Magnetic Properties and Microstructure of Ce-Cu-Al Low Melting Alloy Bonding Sm2Fe17N3 Magnet Fabricated by the Hot-Pressing Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Preparation
2.2. Performance Characterization
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sagawa, M.; Fujimura, S.; Togawa, N. New material for permanent magnets on a base of Nd and Fe. J. Appl. Phys. 1984, 55, 2083–2087. [Google Scholar] [CrossRef]
- Takagi, K.; Hirayama, Y.; Okada, S. Novel powder processing technologies for production of rare-earth permanent magnets. Sci. Technol. Adv. Mater. 2021, 22, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Su, R.; Wen, L.; Li, W.; Liu, X.; Zhang, Z.; Zhao, R.; Han, Y.; Zhang, X.; Li, W. Intrinsically High Magnetic Performance in Core–Shell Structural (Sm,Y)Fe12-Based Permanent Magnets. Adv. Mater. 2022, 34, 2203503. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; He, J.; Li, W.; Liu, X.; Zhang, J.; Wen, L.; Zhang, Z.; Hu, J.; Zhang, J.; Liao, X.; et al. Understanding the Role of Element Grain Boundary Diffusion Mechanism in Nd–Fe–B Magnets. Adv. Funct. Mater. 2022, 32, 2109529. [Google Scholar] [CrossRef]
- Gutfleisch, O.; Willard, M.A.; Brück, E. Magnetic Materials and Devices for the 21st Century: Stronger, Lighter, and More Energy Efficient. Adv. Mater. 2011, 23, 821–842. [Google Scholar] [CrossRef]
- Coey, J.M.D.; Hong, S.; Hurley, D.P.F. Intrinsic magnetic properties of new rare-earth iron intermetallic series. J. Magn. Magn. Mater. 1991, 101, 310–316. [Google Scholar] [CrossRef]
- Schnitzke, K.; Schultz, L.; Wecker, J. High coercivity in Sm2Fe17Nx magnets. Appl. Phys. Lett. 1990, 57, 2853–2855. [Google Scholar] [CrossRef]
- Coey, J.M.D.; Lawler, J.F.; Sun, H. Nitrogenation of R2Fe17compounds: R=rare earth. J. Appl. Phys. 1991, 69, 3007–3010. [Google Scholar] [CrossRef]
- Coey, J.M.D. Perspective and Prospects for Rare Earth Permanent Magnets. Engineering 2020, 6, 119–131. [Google Scholar] [CrossRef]
- Zheng, J.W.; Tian, S.J.; Liu, K.H. Preparation of submicron-sized Sm2Fe17N3 fine powder by ultrasonic spray pyrolysis-hydrogen reduction (USP-HR) and subsequent reduction–diffusion process. AIP Adv. 2020, 10, 055119. [Google Scholar] [CrossRef]
- Coey, J.M.D.; Hong, S. Improved magnetic properties by treatment of iron-based rare earth intermetallic compounds in anmonia. J. Magn. Magn. Mater. 1990, 87, L251–L254. [Google Scholar] [CrossRef]
- Cabral, F.A.O.; Gama, S.; de Morais, E. Study of thermal decomposition mechanism of the Sm2 Fe17N3 phase. IEEE Trans. Magn. 1996, 32, 4365–4367. [Google Scholar] [CrossRef]
- Takagi, K.; Soda, R.; Jinno, M. Possibility of high-performance Sm2Fe17N3 sintered magnets by low-oxygen powder metallurgy process. J. Magn. Magn. Mater. 2020, 506, 166811. [Google Scholar] [CrossRef]
- Mashimo, T.; Tashiro, S.; Hirosawa, S. Fully dense Sm2Fe17Nxpermanent magnets prepared by shock compression. J. Appl. Phys. 1996, 80, 356–361. [Google Scholar] [CrossRef]
- Mashimo, T.; Huang, X.S.; Hirosawa, S. Effects of decomposition on the magnetic property of shock-consolidated Sm2Fe17Nx bulk magnets. J. Mater. Process. Technol. 1999, 85, 138–141. [Google Scholar] [CrossRef]
- Chiba, A.; Hokamoto, K.; Sugimoto, S. Explosive consolidation of Sm–Fe–N and Sm–Fe–N/(Ni, Co) magnetic powders. J. Magn. Magn. Mater. 2007, 310, e881–e883. [Google Scholar] [CrossRef]
- Saito, T.; Fukui, M.; Takeishi, H. Sm–Fe–N bulk magnets produced by compression shearing method. Scr. Mater. 2005, 53, 1117–1121. [Google Scholar] [CrossRef]
- Saito, T.; Sato, H.; Fukui, M. Magnetic properties of Sm–Fe–N bulk magnets produced by compression shearing method. J. Appl. Phys. 2006, 99, 08B509. [Google Scholar] [CrossRef]
- Saito, T.; Sato, H.; Takeishi, H. Anisotropic Sm–Fe–N magnets produced by compression shearing method. Appl. Phys. Lett. 2006, 89, 162511. [Google Scholar] [CrossRef]
- Saito, T.; Kitazima, H. Hard magnetic properties of anisotropic Sm–Fe–N magnets produced by compression shearing method. J. Magn. Magn. Mater. 2011, 323, 2154–2157. [Google Scholar] [CrossRef]
- Handstein, A.; Wendhausen, P.A.P.; Gebel, B.; Pitschke, W.; Mayerhofer, O.; Kirchmayr, H.; Muller, K.H. Influence of hot isostatic pressing on the magnetic properties of melt-spun Sm/sub 2/Fe/sub 17/N/sub x/ magnets. IEEE Trans. Magn. 1993, 29, 2821–2823. [Google Scholar] [CrossRef]
- Ito, S.; Kikuchi, M.; Fujii, T. HIP sintering and magnetic properties of Sm2Fe17N3 with Zn additive. J. Magn. Magn. Mater. 2004, 270, 15–21. [Google Scholar] [CrossRef]
- Zhang, D.T.; Yue, M.; Zhang, J.X. Study on bulk Sm2Fe17Nxsintered magnets prepared by spark plasma sintering. Powder Met. 2007, 50, 215–218. [Google Scholar] [CrossRef]
- Saito, T. Magnetic properties of anisotropic Sm–Fe–N bulk magnets produced by spark plasma sintering method. J. Magn. Magn. Mater. 2008, 320, 1893–1897. [Google Scholar] [CrossRef]
- Saito, T. Magnetic properties of Sm–Fe–N anisotropic magnets produced by magnetic-field-assisted spark plasma sintering. Mater. Sci. Eng. B 2010, 167, 75–79. [Google Scholar] [CrossRef]
- Saito, T. Production of Sm–Fe–N bulk magnets by spark plasma sintering method. J. Magn. Magn. Mater. 2014, 369, 184–188. [Google Scholar] [CrossRef]
- Matsuura, M.; Nishijima, Y.; Tezuka, N. Increase of energy products of Zn-bonded Sm-Fe-N magnets with low oxygen content. J. Magn. Magn. Mater. 2018, 467, 64–68. [Google Scholar] [CrossRef]
- Yamaguchi, W.; Soda, R.; Takagi, K. Metal-coated Sm2Fe17N3 magnet powders with an oxide-free direct metal-metal interface. J. Magn. Magn. Mater. 2020, 498, 166101. [Google Scholar] [CrossRef]
- Yamaguchi, W.; Takagi, K. Effects of nonmagnetic overlay metals on coercivity of Sm2Fe17N3 magnet powders. J. Magn. Magn. Mater. 2020, 516, 167327. [Google Scholar] [CrossRef]
- Otani, Y.; Moukarika, A.; Sun, H. Metal bonded Sm2Fe17N3−δmagnets. J. Appl. Phys. 1991, 69, 6735–6737. [Google Scholar] [CrossRef]
- Kuhrt, C.; O’Donnell, K.; Katter, M. Pressure-assisted zinc bonding of microcrystalline Sm2Fe17Nxpowders. Appl. Phys. Lett. 1992, 60, 3316–3318. [Google Scholar] [CrossRef]
- Matsunami, R.; Matsuura, M.; Tezuka, N. Preparation of Sm−Fe−N Bulk Magnets with High Maximum Energy Products. J. Magn. Soc. Jpn. 2020, 44, 64–69. [Google Scholar] [CrossRef]
- Makita, K.; Hirosawa, S. Coercivity of Zn evaporation-coated Sm2Fe17Nx fine powder and its bonded magnets. J. Alloy. Compd. 1997, 260, 236–241. [Google Scholar] [CrossRef]
- Otogawa, K.; Takagi, K.; Asahi, T. Consolidation of Sm2Fe17N3 magnets with Sm-based eutectic alloy binder. J. Alloy. Compd. 2018, 746, 19–26. [Google Scholar] [CrossRef]
- Lu, C.; Zhu, J.; Gong, J. A method to improving the coercivity of sintered anisotropic Sm-Fe-N magnets. J. Magn. Magn. Mater. 2018, 461, 48–52. [Google Scholar] [CrossRef]
- Zhou, S.X.; Hoier, R.; Andersen, S. Magnetic properties and thermal stability of Nd-Fe-B based magnets with Co and Al. Mater. Forum 1993, 17, 27–34. [Google Scholar]
- Filip, O.; El-Aziz, A.M.; Hermann, R. Effect of Al additives and annealing time on microstructure and corrosion resistance of Nd–Fe–B alloys. Mater. Lett. 2001, 51, 213–218. [Google Scholar] [CrossRef]
- Duan, Z.; Xuan, H.; Su, J. Effects of Al Nanopowder Intergranular Addition on the Magnetic Properties and Temperature Stability of Sintered Nd-Fe-B Magnet. J. Supercond. Nov. Magn. 2022, 35, 547–553. [Google Scholar] [CrossRef]
- Zeng, H.; Liu, Z.; Li, W. Significantly enhancing the coercivity of NdFeB magnets by ternary Pr-Al-Cu alloys diffusion and understanding the elements diffusion behavior. J. Magn. Magn. Mater. 2019, 471, 97–104. [Google Scholar] [CrossRef]
- Soda, R.; Takagi, K.; Jinno, M. Anisotropic Sm2Fe17N3 sintered magnets without coercivity deterioration. AIP Adv. 2016, 6, 115108. [Google Scholar] [CrossRef]
- Yamaguchi, W.; Soda, R.; Takagi, K. Role of Surface Iron Oxides in Coercivity Deterioration of Sm2Fe17N3 Magnet Associated with Low Temperature Sintering. Mater. Trans. 2019, 60, 64–69. [Google Scholar] [CrossRef]
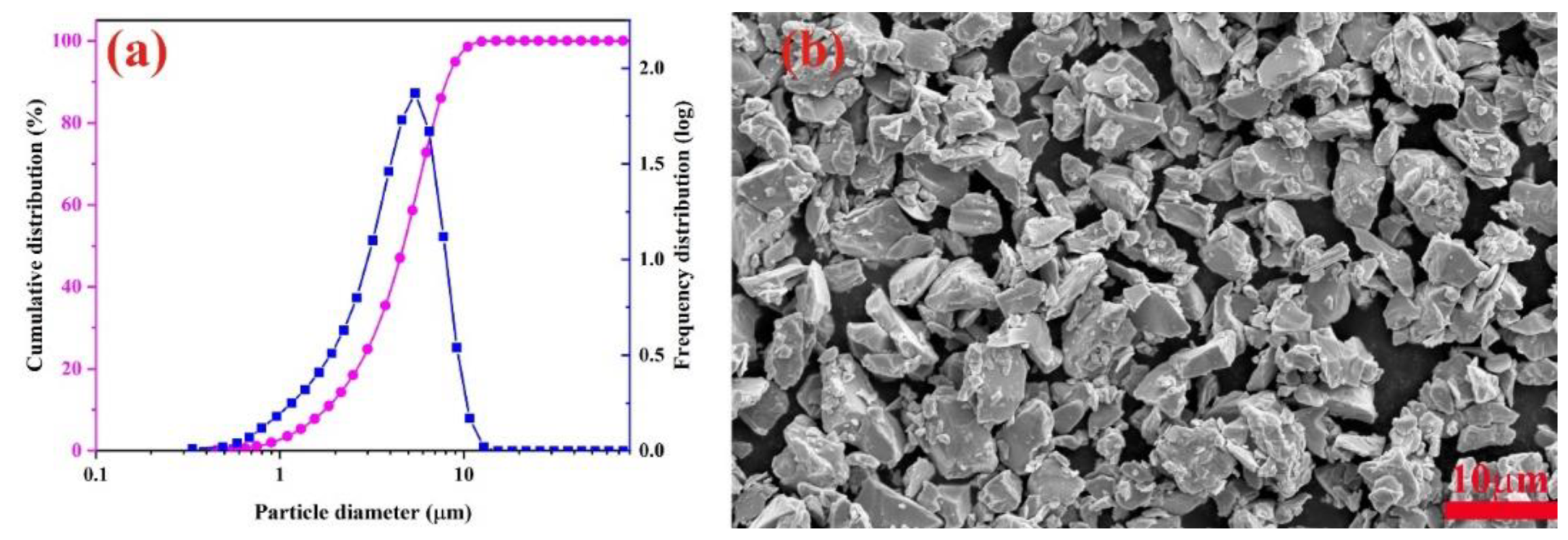

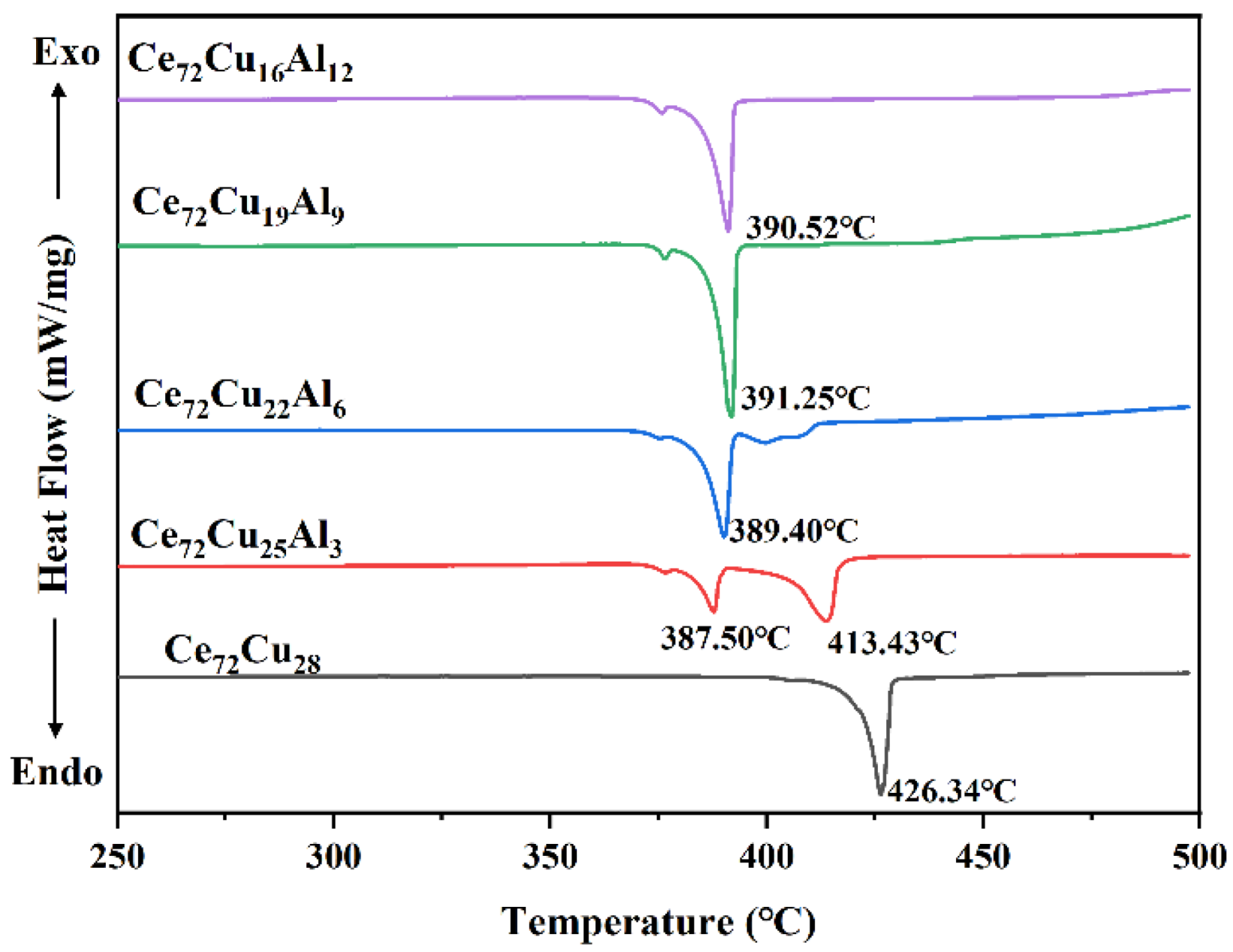
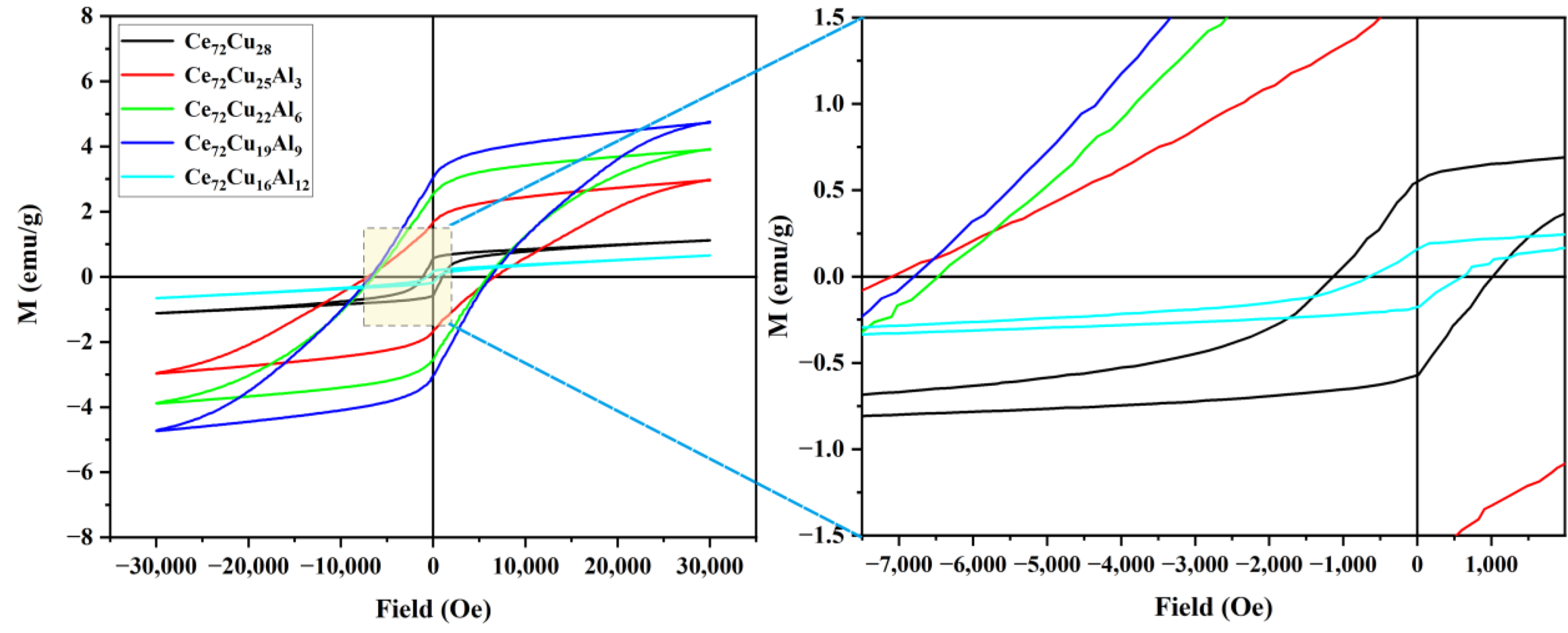
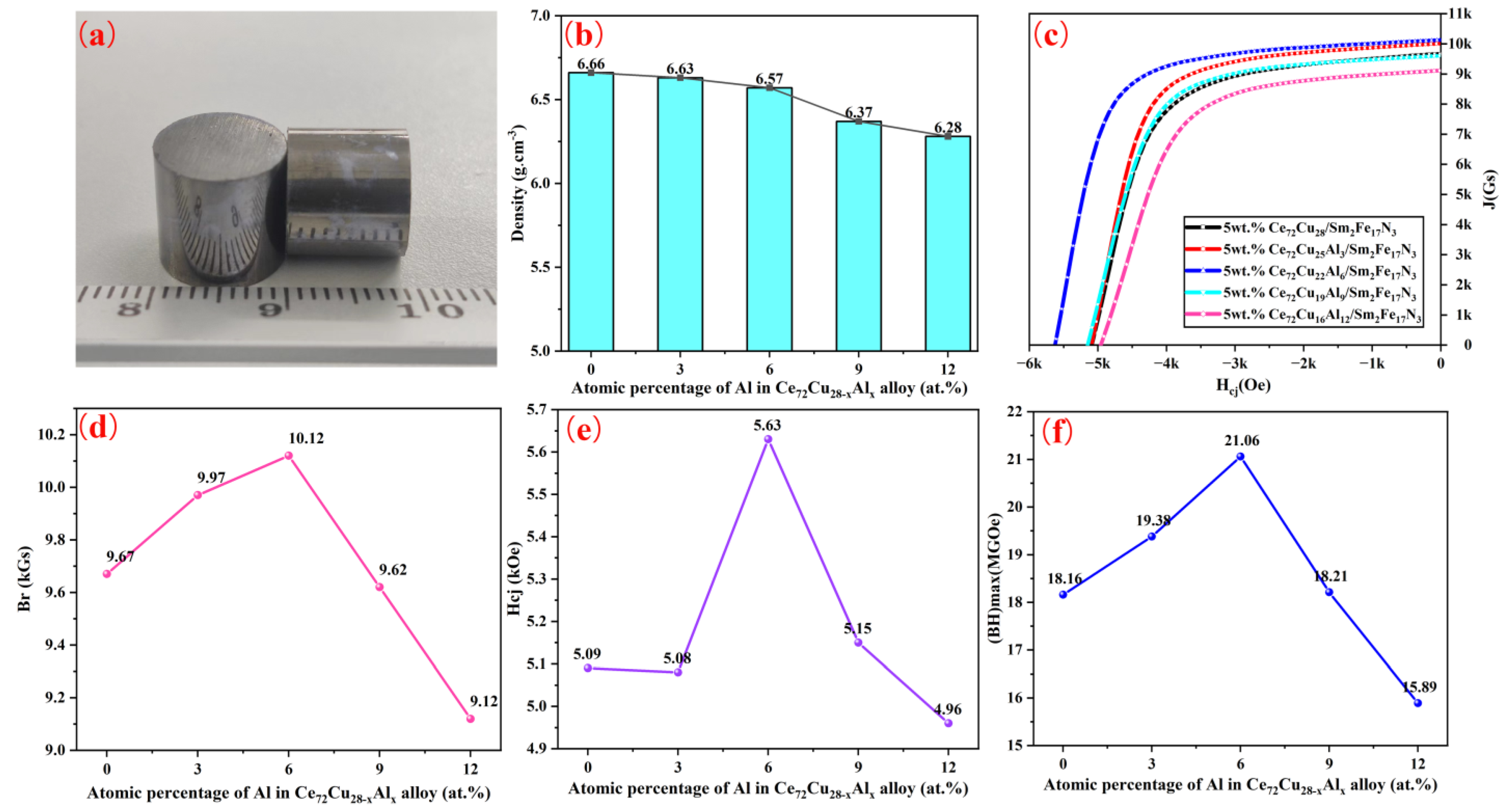



| Ce72Cu28-xAlx | x = 0 | x = 3 | x = 6 | x = 9 | x = 12 | |
|---|---|---|---|---|---|---|
| Oxygen content/ppm | quenching strips | 1319.79 | 1225.04 | 1466.27 | 1207.89 | 1194.59 |
| powders | 85,399.27 | 97,532.32 | 90,098.35 | 82,642.71 | 73,331.51 | |
| Oxygen Content of Binder (ppm) | Br (kGs) | Hcj (kOe) | (BH)max (MGOe) | Hk/Hcj (%) | |
|---|---|---|---|---|---|
| Zn | 6436.33 | 10.37 | 6.16 | 22.77 | 72.60 |
| Zn | 13,109.93 | 10.24 | 5.17 | 20.14 | 67.90 |
| Ce72Cu22Al6 | 90,098.35 | 10.12 | 5.63 | 21.06 | 74.10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, J.; Yu, S.; Huang, H.; Li, R.; Cai, W.; Chen, H.; Li, J.; Qiao, L.; Ying, Y.; Li, W.; et al. Magnetic Properties and Microstructure of Ce-Cu-Al Low Melting Alloy Bonding Sm2Fe17N3 Magnet Fabricated by the Hot-Pressing Method. Magnetochemistry 2022, 8, 149. https://doi.org/10.3390/magnetochemistry8110149
Zheng J, Yu S, Huang H, Li R, Cai W, Chen H, Li J, Qiao L, Ying Y, Li W, et al. Magnetic Properties and Microstructure of Ce-Cu-Al Low Melting Alloy Bonding Sm2Fe17N3 Magnet Fabricated by the Hot-Pressing Method. Magnetochemistry. 2022; 8(11):149. https://doi.org/10.3390/magnetochemistry8110149
Chicago/Turabian StyleZheng, Jingwu, Shitong Yu, Heng Huang, Rongyao Li, Wei Cai, Haibo Chen, Juan Li, Liang Qiao, Yao Ying, Wangchang Li, and et al. 2022. "Magnetic Properties and Microstructure of Ce-Cu-Al Low Melting Alloy Bonding Sm2Fe17N3 Magnet Fabricated by the Hot-Pressing Method" Magnetochemistry 8, no. 11: 149. https://doi.org/10.3390/magnetochemistry8110149
APA StyleZheng, J., Yu, S., Huang, H., Li, R., Cai, W., Chen, H., Li, J., Qiao, L., Ying, Y., Li, W., Yu, J., & Che, S. (2022). Magnetic Properties and Microstructure of Ce-Cu-Al Low Melting Alloy Bonding Sm2Fe17N3 Magnet Fabricated by the Hot-Pressing Method. Magnetochemistry, 8(11), 149. https://doi.org/10.3390/magnetochemistry8110149








