A Critical Review of Lithium-Ion Battery Recycling Processes from a Circular Economy Perspective
Abstract
1. Introduction: The Societal and Economic Importance of Lithium-Ion Batteries
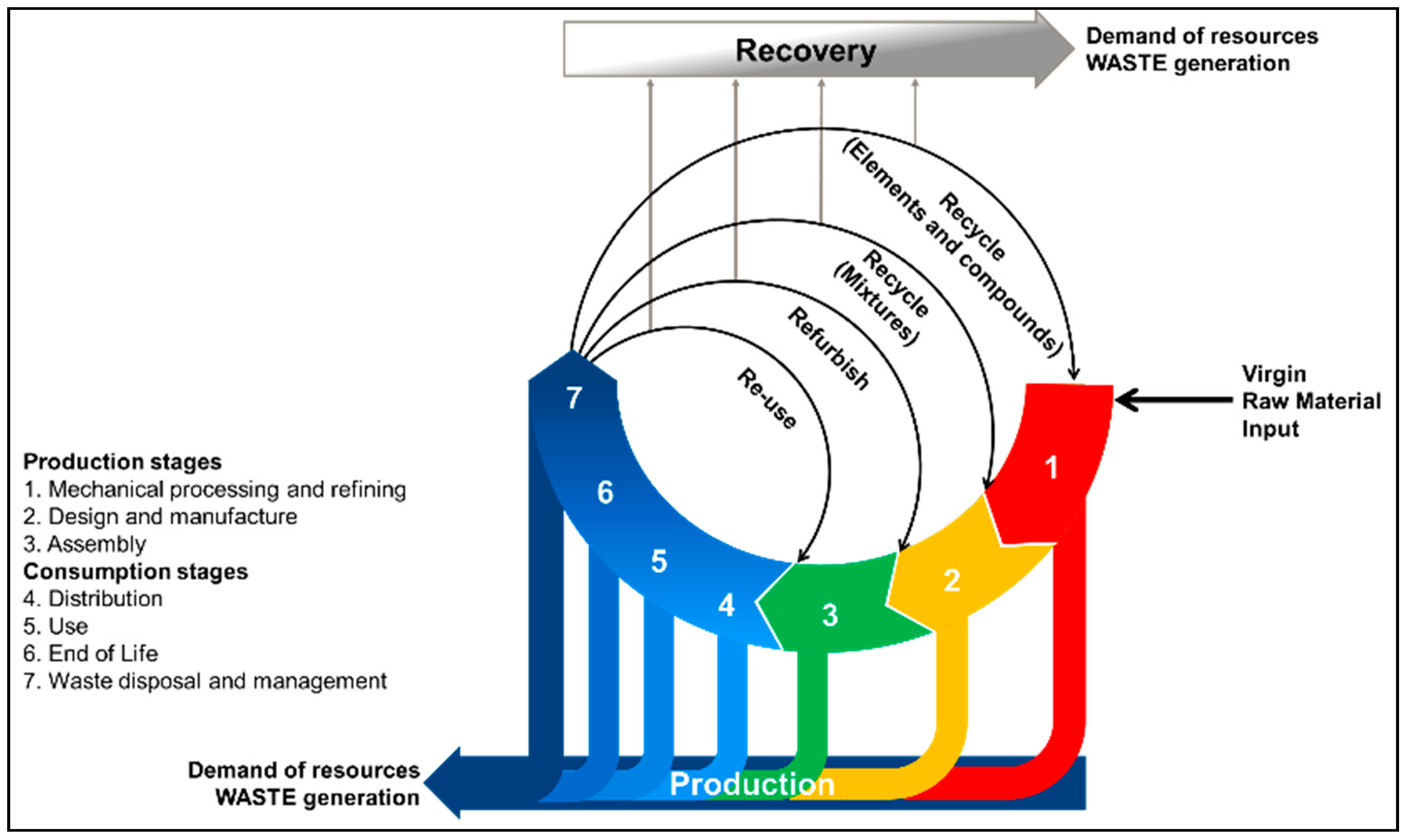
Methodology and Scope
- Established industrial processes with LIBs as the major or unique feed;
- Emerging technologies designed for LIB recycling;
- Processes which do not use LIBs as either a major or unique fraction of the feed, but accept them as secondary feed.
- Established: Umicore Valéas™ (Umicore, Bruxelles, Belgium), Retriev Technologies (Retriev), Recupyl Valibat (Recupyl), Akkuser, and Sumitomo–Sony (Sumitomo).
- Emerging: LithoRec, Accurec, Battery Resources, Steven Loop: OnTo Technology (OnTo), Aalto University Process.
- Other industrial processes accepting LIBs: BatRec, Inmetco, and Glencore.
- What type of battery feed does the process accept?
- What are the main recoveries? What is their final quality?
- Which processes, i.e., hydro- or pyrometallurgy and/or mechanical processing, are used? What are their limitations?
- What other material inputs does the process require?
2. Challenges in LIB Recycling
Compound Challenges
3. Description of LIB Recycling Processes
3.1. Established LIB Recycling Processes
3.1.1. Retriev Technologies
3.1.2. Sumitomo–Sony
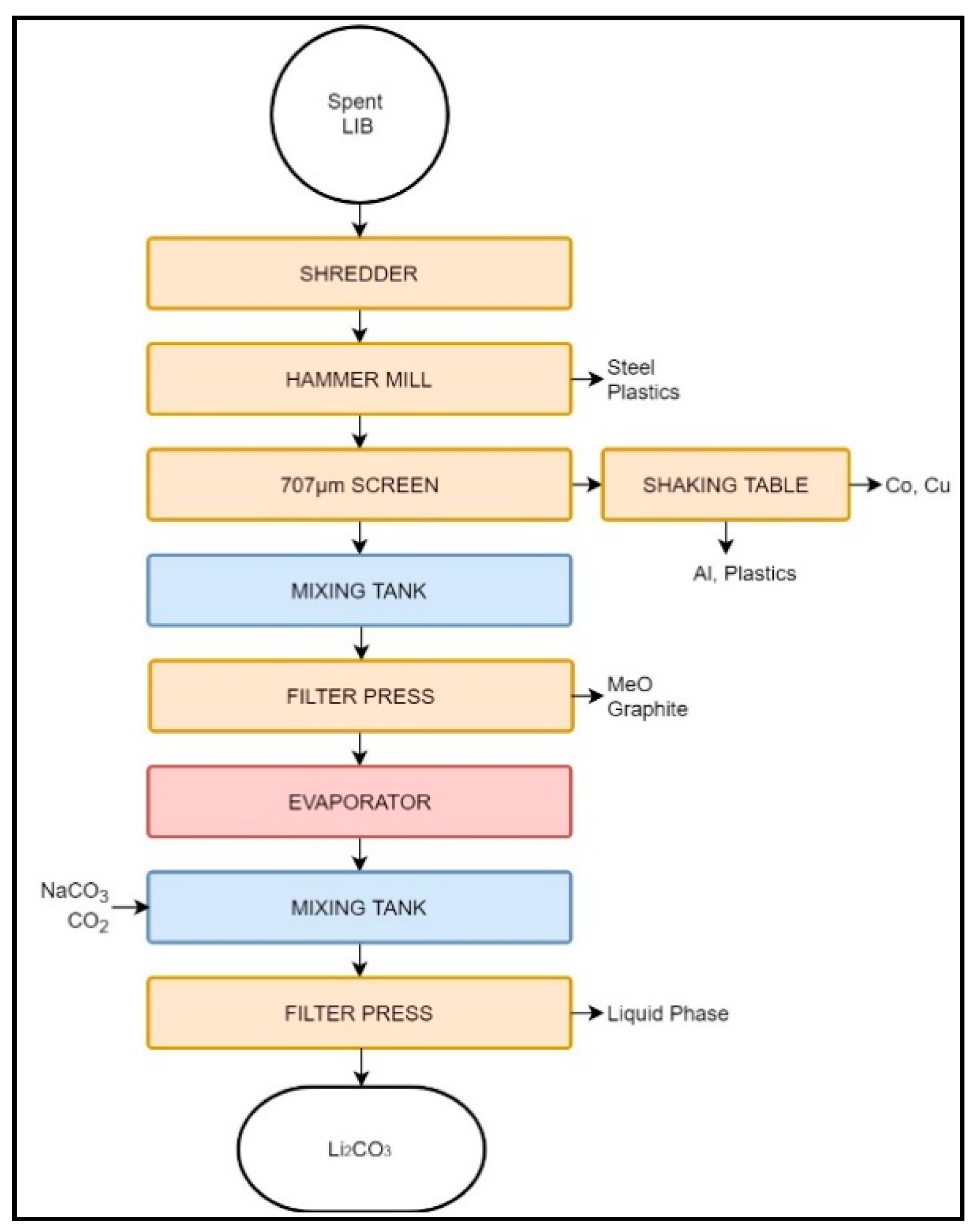
3.1.3. Recupyl Valibat
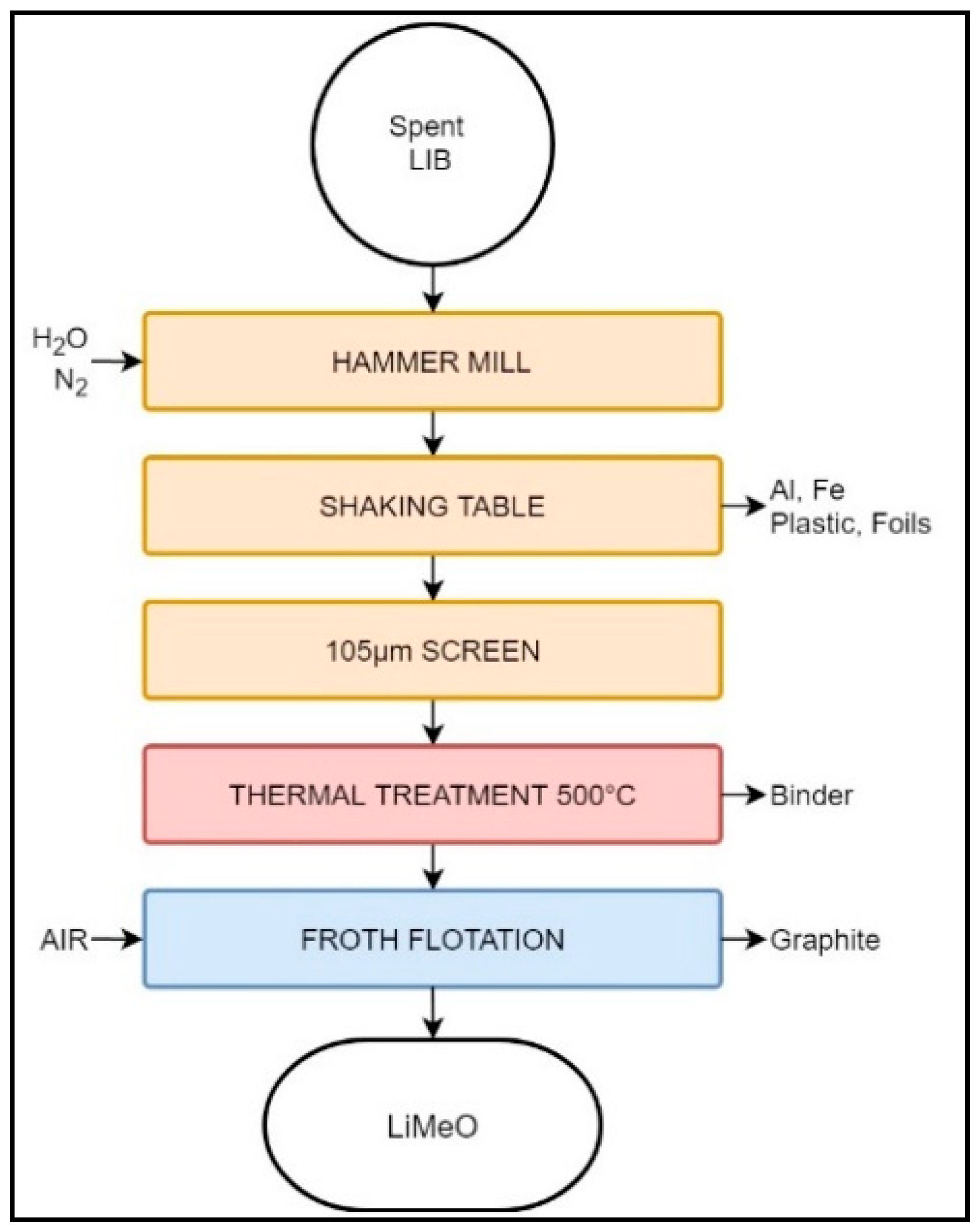
3.1.4. Akkuser
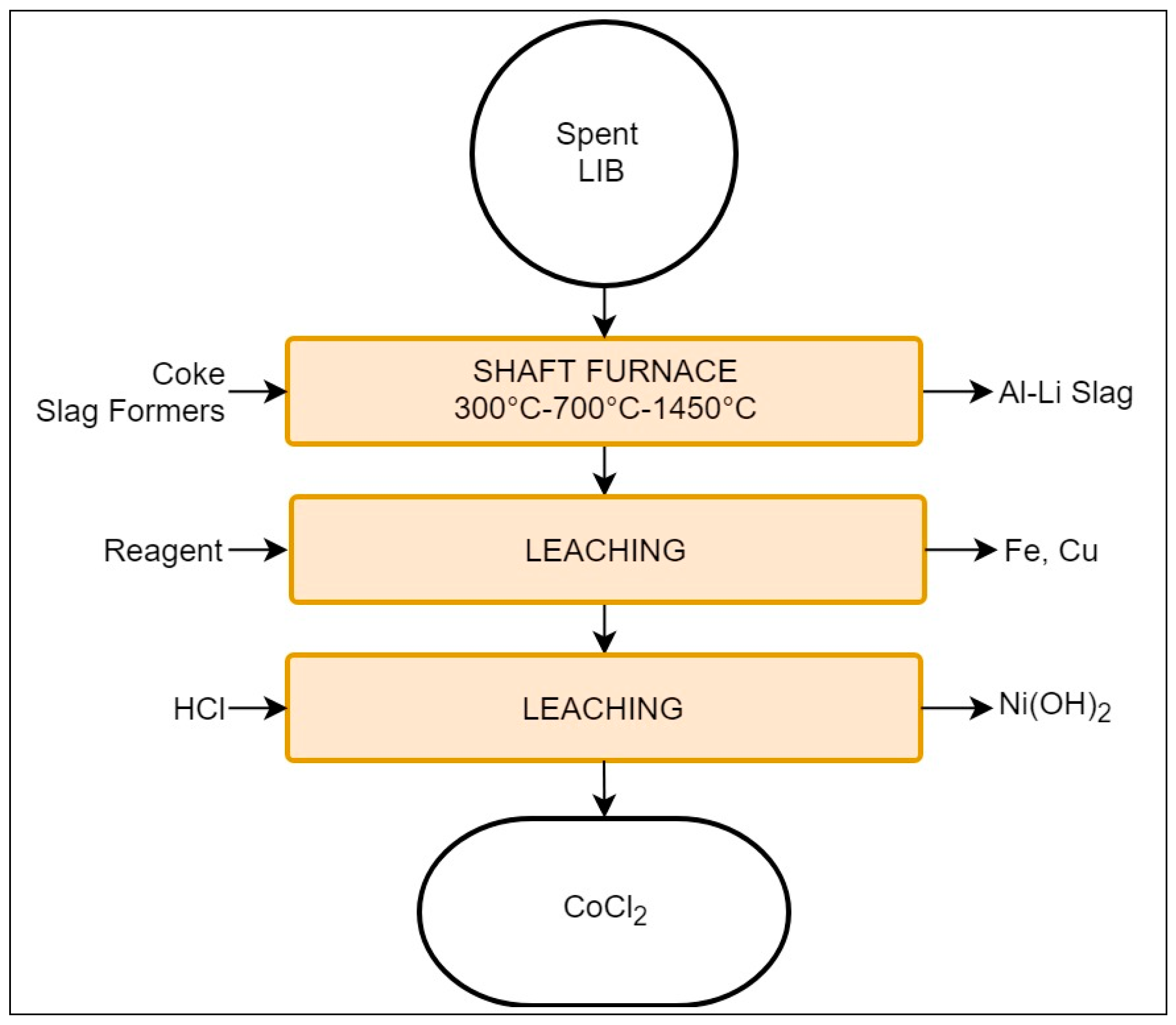
3.1.5. Umicore Valéas™ (Bruxelles, Belgium)
- Low temperature section: evaporation of electrolyte;
- Medium temperature: plastics pyrolysis;
- High temperature: smelting and reduction (1200–1450 °C).
3.1.6. Other Industrial Processes Accepting LIBs
Batrec
Inmetco
Glencore
3.2. Emerging LIB Recycling Processes
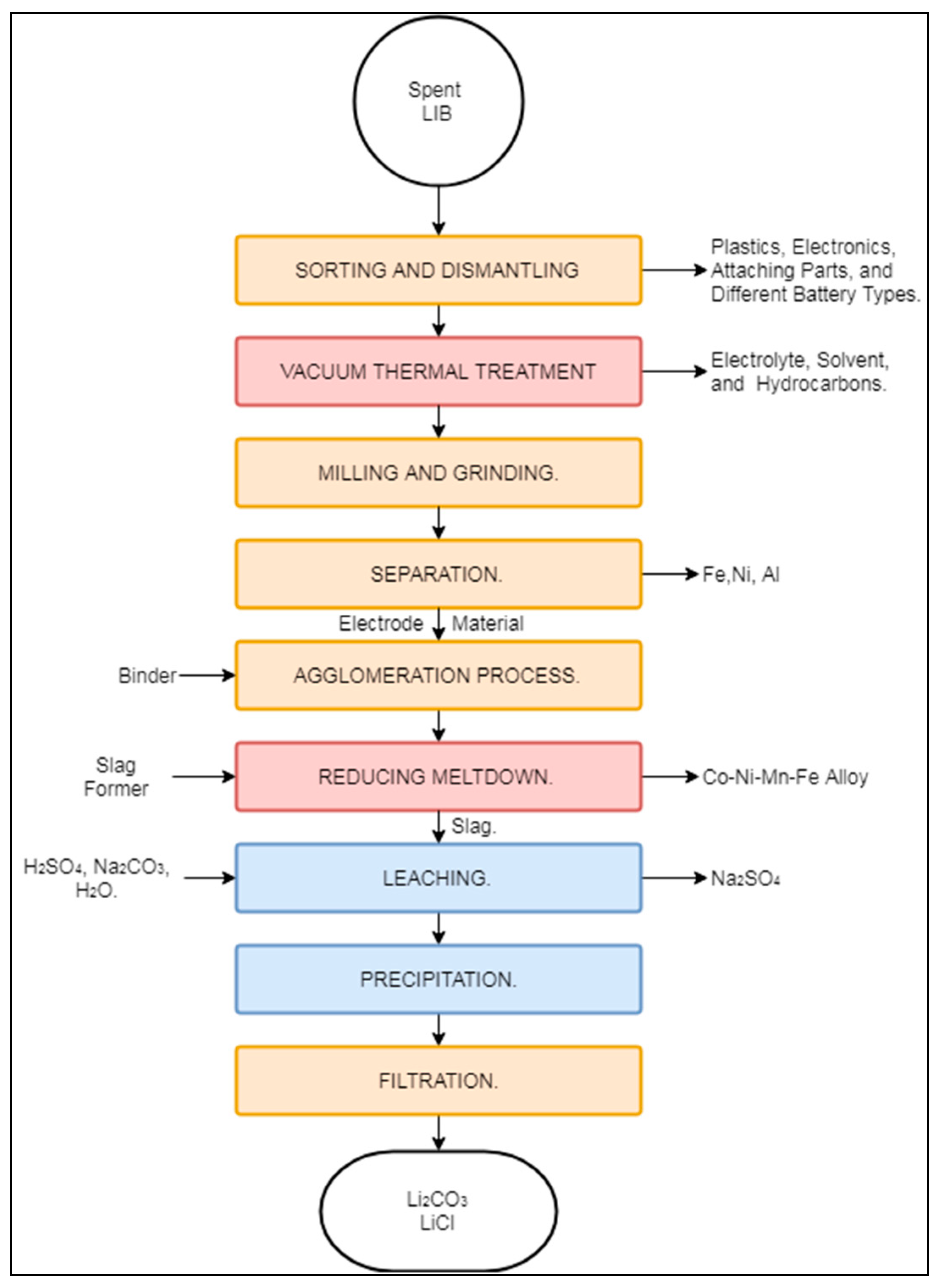
3.2.1. Accurec
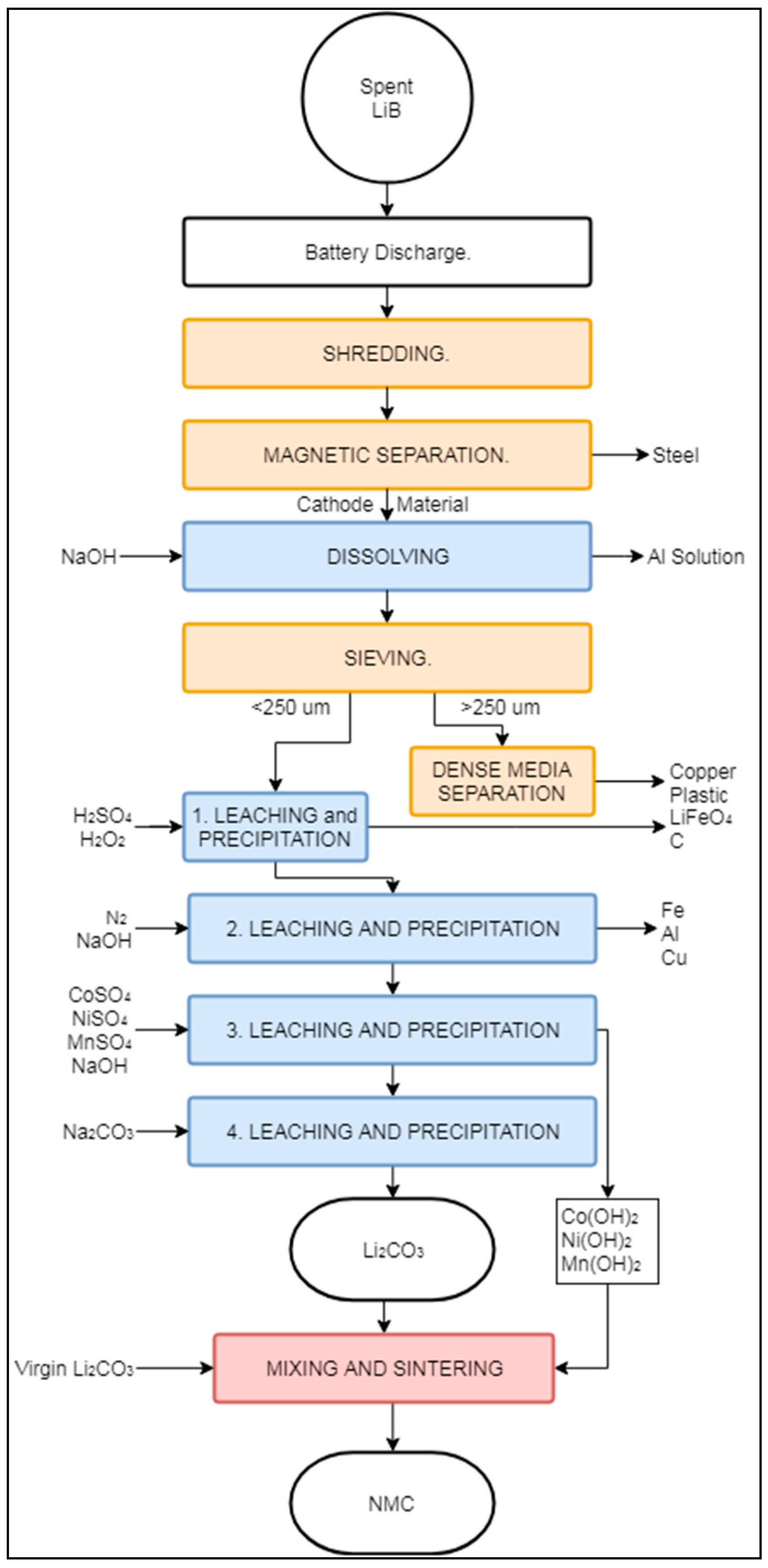
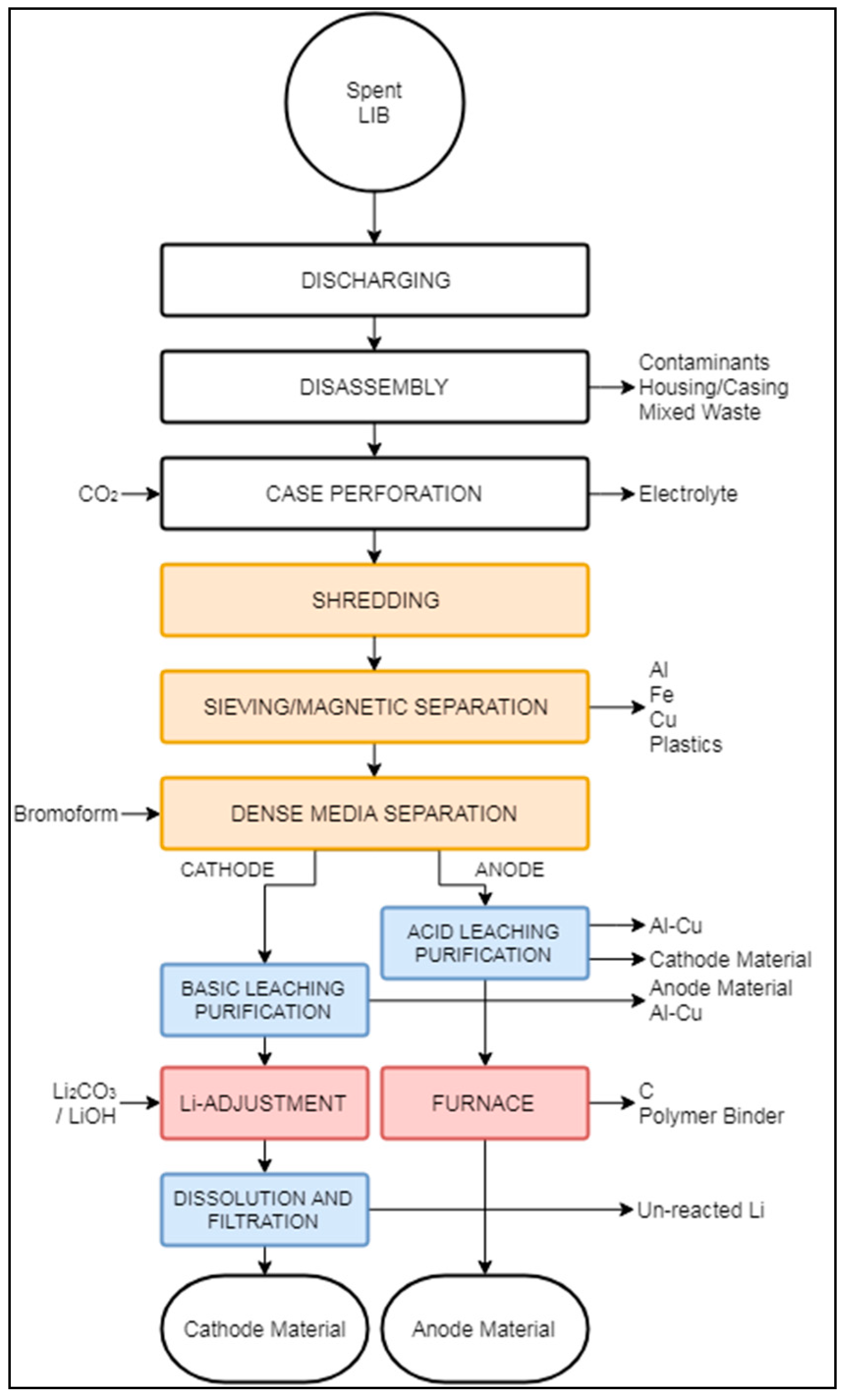
3.2.2. Battery Resources “Closed Loop” Process
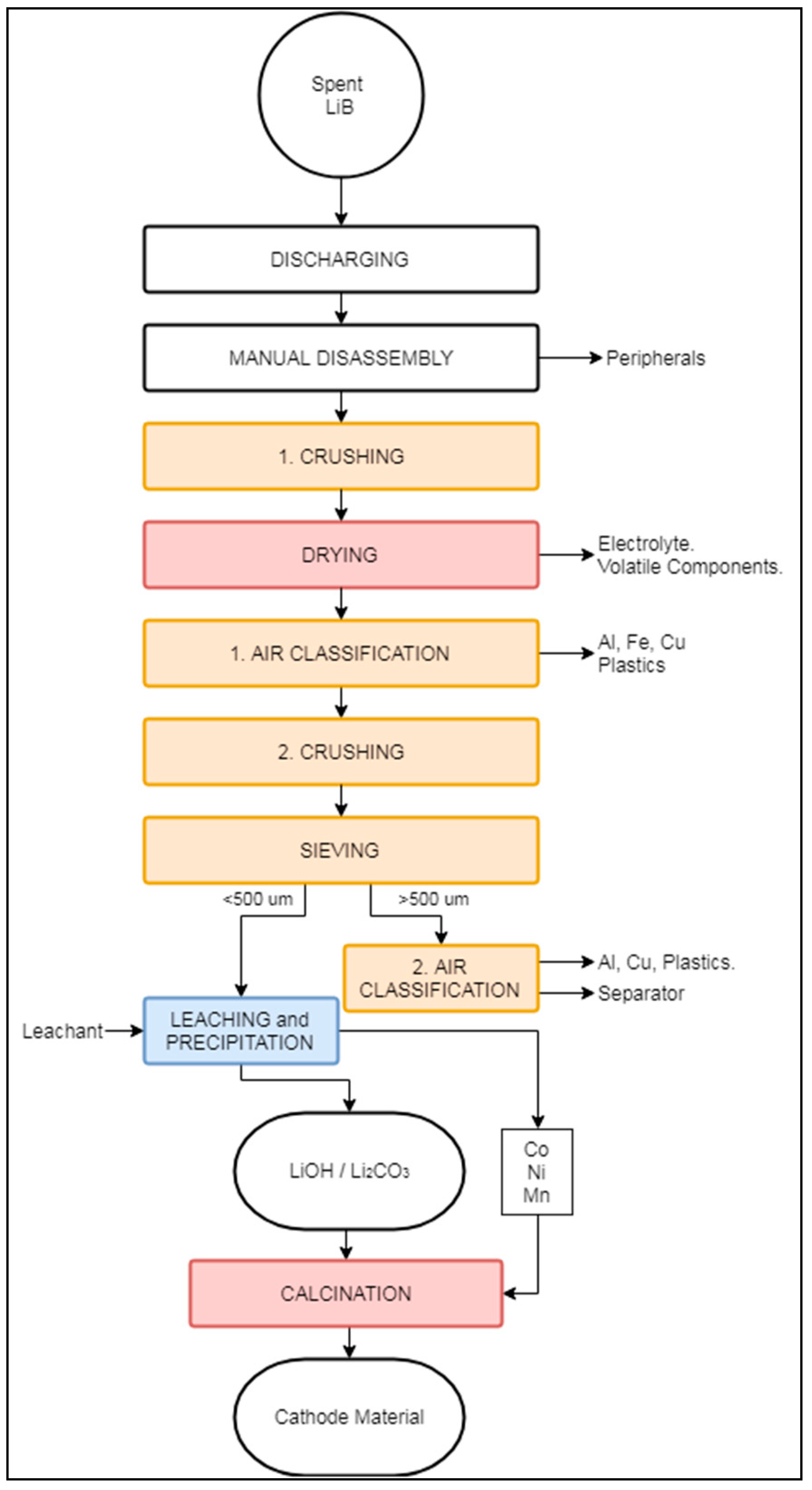
3.2.3. LithoRec
3.2.4. OnTo Technology
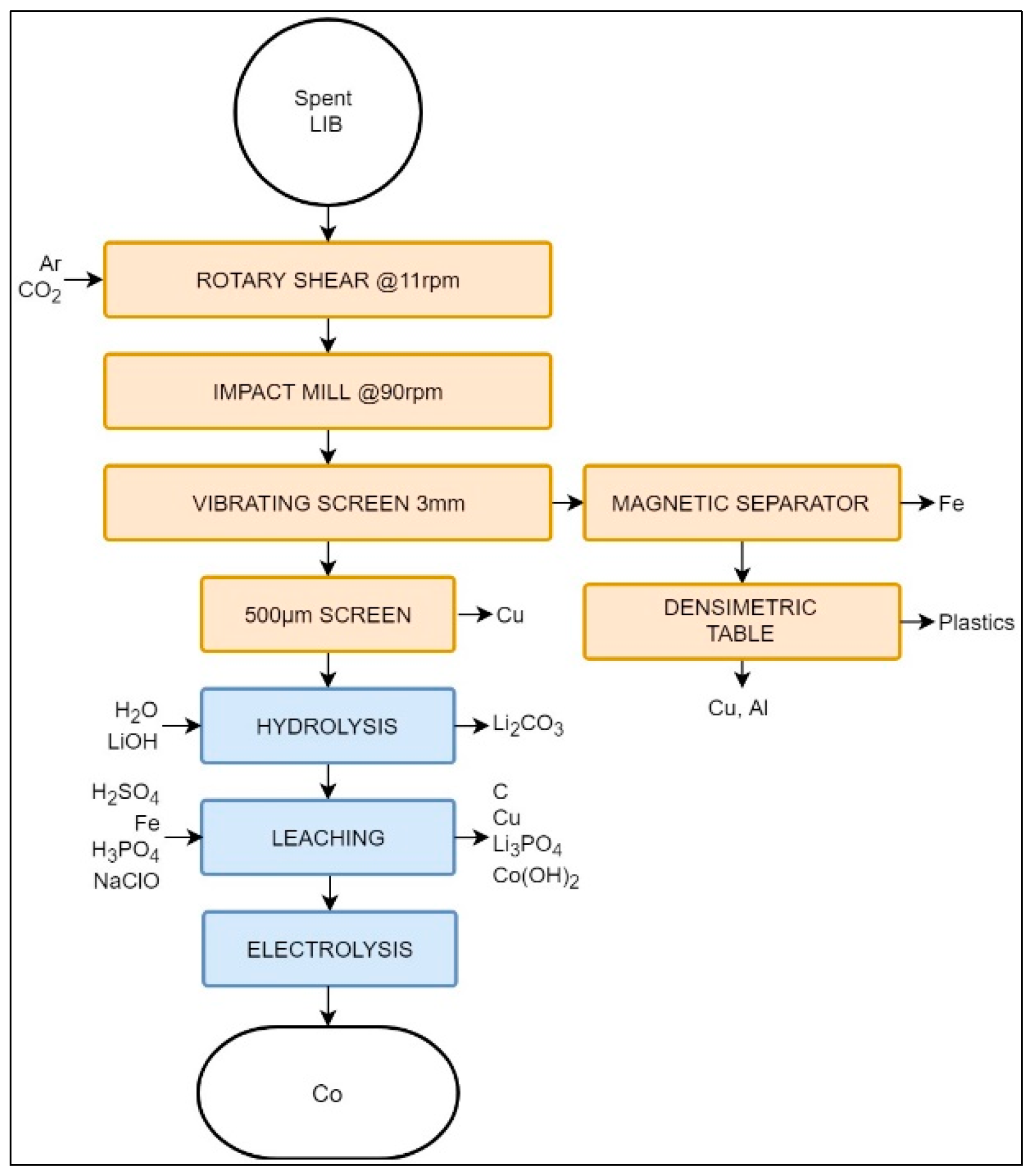
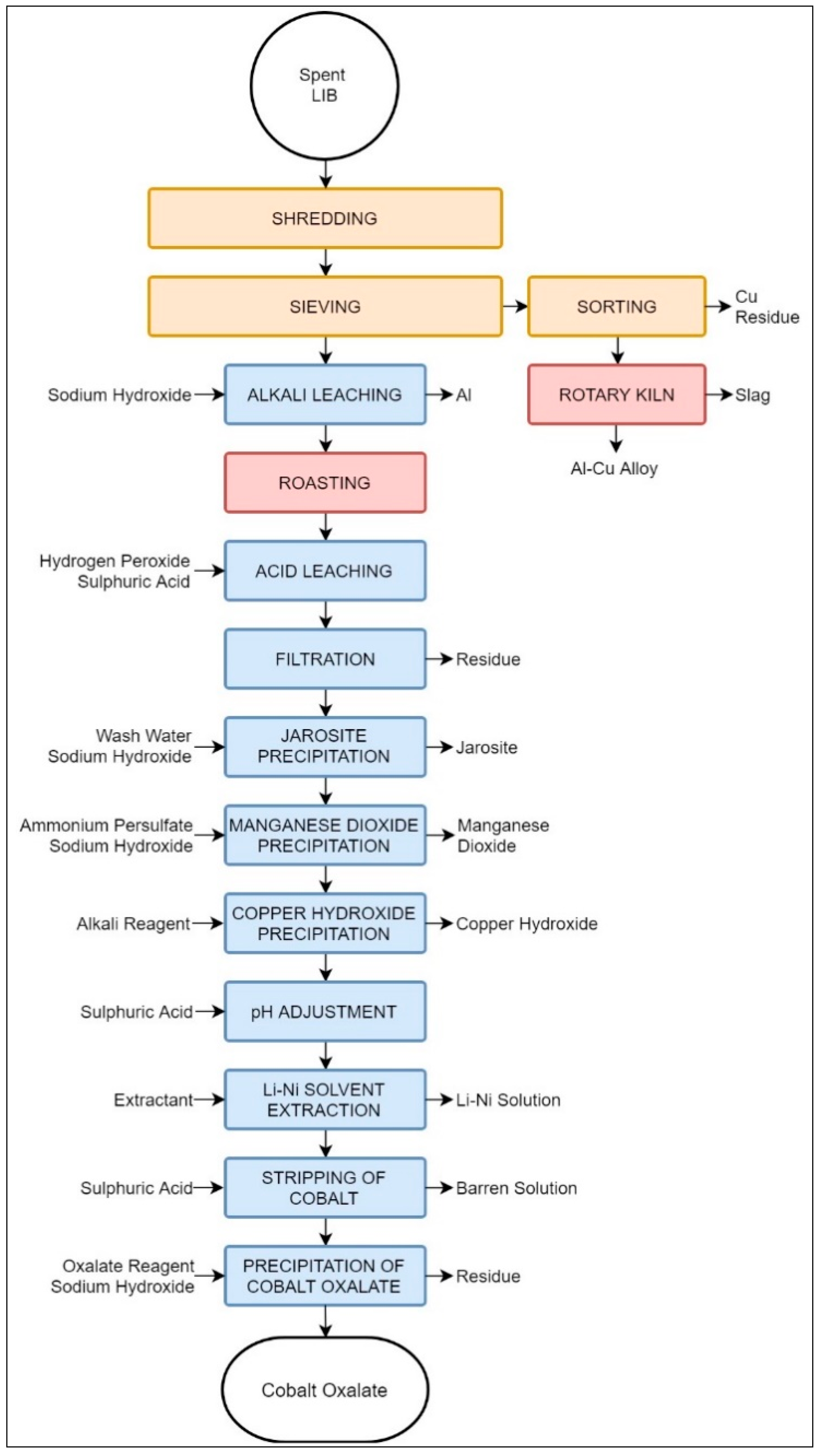
3.2.5. Laboratory Process Suggested by Aalto University
4. Discussion
4.1. Recycling Processes
4.2. Recovery of Materials
4.2.1. Cathode Active Material
4.2.2. Lithium Recovery
4.2.3. Metals Recovery
4.2.4. Graphite Recovery
5. Conclusions and Recommendations
- Energy cost calculations,
- Plant efficiency.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tarascon, J.M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Linden, D.; Reddy, B.T. (Eds.) Linden’s Handbook of Batteries, 4th ed.; McGraw Hill: New York, NY, USA, 2011. [Google Scholar]
- Aditya, J.P.; Ferdowsi, M. Comparison of NiMH and Li-ion batteries in automotive applications. In Proceedings of the 2008 IEEE Vehicle Power and Propulsion Conference, Harbin, China, 3–5 September 2008; pp. 1–6. [Google Scholar]
- Hameer, S.; Van Niekerk, J.L. A review of large-scale electrical energy storage. Int. J. Energy Res. 2015, 39, 1179–1195. [Google Scholar] [CrossRef]
- EBRA. 2012: Noticeable Growth of the Quantity of Batteries Recycled; European Battery Recycling Association: Brussels, Belgium, 2012. [Google Scholar]
- EUR-Lex. Directive 2006/66/EC of the European Parliament and of the Council of 6 September 2006 on batteries and accumulators and waste batteries and accumulators and repealing Directive 91/157/EEC. Off. J. Eur. Union 2006, 266, 1–14. [Google Scholar]
- European Commission. Report on Critical Raw Materials for the EU; European Commission: Brussels, Belgium, 2014. [Google Scholar]
- Roper, L.D. Tesla Model S; Virginia Polytechnic Institute and State University: USA, 2019. Available online: http://www.roperld.com/science/TeslaModelS.htm (accessed on 13 June 2019).
- Bernhart, W. The Lithium-Ion Battery Value Chain—Status, Trends and Implications. In Lithium-Ion Batteries; Elsevier: Rome, Italy, 2014; pp. 553–565. [Google Scholar]
- Pillot, C. The Rechargeable Battery Market and Main Trends 2014–2025. In Proceedings of the Advanced Automotive Battery Conference, Chicago, IL, USA, 21 September 2016. [Google Scholar]
- Mohammadi, F. Electric Vehicle Battery Market Analysis: Lithium-Ion. In Proceedings of the 1st International Conference on Modern Approaches in Engineering Science, Tbilisi, Georgia, 21 November 2018. [Google Scholar]
- Pillot, C. The Rechargeable Battery Market 2017–2025. Avicenne Energy 2018, 6, 26–29. [Google Scholar]
- HDR. Human Development Reports; United Nations Development Programme: New York, NY, USA, 2015. [Google Scholar]
- Earle, S.; Panchuk, K. Physical Geology, 2nd ed.; BC Campus: Victoria, BC, Canada, 2019. [Google Scholar]
- Heelan, J.; Gratz, E.; Zheng, Z.; Wang, Q.; Chen, M.; Apelian, D. Current and Prospective Li-Ion Battery Recycling and Recovery Processes. JOM 2016, 68, 2632–2638. [Google Scholar] [CrossRef]
- Ledung, G. State-of-the-Art in Reuse and Recycling of Lithium-ion Batteries-A Research Review by Hans Eric Melin, Circular Energy Storage; The Swedish Energy Agency: Eskilstuna, Sweden, 2019. [Google Scholar]
- Georgi-Maschler, T.; Friedrich, B.; Weyhe, R.; Heegn, H.; Rutz, M. Development of a recycling process for Li-ion batteries. J. Power Sources 2012, 207, 173–182. [Google Scholar] [CrossRef]
- Wang, X.; Gaustad, G.; Babbitt, C.W.; Richa, K. Economies of scale for future lithium-ion battery recycling infrastructure. Resour. Conserv. Recycl. 2014, 83, 53–62. [Google Scholar] [CrossRef]
- Cerdas, F.; Titscher, P.; Bognar, N.; Schmuch, R.; Winter, M.; Kwade, A.; Herrmann, C. Exploring the Effect of Increased Energy Density on the Environmental Impacts of Traction Batteries: A Comparison of Energy Optimized Lithium-Ion and Lithium-Sulfur Batteries for Mobility Applications. Energies 2018, 11, 150. [Google Scholar] [CrossRef]
- Kim, H.C.; Wallington, T.J.; Arsenault, R.; Bae, C.; Ahn, S.; Lee, J. Cradle-to-Gate Emissions from a Commercial Electric Vehicle Li-Ion Battery: A Comparative Analysis. Environ. Sci. Technol. 2016, 50, 7715–7722. [Google Scholar] [CrossRef]
- Ellingsen, L.A.W.; Majeau-Bettez, G.; Singh, B.; Srivastava, A.K.; Valøen, L.O.; Strømman, A.H.; Majeau-Bettez, G. Life Cycle Assessment of a Lithium-Ion Battery Vehicle Pack. J. Ind. Ecol. 2013, 18, 113–124. [Google Scholar] [CrossRef]
- Xu, J.; Thomas, H.; Francis, R.W.; Lum, K.R.; Wang, J.; Liang, B. A review of processes and technologies for the recycling of lithium-ion secondary batteries. J. Power Sources 2008, 177, 512–527. [Google Scholar] [CrossRef]
- Pillot, C. Future Trends in the Rechargeable Battery Market; Avicenne Energy: Montreux, Switzerland, 2015. [Google Scholar]
- Kivi, J. Battery Recycling, Metal Recovery and Waste Processing State of the Art; Metallialan Ympäristö ja Kiertotalous: Espoo, Finland, 2016. [Google Scholar]
- He, L.P.; Sun, S.Y.; Song, X.F.; Yu, J.G. Recovery of cathode materials and Al from spent lithium-ion batteries by ultrasonic cleaning. Waste Manag. 2015, 46, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, A.J.; Matos, J.F.; Bernardes, A.M.; Müller, I.L. Beneficiation of cobalt, copper and aluminum from wasted lithium-ion batteries by mechanical processing. Int. J. Miner. Process. 2015, 145, 77–82. [Google Scholar] [CrossRef]
- Espinosa, D.C.R.; Bernardes, A.M.; Tenório, J.A.S. An overview on the current processes for the recycling of batteries. J. Power Sources 2004, 135, 311–319. [Google Scholar] [CrossRef]
- Geissdoerfer, M.; Savaget, P.; Bocken, N.M.; Hultink, E.J. The Circular Economy—A new sustainability paradigm? J. Clean. Prod. 2017, 143, 757–768. [Google Scholar] [CrossRef]
- Reuter, M.A.; Worrell, E. Handbook of Recycling; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Hawkins, T.R.; Singh, B.; Strømman, A.H.; Majeau-Bettez, G.; Majeau-Bettez, G. Comparative Environmental Life Cycle Assessment of Conventional and Electric Vehicles. J. Ind. Ecol. 2012, 17, 53–64. [Google Scholar] [CrossRef]
- van Schalkwyk, R.F.; Reuter, M.; Gutzmer, J.; Stelter, M. Challenges of digitalizing the circular economy: Assessment of the state-of-the-art of metallurgical carrier metal platform for lead and its associated technology elements. J. Clean. Prod. 2017, 186, 585–601. [Google Scholar] [CrossRef]
- Martínez, O.V.; Boogaart, K.V.D.; Lundström, M.; Santasalo-Aarnio, A.; Reuter, M.; Serna-Guerrero, R. Statistical entropy analysis as tool for circular economy: Proof of concept by optimizing a lithium-ion battery waste sieving system. J. Clean. Prod. 2019, 212, 1568–1579. [Google Scholar] [CrossRef]
- Melin, H.E. The Lithium-Ion Battery End-of-Life Market—A Baseline Study; World Economic Forum: Cologny, Switzerland, 2018; pp. 1–11. [Google Scholar]
- Pistoia, G. (Ed.) Lithium-Ion Batteries Advances and Applications, 1st ed.; Elsevier BV: Oxford, UK, 2014. [Google Scholar]
- Bernardes, A.; Espinosa, D.; Tenório, J.; Bernardes, A.; Espinosa, D.C.R. Recycling of batteries: A review of current processes and technologies. J. Power Sources 2004, 130, 291–298. [Google Scholar] [CrossRef]
- Ferella, F.; De Michelis, I.; Vegliò, F. Process for the recycling of alkaline and zinc–carbon spent batteries. J. Power Sources 2008, 183, 805–811. [Google Scholar] [CrossRef]
- Ruffino, B.; Zanetti, M.; Marini, P. A mechanical pre-treatment process for the valorization of useful fractions from spent batteries. Resour. Conserv. Recycl. 2011, 55, 309–315. [Google Scholar] [CrossRef]
- Ordóñez, J.; Gago, E.; Girard, A. Processes and technologies for the recycling and recovery of spent lithium-ion batteries. Renew. Sustain. Energy Rev. 2016, 60, 195–205. [Google Scholar] [CrossRef]
- Sayilgan, E.; Kukrer, T.; Civelekoglu, G.; Ferella, F.; Akcil, A.; Veglio, F.; Kitis, M. A review of technologies for the recovery of metals from spent alkaline and zinc–carbon batteries. Hydrometallurgy 2009, 97, 158–166. [Google Scholar] [CrossRef]
- Vezzini, A. Manufacturers, Materials and Recycling Technologies. In Lithium-Ion Batteries; Elsevier BV: Rome, Italy, 2014; pp. 529–551. [Google Scholar]
- Zeng, X.; Li, J.; Singh, N. Recycling of Spent Lithium-Ion Battery: A Critical Review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1129–1165. [Google Scholar] [CrossRef]
- Chen, L.; Tang, X.; Zhang, Y.; Li, L.; Zeng, Z.; Zhang, Y. Process for the recovery of cobalt oxalate from spent lithium-ion batteries. Hydrometallurgy 2011, 108, 80–86. [Google Scholar] [CrossRef]
- Porvali, A.; Aaltonen, M.; Ojanen, S.; Velazquez-Martinez, O.; Eronen, E.; Liu, F.; Wilson, B.P.; Serna-Guerrero, R.; Lundström, M. Mechanical and hydrometallurgical processes in HCl media for the recycling of valuable metals from Li-ion battery waste. Resour. Conserv. Recycl. 2019, 142, 257–266. [Google Scholar] [CrossRef]
- Martinez, V.; Porvali, A.; Boogaart, V.D.; Aarnio, S.; Lundström, M.; Reuter, M.; Guerrero, S.; Velázquez-Martinez, O.; Boogaart, K.G.V.D.; Santasalo-Aarnio, A.; et al. On the Use of Statistical Entropy Analysis as Assessment Parameter for the Comparison of Lithium-Ion Battery Recycling Processes. Batteries 2019, 5, 41. [Google Scholar] [CrossRef]
- Nishi, Y. Lithium ion secondary batteries; past 10 years and the future. J. Power Sources 2001, 100, 101–106. [Google Scholar] [CrossRef]
- Nitta, N.; Wu, F.; Lee, J.T.; Yushin, G. Li-ion battery materials: Present and future. Mater. Today 2015, 18, 252–264. [Google Scholar] [CrossRef]
- Dahn, J.; Ehrlich, G.M. Lithium-Ion Batteries. In Linden’s Handbook of Batteries, 4th ed.; Reddy, T.B., Ed.; McGraw Hill: New York, NY, USA, 2011; pp. 1–79. [Google Scholar]
- Scrosati, B.; Garche, J. Lithium batteries: Status, prospects and future. J. Power Sources 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Blomgren, G.E. The Development and Future of Lithium Ion Batteries. J. Electrochem. Soc. 2017, 164, A5019–A5025. [Google Scholar] [CrossRef]
- Grützke, M.; Mönnighoff, X.; Horsthemke, F.; Kraft, V.; Nowak, S.; Winter, M. Extraction of lithium-ion battery electrolytes with liquid and supercritical carbon dioxide and additional solvents. RSC Adv. 2015, 5, 43209–43217. [Google Scholar] [CrossRef]
- Sloop, S.E. System and Method for Removing and Electrolyte from Energy Storage and/or Conversion Device Using a Supercritical Fluid. U.S. Patent 7,858,216 B2, 28 December 2010. [Google Scholar]
- Nanografi. PVDF Binder for Li-ion Battery Electrodes. 2018. Available online: https://nanografi.com/blog/pvdf-binder-for-liion-battery-electrodes/ (accessed on 18 June 2019).
- Christmann, P.; Gloaguen, E.; Labbé, J.F.; Melleton, J.; Piantone, P. Global Lithium Resources and Sustainability Issues. In Lithium Process Chemistry; Elsevier: Amsterdam, The Netherlands, 2015; pp. 1–40. [Google Scholar]
- Broussely, M.; Biensan, P.; Bonhomme, F.; Blanchard, P.; Herreyre, S.; Nechev, K.; Staniewicz, R. Main aging mechanisms in Li ion batteries. J. Power Sources 2005, 146, 90–96. [Google Scholar] [CrossRef]
- Van Beukering, P.; Kuik, O.; Oosterhuis, F. The Economics of Recycling. In Handbook of Recycling; Elsevier: Amsterdam, The Netherlands, 2014; pp. 479–489. [Google Scholar]
- Li, J.; Wang, G.; Xu, Z. Environmentally-friendly oxygen-free roasting/wet magnetic separation technology for in situ recycling cobalt, lithium carbonate and graphite from spent LiCoO 2/graphite lithium batteries. J. Hazard. Mater. 2016, 302, 97–104. [Google Scholar] [CrossRef]
- Corrigan, D.A.; Masias, A. Batteries for electric and hybrid vehicles. In Linden’s Handbook of Batteries, 4th ed.; Reddy, T.B., Ed.; McGraw Hill: New York, NY, USA, 2011; pp. 3–48. [Google Scholar]
- U.S. Geological Survey. Mineral Commodity Summaries 2018; Unites States Geological Survey: Reston, VA, USA, 2018.
- Pagliaro, M.; Meneguzzo, F. Lithium battery reusing and recycling: A circular economy insight. Heliyon 2019, 5, e01866. [Google Scholar] [CrossRef]
- Dunn, J.B.; James, C.; Gaines, L.; Gallagher, K.; Dai, Q.; Kelly, J.C. Material and Energy Flows in the Production of Cathode and Anode Materials for Lithium Ion Batteries; Argonne National Laboratory: Chicago, IL, USA, 2015. [Google Scholar]
- Wills, B.A.; Finch, J.A.; Wills, B.A.; Finch, J.A. Introduction. In Wills’ Mineral Processing Technology; Butterworth-Heinemann: Oxford, UK, 2016; pp. 1–27. [Google Scholar]
- Kaya, M. Recovery of Metals from Electronic Waste by Physical and Chemical Recycling Processes. Int. J. Chem. Mol. Nucl. Mater. Metall. Eng. 2016, 10, 232–243. [Google Scholar] [CrossRef]
- Schubert, W.; Tomas, J. Chapter 24 Liberation of Valuables Embedded in Particle Compounds and Solid Waste. In Handbook of Powder Technology; Elsevier: Amsterdam, The Netherlands, 2007; Volume 12, pp. 989–1018. [Google Scholar]
- Clark, J.H.; Farmer, T.J.; Herrero-Davila, L.; Sherwood, J. ChemInform Abstract: Circular Economy Design Considerations for Research and Process Development in the Chemical Sciences. Green Chem. 2016, 18, 3914–3934. [Google Scholar] [CrossRef]
- Wills, B.A.; Napier-Munn, T. Mineral Processing Technology An Introduction to the Practical Aspects of Ore Treatment and Mineral Recovery, 7th ed.; Elsevier Science & Technology Books: Oxford, UK, 2006. [Google Scholar]
- McLaughlin, W.; Adams, T.S. Li Reclamation Process. U.S. Patent 5,888,463, 30 March 1999. [Google Scholar]
- Comission for Environmental Cooperation. Environmentally Sound Management of End-of-Life Batteries from Electric-Drive Vehicles in North America; Comission for Environmental Cooperation: Montreal, Canada, 2015. [Google Scholar]
- Lv, W.; Wang, Z.; Cao, H.; Sun, Y.; Zhang, Y.; Sun, Z.H. A Critical Review and Analysis on the Recycling of Spent Lithium-Ion Batteries. ACS Sustain. Chem. Eng. 2018, 6, 1504–1521. [Google Scholar] [CrossRef]
- Retriev Technologies. Lithium-Ion, 2019. Available online: https://www.retrievtech.com/lithiumion (accessed on 19 June 2019).
- Gaines, L.L.; Dunn, J.B. Lithium-Ion Battery Environmental Impacts. In Lithium-Ion Batteries; Elsevier BV: Rome, Italy, 2014; pp. 483–508. [Google Scholar]
- Smith, W.N.; Swoffer, S. Process for Recovering and Regenerating Lithium Cathode Material from Lithium-Ion Batteries. U.S. Patent 8,882,007 B1, 11 November 2014. [Google Scholar]
- Cardarelli, F.; Dube, J. Method for Rrecycling Spent Llithium Metal Polymer Rechargeable Batteries and Related Materials. U.S. Patent 7,192,564 B2, 20 March 2007. [Google Scholar]
- Al-Thyabat, S.; Nakamura, T.; Shibata, E.; Iizuka, A. Adaptation of minerals processing operations for lithium-ion (LiBs) and nickel metal hydride (NiMH) batteries recycling: Critical review. Miner. Eng. 2013, 45, 4–17. [Google Scholar] [CrossRef]
- Tedjar, F.; Foudraz, J.C. Method for the Mixed Recycling of Lithium-Based Anode Batteries and Cells. U.S. Patent 7,820,317 B2, 26 October 2010. [Google Scholar]
- Pudas, J.; Erkkila, A.; Viljamaa, J. Battery Recycling Method. U.S. Patent 8,979,006 B2, 17 March 2015. [Google Scholar]
- Zhang, T.; He, Y.; Wang, F.; Ge, L.; Zhu, X.; Li, H. Chemical and process mineralogical characterizations of spent lithium-ion batteries: An approach by multi-analytical techniques. Waste Manag. 2014, 34, 1051–1058. [Google Scholar] [CrossRef]
- Cheret, D.; Santen, S. Battery Recycling. U.S. Patent 7,916,9206 B2, 30 January 2007. [Google Scholar]
- Batrec, “Batrec”. Available online: http://www.batrec.ch/en/ (accessed on 20 June 2019).
- Zenger, T.; Krebs, A.; van Deutekom, H.J.H. Method of and Apparatus for Dismantling and Storage of Objects Comprising Alkali Metals, Such as Alkali Metal Containing Batteries. U.S. Patent 7,833,646 B2, 16 November 2010. [Google Scholar]
- Vadenbo, C.O. Prospective Environmental Assessment of Lithium Recovery in Battery Recycling; Institute of Environmental Decisions: Zurich, Switzerland, 2009. [Google Scholar]
- Rombach, E.; Friedrich, B. Recycling of Rare Metals. In Handbook of Recycling; Elsevier: Boston, MA, USA, 2014; pp. 125–150. [Google Scholar]
- Accurec Recycling GmbH, “Accurec,” 2019. Available online: https://accurec.de/lithium (accessed on 30 October 2019).
- Meshram, P.; Pandey, B.; Mankhand, T. Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review. Hydrometallurgy 2014, 150, 192–208. [Google Scholar] [CrossRef]
- Gratz, E.; Sa, Q.; Apelian, D.; Wang, Y. A closed loop process for recycling spent lithium ion batteries. J. Power Sources 2014, 262, 255–262. [Google Scholar] [CrossRef]
- Zou, H.; Gratz, E.; Apelian, D.; Wang, Y. A novel method to recycle mixed cathode materials for lithium ion batteries. Green Chem. 2013, 15, 1183. [Google Scholar] [CrossRef]
- Sa, Q.; Gratz, E.; He, M.; Lu, W.; Apelian, D.; Wang, Y. Synthesis of high performance LiNi1/3Mn1/3Co1/3O2 from lithium ion battery recovery stream. J. Power Sources 2015, 282, 140–145. [Google Scholar] [CrossRef]
- Baron, A. A Battery of Success, 2016. Available online: https://www.wpi.edu/news/battery-successes (accessed on 23 August 2019).
- National Science Foundation. SBIR Phase II: A Closed Loop Process for the Recycle of End-of-Life Li-Ion Batteries. 2017. Available online: https://www.nsf.gov/awardsearch/showAward?AWD_ID=1738027&HistoricalAw (accessed on 23 August 2019).
- Battery Resources, “Battery Resources News”. 2019. Available online: https://www.batteryresourcers.com/news (accessed on 23 August 2019).
- Weyhe, R.; Pan, Q. High Energy Lithium Suplhur Cells and Batteries; European Commission: Brussels, Belgium, 2016; pp. 1–30. [Google Scholar]
- Diekmann, J.; Hanisch, C.; Loellhoeffel, T.; Schälicke, G.; Kwade, A. Ecologically Friendly Recycling of Lithium-Ion Batteries—The LithoRec-Process. ECS Trans. 2016, 73, 1–9. [Google Scholar] [CrossRef]
- Diekmann, J.; Hanisch, C.; Frob, L.; Sch, G.; Loellhoeffel, T.; Kwade, A. Ecological Recycling of Lithium-Ion Batteries from Electric Vehicles with Focus on Mechanical Processes. J. Electrochem. Soc. 2017, 164, 6184–6191. [Google Scholar] [CrossRef]
- Duesenfeld. 2019. Available online: https://www.duesenfeld.com/recycling_en.html (accessed on 23 August 2019).
- Träger, T.; Friedrich, B.; Weyhe, R. Recovery Concept of Value Metals from Automotive Lithium-Ion Batteries. Chem. Ing. Tech. 2015, 87, 1550–1557. [Google Scholar] [CrossRef]
- Hanisch, C.; Diekmann, J.; Stieger, A.; Haselrieder, W.; Kwade, A. Recycling of Lithium-Ion Batteries. In Handbook of Clean Energy Systems; Yan, J., Cabeza, L.F., Sioshansi, R., Eds.; John Wiley & Sons, Ltd.: West Sussex, UK, 2015. [Google Scholar]
- Krüger, S.; Hanisch, C.; Kwade, A.; Winter, M.; Nowak, S. Effect of impurities caused by a recycling process on the electrochemical performance of Li[Ni0.33Co0.33Mn0.33]O2. J. Electroanal. Chem. 2014, 726, 91–96. [Google Scholar] [CrossRef]
- Hailey, P.; Kepler, K. Direct Recycling Technology for Plug-In electric Vehicle Lithium-Ion Battery Packs; Farasis Energy Inc.: Hayward, CA, USA, 2015. [Google Scholar]
- Sloop, S.E. Battery Materials Recycling; OnTo Technology LLC: Bend, OR, USA, 2015; p. 20. [Google Scholar]
- Gaines, L.; Sullivan, J.; Burnham, A.; Belharouak, I. Life-Cycle Analysis of Production and Recycling of Lithium Ion Batteries. Transp. Res. Rec. J. Transp. Res. Board 2011, 2252, 57–65. [Google Scholar] [CrossRef]
- Sloop, S.E.; Parker, R. System and Method for Processing an End-of-Life or Reduced Performance Energy Storage and/or Conversion Device Using a Supercritical Fluid. U.S. Patent 8,067,107 B2, 29 November 2011. [Google Scholar]
- Sloop, S.E. Reintroduction of Lithium into Recycled Battery Materials. U.S. Patent 8,846,225 B2, 30 September 2014. [Google Scholar]
- Sloop, S.E. Reintroduction of Lithium into Recycled Battery Materials. U.S. Patent 9,287,552 B2, 15 March 2016. [Google Scholar]
- Sloop, S.E. Recycling Positive-Electrode Material of a Lithium-Ion Battery. U.S. Patent 2016/004,3450 A1, 11 February 2016. [Google Scholar]
- Sloop, S.E.; Bend, M.A. Recycling and Reconditioning of Battery Electrode Materials. U.S. Patent 9,484,606 B1, 1 November 2016. [Google Scholar]
- Dunn, J.B.; Gaines, L.; Sullivan, J.; Wang, M.Q. Impact of Recycling on Cradle-to-Gate Energy Consumption and Greenhouse Gas Emissions of Automotive Lithium-Ion Batteries. Environ. Sci. Techol. 2012, 46, 12704–12710. [Google Scholar] [CrossRef]
- Zhang, T.; He, Y.; Ge, L.; Fu, R.; Zhang, X.; Huang, Y. Characteristics of wet and dry crushing methods in the recycling process of spent lithium-ion batteries. J. Power Sources 2013, 240, 766–771. [Google Scholar] [CrossRef]
- Ekberg, C.; Petranikova, M. (Eds.) Lithium Process Chemistry; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Pudas, J. Best Battery Recycling, Dry Technology; Akkuser: Nivala, Finland, 2009. [Google Scholar]
- Agency for Toxic Substances & Disease Registry. ATSDR, 2015. Available online: https://www.atsdr.cdc.gov/phs/phs.asp?id=711&tid=128 (accessed on 15 August 2019).
- Dunn, J.; Gaines, L.; Barnes, M.; Wang, M.; Sullivan, J. Material and Energy Flows in the Materials Production, Assembly, and End-of-Life Stages of the Automotive Lithium-Ion Battery Life Cycle; Argonne National Laboratory: Chicago, IL, USA, 2012. [Google Scholar]
- Rothermel, S.; Evertz, M.; Kasnatscheew, J.; Qi, X.; Grützke, M.; Winter, M.; Nowak, S.; Kasnascheetw, J. Graphite Recycling from Spent Lithium-Ion Batteries. ChemSusChem 2016, 9, 3473–3484. [Google Scholar] [CrossRef] [PubMed]
- Boyden, A.; Soo, V.K.; Doolan, M. The Environmental Impacts of Recycling Portable Lithium-Ion Batteries. Procedia CIRP 2016, 48, 188–193. [Google Scholar] [CrossRef]
- Dunn, J.B.; Gaines, L.; Kelly, J.C.; James, C.; Gallagher, K.G. The significance of Li-ion batteries in electric vehicle life-cycle energy and emissions and recycling’s role in its reduction. J. Ind. Ecol. 2015, 19, 518–519. [Google Scholar] [CrossRef]
- European Comission. Report on Non-Critical Raw Material Profiles; European Commission: Brussels, Belgium, 2014. [Google Scholar]
- Glöser, S.; Espinoza, L.T.; Gandenberger, C.; Faulstich, M. Raw material criticality in the context of classical risk assessment. Resour. Policy 2015, 44, 35–46. [Google Scholar] [CrossRef]
- Oliveira, L.; Messagie, M.; Rangaraju, S.; Sanfelix, J.; Rivas, M.H.; Van Mierlo, J. Key issues of lithium-ion batteries—From resource depletion to environmental performance indicators. J. Clean. Prod. 2015, 108, 354–362. [Google Scholar] [CrossRef]
- Martin, G.; Rentsch, L.; Höck, M.; Bertau, M. Lithium market research—Global supply, future demand and price development. Energy Storage Mater. 2017, 6, 171–179. [Google Scholar] [CrossRef]
- Reuter, M.A.; Hudson, C.; van Schaik, A.; Heiskanen, K.; Meskers, C.; Hagelüken, C. Metal Recycling: Opportunities, Limits, Infrastructure, A Report of the Working Group on the Global Metal Flows to the Inter- national Resource Panel; United Nations Environmental Programme: Paris, France, 2013. [Google Scholar]
- Tran, T.; Luong, V.T. Lithium Production Processes. In Lithium Process Chemistry; Elsevier: Amsterdam, The Netherlands, 2015; pp. 81–124. [Google Scholar]
- Hanisch, C.; Haselrieder, W.; Kwade, A. Recovery of Active Materials from Spent Lithium-Ion Electrodes and Electrode Production Rejects. In Glocalized Solutions for Sustainability in Manufacturing; Springer Science and Business Media LLC: Berlin, Germany, 2011; pp. 85–89. [Google Scholar]
- Moradi, B.; Botte, G.G. Recycling of graphite anodes for the next generation of lithium ion batteries. J. Appl. Electrochem. 2016, 46, 123–148. [Google Scholar] [CrossRef]
- Llamas, A.A.; Delgado, A.V.; Capilla, A.V.; Cuadra, C.T.; Hultgren, M.; Peltomäki, M.; Roine, A.; Stelter, M.; Reuter, M. Simulation-based exergy, thermo-economic and environmental footprint analysis of primary copper production. Miner. Eng. 2019, 131, 51–65. [Google Scholar] [CrossRef]
- Michaux, B.; Hannula, J.; Rudolph, M.; Reuter, M.; Boogaart, K.V.D.; Möckel, R.; Kobylin, P.; Hultgren, M.; Peltomäki, M.; Roine, A.; et al. Water-saving strategies in the mining industry—The potential of mineral processing simulators as a tool for their implementation. J. Environ. Manag. 2019, 234, 546–553. [Google Scholar] [CrossRef]
- Jose-Luis, P.; Abadias, A.; Valero, A.; Valero, A.; Reuter, M. The energy needed to concentrate minerals from common rocks: The case of copper ore. Energy 2019, 181, 494–503. [Google Scholar] [CrossRef]
- Reuter, M.A.; Van Schaik, A.; Gutzmer, J.; Bartie, N.; Abadías-Llamas, A. Challenges of the Circular Economy: A Material, Metallurgical, and Product Design Perspective. Annu. Rev. Mater. Res. 2019, 49, 253–274. [Google Scholar] [CrossRef]

| Battery Component | %w/w | Most Commonly Used Material |
|---|---|---|
| Case | ~25% | Steel/plastics |
| Cathode | ~27% | LiCoO2, LiNixMnyCozO2, LiMn2O4, LiNiO2, LiFePO4 |
| Anode | ~17% | Graphite/Li4Ti5O12 |
| Copper and aluminium foils and current collectors | ~13% | Cu/Al |
| Electrolyte | ~10% | Solution of LiPF6, LiBF4, LiClO4, and LiSO2 dissolved in propylene carbonate, ethylene carbonate, or dimethyl sulfoxide |
| Separator | ~4% | Microporous polypropylene |
| Binder | ~4% | Polivinylidene Difluoride (PVDF) |
| Process | Feed | Pre-Processing | Mechanical Processing | Pyro | Hydro | Main Recoveries | Secondary Recoveries | Use of Main Recoveries | Use of Secondary Recoveries | Losses |
|---|---|---|---|---|---|---|---|---|---|---|
| Umicore ValÉas™ | LIB, NiMH | Dismantling | - | Shaft furnace | Leaching solvent extraction | Co, Ni, Cu, Fe CoCl2 | Slag: Al, Si, Ca, Fe, Li, Mn, REE | Metal industry, cathode production | Construction industry | Electrolyte, plastics, graphite |
| Sumitomo–Sony | LIB. | Sorting Dismantling | - | Calcination | Hydro | CoO | Co–Ni–Fe alloy Cu, Al, Fe | Battery industry | Metal industry | Electrolyte, plastics, Li, Ni, graphite |
| Retriev Technologies | LIB Primary Li | Dismantling | Wet comminution, screening, shaking table, filtration, | Precipitation | Li2CO3 MeO | Steel Cu, Co, Al | Cathode production, metal industry | Metal industry | Plastic | |
| Recupyl Valibat | LIB Primary Li | - | crushing, vibrating screen, secondary screen, magnetic separator, densimetric table, | - | Hydrolysis leaching | Li2CO3, LiCO2, Li3PO4 | Steel Cu, Al Cu, MeO, C | Cathode Production. | Metal industry | Cu, graphite |
| Akkuser | LIB | Sorting | 1st cutting, air filtration, cutting, magnetic separator | - | - | Co, Cu powder Fe | Non-ferrous metals | Various including metal industry | Metal industry | Plastic |
| Accurec | LIB | Sorting Dismantling | Milling, separation, agglomeration, filtration, ambient | Vacuum thermal treatment, reduction | H2SO4 | Li2CO3 Co-Alloy | Metallic alloy | Production of cathode material | Metal recovery | Electrolyte, polymers, graphite |
| Battery Resources | LIB | Discharge | Shredding, magnetic separation, sieving, dense media; ambient | Sintering | Leaching by: NaOH, H2O2, H2SO4, and Na2CO3 | Li2CO3 NMC(OH)2 | Ferrous metals | Battery cell production | Metal industry | Electrolyte |
| LithoRec | LIB | Discharge Manual disassembly | Two-stage crushing, two-stage air classification; inert. | Drying, calcination | Undisclosed leaching agent | Li2CO3 metal oxides | Al–Cu, plastic fractions | Battery cell production | Metal industry | Electrolyte |
| OnTo | LIB Primary Li | Discharge Dismantling | Shredding, sieving dense media separation; supercritical CO2 | Present (undisclosed). | Leaching purification, dissolution | Refurbished cell cathode powder | Ferrous and non-ferrous metals | Battery cell production | Metal industry | Binder, graphite |
| Aalto University | LIB | - | Shredding, sieving | Aluminium smelter | Leaching | CoC2O4 Al | Jarosite, manganese dioxide, copper hydroxide, Li–Ni solution. | Reprocessing required | Reprocessing required | Graphite, binder plastic, Cu, water |
| Process | Co Recovery | Quality of Recovery |
|---|---|---|
| Umicore | CoCl2 | Ready for LiCoO2 synthesis |
| Sumitomo–Sony | CoO | Recovery needs pyrometallurgical processs before cathode synthesis |
| Retriev Technologies | MeO + cake | Elemental cobalt |
| Recupyl | LCO/Co(OH)2/Co | final product |
| Akkuser | Co + graphite | Recovery requires hydro- and/or pyrometallurgical processes before cathode synthesis |
| Accurec | Co alloy | Elemental cobalt |
| Battery Resources | NMC(OH)2 | Ready for cathode synthesis |
| LithoRec | MeO | Ready for cathode synthesis |
| OnTo | Cathode powder (refurbished) | Final product |
| Aalto University | CoC2O4 | Recovery requires processing |
| Process | Li Product | Recovery Method | Efficiency | Recovery Quality | Target Use of Recovery |
|---|---|---|---|---|---|
| Umicore | Not recovered | - | - | - | - |
| Sumitomo–Sony | Not recovered | - | - | - | - |
| Retriev Technologies | Li2CO3 | Precipitation with Na2CO3/CO2 | 90% | No data | Metal manufacture |
| Recupyl | Li2CO3/Li3PO4 | Precipitation with CO2 | No Data | No data | Not discussed |
| Akkuser | Not recovered | - | - | - | - |
| Accurec | Li2CO3 | Precipitation | 76–90% | >99% | Glass production/cathode synthesis |
| Battery Resources | Li2CO3 | Precipitation with Na2CO3 | 67–80% | “High Purity” | Cathode powder synthesis |
| LithoRec | Li2CO3/LiOH | Precipitation | 85–95% | No Data | Cathode powder synthesis |
| OnTo | Li2CO3/cathode | Undisclosed | Undisclosed | 99% | Battery production |
| Aalto University | Li–Ni solution | Leaching | 93–97% | 0.1% | Not discussed |
| Process | Fe Casing | Al and Cu Foils | Losses |
|---|---|---|---|
| Umicore | Fe: recovered in alloy | Cu: recovered in alloy | Al, polymer |
| Sumitomo–Sony | Fe: recovered in alloy | Cu: recovered.in alloy | Al, polymer |
| Retriev Technologies | Fe, Al, and plastics: recovered in shaking table | Not mentioned. | Not mentioned |
| Recupyl | Fe: recovered with magnetic separator | Al, Cu, and plastics: recovered with density separation | Not mentioned |
| Akkuser | Fe: recovered with magnetic separator Plastics: recovered via air filtration | Al and Cu: recovered from fin powder | Fraction of Cu and Al |
| Accurec | Fe: recovered with magnetic separator | Al and Cu: recovered via air separator | Polymer |
| Battery Resources | Fe: recovered with magnetic separator | Al: recovered during leaching Cu: recovered via dense media separation and precipitation | Not mentioned |
| LithoRec | Fe, Al, and plastics: recovered via air separator | Al and Cu: recovered via sieves and zig-zag sifter | Not mentioned |
| OnTo | Fe, Al, and plastics: recovered | Al and Cu: recovered | Not mentioned |
| Aalto University | Fe: extracted at pre-processing | Rotary kiln | Cu, graphite. |
| Process | Graphite |
|---|---|
| Umicore | Used as reducing agent in furnace |
| Sumitomo–Sony | Lost during calcination |
| Retriev Technologies | Recovered during filtration as: MeO–graphite cake |
| Recupyl | Filtered off in leaching step |
| Akkuser | Remain as fraction of the black mass |
| Accurec | Used as reduction agent |
| Battery Resources | Extraction, no use expressed |
| LithoRec | Extraction, no use expressed |
| OnTo | Recovered by leaching for LIB purposes |
| Aalto University | Lost in rotary kiln |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Velázquez-Martínez, O.; Valio, J.; Santasalo-Aarnio, A.; Reuter, M.; Serna-Guerrero, R. A Critical Review of Lithium-Ion Battery Recycling Processes from a Circular Economy Perspective. Batteries 2019, 5, 68. https://doi.org/10.3390/batteries5040068
Velázquez-Martínez O, Valio J, Santasalo-Aarnio A, Reuter M, Serna-Guerrero R. A Critical Review of Lithium-Ion Battery Recycling Processes from a Circular Economy Perspective. Batteries. 2019; 5(4):68. https://doi.org/10.3390/batteries5040068
Chicago/Turabian StyleVelázquez-Martínez, Omar, Johanna Valio, Annukka Santasalo-Aarnio, Markus Reuter, and Rodrigo Serna-Guerrero. 2019. "A Critical Review of Lithium-Ion Battery Recycling Processes from a Circular Economy Perspective" Batteries 5, no. 4: 68. https://doi.org/10.3390/batteries5040068
APA StyleVelázquez-Martínez, O., Valio, J., Santasalo-Aarnio, A., Reuter, M., & Serna-Guerrero, R. (2019). A Critical Review of Lithium-Ion Battery Recycling Processes from a Circular Economy Perspective. Batteries, 5(4), 68. https://doi.org/10.3390/batteries5040068





