Abstract
Two dinuclear lanthanide complexes [Dy2(L1)6(L2)2]·2EtOH (1) and [Tb2(L1)6(L2)2]·2EtOH (2) (HL1 = phenoxyacetic acid and L2 = 2,2′-bipyridine) were synthesized and the crystal structures were determined. In both complexes, the lanthanide centers are nine-coordinated and have a muffin geometry. Detailed magnetic study reveals the presence of field-induced single molecule magnet (SMM) behavior for complex 1, whereas complex 2 is non-SMM in nature. Further magnetic study with 1′, yttrium doped magnetically diluted sample of 1, disclosed the presence of Orbach and Raman relaxation processes with effective energy barrier, ∆E = 16.26 cm−1 and relaxation time, τo = 2.42 × 10−8 s. Luminescence spectra for complexes 1 and 2 in acetonitrile were studied which show characteristic emission peaks for DyIII and TbIII ions, respectively.
1. Introduction
Limitless research interest has been involved in the investigation of single-molecule magnets (SMMs) or molecule-based magnetic materials to date. It has been claimed that this special class of materials could be explored in terms of several potential applications, such as high-density data storage, quantum computing, molecular spintronics, cryogenic magnetic refrigeration, fabrication of nanoscopic molecular devices, etc. [1,2,3,4,5,6]. The rich quantum effect of SMMs leads to tunneling of magnetization, quantum coherence, or quantum superposition at a molecular level. The unique magnetic bistability as well as blocking of magnetization are the resultants of the high spin ground state and inherent magnetic anisotropy of SMMs [7,8,9]. Now recent research trends show the prior use of lanthanides to design and construct attractive SMMs, especially for their unquenched orbital angular momentum, large spin ground state, crystal field effect, and single-ion anisotropy [10,11,12]. There are several exciting lanthanide-based SMMs that have been reported so far [13,14].
Recently, Mills and coworkers reported a dysprosocenium complex showing magnetic hysteresis up to 60 K [15]. Zheng and coworkers reported a pentagonal bipyramidal dysprosium complex with a very high effective energy barrier [16]. On the other hand, Tang and coworkers reported an equatorially coordinated erbium mononuclear SMM [17]. A dysprosium metallocene cation [(CpiPr5) Dy(Cp*)]+, having magnetic blocking temperature above 77 K, was reported by the Layfield group [18].
However, studying the magnetic behavior of a multi-nuclear lanthanide system is still a difficult job. The complexity in a lanthanide SMM arises from the single ion anisotropy as well as from the magnetic exchange interaction between the metal centers simultaneously [19]. These two important parameters always heavily affect the magnetic relaxations pathways. In this regard, smallest lanthanide clusters, namely dinuclear lanthanide SMMs, could be a suitable system to study the interference between single ion anisotropy and magnetic exchange interactions in detail [20,21,22,23,24]. In this sense, some exciting reports have been documented in the literature. Mallah et al. described how the easy axis of magnetization in a dysprosium center leads to the slow magnetic relaxation for an antiferromagnetically coupled binuclear complex [25]. Meanwhile, a systematic study has been done on the anisotropic energy barriers in a series of dinuclear dysprosium SMMs through electron-withdrawing effects by Murugesu and co-workers [26]. Winpenny and coworkers thoroughly investigated the magnetic coupling between two lanthanide centers in several dinuclear single molecule magnets in detail [27,28].
But designing a lanthanide complex for dinuclear SMMs is quite a difficult task because of the high coordination number and diverse coordination geometries of lanthanide ions. Aromatic carboxylic acids could be a suitable choice but the strong oxophilic nature of lanthanide ions generally leads to extended higher dimensional structures via different coordination modes [29,30]. So the use of ancillary ligands like 2,2′-bipyridine, 4,4′-bipyridine, 1,10-phenanthroline, or their derivatives could solve this issue by occupying the coordination sites in an efficient manner [31,32,33,34]. Additionally, such an ancillary ligand could introduce stability and fascinating luminescence properties in the resultant complexes [32].
In this regard, phenoxyacetic acid (HL1) and 2,2′-bipyridine (L2) were used to construct dinuclear dysprosium and terbium complexes and detailed study for the magnetic behavior of the complexes were performed. Furthermore, it is well known that lanthanide complexes have interesting photophysical properties which can be explored to various electronic and optical applications [35,36,37,38]. So, solution-state photoluminescence studies of the complexes were also performed.
2. Results
Two lanthanide complexes [Dy2(L1)6(L2)2]·2EtOH (1) and [Tb2(L1)6(L2)2]·2EtOH (2) were prepared by the reaction of phenoxyacetic acid (HL1), 2,2′-bipyridine (L2) and Ln-chloride salt in 3:1:1 molar ratio in ethanol at 90 °C. For the purpose of study of magnetically diluted sample with yttrium ion, complexes [Y2(L1)6(L2)2]·2EtOH (3) and [(Y0.82Dy0.18)2(L1)6(L2)2]·2EtOH (1′) were also prepared following the similar procedure. The broad band, in the range 3422–3425 cm−1, appearing in the IR spectra (vide infra) of the complexes 1–3 and 1′ can be assigned to the stretching(s) of O–H of ethanol. The strongly intense band in the range 1572–1574 cm−1 for the complexes 1–3 and 1′ arises due to the asymmetric stretching vibrations of the carboxylate moieties, whereas the strong band in the range 1429–1434 cm−1 can be assigned to the symmetric stretching vibrations of the same.
2.1. Description of Crystal Structures
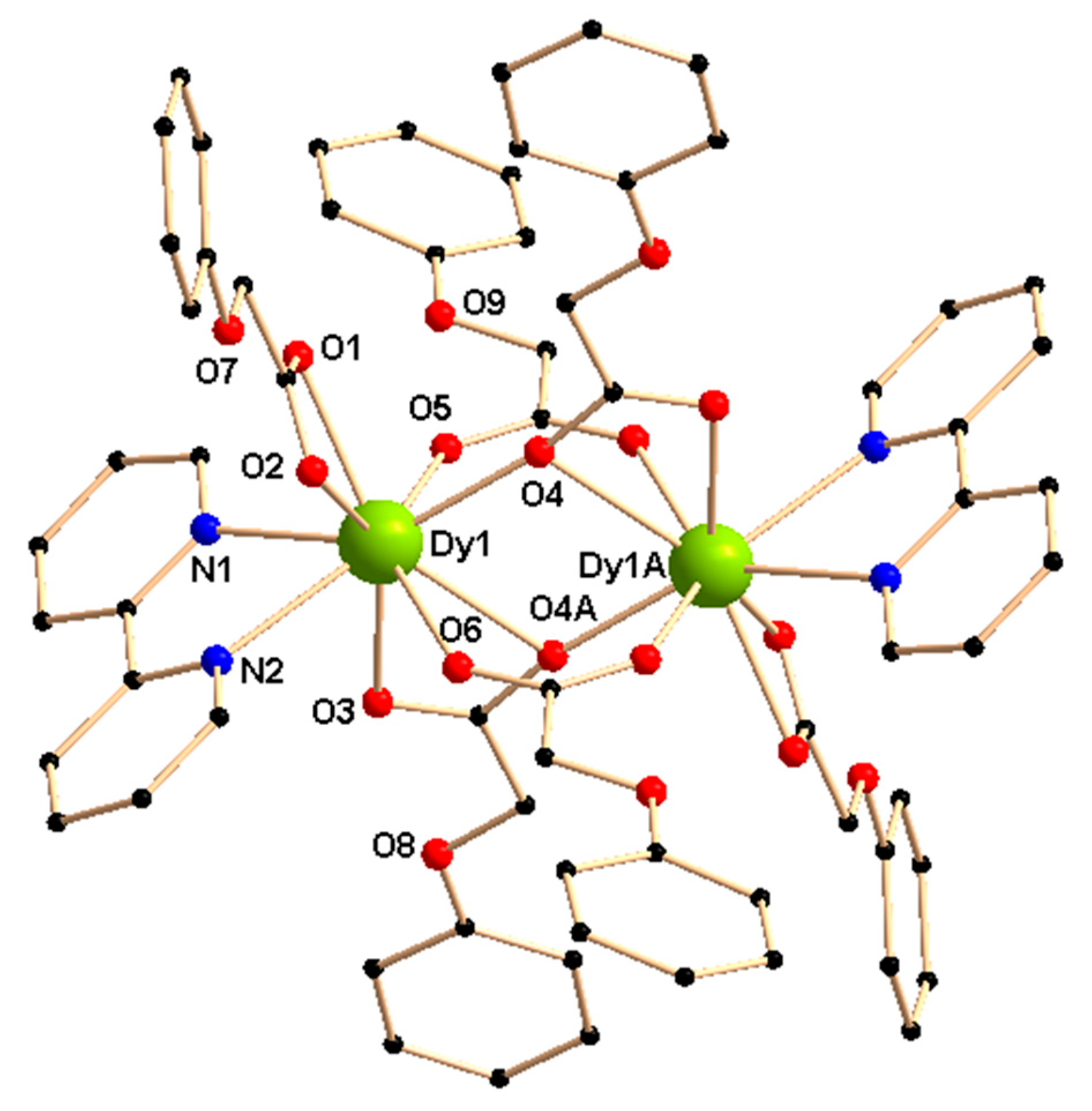
Complexes 1–3 are iso-structural and crystallize in the triclinic space group Pī (Figure 1 and Table S1). The centro-symmetric discrete dimeric structures contain nine-coordinated lanthanide/yttrium centers that adopt a muffin geometry, determined by SHAPE 2.1 [39] (Table S2). There are two lanthanide(III)/yttrium(III) centers, six phenoxyacetate ligands (L1—), and two 2,2′-bipyridine ligands (L2) present in the dinuclear structures.
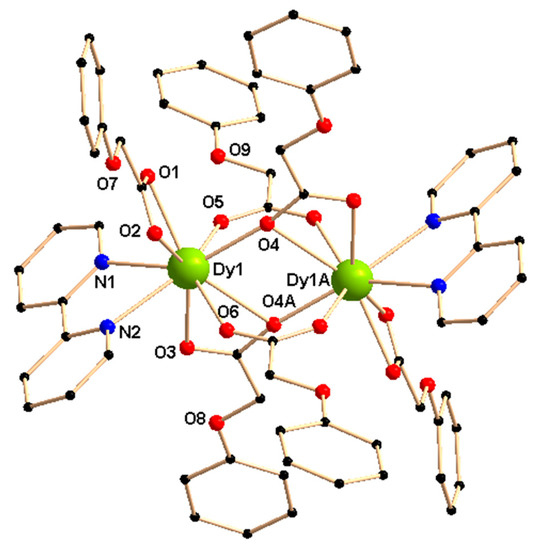
Figure 1.
Crystal structure of [Dy2(L1)6(L2)2]·2EtOH (1). Solvent ethanol molecules are deleted for clarity. Symmetry, A, –x, –y, 2–z.
Two lanthanide/yttrium centers are bridged by two L1− through μ-ɳ1: ɳ1-bidentate carboxylate bridging mode and thus two coordination positions of each center are satisfied. Two L1− having carboxylate moieties with μ-ɳ2: ɳ1-tridentate bridging modes are present in the coordination sphere of the lanthanide/yttrium centers and satisfy three coordination sites of each center. On the other hand, one L1− with ɳ2-chelating carboxylate moiety and one neutral 2,2′-bipyridine (L2) ligand fulfill the remaining four coordination positions of each lanthanide/yttrium center. Thus the +III charges of each lanthanide/yttrium center(s) in the dinuclear structure are balanced entirely by coordinated L1− ligands.
Selected bond lengths and bond angles in the coordination environment of the lanthanide/yttrium centers in 1–3 are listed in Table 1 and Table S3, respectively. The bonds involving the lanthanide/yttrium ions and μ- ɳ1: ɳ1-bidentate bridging carboxylates are shorter than those involving the chelating carboxylates: the range of LnIII/YIII—O(carboxylate) bond distances for the chelating carboxylates are 2.411–2.479 Å in 1–3, whereas for μ-ɳ1: ɳ1-bidentate bridging carboxylates the distances are within 2.307–2.337 Å. Three types of bonds, involving the lanthanide/yttrium ion(s) and μ-ɳ2: ɳ1-tridentate bridging carboxylates (Figure 1) in the complexes 1–3, lie in the range 2.415–2.443, 2.303–2.337 and 2.677–2.687 Å. On the other hand, the bond lengths between the neutral 2,2′-bipyridine ligands (L2) and the lanthanide/yttrium center(s) lie in the range of 2.507–2.569 Å in 1–3.

Table 1.
Selected bond lengths in Å for DyIII, TbIII and YIII center(s) in 1, 2 and 3, respectively. Symmetry: A, –x, –y, 2–z for 1; A, –x, 1–y, 2–z for 2 and A, –x, 1–y, –z for 3.
The intra-molecular LnIII····LnIII or YIII····YIII separation is 3.963, 3.978, and 3.976 Å for 1–3, respectively. The smallest value for intermolecular LnIII····LnIII separation is 8.125 and 8.121 Å for 1 and 2, respectively, while the equivalent distance is 8.113 Å in case of the yttrium analogue 3. π····π stacking interactions between the benzene rings of μ-ɳ2:ɳ1-tridentate bridging phenoxyacetates of two neighboring molecules are present in 1–3 and the π····π stacking distances lie in the range 3.961–3.985 Å (Figure S1). Inter-molecular π····π stacking interactions are also found between the 2,2′-bipyridine moieties of neighboring molecules with the π····π stacking distance, 3.772, 3.777, and 3.780 Å for 1–3, respectively.
The dinuclear structure in 1–3 contains two ethanol molecules as solvents of crystallization. There is a possibility of weak hydrogen bonding interaction between the hydroxyl hydrogen of the ethanol molecule with the oxygen (O1) of the chelating carboxylate. The range of D···A distance in this case is 2.883–2.996 Å for 1–3. However, this hydrogen bonding interaction does not contribute to form a supra-molecular entity.
2.2. Magnetic Properties
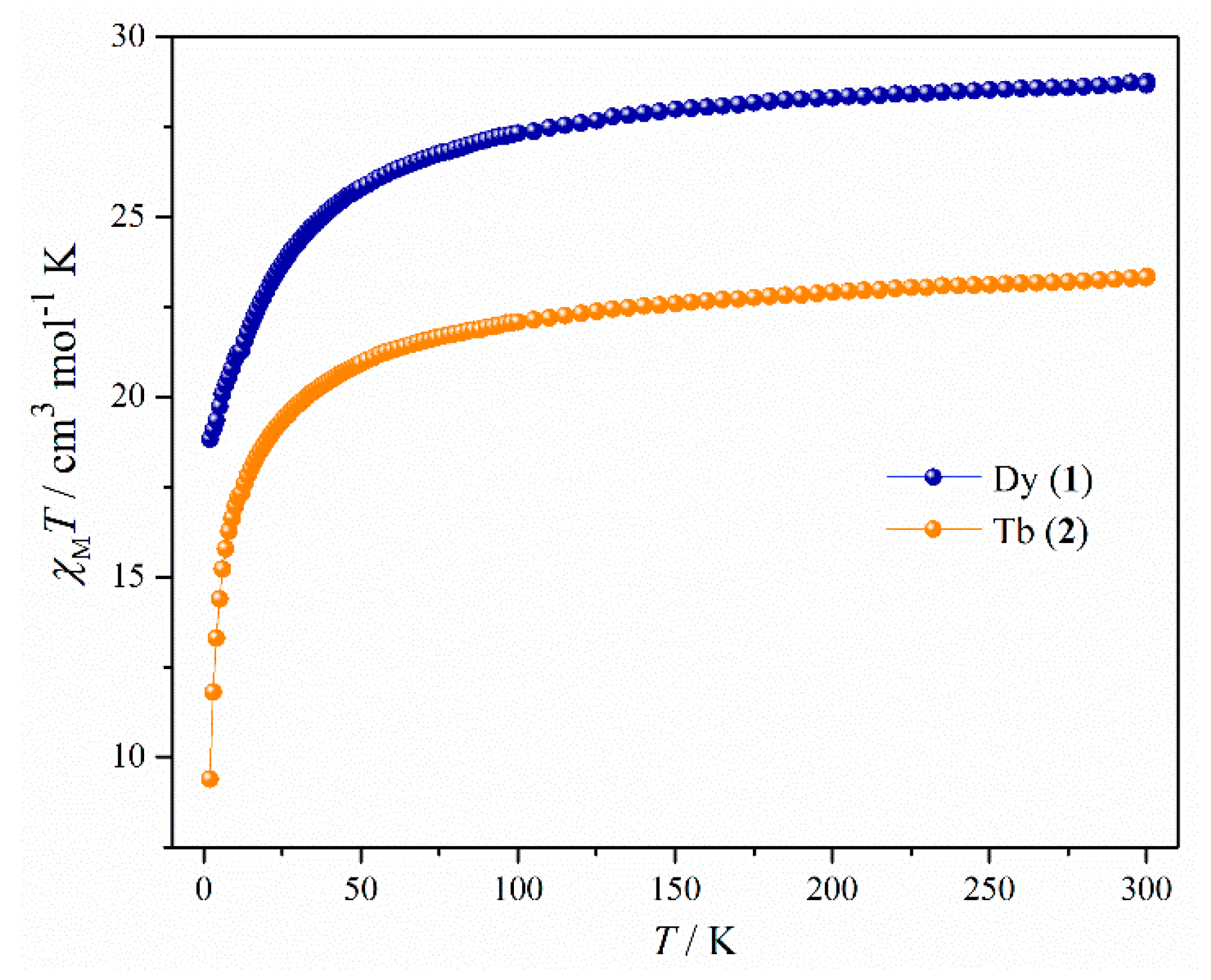
For both the complexes, detailed magnetic studies were performed on polycrystalline powdered samples. In dc (direct current) magnetic measurements, the room temperature χMT value of complex 1 was found to be 28.75 cm3 mol−1 K which is very close to the theoretical one (28.34 cm3 mol−1 K, 6H15/2, g = 4/3) for two uncoupled dysprosium ions (Figure 2). From 300 K, the χMT values gradually decreased up to around 55 K. After that a sudden decrease was observed and the χMT value reached 18.83 cm3 mol−1 K at 1.9 K. The shape of the χMT plot may not imply the presence of weak antiferromagnetic interactions between metal centers due to depopulation of stark sublevels, large orbital angular momentum, and strong spin-orbit coupling for the dysprosium ions [40,41,42]. The room temperature χMT value (23.34 cm3 mol−1 K) for complex 2 was also consistent with the theoretical one (23.63 cm3 mol−1 K, 7F6, g = 3/2) for two uncoupled terbium ions (Figure 2). A slow decrease in χMT values was observed up to around 60 K. After that χMT rapidly decreased to a value of 9.38 cm3 K mol−1 at 1.9 K. So in this case as well, the overall plot might not have indicated antiferromagnetic interaction in 2 [40,41,42]. However, while the decreasing of the χMT is mainly attributed to the depopulation of the mJ sublevels, and strong magnetic anisotropy, the presence of intra and or intermolecular antiferromagnetic interaction (exchange and/or dipolar) cannot be ruled out without magnetic dilution and/or computational results.
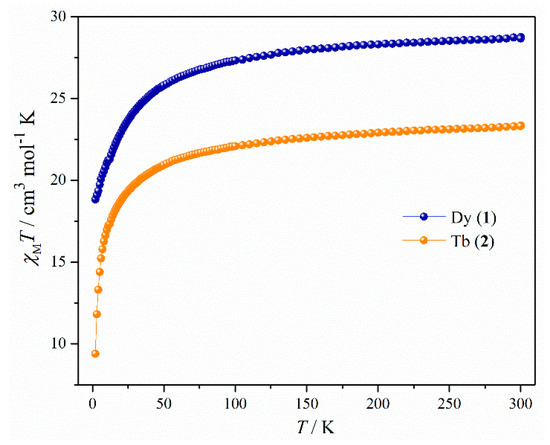
Figure 2.
Temperature dependence of the χMT products in 1000 Oe for complexes 1 and 2.
For complexes 1 and 2, the isothermal magnetization measurements were performed with the increasing magnetic field (0 Oe to 50 kOe) at 1.8 K (Figure S2). A rapid increase in the magnetization is observed at the low field region, afterwards a slow increase leads to an unsaturation even at 50 kOe. So the unsaturation in the magnetization plots surely suggests the existence of magnetic anisotropy and/or low-lying excited states for both complexes [43,44].
The ac (alternating current) magnetic measurements were performed for both the complexes to investigate their SMM properties. For complex 1, no peak maximum was observed in the out of phase ac susceptibility for a temperature range of 1.85 K–10.87 K (in presence of an oscillating ac field of 3 Oe and frequency range 1-1000 Hz) without an external dc magnetic field (Figure S3). Now the absence of peak maxima in the out of phase ac susceptibility indicates the fast zero-field quantum tunneling of magnetization (QTM) [45,46]. So to suppress the effect of QTM, ac magnetic susceptibility measurements were performed under several (0 Oe to 3000 Oe) dc magnetic fields at 1.85 K for the frequency range 0.1–1000 Hz (Figure S4). Now at two optimized dc fields, ac magnetic susceptibility measurements were performed to inspect the magnetic relaxation (Figure S5). However, the occurrence of multiple peak maxima in the out of phase ac susceptibility clearly indicated some inter- and intra-molecular exchange interactions or dipolar interactions for 1 [47,48].
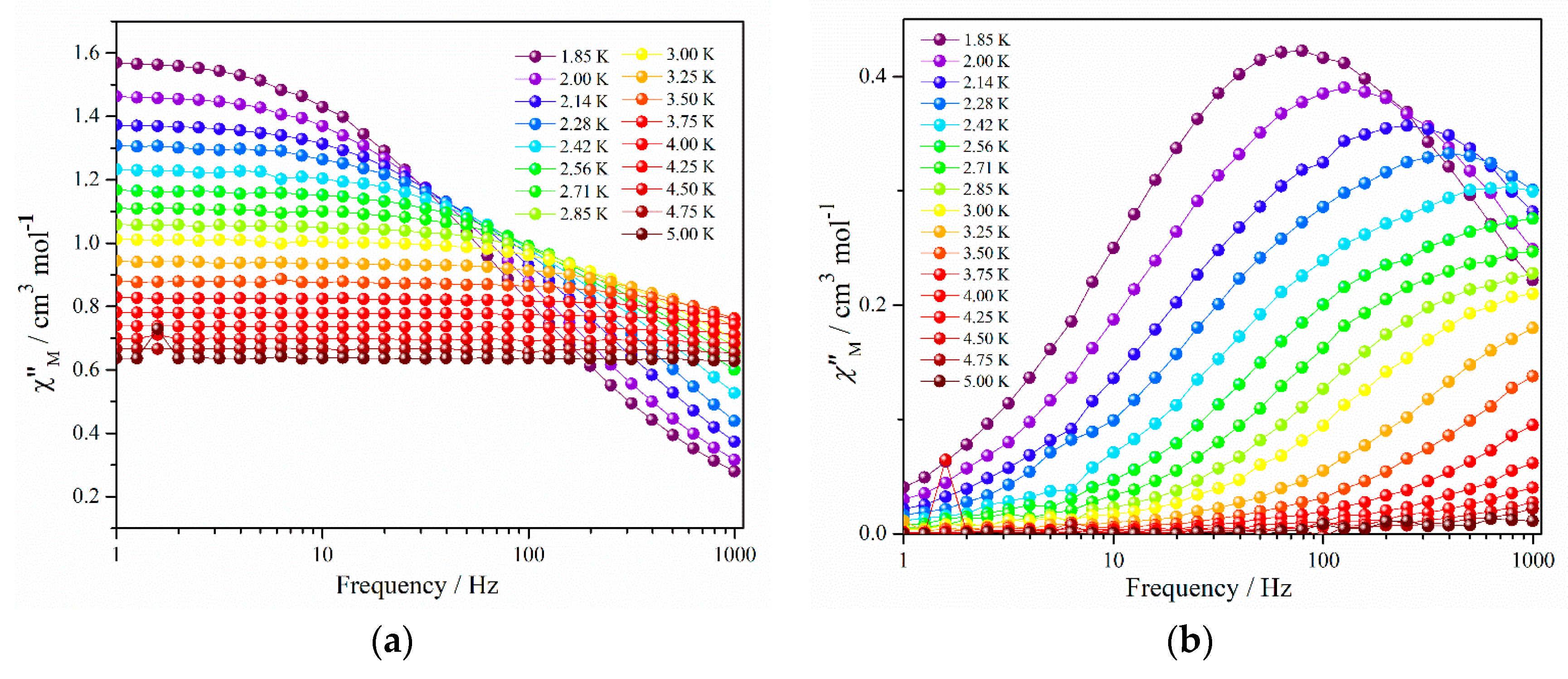
Now to minimize the effect of magnetic exchange interactions (or dipolar interactions), yttrium doped magnetically diluted sample (1′) of 1 was prepared by a molar ratio of 2:8 (Dy:Y). No peak maxima in out of phase magnetic susceptibility was observed for 1′ at zero magnetic field (Figure S6) and this suggests the effect of QTM. So an optimized field of 1000 Oe was applied to perform the ac magnetic measurements (Figure S7 and Figure 3).
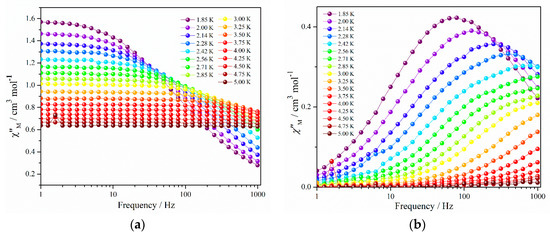
Figure 3.
Frequency dependence of the (a) in phase and (b) out of phase components of the ac magnetic susceptibility for complex 1′ under 1000 Oe dc field.
Appearance of clear frequency dependent peak maxima in the out of phase ac magnetic susceptibility confirms the SMM behavior.
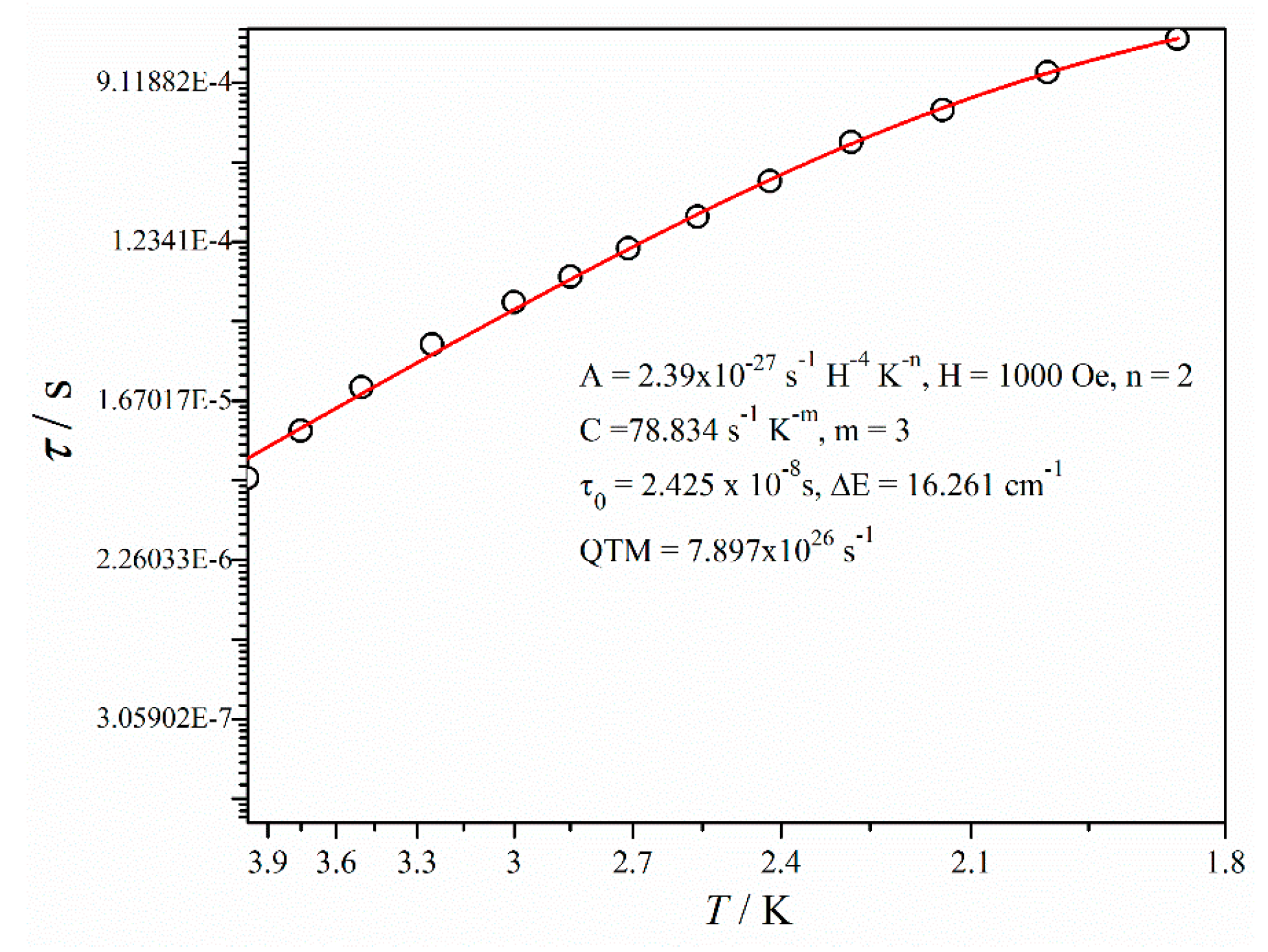
The ac susceptibility data were analyzed with Cole-Cole model for a temperature range of 1.85–4.00 K (Figure S8) [49,50]. The set of α values (0.27–0.42) directs the broad distribution of magnetic relaxation process (Table S4). To extract the energy barrier and relaxation time, the following Equation (1) was used:
where, the first and second term signifies the direct and Raman process whereas the third and fourth term denotes Orbach relaxation and QTM respectively [12,51,52]. The overall magnetic relaxation follows a combination of Raman and Orbach processes here. The m value was fixed to be 3 for the best fitting. Generally, the expected value of m is 9 for a Kramers ion like dysprosium, but values between 2 and 7 are acceptable for acoustic and optical phonon Raman [53,54]. The extracted anisotropic energy barrier is ΔE = 16.261 cm−1 with the relaxation time (τ0) = 2.425 × 10−8 s (Figure 4 and Table S5).
τ−1 = AH4Tn + CTm + τ0−1exp(−∆E/kBT) + 1/QTM
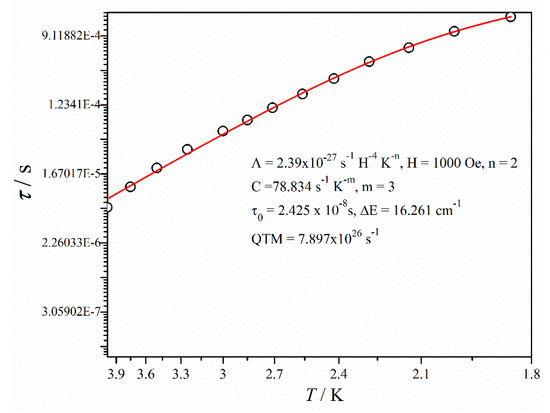
Figure 4.
Temperature dependence of relaxation time for complex 1′.
In case of complex 1, we performed the ac magnetic susceptibility measurements for a higher frequency range of 10–10,000 Hz (Figures S9 and S10). Clear peak maxima were observed in out of phase ac susceptibility for an optimized field of 600 Oe (Figure S10). This observation confirms the presence of slow magnetic relaxation at an optimized field of higher frequency as well.
For the complex 2 (having a non-Kramers ion, Tb3+), frequency dependency was not observed in the out of phase ac susceptibilities under the zero field or in several applied dc magnetic fields (Figures S11 and S12). So for terbium (a non-Kramers ion) based complex 2, the integer J ground states were split by the transverse anisotropy. As a result of under-barrier magnetic relaxation mechanism (or spin-parity), non-SMM behavior was observed [40,55].
2.3. Photophysical Properties
The absorption spectra of complexes 1 and 2 and the free ligands, HL1 and L2 were studied in acetonitrile at room temperature and the spectral data are listed in Table S6. The presence of absorption band at 277 nm (ε = 31,776 M−1 cm−1 for complex 1 and ε = 34,031 M−1 cm−1 for complex 2) for both the complexes may be attributed to the π→π* transition of the ligands. On the other hand, the absorption spectra of free HL1 and L2 in acetonitrile show bands of lower absorption intensity than that of the complexes (λmax = 276 nm; ε = 1224 M−1 cm−1 for HL1 and λmax = 281 nm; ε = 14,785 M−1 cm−1 for L2). This could be due to the coordination interaction between the lanthanide ions with the ligands to form a more extensive π→π* conjugated system [56,57].
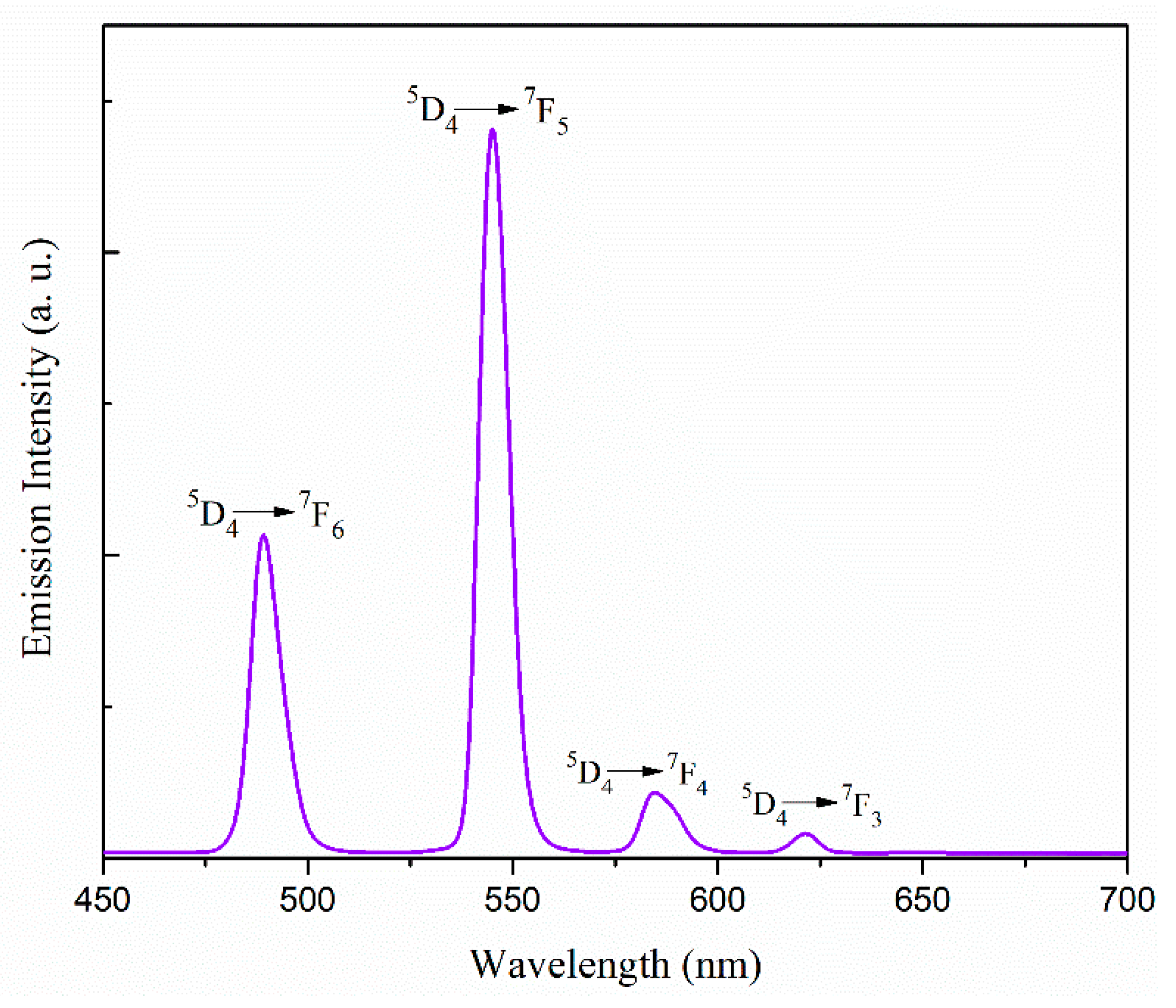
The emission spectra in acetonitrile were obtained at room temperature under the excitation wavelength of 300 nm for complex 1 and 277 nm for complex 2. 1 × 10−4 (M) solution of the complexes were used for this purpose. The luminescence spectra of complexes 1 and 2 are shown in Figure S13 and Figure 5, respectively. For complex 1 two prominent peaks appear at 480 and 573 nm, which can be assigned to the 4F9/2 → 6H15/2 and 4F9/2 → 6H13/2 transitions of the DyIII ion [58,59,60]. On the other hand, there are four emission peaks at 489, 545, 585, and 621 nm in the luminescence spectrum of complex 2. These emission bands correspond to the transition between the first excited state and the ground state multiplets of the TbIII ion, namely 5D4 → 7F6 (489 nm), 5D4 → 7F5 (545 nm), 5D4 → 7F4 (585 nm), and 5D4 → 7F3 (621 nm) transitions [59,60,61,62,63]. The strongest emission at 545 nm, due to 5D4 → 7F5 transition, is the characteristic transition in a terbium containing complex.
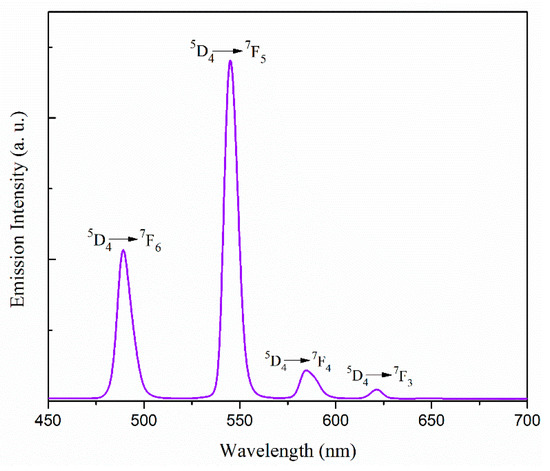
Figure 5.
Emission spectrum of complex 2 in acetonitrile at room temperature.
It is evident from the above results that the ligands are able to serve as antenna for the DyIII and TbIII ions, i.e., the ligands can absorb energy and transfer it to the lanthanide ions. Such intra-molecular energy transfer leads to the characteristic emission of the DyIII and TbIII ions.
3. Conclusions
In this work, two new lanthanide based dinuclear complexes were synthesized and characterized in detail. Here the synthetic strategy was adopted through the using of a chelating carboxylate ligand with the ancillary ligand, 2,2′-bipyridine. Field induced single molecule magnetic behavior is observed for the dysprosium based complex. Furthermore, the magnetic dilution study reveals the magnetic relaxation is happening through a combined process of Raman and Orbach mechanisms. On the other hand, in the solution-state luminescence spectrum, both dysprosium and terbium complexes show characteristic emission peaks for DyIII and TbIII ions, respectively. The dinucelar lanthanide complexes with carboxylato ligands and 2,2′-bipyridine are able to exhibit different interesting properties at the molecular level, finding potential applications in the field of molecular magnetism, coordination chemistry, and molecular spintronics research.
4. Materials and Methods
All reagents and solvents were purchased from commercial sources and used as received. Elemental analyses (C, H and N) were performed at the Research and Analytical Center for Giant Molecules, Tohoku University. IR spectra of the samples were acquired at room temperature with a JASCO FT/IR-4200 spectrophotometer in ATR mode. Magnetic susceptibility measurements were conducted using a Quantum Design SQUID magnetometer MPMS-XL (Quantum Design, San Diego, CA, USA). AC measurements were performed with an ac field amplitude of 3 Oe. High frequency ac data were collected by Quantum Design PPMS MODEL 6000. A polycrystalline sample embedded in n-eicosane was used for the measurements. Absorption spectra of the complexes were measured using a Shimadzu UV-3100 PC UV-VIS-NIR scanning spectrophotometer and the luminescence spectra in acetonitrile were measured using a JASCO FP-8300 spectrofluorometer.
The crystallographic data of 1–3 (CCDC 1921591–1921593) are summarized in Table S1. Diffraction data were collected on a Rigaku Saturn 724+ CCD diffractometer with graphite monochromated Mo-Kα radiation (λ = 0.71073 Å). The data collection temperature was 120 K for 1–3. Data processing was performed using the CrysAlisPro [64] crystallographic software package for 1 and CrystalClear [65] crystallographic software package was used for 2 and 3. All the structures were solved by SIR-92 [66] using direct methods and the structures were refined by full-matrix least-squares based on F2 using SHELXL-2014/7 [67,68] packages. C35 and C36 atoms of the solvent ethanol molecule were disordered over two sites in each of the complexes, 1–3. The disorder was fixed, allowing each individual atom to refine freely and the final occupancy parameters were set as 0.6 and 0.4 for both C35 and C36, respectively in both of the complexes 1 and 3; the same parameters were set to 0.5 and 0.5 for both C35 and C36 in complex 2. The final refinements converged at the R1 values [I > 2σ(I)] 0.0804, 0.0383 and 0.0505 for 1–3, respectively.
Synthesis of [Dy2(L1)6(L2)2]·2EtOH (1): 0.6 mmol (0.091 g) of phenoxyacetic acid (HL1) and 0.2 mmol (0.031 g) of 2,2′-bipyridine (L2) were dissolved in 20 mL of ethanol and the pH of the solution was adjusted to 5–7 by 1 (M) aqueous NaOH solution. Then 5 mL of water/ethanol (1:4) solution of 0.2 mmol (0.075 g) DyCl3·6H2O was added to the colorless mixed ligand solution and mixture was refluxed at 90 °C for 4 h in a round bottomed flask. The resulting colorless solution was then filtered to eliminate any suspended particles and the filtrate was kept at room temperature for slow evaporation. After 3–4 days, diffraction quality single crystals were collected by filtration and washed with cold ethanol. Yield (based on Dy): 0.075 g (46%). Anal. calcd for C72H70N4O20Dy2: C, 52.85; H, 4.31; N, 3.42%. Found: C, 52.75; H, 4.32; N, 3.48%. IR (cm−1): 3425(w), 2928(w), 1636(s), 1594(s), 1574(s), 1493(m), 1463(m), 1451(m), 1429(s), 1334(m), 1243(s), 1171(m), 1082(m), 1060(s), 1014(m), 840(m), 752(s), 689(s), and 640(m).
Synthesis of [Tb2(L1)6(L2)2]·2EtOH (2): This compound was prepared following a similar procedure to that described for 1 except for using TbCl3∙6H2O (0.075 g, 0.2 mmol) instead of DyCl3∙6H2O. Yield (based on Tb): 0.073g (45%). Anal. calcd for C72H70N4O20Tb2: C, 53.08; H, 4.33; N, 3.44%. Found: C, 53.11; H, 4.38; N, 3.38%. IR (cm–1): 3424(w), 2918(w), 1633(s), 1595(s), 1574(s), 1493(m), 1461(m), 1454(m), 1431(s), 1334(m), 1236(s), 1170(m), 1081(m), 1064(s), 1009(m), 839(m), 750(s), 688(s), and 638(m).
Synthesis of [Y2(L1)6(L2)2]·2EtOH (3): This compound was prepared following a similar procedure to that described for 1 except for using YCl3∙6H2O (0.061 g, 0.2 mmol) instead of DyCl3∙6H2O. Yield (based on Y): 0.073 g (49%). Anal. calcd for C72H70N4O20Y2: C, 58.07; H, 4.74; N, 3.76%. Found: C, 57.92; H, 4.73; N, 3.87%. IR (cm–1): 3422(w), 2915(w), 1636(s), 1596(s), 1572(m), 1495(m), 1465(m), 1452(m), 1432(s), 1336(m), 1242(s), 1172(m), 1082(m), 1065(s), 1014(m), 840(m), 749(s), 688(s), and 644(m).
Synthesis of [(Y0.82Dy0.18)2(L1)6(L2)2]·2EtOH (1′): This compound was prepared following a similar procedure to that described for 1 except for using YCl3∙6H2O (0.048 g, 0.16 mmol) and DyCl3∙6H2O (0.015 g, 0.04 mmol) instead of pure DyCl3∙6H2O. The ratio of YIII and DyIII ions from elemental analysis and χMT vs. T plot. (Figure S14). Yield 0.071 g (47%). Anal. calcd for C72H70N4O20Y1.64Dy0.36: C, 57.06; H, 4.65; N, 3.70%. Found: C, 57.02; H, 4.64; N, 3.83%. IR (cm−1): 3424(w), 2915(w), 1638(s), 1595(s), 1573(s), 1493(m), 1461(m), 1452(m), 1434(s), 1335(m), 1240(s), 1171(m), 1081(m), 1063(s), 1012(m), 838(m), 752(s), 689(s), and 643(m).
Supplementary Materials
The following are available online at https://www.mdpi.com/2312-7481/5/4/56/s1, Figure S1: Perspective view of two dimensional sheet caused by intermolecular π···π stacking interactions in crystallographic bc plane for complex 1, Figure S2. Magnetization (M) vs. Field (H) plots at 1.8 K, Figure S3. Frequency and temperature dependency without dc field for complex 1, Figure S4. Frequency dependency of the (a) in phase and (b) out of phase ac susceptibility under indicated dc fields at 1.85K for 1, Figure S5. Frequency dependence of the (a) in phase and (b) out of phase components of the ac magnetic susceptibility for 1 under 1000 Oe dc field and frequency dependence of the (c) in phase and (d) out of phase components of the ac magnetic susceptibility for 1 under 2400 Oe dc field, Figure S6. Frequency and temperature dependency without dc field for complex 1′, Figure S7. Frequency dependency of the (a) in phase and (b) out of phase ac susceptibility under indicated dc fields at 1.85 K for 1′, Figure S8. Cole-Cole plots for 1′ at different temperatures, Figure S9. Frequency dependency of the (a) in phase and (b) out of phase ac susceptibility under indicated dc fields at 1.9 K (the indication for the fields for both plots are shown only in (b) for clarity) for 1 in the high frequency region, Figure S10. Frequency dependency of the (a) in-phase and (b) out-of-phase components of the ac magnetic susceptibility under 600 Oe dc field for 1 in the high frequency region, Figure S11. Frequency and temperature dependency without dc field for complex 2, Figure S12. Frequency dependency of the (a) in phase and (b) out of phase ac susceptibility under indicated dc fields at 1.85 K for 2 (the indication for the fields for both plots are shown only in (b) for clarity), Figure S13. Emission spectrum of complex 1 in acetonitrile at room temperature, Figure S14. Temperature dependence of the χMT products in 1000 Oe for 1′, Table S1. Crystallographic data for 1–3, Table S2. Summary of SHAPE analysis around LnIII center(s) for 1 and 2 and YIII center(s) in 3 (Ln = Dy for 1, Ln = Tb for 2), Table S3. Selected bond angles in ⁰ for DyIII, TbIII and YIII center(s) in 1, 2 and 3, respectively. Symmetry: A, –x, –y, 2–z for 1; A, –x, 1–y, 2–z for 2 and A, –x, 1–y, –z for 3, Table S4. Parameters obtained from Cole-Cole fitting for 1′, Table S5. List of parameters related to the magnetic properties of 1′, Table S6. Spectral parameters of the absorption spectra for complexes 1 and 2. (CCDC 1921591–1921593) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre.
Author Contributions
L.M. designed the project and performed all the synthetic experiments; L.M. solved the crystal structures and analyzed the fluorescence data; L.M. and S.B. collected and analyzed the magnetic data; L.M. and S.B. wrote the paper; M.Y. supervised the overall project.
Funding
This work was partially supported by CREST (JPMJCR12L3), JST. M. Yamashita thanks the supports by 111 project (B18030) from China. S.B. thanks JSPS (P17038) for financial support.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Bogani, L.; Wernsdorfer, W. Molecular spintronics using single-molecule magnets. Nature Mater. 2008, 7, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Domingo, N.; Bellido, E.; Ruiz-Molina, D. Advances on structuring, integration and magnetic characterization of molecular nanomagnets on surfaces and devices. Chem. Soc. Rev. 2012, 41, 258–302. [Google Scholar] [CrossRef] [PubMed]
- Evangelisti, M.; Roubeau, O.; Palacios, E.; Camón, A.; Hooper, T.N.; Brechin, E.K.; Alonso, J.J. Cryogenic Magnetocaloric Effect in a Ferromagnetic Molecular Dimer. Angew. Chem. Int. Ed. 2011, 50, 6606–6609. [Google Scholar] [CrossRef] [PubMed]
- Aromí, G.; Aguilá, D.; Gamez, P.; Luis, F.; Roubeau, O. Design of magnetic coordination complexes for quantum computing. Chem. Soc. Rev. 2012, 41, 537–546. [Google Scholar] [CrossRef]
- Candini, A.; Klyatskaya, S.; Ruben, M.; Wernsdorfer, W.; Affronte, M. Graphene Spintronic Devices with Molecular Nanomagnets. Nano Lett. 2011, 11, 2634–2639. [Google Scholar] [CrossRef]
- Biswas, S.; Mondal, A.K.; Konar, S. Densely Packed Lanthanide Cubane Based 3D Metal−Organic Frameworks for Efficient Magnetic Refrigeration and Slow Magnetic Relaxation. Inorg. Chem. 2016, 55, 2085–2090. [Google Scholar] [CrossRef]
- Ishikawa, N.; Sugita, M.; Ishikawa, T.; Koshihara, S.-Y.; Kaizu, Y. Lanthanide Double-Decker Complexes Functioning as Magnets at the Single-Molecular Level. J. Am. Chem. Soc. 2003, 125, 8694–8695. [Google Scholar] [CrossRef]
- Woodruff, D.N.; Winpenny, R.E.P.; Layfield, R.A. Lanthanide Single-Molecule Magnets. Chem. Rev. 2013, 113, 5110–5148. [Google Scholar] [CrossRef]
- Giusti, A.; Charron, G.; Mazerat, S.; Compain, J.-D.; Mialane, P.; Dolbecq, A.; Rivière, E.; Wernsdorfer, W.; Biboum, R.N.; Keita, B.; et al. Magnetic Bistability of Individual Single-Molecule Magnets Grafted on Single-Wall Carbon Nanotubes. Angew. Chem. Int. Ed. 2009, 48, 4949–4952. [Google Scholar] [CrossRef]
- Sorace, L.; Benelli, C.; Gatteschi, D. Lanthanides in molecular magnetism: old tools in a new field. Chem. Soc. Rev. 2011, 40, 3092–3104. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, X.; Meng, X.; Shi, W.; Cheng, P.; Powell, A.K. Constraining the coordination geometries of lanthanide centers and magnetic building blocks in frameworks: A new strategy for molecular nanomagnets. Chem. Soc. Rev. 2016, 45, 2423–2439. [Google Scholar] [CrossRef] [PubMed]
- Liddle, S.T.; Slageren, J.V. Improving f-element single molecule magnets. Chem. Soc. Rev. 2015, 44, 6655–6669. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Guo, Y.-N.; Tang, J. Recent advances in dysprosium-based single molecule magnets: Structural overview and synthetic strategies. Coord. Chem. Rev. 2013, 257, 1728–1763. [Google Scholar] [CrossRef]
- Tian, H.; Wang, M.; Zhao, L.; Guo, Y.-N.; Guo, Y.; Tang, J.; Liu, Z. A Discrete Dysprosium Trigonal Prism Showing Single-Molecule Magnet Behaviour. Chem. Eur. J. 2012, 18, 442–445. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, C.A.P.; Ortu, F.; Reta, D.; Chilton, N.F.; Mills, D.P. Molecular magnetic hysteresis at 60 kelvin in dysprosocenium. Nature 2017, 548, 439–442. [Google Scholar] [CrossRef]
- Ding, Y.-S.; Chilton, N.F.; Winpenny, R.E.P.; Zheng, Y.-Z. On Approaching the Limit of Molecular Magnetic Anisotropy: A Near-Perfect Pentagonal Bipyramidal Dysprosium (III) Single-Molecule Magnet. Angew. Chem. Int. Ed. 2016, 55, 16071–16074. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, L.; Wang, C.; Xue, S.; Lin, S.-Y.; Tang, J. Equatorially Coordinated Lanthanide Single Ion Magnets. J. Am. Chem. Soc. 2014, 136, 4484–4487. [Google Scholar] [CrossRef]
- Guo, F.-S.; Day, B.M.; Chen, Y.-C.; Tong, M.-L.; Mansikkamäki, A.; Layfield, R.A. Magnetic hysteresis up to 80 kelvin in a dysprosium metallocene single-molecule magnet. Science 2018, 362, 1400–1403. [Google Scholar] [CrossRef]
- Guo, M.; Xu, Y.; Wu, J.; Zhao, L.; Tang, J. Geometry and magnetic interaction modulations in dinuclear Dy2 single-molecule magnets. Dalton Trans. 2017, 46, 8252–8258. [Google Scholar] [CrossRef]
- Habib, F.; Murugesu, M. Lessons learned from dinuclear lanthanide nano-magnets. Chem. Soc. Rev. 2013, 42, 3278–3288. [Google Scholar] [CrossRef]
- Ghosh, S.; Mandal, S.; Singh, M.K.; Liu, C.-M.; Rajaraman, G.; Mohanta, S. Experimental and theoretical exploration of magnetic exchange interactions and single-molecule magnetic behaviour of bis(η1:η2:μ2-carboxylate)GdIII2 /DyIII2 systems. Dalton Trans. 2018, 47, 11455–11469. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Bernot, K.; Pointillart, F.; Poneti, G.; Calvez, G.; Daiguebonne, C.; Guillou, O.; Sessoli, R. A Luminescent and Sublimable DyIII-Based Single-Molecule Magnet. Chem. Eur. J. 2012, 18, 11379–11387. [Google Scholar] [CrossRef] [PubMed]
- Pointillart, F.; Gal, Y.L.; Golhen, S.; Cador, O.; Ouahab, L. Single-Molecule Magnet Behaviour in a Tetrathiafulvalene-Based Electroactive Antiferromagnetically Coupled Dinuclear Dysprosium (III) Complex. Chem. Eur. J. 2011, 17, 10397–10404. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.-N.; Xu, G.-F.; Wernsdorfer, W.; Ungur, L.; Guo, Y.; Tang, J.; Zhang, H.-J.; Chibotaru, L.F.; Powell, A.K. Strong Axiality and Ising Exchange Interaction Suppress Zero-Field Tunneling of Magnetization of an Asymmetric Dy2 Single-Molecule Magnet. J. Am. Chem. Soc. 2011, 133, 11948–11951. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.Y.; Bolvin, H.; Campbell, V.E.; Guillot, R.; Kampf, J.W.; Wernsdorfer, W.; Gendron, F.; Autschbach, J.; Pecoraro, V.L.; Mallah, T. Assessing the exchange coupling in binuclear lanthanide(III) complexes and the slow relaxation of the magnetization in the antiferromagnetically coupled Dy2 derivative. Chem. Sci. 2015, 6, 4148–4159. [Google Scholar] [CrossRef] [PubMed]
- Habib, F.; Brunet, G.; Vieru, V.; Korobkov, I.; Chibotaru, L.F.; Murugesu, M. Significant enhancement of energy barriers in dinuclear dysprosium single-molecule magnets through electron-withdrawing effects. J. Am. Chem. Soc. 2013, 135, 13242–13245. [Google Scholar] [CrossRef]
- Pineda, E.M.; Chilton, N.F.; Marx, R.; Dörfel, M.; Sells, D.O.; Neugebauer, P.; Jiang, S.-D.; Collison, D.; Slageren, J.V.; McInnes, E.J.L.; et al. Direct measurement of dysprosium(III)·dysprosium(III) interactions in a single-molecule magnet. Nat. Commun. 2014, 5, 5243. [Google Scholar] [CrossRef]
- Giansiracusa, M.J.; Moreno-Pineda, E.; Hussain, R.; Marx, R.; Prada, M.M.; Neugebauer, P.; Al-Badran, S.; Collison, D.; Tuna, F.; Slageren, J.V.; et al. Measurement of Magnetic Exchange in Asymmetric Lanthanide Dimetallics: Toward a Transferable Theoretical Framework. J. Am. Chem. Soc. 2018, 140, 2504–2513. [Google Scholar] [CrossRef]
- Ji, B.; Deng, D.; He, X.; Liu, B.; Miao, S.; Ma, N.; Wang, W.; Ji, L.; Liu, P.; Li, X. Syntheses, Structures, Luminescence, and Magnetic Properties of One-dimensional Lanthanide Coordination Polymers with a Rigid 2,2′-Bipyridine-3,3′,6,6′-tetracarboxylic Acid Ligand. Inorg. Chem. 2012, 51, 2170–2177. [Google Scholar] [CrossRef]
- Ye, J.-W.; Wang, J.; Zhang, J.-Y.; Zhang, P.; Wang, Y. Construction of 2-D lanthanide coordination frameworks: syntheses, structures and luminescent property. CrystEngComm 2007, 9, 515–523. [Google Scholar] [CrossRef]
- Lu, Y.-B.; Jiang, X.-M.; Zhu, S.-D.; Du, Z.-Y.; Liu, C.-M.; Xie, Y.-R.; Liu, L.-X. Anion Effects on Lanthanide (III) Tetrazole-1-acetate Dinuclear Complexes Showing Slow Magnetic Relaxation and Photofluorescent Emission. Inorg. Chem. 2016, 55, 3738–3749. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.-M.; Ren, N.; Zhang, J.-J. Lanthanide complexes with 3-methoxybenzoic acid and 5,5′-dimethyl-2,2′-bipyridine: Crystal structures, luminescence and magnetic property. Inorg. Chim. Acta 2018, 480, 140–148. [Google Scholar] [CrossRef]
- Armelao, L.; Dell’Amico, D.B.; Bellucci, L.; Bottaro, G.; Ciattini, S.; Labella, L.; Manfroni, G.; Marchetti, F.; Mattei, C.A.; Samaritani, S. Homodinuclear Lanthanide Complexes with the Divergent Heterotopic 4,4′-Bipyridine N-Oxide (bipyMO) Ligand. Eur. J. Inorg. Chem. 2018, 4421–4428. [Google Scholar] [CrossRef]
- Chen, G.-J.; Guo, Y.-N.; Tian, J.-L.; Tang, J.; Gu, W.; Liu, X.; Yan, S.-P.; Cheng, P.; Liao, D.-Z. Enhancing Anisotropy Barriers of Dysprosium (III) Single-Ion Magnets. Chem. Eur. J. 2012, 18, 2484–2487. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.-H.; Li, Q.-W.; Chen, Y.-C.; Liu, J.-L.; Tong, M.-L. Luminescent single-molecule magnets based on lanthanides: Design strategies, recent advances and magneto-luminescent studies. Coord. Chem. Rev. 2019, 378, 365–381. [Google Scholar] [CrossRef]
- Long, J.; Guari, Y.; Ferreira, R.A.S.; Carlos, L.D.; Larionova, J. Recent advances in luminescent lanthanide based Single-Molecule Magnets. Coord. Chem. Rev. 2018, 363, 57–70. [Google Scholar] [CrossRef]
- Wu, D.-F.; Liu, Z.; Ren, P.; Liu, X.-H.; Wang, N.; Cui, J.-Z.; Gao, H.-L. A new family of dinuclear lanthanide complexes constructed from an 8-hydroxyquinoline Schiff base and β-diketone: Magnetic properties and near-infrared luminescence. Dalton Trans. 2019, 48, 1392–1403. [Google Scholar] [CrossRef]
- Biswas, S.; Jena, H.S.; Goswami, S.; Sanda, S.; Konar, S. Synthesis and Characterization of Two Lanthanide (Gd3+ and Dy3+)-Based Three-Dimensional Metal Organic Frameworks with Squashed Metallomacrocycle Type Building Blocks and Their Magnetic, Sorption, and Fluorescence Properties Study. Cryst. Growth Des. 2014, 14, 1287–1295. [Google Scholar] [CrossRef]
- Alvarez, S.; Alemany, P.; Casanova, D.; Cirera, J.; Llunell, M.; Avnir, D. Shape maps and polyhedral interconversion paths in transition metal chemistry. Coord. Chem. Rev. 2005, 249, 1693–1708. [Google Scholar] [CrossRef]
- Biswas, S.; Mandal, L.; Shen, Y.; Yamashita, M. Exploration of SMM behavior of Ln2-complexes derived from thianaphthene-2-carboxylic acid. Dalton Trans. 2019, 48, 14096–14102. [Google Scholar] [CrossRef]
- Biswas, S.; Jena, H.S.; Sanda, S.; Konar, S. Proton-Conducting Magnetic Coordination Polymers. Chem. Eur. J. 2015, 21, 13793–13801. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.; Xu, Z.-L.; Bao, S.-S.; Wang, T.-T.; Zheng, Z.-H.; Ferreira, R.A.S.; Zheng, L.-M.; Carlos, L.D. Lanthanide salen-type complexes exhibiting single ion magnet and photoluminescent properties. Dalton Trans. 2016, 45, 2974–2982. [Google Scholar] [CrossRef] [PubMed]
- Jeletic, M.; Lin, P.-H.; Roy, J.J.L.; Korobkov, I.; Gorelsky, S.I.; Murugesu, M. An Organometallic Sandwich Lanthanide Single-Ion Magnet with an Unusual Multiple Relaxation Mechanism. J. Am. Chem. Soc. 2011, 133, 19286–19289. [Google Scholar] [CrossRef] [PubMed]
- Habib, F.; Lin, P.-H.; Long, J.; Korobkov, I.; Wernsdorfer, W.; Murugesu, M. The Use of Magnetic Dilution to Elucidate the Slow Magnetic Relaxation Effects of a Dy2 Single-Molecule Magnet. J. Am. Chem. Soc. 2011, 133, 8830–8833. [Google Scholar] [CrossRef]
- Gavey, E.L.; Hareri, M.A.; Regier, J.; Carlos, L.D.; Ferreira, R.A.S.; Razavi, F.S.; Rawson, J.M.; Pilkington, M. Placing a crown on DyIII—A dual property LnIII crown ether complex displaying optical properties and SMM behaviour. J. Mater. Chem. C 2015, 3, 7738–7747. [Google Scholar] [CrossRef]
- Jiang, Z.-X.; Liu, J.-L.; Chen, Y.-C.; Liu, J.; Jia, J.-H.; Tong, M.-L. Lanthanoid single-ion magnets with the LnN10 coordination geometry. Chem. Commun. 2016, 52, 6261–6264. [Google Scholar]
- Morita, T.; Katoh, K.; Breedlove, B.K.; Yamashita, M. Controlling the Dipole−Dipole Interactions between Terbium (III) Phthalocyaninato Triple-Decker Moieties through Spatial Control Using a Fused Phthalocyaninato Ligand. Inorg. Chem. 2013, 52, 13555–13561. [Google Scholar] [CrossRef]
- Blagg, R.J.; Ungur, L.; Tuna, F.; Speak, J.; Comar, P.; Collison, D.; Wernsdorfer, W.; McInnes, E.J.L.; Chibotaru, L.F.; Winpenny, R.E.P. Magnetic relaxation pathways in lanthanide single-molecule magnets. Nat. Chem. 2013, 5, 673–678. [Google Scholar] [CrossRef]
- Mori, F.; Nyui, T.; Ishida, T.; Nogami, T.; Choi, K.-Y.; Nojiri, H. Oximate-Bridged Trinuclear Dy-Cu-Dy Complex Behaving as a Single-Molecule Magnet and Its Mechanistic Investigation. J. Am. Chem. Soc. 2006, 128, 1440–1441. [Google Scholar] [CrossRef]
- Liang, Z.; Damjanović, M.; Kamila, M.; Cosquer, G.; Breedlove, B.K.; Enders, M.; Yamashita, M. Proton Control of the Lanthanoid Single-Ion Magnet Behavior of a Double-Decker Complex with an Indolenine-Substituted Annulene Ligand. Inorg. Chem. 2017, 56, 6512–6521. [Google Scholar] [CrossRef]
- Mandal, L.; Biswas, S.; Cosquer, G.; Shen, Y.; Yamashita, M. Anion-driven structures and SMM behavior of dinuclear terbium and ytterbium complexes. Dalton Trans. 2018, 47, 17493–17499. [Google Scholar] [CrossRef]
- Meihaus, K.R.; Minasian, S.G.; Lukens, W.W.; Kozimor, J.S.A.; Shuh, D.K.; Tyliszczak, T.; Long, J.R. Influence of Pyrazolate vs N-Heterocyclic Carbene Ligands on the Slow Magnetic Relaxation of Homoleptic Trischelate Lanthanide (III) and Uranium (III) Complexes. J. Am. Chem. Soc. 2014, 136, 6056–6068. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, J.J.; Yuan, A.; Song, Y. Slow magnetic relaxation in luminescent mononuclear dysprosium (III) and erbium (III) pentanitrate complexes with the same LnO10 coordination geometry. Dalton Trans. 2017, 46, 15812–15818. [Google Scholar] [CrossRef]
- Kalita, P.; Goura, J.; Martinez, J.M.H.; Colacio, E.; Chandrasekhar, V. Homodinuclear {LnIII2} (LnIII = GdIII, TbIII, HoIII, and DyIII) Complexes: Field-Induced SMM Behavior of the DyIII and TbIII Analogues. Eur. J. Inorg. Chem. 2019, 2019, 212–220. [Google Scholar] [CrossRef]
- Boulon, M.-E.; Cucinotta, G.; Luzon, J.; Degl’Innocenti, C.; Perfetti, M.; Bernot, K.; Calvez, G.; Caneschi, A.; Sessoli, R. Magnetic Anisotropy and Spin-Parity Effect Along the Series of Lanthanide Complexes with DOTA. Angew. Chem. Int. Ed. 2013, 52, 350–354. [Google Scholar] [CrossRef]
- Xiao, Q.; Yanbin, Z.; Xia, L. Synthesis, Crystal structure and fluorescence of a new europium complex with 2-bromobenzoate and 2,2′-bipyridine. J. Rare Earths 2009, 27, 797–800. [Google Scholar]
- Zhao, Y.-F.; Chu, H.-B.; Bai, F.; Gao, D.-Q.; Zhang, H.-X.; Zhou, Y.-S.; Wei, X.-Y.; Shan, M.-N.; Li, H.-Y.; Zhao, Y.-L. Synthesis, crystal structure, luminescent property and antibacterial activity of lanthanide ternary complexes with 2,4,6-tri(2-pyridyl)-s-triazine. J. Organomet. Chem. 2012, 716, 167–174. [Google Scholar] [CrossRef]
- Liu, C.-S.; Du, M.; Sañudo, E.C.; Echeverria, J.; Hu, M.; Zhang, Q.; Zhou, L.-M.; Fang, S.-M. A luminescent linear trinuclear DyIII complex exhibiting slow magnetic relaxation of single ion origin. Dalton Trans. 2011, 40, 9366–9369. [Google Scholar] [CrossRef]
- Wen, H.-R.; Xie, X.-R.; Liu, S.-J.; Bao, J.; Wang, F.-F.; Liu, C.-M.; Liao, J.-S. Homochiral luminescent lanthanide dinuclear complexes derived from a chiral carboxylate. RSC Adv. 2015, 5, 98097–98104. [Google Scholar] [CrossRef]
- Zhao, J.; Zhu, G.-H.; Xie, L.-Q.; Wu, Y.-S.; Wu, H.-L.; Zhou, A.-J.; Wu, Z.-Y.; Wang, J.; Chen, Y.-C.; Tong, M.-L. Magnetic and luminescent properties of lanthanide coordination polymers with asymmetric biphenyl-3,2′,5′-tricarboxylate. Dalton Trans. 2015, 44, 14424–14435. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Z.-Y.; Zou, Y.-Q. Synthesis, Structure and Luminescence Properties of Four Novel Terbium 2-Fluorobenzoate Complexes. Eur. J. Inorg. Chem. 2005, 2005, 2909–2918. [Google Scholar] [CrossRef]
- Samuel, A.P.S.; Xu, J.; Raymond, K.N. Predicting Efficient Antenna Ligands for Tb (III) Emission. Inorg. Chem. 2009, 48, 687–698. [Google Scholar] [CrossRef]
- Li, Q.; Li, T.; Wu, J. Luminescence of Europium (III) and Terbium (III) Complexes Incorporated in Poly (Vinyl Pyrrolidone) Matrix. J. Phys. Chem. B 2001, 105, 12293–12296. [Google Scholar] [CrossRef]
- CrysAlisPro, Version 1.171.38.46; Rigaku Oxford Diffraction: Tokyo, Japan, 2015.
- Crystal Clear-SM, version 1.4.0 SP1; Rigaku Corporation: Tokyo, Japan, 2008.
- Altomare, A.; Burla, M.C.; Camalli, M.; Cascarano, G.L.; Giacovazzo, C.; Guagliardi, A.; Moliterni, A.G.G.; Polidori, G.; Spagna, R. SIR97: A new tool for crystal structure determination and refinement. J. Appl. Crystallogr. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXL-2014/7, Crystal Structure Refinement Program. University of Göttingen. Acta Crystallogr. Sect. C 2014, 71, 3–8. [Google Scholar]
- Farrugia, L.J. WinGX and ORTEP for Windows: An update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).