Interface Stabilization of Aqueous Aluminum Batteries via Non-Flammable Co-Solvent
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials Preparation
2.2. Material Characterization
2.3. Electrochemical Measurements
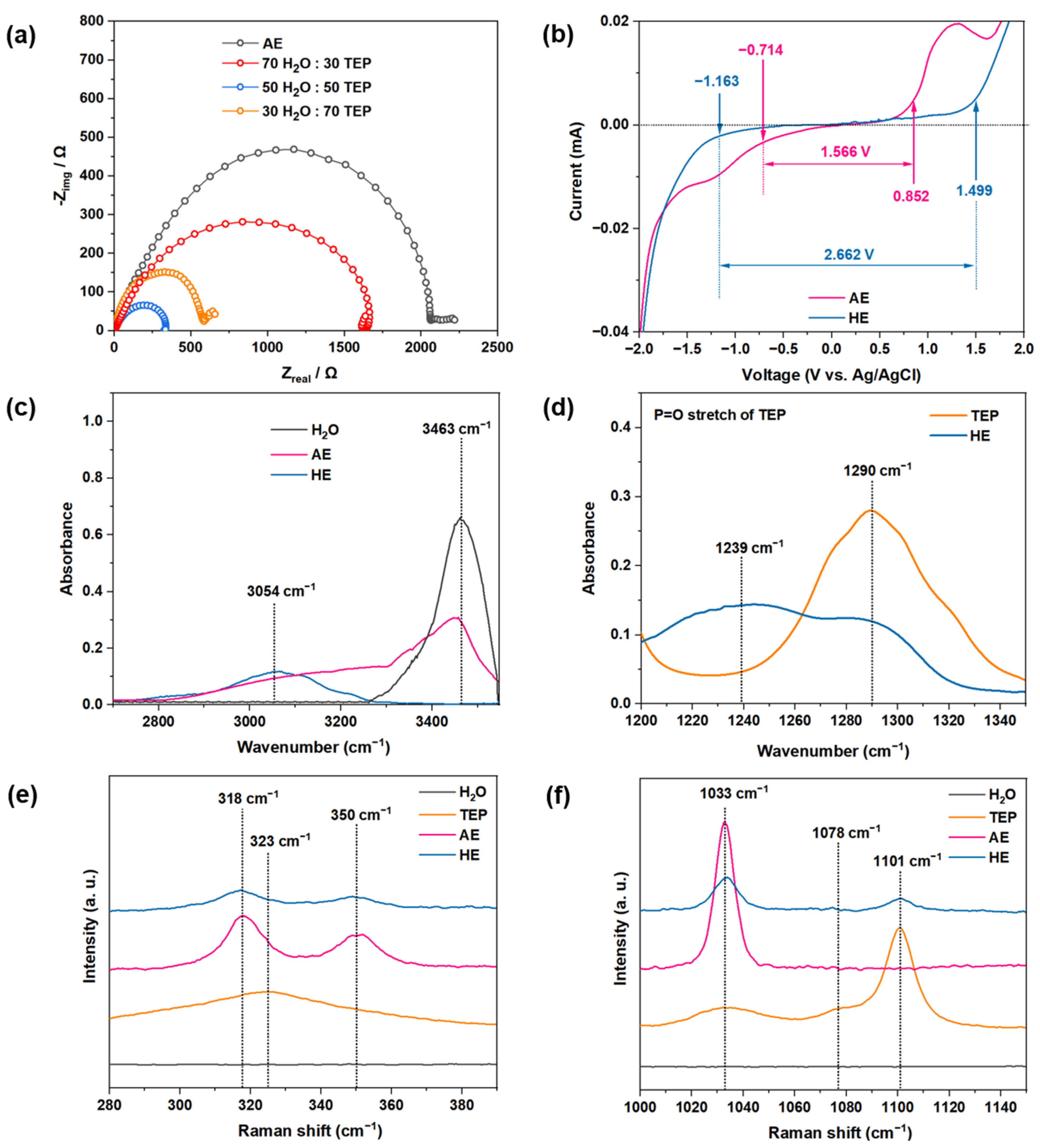
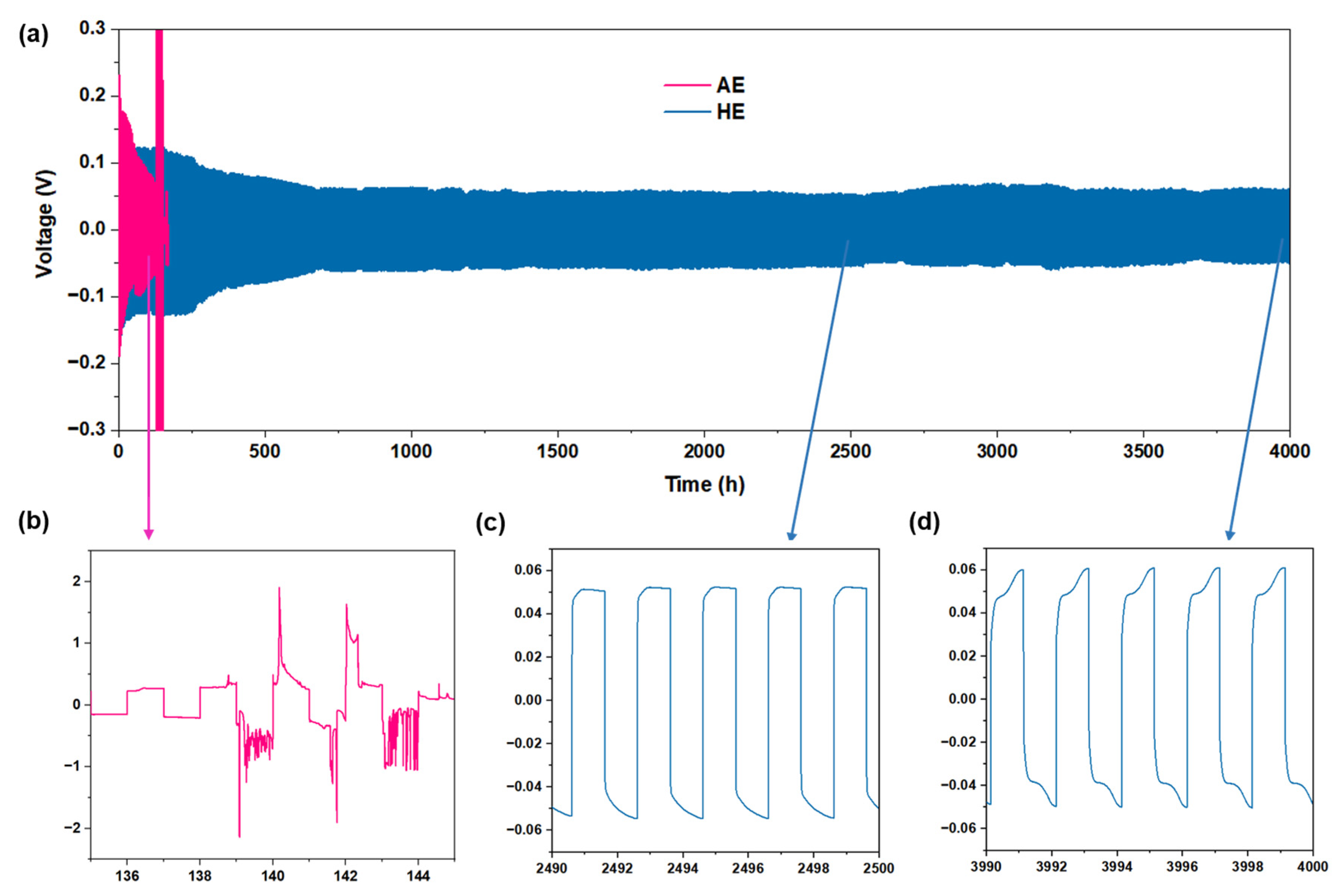
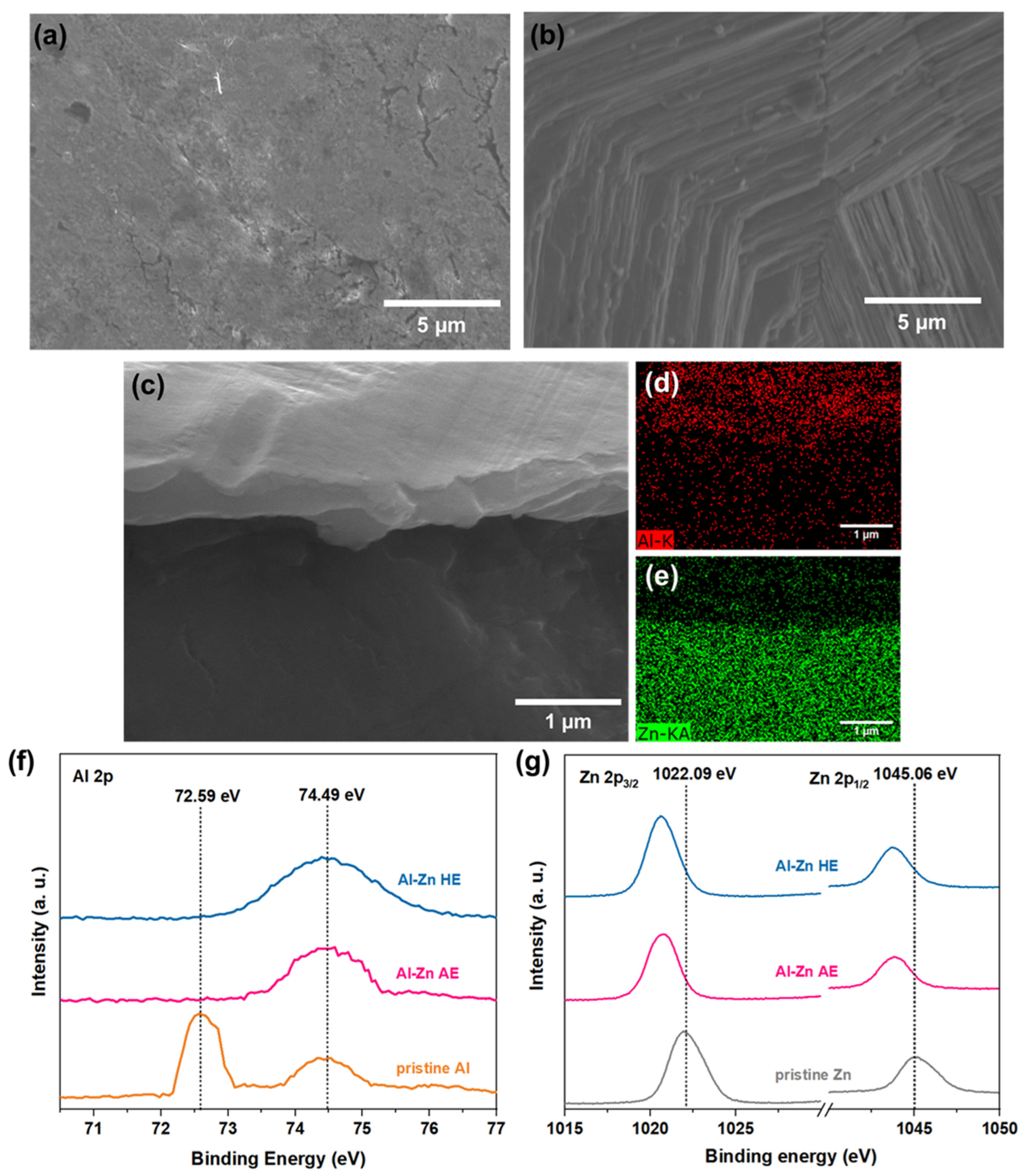
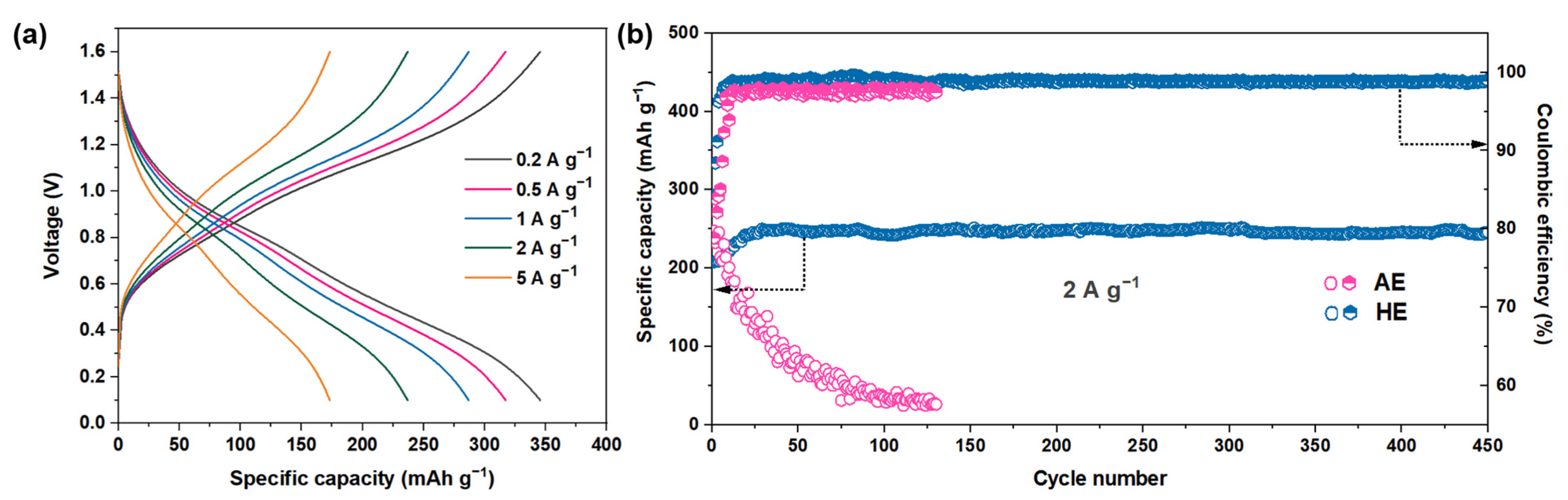
3. Results and Discussion
4. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Kim, K.-I. Progresses, Challenges and Prospects for Aluminum Anode in Aqueous Secondary Batteries. J. Electrochem. Sci. Technol. 2025, 16, 37–53. [Google Scholar] [CrossRef]
- Chao, D.; Zhou, W.; Xie, F.; Ye, C.; Li, H.; Jaroniec, M.; Qiao, S.Z. Roadmap for Advanced Aqueous Batteries: From Design of Materials to Applications. Sci. Adv. 2020, 6, eaba4098. [Google Scholar] [CrossRef]
- Blanc, L.E.; Kundu, D.; Nazar, L.F. Scientific Challenges for the Implementation of Zn-ion Batteries. Joule 2020, 4, 771–799. [Google Scholar] [CrossRef]
- Jia, H.; Wang, Z.; Tawiah, B.; Wang, Y.; Chan, C.Y.; Fei, B.; Pan, F. Recent Advances in Zinc Anodes for High-performance Aqueous Zn-ion Batteries. Nano Energy 2020, 70, 104523. [Google Scholar] [CrossRef]
- Ru, Y.; Zheng, S.; Xue, H.; Pang, H. Potassium Cobalt Hexacyanoferrate Nanocubic Assemblies for High-performance Aqueous Aluminum Ion Batteries. Chem. Eng. J. 2020, 382, 122853. [Google Scholar] [CrossRef]
- Butterwick, L.; Smith, G.D.W. Aluminum Recovery from Consumer Waste- I. Technology Review. Resour. Conserv. Recycl. 1986, 9, 281–292. [Google Scholar] [CrossRef]
- Kim, D.J.; Yoo, D.-J.; Otley, M.T.; Prokofjevs, A.; Pezzato, C.; Owczarek, M.; Lee, S.J.; Choi, J.W.; Stoddart, J.F. Rechargeable Aluminum Organic Batteries. Nat. Energy 2019, 4, 51–59. [Google Scholar] [CrossRef]
- Lin, Z.J.; Mao, M.L.; Yang, C.X.; Tong, Y.X.; Li, Q.H.; Yue, J.M.; Yang, G.J.; Zhang, Q.H.; Hong, L.; Yu, X.Q.; et al. Amorphous Anion-rich Titanium Polysulfides for Aluminum-ion Batteries. Sci. Adv. 2021, 7, eabg6314. [Google Scholar] [CrossRef]
- Pang, Q.; Meng, J.; Gupta, S.; Hong, X.; Kwok, C.Y.; Zhao, J.; Jin, Y.; Xu, L.; Karahan, O.; Wang, Z.; et al. Fast-charging Aluminum-chalcogen Batteries Resistant to Dendritic Shorting. Nature 2022, 608, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wang, W.; Lei, H.; Wang, M.; Jiao, S. Alternate Storage of Opposite Charges in Multisites for High-energy-density Al-MOF Batteries. Adv. Mater. 2022, 34, 2110109. [Google Scholar] [CrossRef]
- Ji, X. A Paradigm of Storage Batteries. Energy Environ. Sci. 2019, 12, 3203–3224. [Google Scholar] [CrossRef]
- Chen, Y.; Kang, Y.; Zhao, Y.; Wang, L.; Liu, J.; Li, Y.; Liang, Z.; He, X.; Li, X.; Tavajohi, N.; et al. A Review of Lithium-ion Battery Safety Concerns. J. Energy Chem. 2021, 59, 83–99. [Google Scholar] [CrossRef]
- Chayambuka, K.; Mulder, G.; Danilov, D.L.; Notten, P.H. From Li-ion Batteries toward Na-ion Chemistries: Challenges and Opportunities. Adv. Energy Mater. 2020, 10, 2001310. [Google Scholar] [CrossRef]
- Wang, Q.; Mao, B.; Stoliarov, S.I.; Sun, J. A Review of Lithium Ion Battery Failure Mechanisms and Fire Prevention Strategies. Prog. Energy Combust. Sci. 2019, 73, 95–131. [Google Scholar] [CrossRef]
- Angell, M.; Pan, C.-J.; Rong, Y.; Yuan, C.; Lin, M.-C.; Hwang, B.-J.; Dai, H. High Coulombic Efficiency Aluminum-ion Battery Using an AlCl3-Urea Ionic Liquid Analog Electrolyte. Proc. Natl. Acad. Sci. USA 2017, 114, 834–839. [Google Scholar] [CrossRef]
- Lin, M.-C.; Gong, M.; Lu, B.; Wu, Y.; Wang, D.-Y.; Guan, M.; Angell, M.; Chen, C.; Yang, J.; Hwang, B.-J.; et al. An ultrafast rechargeable aluminium-ion battery. Nature 2015, 520, 324–328. [Google Scholar] [CrossRef]
- Garcia-Mendez, R.; Zheng, J.; Bock, D.C.; Jaye, C.; Fischer, D.A.; Marschilok, A.C.; Takeuchi, K.J.; Takeuchi, E.S.; Archer, L.A. Understanding the Reversible Electrodeposition of Aluminum in Low-cost Room-temperature Molten Salts. Cell Rep. Phys. Sci. 2023, 4, 101452. [Google Scholar] [CrossRef]
- Pastel, G.R.; Chen, Y.; Pollard, T.P.; Schroeder, M.A.; Bowden, M.E.; Zheng, A.; Hahn, N.T.; Ma, L.; Murugesan, V.; Ho, J.; et al. A Sobering Examination of the Feasibility of Aqueous Aluminum Batteries. Energy Environ. Sci. 2022, 15, 2460–2469. [Google Scholar] [CrossRef]
- Dong, T.; Ng, K.L.; Wang, Y.; Voznyy, O.; Azimi, G. An Aqueous Rechargeable Al-ion Battery Based on Cobalt Hexacyanoferrate and Al Metal. Adv. Energy Mater. 2021, 11, 2100077. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, Z.; Xu, T.; Wang, W.; Wang, B.; Yu, X. Solvation Structure Regulation for Highly Reversible Aqueous Al Metal Batteries. J. Am. Chem. Soc. 2024, 146, 2257–2266. [Google Scholar] [CrossRef]
- Trachtman, M.; Markham, G.D.; Glusker, J.P.; George, P.; Bock, C.W. Interactions of Metal Ions with Water: Ab Initio Molecular Orbital Studies of Structure, Bonding Enthalpies, Vibrational Frequencies and Charge Distributions. 1. Monohydrates. Inorg. Chem. 1998, 37, 4421–4431. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Lv, C.; Wang, L.; Cui, W.; Zhang, L.; Dinh, K.N.; Tan, H.; Wu, C.; Wu, T.; Ren, Y.; et al. Architecting a Stable High-energy Aqueous Al-ion Battery. J. Am. Chem. Soc. 2024, 142, 15295–15304. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, C.; Chai, L.; Zhang, W.; Li, Z. Bimetallic Rechargeable Al/Zn Hybrid Aqueous Batteries Based on Al–Zn Alloys with Composite Electrolytes. Adv. Mater. 2022, 34, 2206099. [Google Scholar] [CrossRef]
- Dou, Q.; Yao, N.; Pang, W.K.; Park, Y.; Xiong, P.; Han, X.; Rana, H.H.; Chen, X.; Fu, Z.-H.; Thomsen, L.; et al. Unveiling Solvation Structure and Desolvation Dynamics of Hybrid Electrolytes for Ultralong Cyclability and Facile Kinetics of Zn-Al Alloy Anodes. Energy Environ. Sci. 2022, 15, 4572–4583. [Google Scholar] [CrossRef]
- Ran, Q.; Shi, H.; Meng, H.; Zeng, S.-P.; Wan, W.-B.; Zhang, W.; Lang, X.-Y.; Jiang, Q. Aluminum-copper Alloy Anode Materials for High-energy Aqueous Aluminum Batteries. Nat. Commun. 2022, 13, 576. [Google Scholar] [CrossRef]
- Jia, B.E.; Hu, E.; Hu, Z.; Liew, J.J.; Hong, Z.; Guo, Y.; Srinviassan, M.; Zhu, Q.; Xu, J.; Chen, J.; et al. Laminated Tin-aluminum Anodes to Build Practical Aqueous Aluminum Batteries. Energy Storage Mater. 2024, 65, 103141. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, C.; Luo, P.; Xin, Y.; Ge, Y.; Tian, H. An Artiricial Aluminum-tin Alloy Layer on Aluminum Metal Anodes for Ultra-stable Rechargeable Aluminum-ion Batteries. Nanoscale 2024, 16, 13171–13182. [Google Scholar] [CrossRef]
- Chen, J.; Vatamanu, J.; Xing, L.; Borodin, O.; Chen, H.; Guan, X.; Liu, X.; Xu, K.; Li, W. Improving Electrochemical Stability and Low-temperature Performance with Water/acetonitrile Hybrid Electrolytes. Adv. Energy Mater. 2020, 10, 1902654. [Google Scholar] [CrossRef]
- Shi, J.; Xia, K.; Liu, L.; Liu, C.; Zhang, Q.; Li, L.; Zhou, X.; Liang, J.; Tao, Z. Ultrahigh Coulombic Efficiency and long-life Aqueous Zn Anodes Enabled by Electrolyte Additive of Acetonitrile. Electrochim. Acta 2020, 358, 136937. [Google Scholar] [CrossRef]
- Yang, D.; Watanabe, M.; Takagaki, A.; Ishihara, T. High Voltage and Capacity Dual-ion Battery Using Acetonitrile-aqueous Hybrid Electrolyte with Concentrated LiFSI-LiTFSI. J. Electrochem. Soc. 2022, 169, 120516. [Google Scholar] [CrossRef]
- Dou, Q.; Lei, S.; Wang, D.-W.; Zhang, Q.; Xiao, D.; Guo, H.; Wang, A.; Yang, H.; Li, Y.; Shi, S.; et al. Safe and High-rate Supercapacitors Based on An “Acetonitrile/water in Salt” Hybrid Electrolyte. Energy Environ. Sci. 2018, 11, 3212–3219. [Google Scholar] [CrossRef]
- Wang, F.; Borodin, O.; Ding, M.S.; Gobet, M.; Vatamanu, J.; Fan, X.; Gao, T.; Eidson, N.; Liang, Y.; Sun, W.; et al. Hybrid Aqueous/non-aqueous Electrolyte for Safe and High-energy Li-ion Batteries. Joule 2018, 2, 927–937. [Google Scholar] [CrossRef]
- Miao, L.; Wang, R.; Di, S.; Qian, Z.; Zhang, L.; Xin, W.; Liu, M.; Zhu, Z.; Chu, S.; Du, Y.; et al. Aqueous Electrolytes with Hydrophobic Organic Cosolvents for Stabilizing Zinc Metal Anodes. ACS Nano 2022, 16, 9667–9678. [Google Scholar] [CrossRef]
- Kakoty, B.; Vengarathody, R.; Mukherji, S.; Ahuja, V.; Joseph, A.; Narayana, C.; Balasubramanian, S.; Senguttuvan, P. Two for One: Propylene Carbonate Co-solvent for High Performance Aqueous Zinc-ion Batteries- Remedies for Persistent Issues at Both Electrodes. J. Mater. Chem. A 2022, 10, 12597–12607. [Google Scholar] [CrossRef]
- Ren, H.; Li, S.; Xu, L.; Wang, L.; Liu, X.; Wang, L.; Liu, Y.; Zhang, L.; Zhang, H.; Gong, Y.; et al. Tailoring Water-in-DMSO Electrolyte for Ultra-Stable Rechargeable Zinc Batteries. Angew. Chem. 2025, 137, e202423302. [Google Scholar] [CrossRef]
- Jia, H.; Jiang, X.; Wang, Y.; Lam, Y.; Shi, S.; Liu, G. Hybrid Co-solvent-induced High-entropy Electrolyte: Regulating of Hydrated Zn2+ Solvation Structures for Excellent Reversibility and Wide Temperature Adaptability. Adv. Energy Mater. 2024, 14, 2304285. [Google Scholar] [CrossRef]
- Greenburg, L.C.; Holoubek, J.; Cui, Y.; Zhang, P.; Ai, H.; Zhang, E.; Liu, C.; Feng, G.; Cui, Y. Crowding Agent Stabilizes Aqueous Electrolyte for Reversible Iron Metal Anode. ACS Energy Lett. 2025, 10, 1022–1029. [Google Scholar] [CrossRef]
- Wang, F.; Borodin, O.; Gao, T.; Fan, X.; Sun, W.; Han, F.; Faraone, A.; Dura, J.A.; Xu, K.; Wang, C. Highly Reversible Zinc Metal Anode for Aqueous Batteries. Nat. Mater. 2018, 17, 543–549. [Google Scholar] [CrossRef]
- Naveed, A.; Yang, H.; Yang, J.; Nuli, Y.; Wang, J. Highly Reversible and Rechargeable Safe Zn Batteries Based on A Triethyl Phosphate Electrolyte. Angew. Chem. Int. Ed. 2019, 58, 2760–2764. [Google Scholar] [CrossRef]
- Ali, A.F.; Mustarelli, P.; Magistris, A. Optimal Synthesis of Organo-phosphate Precursors for Sol-gel Preparations. Mater. Res. Bull. 1998, 33, 697–710. [Google Scholar] [CrossRef]
- Rulmont, A.; Cahay, R.; Duyckaerts, M.L.; Tarte, P. Vibrational Spectroscopy of Phosphate: Some General Correlations between Structure and Spectra. Eur. J. Solid State Inorg. Chem. 1991, 28, 207–219. [Google Scholar]
- Kim, D.J.; Hwang, H.Y.; Nam, S.Y. Characterization of Sulfonated Poly(Arylene Ether Sulfone) (SPAES)/Silica-phosphate Sol-gel Composite Membrane: Effects of the Sol-Gel Composition. Macromol. Res. Up 2013, 21, 1194–1200. [Google Scholar] [CrossRef]
- Rudolph, W.W.; Mason, R.; Pye, C.C. Aluminum(III) Hydration in Aqueous Solution. A Raman Spectroscopic Investigation and an Ab Initio Molecular Orbital Study of Aluminum(III) Water Clusters. Phys. Chem. Chem. Phys. 2000, 2, 5030–5040. [Google Scholar] [CrossRef]
- Rudolph, W.W.; Irmer, G. Hydration and Ion Pair Formation in Common Aqueous La(III) Salt Solutions- a Raman Scattering and DFT Study. Dalton Trans. 2015, 44, 295–305. [Google Scholar] [CrossRef]
- Kumar, S.; Rama, P.; Yang, G.; Lieu, W.Y.; Chinnadurai, D.; Seh, Z.W. Additive-driven Interfacial Engineering of Aluminum Metal Anode for Ultralong Cycling Life. Nanomicro Lett. 2023, 15, 21. [Google Scholar] [CrossRef]
- Latos, P.; Culkin, A.; Barteczko, N.; Boncel, S.; Jurczyk, S.; Brown, L.C.; Nockemann, P.; Chrobok, A.; Swadźba-Kwaśny, M. Water-tolerant Trifloaluminate Ionic Liquids: New and Unique Lewis Acidic Catalysts for the Synthesis of Chromane. Front. Chem. 2018, 6, 535. [Google Scholar] [CrossRef]
- Ejigu, A.; Le Fevre, L.W.; Elgendy, A.; Spencer, B.F.; Bawn, C.; Dryfe, R.A. Optimization of Electrolytes for High-performance Aqueous Aluminum-ion Batteries. ACS Appl. Mater. Interfaces 2022, 14, 25232–25245. [Google Scholar] [CrossRef]
- Czamara, K.; Majzner, K.; Pacia, M.Z.; Kochan, K.; Kaczor, A.; Baranska, E.M. Raman Spectroscopy of Lipids: A Review. J. Raman Spectrosc. 2015, 46, 4–20. [Google Scholar] [CrossRef]
- Ta, K.M.; Cooke, D.J.; Gillie, L.J.; Parker, S.C.; Seal, S.; Wilson, P.B.; Phillips, R.M.; Skelton, J.M.; Molinari, M. Infrared and Raman Diagnostic Modeling of Phosphate Adsorption on Ceria Nanoparticles. J. Phys. Chem. C 2023, 127, 20183–20193. [Google Scholar] [CrossRef]
- Latreche, M.; Willart, J.F.; Paccou, L.; Guinet, Y.; Danède, F.; Hédoux, A. Contribution of Low-frequency Raman Spectroscopy to Determine the Solubility Curves of Drugs in Polymers: The Case of Paracetamol/PVP. Eur. J. Pharm. Biopharm. 2020, 154, 222–227. [Google Scholar] [CrossRef]
- Hadjiivanov, K.I.; Panayotov, D.A.; Mihaylov, M.Y.; Ivanova, E.Z.; Chakarova, K.K.; Andonova, S.M.; Drenchev, N.L. Power of Infrared and Raman Spectroscopies to Characterize Metal-organic Frameworks and Investigate Their Interaction with Guest Molecules. Chem. Rev. 2020, 121, 1286–1424. [Google Scholar] [CrossRef]
- Fales, B.S.; Fujamade, N.O.; Nei, Y.W.; Oomens, J.; Rodgers, M.T. Infrared Multiple Photon Dissociation Action Spectroscopy and Theoretical Studies of Diethyl Phosphate Complexes: Effects of Protonation and Sodium Cationization on Structure. J. Am. Soc. Mass Spectrom. 2011, 22, 81–92. [Google Scholar] [CrossRef][Green Version]
- Horvath, A.; Anaredy, R.S.; Shaw, S.K. Solvents and Stabilization in Ionic Liquid Films. Langmuir 2022, 38, 9372–9381. [Google Scholar] [CrossRef]
- Murugavel, R.; Choudhury, A.; Walawalkar, M.G.; Pothiraja, R.; Rao, C.N.R. Metal Complexes of Organophosphate Esters and Open-framework Metal Phosphates: Synthesis, Structure, Transformations, and Applications. Chem. Rev. 2008, 108, 3549–3655. [Google Scholar] [CrossRef]
- Feliu, S., Jr.; Barranco, V. XPS Study of the Surface Chemistry of Conventional Hot-dip Galvanised Pure Zn, Galvanneal and Zn-Al Alloy Coatings on Steel. Acta Mater. 2003, 51, 5413–5424. [Google Scholar] [CrossRef]
- Bonova, L.; Zahoranová, A.; Kováčik, D.; Zahoran, M.; Mičušík, M.; Černák, M. Atmospheric Pressure Plasma Treatment of Flat Aluminum Surface. Appl. Surf. Sci. 2015, 331, 79–86. [Google Scholar] [CrossRef]
- Han, A.; Zhang, J.; Sun, W.; Chen, W.; Zhang, S.; Han, Y.; Feng, Q.; Zheng, L.; Gu, L.; Chen, C.; et al. Isolating Contiguous Pt Atoms and Forming Pt-Zn Intermetallic Nanoparticles to Regulate Selectivity in 4-Nitrophenylacetylene Hydrogenation. Nat. Commun. 2019, 10, 3787. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Cui, X.; Wei, S.; Zhang, Q.; Gu, L.; Meng, F.; Fan, J.; Zheng, W. Highly Active Zigzag-like Pt-Zn Alloy Nanowires with High-index Facets for Alcohol Electrooxidation. Nano Res. 2019, 12, 1173–1179. [Google Scholar] [CrossRef]
- Gong, M.; Deng, Z.; Xiao, D.; Han, L.; Hao, T.; Lu, Y.; Shen, T.; Liu, X.; Lin, R.; Huang, T.; et al. One-nanometer-thick Pt3Ni Bimetallic Nanowires Advanced Oxygen Reduction Reaction: Integrating Multiple Advantages into One Catalyst. Acs Catal. 2019, 9, 4488–4494. [Google Scholar] [CrossRef]
- Yang, C.; Wu, Z.; Zhang, G.; Sheng, H.; Tian, J.; Duan, Z.; Sohn, H.; Kropf, A.J.; Wu, T.; Krause, T.R.; et al. Promotion of Pd Nanoparticles by Fe and Formation of a Pd3Fe Intermetallic Alloy for Propane Dehydrogenation. Catal. Today 2019, 323, 123–128. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.-i. Interface Stabilization of Aqueous Aluminum Batteries via Non-Flammable Co-Solvent. Batteries 2025, 11, 324. https://doi.org/10.3390/batteries11090324
Kim K-i. Interface Stabilization of Aqueous Aluminum Batteries via Non-Flammable Co-Solvent. Batteries. 2025; 11(9):324. https://doi.org/10.3390/batteries11090324
Chicago/Turabian StyleKim, Keun-il. 2025. "Interface Stabilization of Aqueous Aluminum Batteries via Non-Flammable Co-Solvent" Batteries 11, no. 9: 324. https://doi.org/10.3390/batteries11090324
APA StyleKim, K.-i. (2025). Interface Stabilization of Aqueous Aluminum Batteries via Non-Flammable Co-Solvent. Batteries, 11(9), 324. https://doi.org/10.3390/batteries11090324





