Porada, M. Electrochemical Behavior of Zinc Particles with Silica Based Coatings as Anode Material for Zinc Air Batteries with Improved Discharge Capacity. J. Power Sources 2017, 351, 115–122. [Google Scholar] [CrossRef]
- Stock, D.; Dongmo, S.; Janek, J.; Schröder, D. Benchmarking Anode Concepts: The Future of Electrically Rechargeable Zinc-Air Batteries. ACS Energy Lett. 2019, 4, 1287–1300. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, Y.J.; Eom, S.W.; Choi, N.S.; Kim, K.W.; Cho, S.B. Improvement in Self-Discharge of Zn Anode by Applying Surface Modification for Zn-Air Batteries with High Energy Density. J. Power Sources 2013, 227, 177–184. [Google Scholar] [CrossRef]
- Sun, W.; Ma, M.; Zhu, M.; Xu, K.; Xu, T.; Zhu, Y.; Qian, Y. Chemical Buffer Layer Enabled Highly Reversible Zn Anode for Deeply Discharging and Long-Life Zn–Air Battery. Small 2022, 18, 2106604. [Google Scholar] [CrossRef]
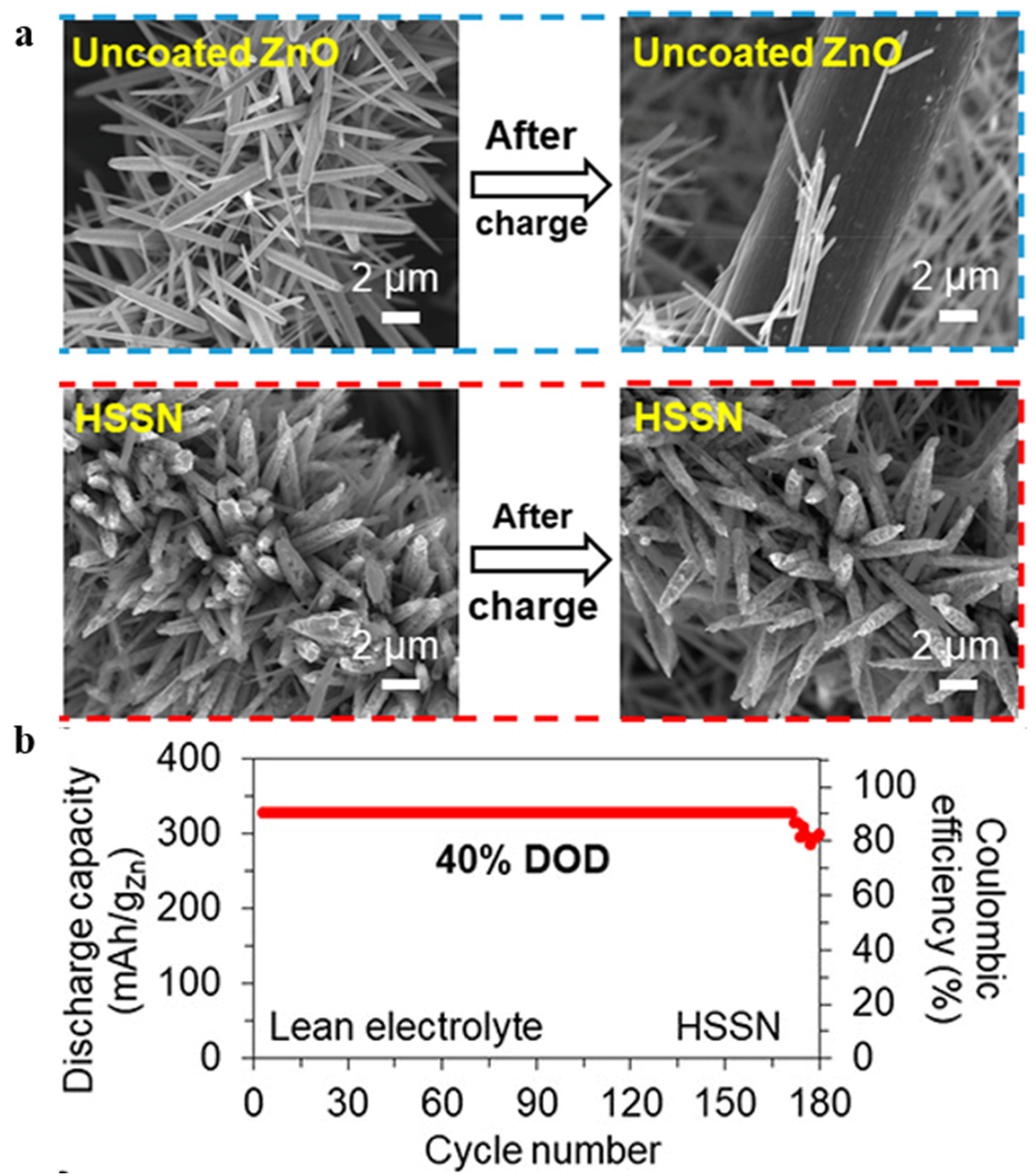
- Zhang, Y.; Wu, Y.; Ding, H.; Yan, Y.; Zhou, Z.; Ding, Y.; Liu, N. Sealing ZnO Nanorods for Deeply Rechargeable High-Energy Aqueous Battery Anodes. Nano Energy 2018, 53, 666–674. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Y.; You, W.; Tian, M.; Huang, P.W.; Zhang, Y.; Sun, Z.; Ma, Y.; Hao, T.; Liu, N. Deeply Rechargeable and Hydrogen-Evolution-Suppressing Zinc Anode in Alkaline Aqueous Electrolyte. Nano Lett. 2020, 20, 4700–4707. [Google Scholar] [CrossRef]
- Kim, Y.J.; Ryu, K.S. The Surface-Modified Effects of Zn Anode with CuO in Zn-Air Batteries. Appl. Surf. Sci. 2019, 480, 912–922. [Google Scholar] [CrossRef]
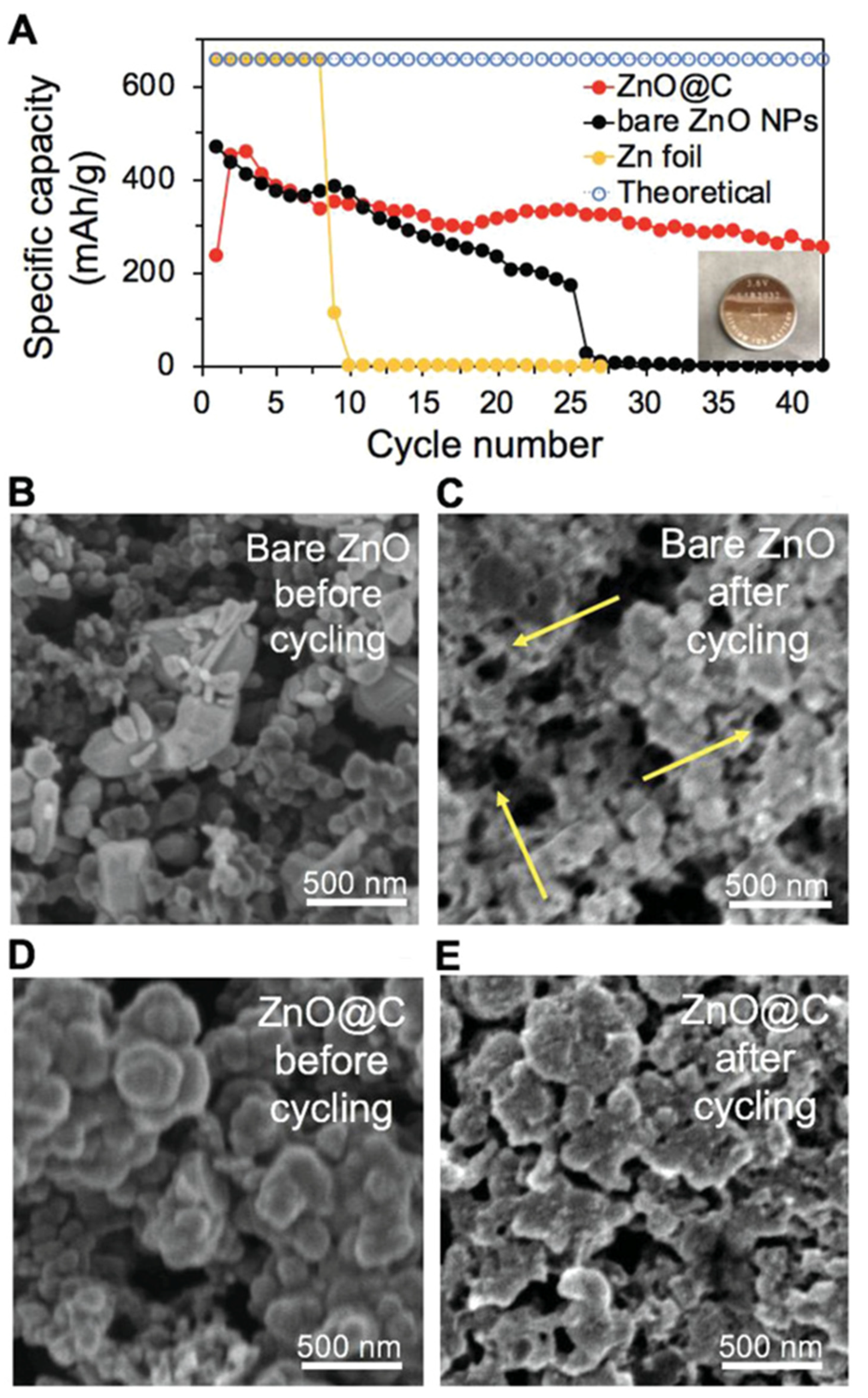
- Wu, Y.; Zhang, Y.; Ma, Y.; Howe, J.D.; Yang, H.; Chen, P.; Aluri, S.; Liu, N. Ion-Sieving Carbon Nanoshells for Deeply Rechargeable Zn-Based Aqueous Batteries. Adv. Energy Mater. 2018, 8, 1802470. [Google Scholar] [CrossRef]
- Leong, K.W.; Wang, Y.; Ni, M.; Pan, W.; Luo, S.; Leung, D.Y.C. Rechargeable Zn-Air Batteries: Recent Trends and Future Perspectives. Renew. Sustain. Energy Rev. 2022, 154, 111771. [Google Scholar] [CrossRef]
- Schmid, M.; Willert-Porada, M. Zinc Particles Coated with Bismuth Oxide Based Glasses as Anode Material for Zinc Air Batteries with Improved Electrical Rechargeability. Electrochim. Acta 2018, 260, 246–253. [Google Scholar] [CrossRef]
- Nam Jo, Y.; Santhoshkumar, P.; Prasanna, K.; Vediappan, K.; Woo Lee, C. Improving Self-Discharge and Anti-Corrosion Performance of Zn-Air Batteries Using Conductive Polymer-Coated Zn Active Materials. J. Ind. Eng. Chem. 2019, 76, 396–402. [Google Scholar] [CrossRef]
- Deyab, M.A.; Mele, G. Polyaniline/Zn-Phthalocyanines Nanocomposite for Protecting Zinc Electrode in Zn-Air Battery. J. Power Sources 2019, 443, 227264. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, D.; Huang, G.H.; Zhou, L.; Huang, B. Preparation and Properties of ZnO/PVA/β-CD/PEG Composite Electrode for Rechargeable Zinc Anode. J. Electroanal. Chem. 2018, 827, 85–92. [Google Scholar] [CrossRef]
- Stock, D.; Dongmo, S.; Walther, F.; Sann, J.; Janek, J.; Schröder, D. Homogeneous Coating with an Anion-Exchange Ionomer Improves the Cycling Stability of Secondary Batteries with Zinc Anodes. ACS Appl. Mater. Interfaces 2018, 10, 8640–8648. [Google Scholar] [CrossRef] [PubMed]
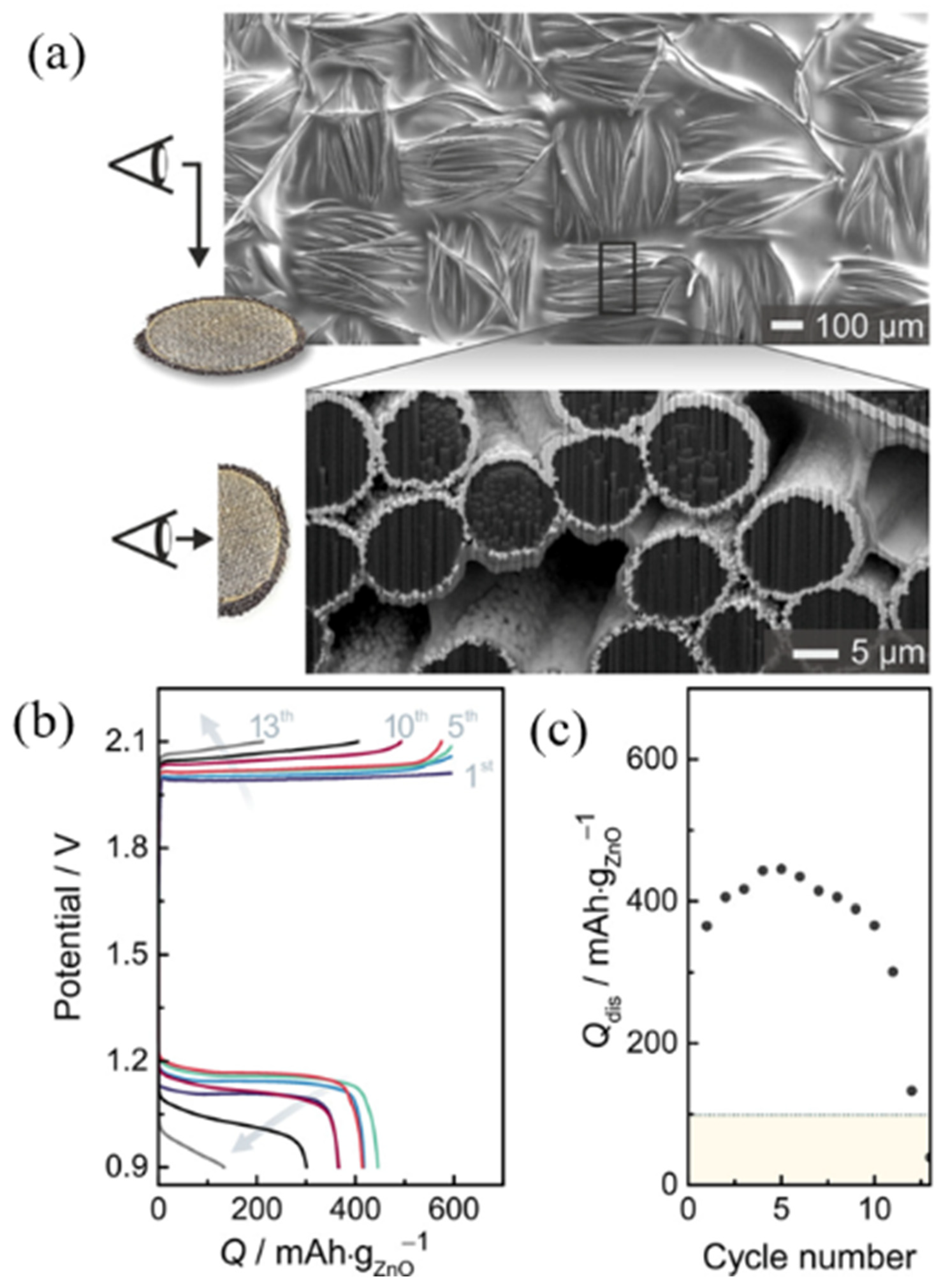
- Stock, D.; Dongmo, S.; Damtew, D.; Stumpp, M.; Konovalova, A.; Henkensmeier, D.; Schlettwein, D.; Schröder, D. Design Strategy for Zinc Anodes with Enhanced Utilization and Retention: Electrodeposited Zinc Oxide on Carbon Mesh Protected by Ionomeric Layers. ACS Appl. Energy Mater. 2018, 1, 5579–5588. [Google Scholar] [CrossRef]
- Stock, D.; Dongmo, S.; Miyazaki, K.; Abe, T.; Janek, J.; Schröder, D. Towards Zinc-Oxygen Batteries with Enhanced Cycling Stability: The Benefit of Anion-Exchange Ionomer for Zinc Sponge Anodes. J. Power Sources 2018, 395, 195–204. [Google Scholar] [CrossRef]
- Chang, J.; Wang, G.; Yang, Y. Recent Advances in Electrode Design for Rechargeable Zinc–Air Batteries. Small Sci. 2021, 1, 2100044. [Google Scholar] [CrossRef]
- Parker, J.F.; Ko, J.S.; Rolison, D.R.; Long, J.W. Translating Materials-Level Performance into Device-Relevant Metrics for Zinc-Based Batteries. Joule 2018, 2, 2519–2527. [Google Scholar] [CrossRef]
- Mainar, A.R.; Colmenares, L.C.; Grande, H.J.; Blázquez, J.A. Enhancing the Cycle Life of a Zinc–Air Battery by Means of Electrolyte Additives and Zinc Surface Protection. Batteries 2018, 4, 46. [Google Scholar] [CrossRef]
- Tran, T.N.T.; Chung, H.; Ivey, D.G. A Study of Alkaline Gel Polymer Electrolytes for Rechargeable Zinc–Air Batteries. Electrochim. Acta 2019, 327, 135021. [Google Scholar] [CrossRef]
- Liu, X.; Fan, X.; Liu, B.; Ding, J.; Deng, Y.; Han, X.; Zhong, C.; Hu, W. Mapping the Design of Electrolyte Materials for Electrically Rechargeable Zinc–Air Batteries. Adv. Mater. 2021, 33, 2006461. [Google Scholar] [CrossRef]
- Schröder, D.; Sinai Borker, N.N.; König, M.; Krewer, U. Performance of Zinc Air Batteries with Added K2CO3 in the Alkaline Electrolyte. J. Appl. Electrochem. 2015, 45, 427–437. [Google Scholar] [CrossRef]
- Liu, S.; Han, W.; Cui, B.; Liu, X.; Zhao, F.; Stuart, J.; Licht, S. A Novel Rechargeable Zinc-Air Battery with Molten Salt Electrolyte. J. Power Sources 2017, 342, 435–441. [Google Scholar] [CrossRef]
- Li, Y.; Fan, X.; Liu, X.; Qu, S.; Liu, J.; Ding, J.; Han, X.; Deng, Y.; Hu, W.; Zhong, C. Long-Battery-Life Flexible Zinc-Air Battery with near-Neutral Polymer Electrolyte and Nanoporous Integrated Air Electrode. J. Mater. Chem. A 2019, 7, 25449–25457. [Google Scholar] [CrossRef]
- Xiao, C.; Yao, R.; Zhu, H.; Qian, L.; Yang, C. Choline Chloride Enhances the Electrochemical Stability of Zinc Plating/Stripping. Chem. Commun. 2022, 58, 10088–10090. [Google Scholar] [CrossRef] [PubMed]
- Ingale, P.; Sakthivel, M.; Drillet, J.-F. Test of Diethylmethylammonium Trifluoromethanesulfonate Ionic Liquid as Electrolyte in Electrically Rechargeable Zn/Air Battery. J. Electrochem. Soc. 2017, 164, H5224–H5229. [Google Scholar] [CrossRef]
- Hwang, H.J.; Chi, W.S.; Kwon, O.; Lee, J.G.; Kim, J.H.; Shul, Y.G. Selective Ion Transporting Polymerized Ionic Liquid Membrane Separator for Enhancing Cycle Stability and Durability in Secondary Zinc-Air Battery Systems. ACS Appl. Mater. Interfaces 2016, 8, 26298–26308. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Lee, D.U.; Hassan, F.M.; Bai, Z.; Park, M.G.; Chen, Z. Flexible High-Energy Polymer-Electrolyte-Based Rechargeable Zinc-Air Batteries. Adv. Mater. 2015, 27, 5617–5622. [Google Scholar] [CrossRef]
- Wang, F.; Borodin, O.; Gao, T.; Fan, X.; Sun, W.; Han, F.; Faraone, A.; Dura, J.A.; Xu, K.; Wang, C. Highly Reversible Zinc Metal Anode for Aqueous Batteries. Nat. Mater. 2018, 17, 543–549. [Google Scholar] [CrossRef]
- Sun, W.; Wang, F.; Zhang, B.; Zhang, M.; Küpers, V.; Ji, X.; Theile, C.; Bieker, P.; Xu, K.; Wang, C.; et al. A Rechargeable Zinc-Air Battery Based on Zinc Peroxide Chemistry. Science 2021, 371, 46–51. [Google Scholar] [CrossRef]
- Chen, P.; Zhang, K.; Tang, D.; Liu, W.; Meng, F.; Huang, Q.; Liu, J. Recent Progress in Electrolytes for Zn–Air Batteries. Front. Chem. 2020, 8, 372. [Google Scholar] [CrossRef]
- Sumboja, A.; Lübke, M.; Wang, Y.; An, T.; Zong, Y.; Liu, Z. All-Solid-State, Foldable, and Rechargeable Zn-Air Batteries Based on Manganese Oxide Grown on Graphene-Coated Carbon Cloth Air Cathode. Adv. Energy Mater. 2017, 7, 1700927. [Google Scholar] [CrossRef]
- Hao, J.; Li, X.; Zeng, X.; Li, D.; Mao, J.; Guo, Z. Deeply Understanding the Zn Anode Behaviour and Corresponding Improvement Strategies in Different Aqueous Zn-Based Batteries. Energy Environ. Sci. 2020, 13, 3917–3949. [Google Scholar] [CrossRef]
- Pei, Z.; Yuan, Z.; Wang, C.; Zhao, S.; Fei, J.; Wei, L.; Chen, J.; Wang, C.; Qi, R.; Liu, Z.; et al. A Flexible Rechargeable Zinc–Air Battery with Excellent Low-Temperature Adaptability. Angew. Chem. Int. Ed. 2020, 59, 4793–4799. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Liu, J.; Song, Z.; Han, X.; Deng, Y.; Zhong, C.; Hu, W. Porous Nanocomposite Gel Polymer Electrolyte with High Ionic Conductivity and Superior Electrolyte Retention Capability for Long-Cycle-Life Flexible Zinc–Air Batteries. Nano Energy 2019, 56, 454–462. [Google Scholar] [CrossRef]
- Song, Z.; Ding, J.; Liu, B.; Liu, X.; Han, X.; Deng, Y.; Hu, W.; Zhong, C. A Rechargeable Zn–Air Battery with High Energy Efficiency and Long Life Enabled by a Highly Water-Retentive Gel Electrolyte with Reaction Modifier. Adv. Mater. 2020, 32, 1908127. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Zhang, J.; Song, X.; Zarrin, H.; Tian, X.; Qiao, J.; Rasen, L.; Li, K.; Chen, Z. A Flexible Solid-State Electrolyte for Wide-Scale Integration of Rechargeable Zinc-Air Batteries. Energy Environ. Sci. 2016, 9, 663–670. [Google Scholar] [CrossRef]
- Huang, Y.; Li, Z.; Pei, Z.; Liu, Z.; Li, H.; Zhu, M.; Fan, J.; Dai, Q.; Zhang, M.; Dai, L.; et al. Solid-State Rechargeable Zn//NiCo and Zn–Air Batteries with Ultralong Lifetime and High Capacity: The Role of a Sodium Polyacrylate Hydrogel Electrolyte. Adv. Energy Mater. 2018, 8, 1802288. [Google Scholar] [CrossRef]
- Dou, H.; Xu, M.; Zheng, Y.; Li, Z.; Wen, G.; Zhang, Z.; Yang, L.; Ma, Q.; Yu, A.; Luo, D.; et al. Bioinspired Tough Solid-State Electrolyte for Flexible Ultralong-Life Zinc–Air Battery. Adv. Mater. 2022, 34, 2110585. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, J.; Song, X.; Jiang, G.; Zarrin, H.; Xu, P.; Li, K.; Yu, A.; Chen, Z. Laminated Cross-Linked Nanocellulose/Graphene Oxide Electrolyte for Flexible Rechargeable Zinc–Air Batteries. Adv. Energy Mater. 2016, 6, 1600476. [Google Scholar] [CrossRef]
- Zhao, N.; Wu, F.; Xing, Y.; Qu, W.; Chen, N.; Shang, Y.; Yan, M.; Li, Y.; Li, L.; Chen, R. Flexible Hydrogel Electrolyte with Superior Mechanical Properties Based on Poly(Vinyl Alcohol) and Bacterial Cellulose for the Solid-State Zinc-Air Batteries. ACS Appl. Mater. Interfaces 2019, 11, 15537–15542. [Google Scholar] [CrossRef]
- Wei, Y.; Wang, M.; Xu, N.; Peng, L.; Mao, J.; Gong, Q.; Qiao, J. Alkaline Exchange Polymer Membrane Electrolyte for High Performance of All-Solid-State Electrochemical Devices. ACS Appl. Mater. Interfaces 2018, 10, 29593–29598. [Google Scholar] [CrossRef]
- Wang, M.; Xu, N.; Fu, J.; Liu, Y.; Qiao, J. High-Performance Binary Cross-Linked Alkaline Anion Polymer Electrolyte Membranes for All-Solid-State Supercapacitors and Flexible Rechargeable Zinc-Air Batteries. J. Mater. Chem. A 2019, 7, 11257–11264. [Google Scholar] [CrossRef]
- Xu, M.; Dou, H.; Zhang, Z.; Zheng, Y.; Ren, B.; Ma, Q.; Wen, G.; Luo, D.; Yu, A.; Zhang, L.; et al. Hierarchically Nanostructured Solid-State Electrolyte for Flexible Rechargeable Zinc–Air Batteries. Angew. Chem. Int. Ed. 2022, 61, e202117703. [Google Scholar] [CrossRef]
- Shinde, S.S.; Jung, J.Y.; Wagh, N.K.; Lee, C.H.; Kim, D.H.; Kim, S.H.; Lee, S.U.; Lee, J.H. Ampere-Hour-Scale Zinc–Air Pouch Cells. Nat. Energy 2021, 6, 592–604. [Google Scholar] [CrossRef]
- Zuo, Y.; Wang, K.; Zhao, S.; Wei, M.; Liu, X.; Zhang, P. A High Areal Capacity Solid-State Zinc-Air Battery via Interface Optimization of Electrode and Electrolyte. Chem. Eng. J. 2022, 430, 132996. [Google Scholar] [CrossRef]
- Famprikis, T.; Canepa, P.; Dawson, J.A.; Islam, M.S.; Masquelier, C. Fundamentals of Inorganic Solid-State Electrolytes for Batteries. Nat. Mater. 2019, 18, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Zhang, G.; Du, L.; Yang, D.; Yang, H.; Sun, S. Defect Electrocatalysts and Alkaline Electrolyte Membranes in Solid-State Zinc–Air Batteries: Recent Advances, Challenges, and Future Perspectives. Small Methods 2021, 5, 2000868. [Google Scholar] [CrossRef] [PubMed]
- Balaish, M.; Kraytsberg, A.; Ein-Eli, Y. A Critical Review on Lithium-Air Battery Electrolytes. Phys. Chem. Chem. Phys. 2014, 16, 2801–2822. [Google Scholar] [CrossRef]
- Simons, T.J.; Torriero, A.A.J.; Howlett, P.C.; MacFarlane, D.R.; Forsyth, M. High Current Density, Efficient Cycling of Zn2+ in 1-Ethyl-3-Methylimidazolium Dicyanamide Ionic Liquid: The Effect of Zn2+ Salt and Water Concentration. Electrochem. Commun. 2012, 18, 119–122. [Google Scholar] [CrossRef]
- Kar, M.; Simons, T.J.; Forsyth, M.; MacFarlane, D.R. Ionic Liquid Electrolytes as a Platform for Rechargeable Metal-Air Batteries: A Perspective. Phys. Chem. Chem. Phys. 2014, 16, 18658–18674. [Google Scholar] [CrossRef]
- Ghazvini, M.S.; Pulletikurthi, G.; Cui, T.; Kuhl, C.; Endres, F. Electrodeposition of Zinc from 1-Ethyl-3-Methylimidazolium Acetate-Water Mixtures: Investigations on the Applicability of the Electrolyte for Zn-Air Batteries. J. Electrochem. Soc. 2018, 165, D354–D363. [Google Scholar] [CrossRef]
- Sakthivel, M.; Batchu, S.P.; Shah, A.A.; Kim, K.; Peters, W.; Drillet, J.F. An Electrically Rechargeable Zinc/Air Cell with an Aqueous Choline Acetate Electrolyte. Materials 2020, 13, 2975. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zheng, X.; Voznyy, O.; Comin, R.; Bajdich, M.; García-Melchor, M.; Han, L.; Xu, J.; Liu, M.; Zheng, L.; et al. Homogeneously Dispersed Multimetal Oxygen-Evolving Catalysts. Science 2016, 352, 333–337. [Google Scholar] [CrossRef] [PubMed]
- An, L.; Huang, B.; Zhang, Y.; Wang, R.; Zhang, N.; Dai, T.; Xi, P.; Yan, C.H. Interfacial Defect Engineering for Improved Portable Zinc–Air Batteries with a Broad Working Temperature. Angew. Chem. Int. Ed. 2019, 58, 9459–9463. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Ding, L.; Wang, C.; Meng, Q.; Yuan, Z.; Zhou, Z.; Zhao, S.; Chen, Y. Make It Stereoscopic: Interfacial Design for Full-Temperature Adaptive Flexible Zinc-Air Batteries. Energy Environ. Sci. 2021, 14, 4926–4935. [Google Scholar] [CrossRef]
- Wang, Q.; Feng, Q.; Lei, Y.; Tang, S.; Xu, L.; Xiong, Y.; Fang, G.; Wang, Y.; Yang, P.; Liu, J.; et al. Quasi-Solid-State Zn-Air Batteries with an Atomically Dispersed Cobalt Electrocatalyst and Organohydrogel Electrolyte. Nat. Commun. 2022, 13, 3689. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Xu, X.; Peng, S.; Chen, J.; Yu, D.; Xiao, C.; Li, Y.; Chen, Y.; Hu, X.; Liu, M.; et al. A Flexible and Safe Aqueous Zinc-Air Battery with a Wide Operating Temperature Range from -20 to 70 °C. ACS Sustain. Chem. Eng. 2020, 8, 11501–11511. [Google Scholar] [CrossRef]
- Zhao, C.X.; Liu, J.N.; Yao, N.; Wang, J.; Ren, D.; Chen, X.; Li, B.Q.; Zhang, Q. Can Aqueous Zinc–Air Batteries Work at Sub-Zero Temperatures? Angew. Chem. Int. Ed. 2021, 60, 15281–15285. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Park, K.S. The Li-Ion Rechargeable Battery: A Perspective. J. Am. Chem. Soc. 2013, 135, 1167–1176. [Google Scholar] [CrossRef]
- Tripathi, A.M.; Su, W.N.; Hwang, B.J. In Situ Analytical Techniques for Battery Interface Analysis. Chem. Soc. Rev. 2018, 47, 736–751. [Google Scholar] [CrossRef]
- Wu, F.; Yu, Y. Toward True Lithium-Air Batteries. Joule 2018, 2, 815–817. [Google Scholar] [CrossRef]
- Cao, L.; Li, D.; Pollard, T.; Deng, T.; Zhang, B.; Yang, C.; Chen, L.; Vatamanu, J.; Hu, E.; Hourwitz, M.J.; et al. Fluorinated Interphase Enables Reversible Aqueous Zinc Battery Chemistries. Nat. Nanotechnol. 2021, 16, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Stalin, S.; Archer, L.A. Stabilizing Metal Battery Anodes through the Design of Solid Electrolyte Interphases. Joule 2021, 5, 1119–1142. [Google Scholar] [CrossRef]
- Popovic, J. The Importance of Electrode Interfaces and Interphases for Rechargeable Metal Batteries. Nat. Commun. 2021, 12, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Lee, J.; Park, Y.; Choi, J.W. Aqueous Zinc Ion Batteries: Focus on Zinc Metal Anodes. Chem. Sci. 2020, 11, 2028–2044. [Google Scholar] [CrossRef]
- Liang, G.; Zhi, C. A Reversible Zn-Metal Battery. Nat. Nanotechnol. 2021, 16, 853–855. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Du, X.; Zhao, J.; Wang, Y.; Ju, J.; Chen, Z.; Hu, Z.; Yan, D.; Zhou, X.; Cui, G. Zinc Anode-Compatible in-Situ Solid Electrolyte Interphase via Cation Solvation Modulation. Nat. Commun. 2019, 10, 5374. [Google Scholar] [CrossRef]
- Cao, L.; Li, D.; Deng, T.; Li, Q.; Wang, C. Hydrophobic Organic-Electrolyte-Protected Zinc Anodes for Aqueous Zinc Batteries. Angew. Chem. Int. Ed. 2020, 59, 19292–19296. [Google Scholar] [CrossRef]
- Suo, L.; Borodin, O.; Gao, T.; Olguin, M.; Ho, J.; Fan, X.; Luo, C.; Wang, C.; Xu, K. “Water-in-Salt” Electrolyte Enables High-Voltage Aqueous Lithium-Ion Chemistries. Science 2015, 350, 938–943. [Google Scholar] [CrossRef]
- Zheng, J.; Archer, L.A. Controlling Electrochemical Growth of Metallic Zinc Electrodes: Toward Affordable Rechargeable Energy Storage Systems. Sci. Adv. 2021, 7, eabe0219. [Google Scholar] [CrossRef]
- Zhou, M.; Guo, S.; Li, J.; Luo, X.; Liu, Z.; Zhang, T.; Cao, X.; Long, M.; Lu, B.; Pan, A.; et al. Surface-Preferred Crystal Plane for a Stable and Reversible Zinc Anode. Adv. Mater. 2021, 33, 2100187. [Google Scholar] [CrossRef]
- Zheng, J.; Zhao, Q.; Tang, T.; Yin, J.; Quilty, C.D.; Renderos, G.D.; Liu, X.; Deng, Y.; Wang, L.; Bock, D.C.; et al. Reversible Epitaxial Electrodeposition of Metals in Battery Anodes. Science 2019, 366, 645–648. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, J.; Qi, K.; Yang, Y.; Liu, D.; Wang, T.; Liang, S.; Lu, B.; Zhu, Y.; Zhou, J. An Ion-Sieving Janus Separator toward Planar Electrodeposition for Deeply Rechargeable Zn-Metal Anodes. Adv. Mater. 2022, 34, 2205175. [Google Scholar] [CrossRef] [PubMed]
- Yano, M.; Fujitani, S.; Nishio, K.; Akai, Y.; Kurimura, M. Effect of Additives in Zinc Alloy Powder on Suppressing Hydrogen Evolution. J. Power Sources 1998, 74, 129–134. [Google Scholar] [CrossRef]
- Wongrujipairoj, K.; Poolnapol, L.; Arpornwichanop, A.; Suren, S.; Kheawhom, S. Suppression of Zinc Anode Corrosion for Printed Flexible Zinc-air Battery. Phys. Status Solidi Basic Res. 2017, 254, 1600442. [Google Scholar] [CrossRef]
- Park, M.G.; Lee, D.U.; Seo, M.H.; Cano, Z.P.; Chen, Z. 3D Ordered Mesoporous Bifunctional Oxygen Catalyst for Electrically Rechargeable Zinc-Air Batteries. Small 2016, 12, 2707–2714. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Pan, Z.; Wang, E.; An, L.; Sun, G. Aqueous Metal-Air Batteries: Fundamentals and Applications. Energy Storage Mater. 2020, 27, 478–505. [Google Scholar] [CrossRef]
- Bonnick, P.; Dahn, J.R. A Simple Coin Cell Design for Testing Rechargeable Zinc-Air or Alkaline Battery Systems Service A Simple Coin Cell Design for Testing Rechargeable Zinc-Air. J. Electrochem. Soc. 2012, 159, A981–A989. [Google Scholar] [CrossRef]
- Dongmo, S.; Jakob, J.; Kreissl, A.; Miyazaki, K.; Abe, T.; You, T.; Hu, C.; Schröder, D. Reproducible and Stable Cycling Performance Data on Secondary Zinc Oxygen Batteries. Sci. Data 2020, 7, 395. [Google Scholar] [CrossRef]


| Type of Anode Optimization | Anode Materials | Discharge Capacity/Areal Capacity | DoD/ZU | Cycling Stability | Coulombic Efficiency |
|---|---|---|---|---|---|
| Structural optimization | 3D Zinc sponge anode [29] | 164 mAh/g | 23% | 45 cycles/(24 mA/cm2) | - |
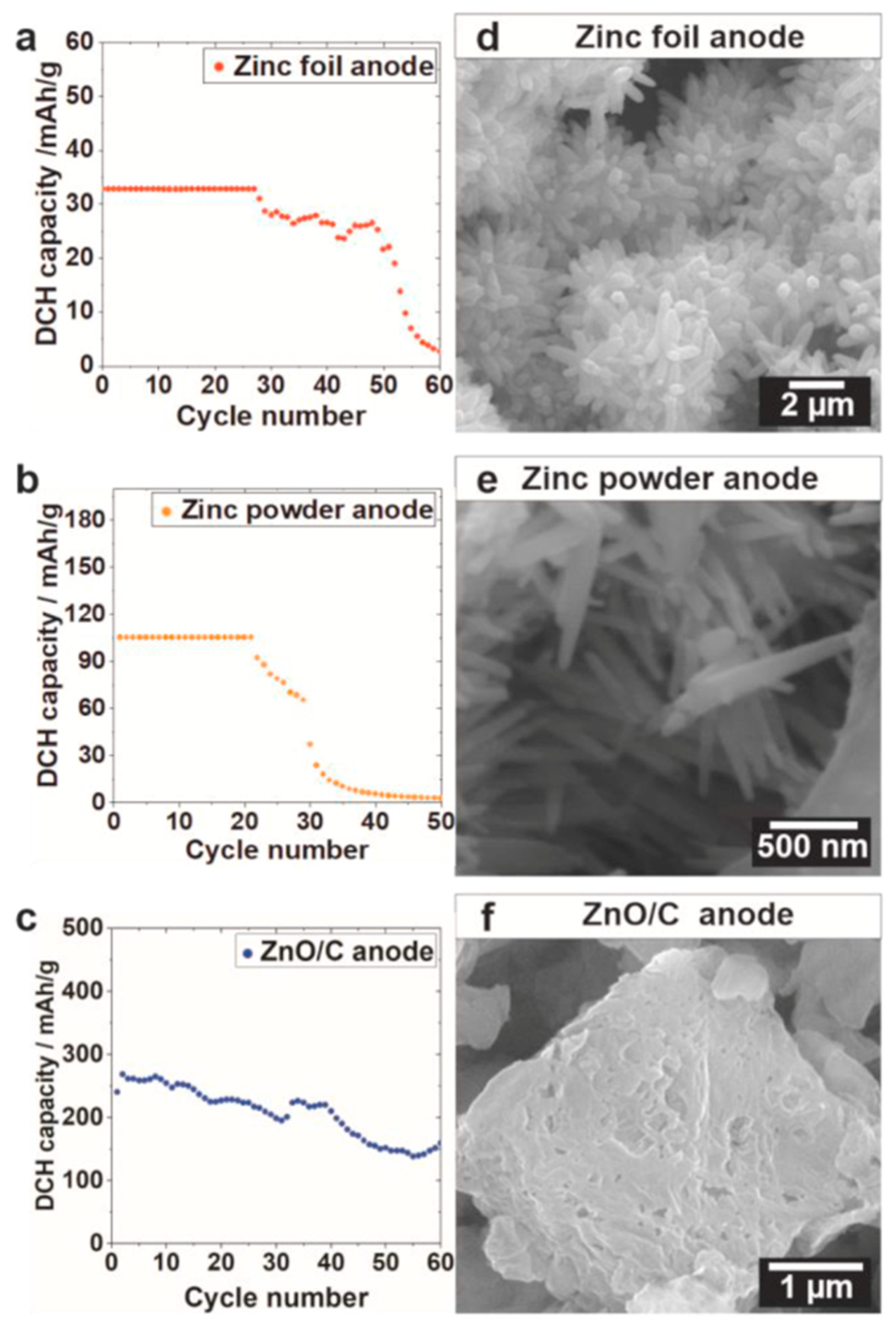
| Structural optimization | 3D ZnO/C [17] | 267 mAh/g | 32% | 60 cycles (0.55 mA/cm2) | 83% |
| Structural optimization | Ag-modified Cu foam/zinc [26] | - | 20% | 80 cycles (25 mA/cm2) | 94% |
| Structural optimization | Nanoporous zinc [30] | 400 mAh/cm2 | 10% | 80 h (10 mA/cm2) | |
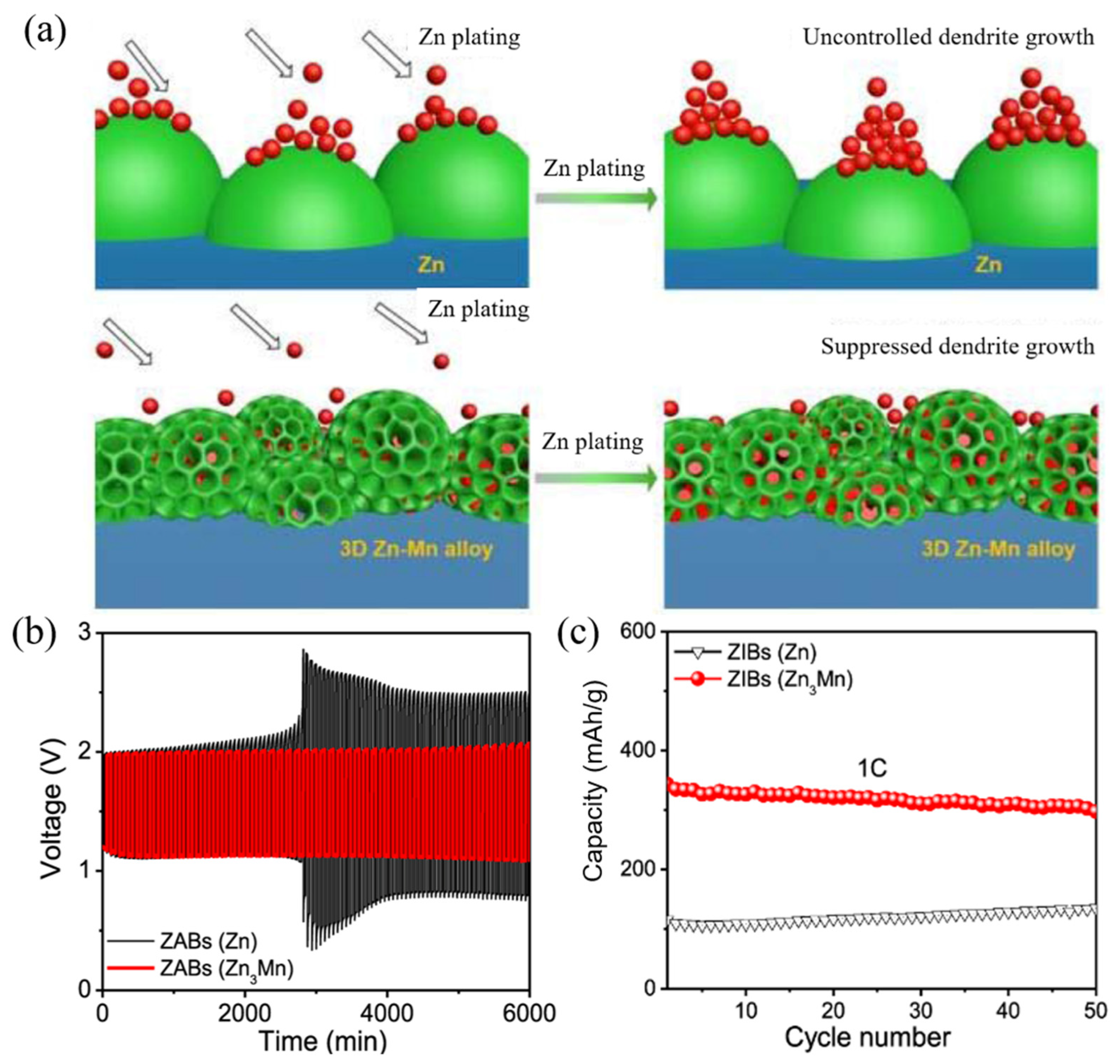
| Structural optimization | Zn3Mn alloy [31] | - | 6000 min (30 mA/cm2) | ||
| Structural optimization | ZnSn10 [32] | 200 h (0.5 mA/cm2) | |||
| Structural optimization | Textured zinc anode [38] | 6 mAh/cm2 | 20% | 250 h (0.1 mA/cm2) | |
| Surface optimization | ZnO@TiO2 [44] | - 616 mAh/g | 40% 100% | 170 cycles 33 cycles | 93.09% 93.5% |
| Surface optimization | ZnO/C [46] | 2.55 mA/cm2 | 100% | 42 cycles (2.55 mA/cm2) | >95% |
| Surface optimization | Bi2O3–ZnO–CaO/Zn [48] | 767 mAh/g | 62%/ZU | 20 cycles | |
| Surface optimization | ZnO/PVA/β-CD/PEG [51] | - | 80 cycles (25 mA/cm2) | ||
| Surface optimization | PANI/Zinc-phthalocyanine [50] | 120 h (10 mA/cm2) | |||
| Surface optimization | ZnO/C with FAA3 coating [53] | 351 mA/g | 53.6% | 13 cycles | 71.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, S.K.; Deckenbach, D.; Schneider, J.J. Secondary Zinc–Air Batteries: A View on Rechargeability Aspects. Batteries 2022, 8, 244. https://doi.org/10.3390/batteries8110244
Yadav SK, Deckenbach D, Schneider JJ. Secondary Zinc–Air Batteries: A View on Rechargeability Aspects. Batteries. 2022; 8(11):244. https://doi.org/10.3390/batteries8110244
Chicago/Turabian StyleYadav, Sudheer Kumar, Daniel Deckenbach, and Jörg J. Schneider. 2022. "Secondary Zinc–Air Batteries: A View on Rechargeability Aspects" Batteries 8, no. 11: 244. https://doi.org/10.3390/batteries8110244
APA StyleYadav, S. K., Deckenbach, D., & Schneider, J. J. (2022). Secondary Zinc–Air Batteries: A View on Rechargeability Aspects. Batteries, 8(11), 244. https://doi.org/10.3390/batteries8110244







