Seed Germination within Genus Rosa: The Complexity of the Process and Influencing Factors
Abstract
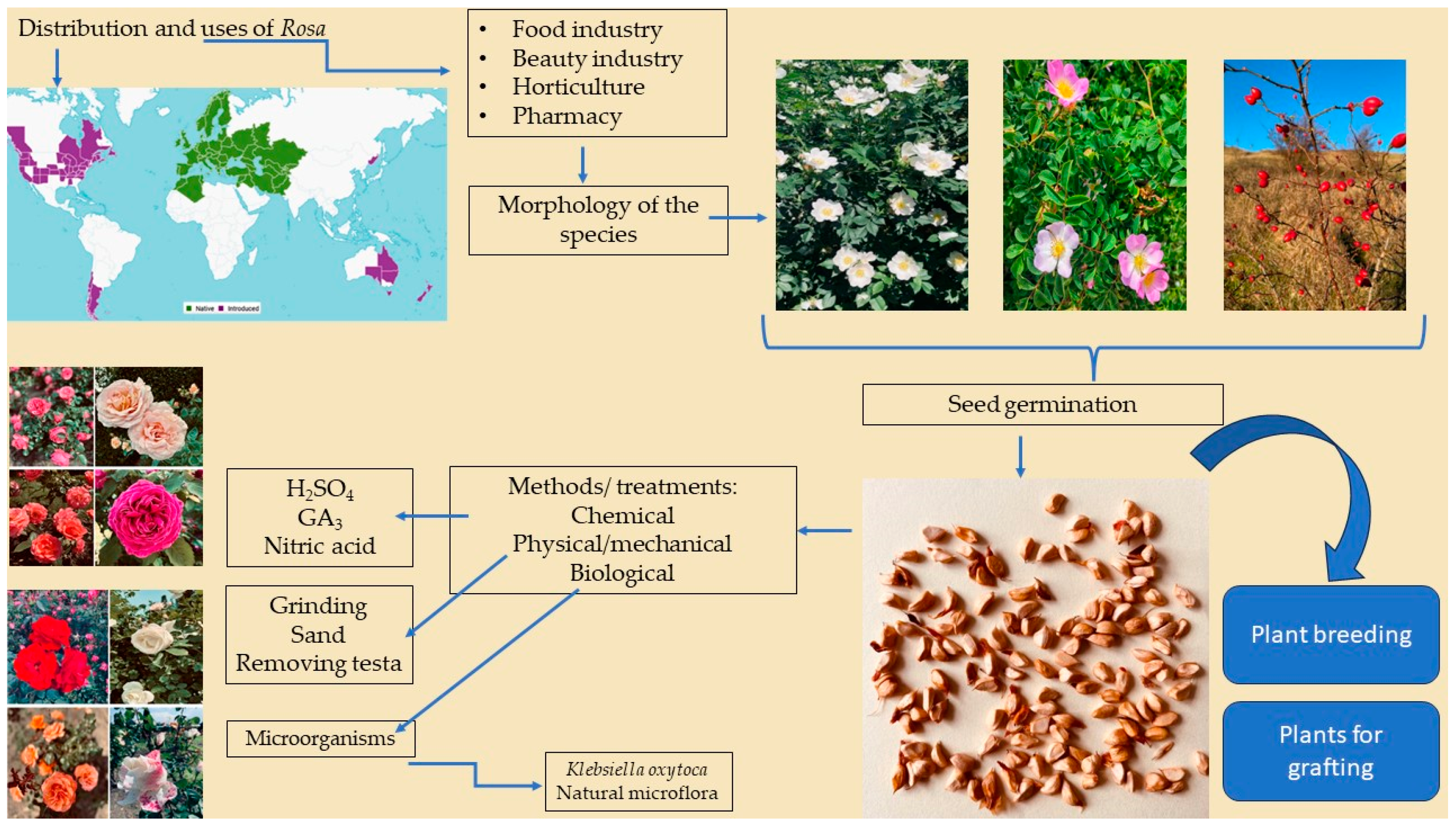
1. Introduction
1.1. Distribution of the Genus Rosa
1.2. Botanical Aspect of the Species
1.3. Importance of the Species
1.3.1. Chemical Composition of the Hips
1.3.2. Health Benefits of Rose Hips
1.3.3. Horticultural and Economical Importance of the Species
2. Seed Characteristics and Germination within the Rosa Genus
3. Treatments Used to Stimulate Seed Germination in Species of the Genus Rosa
3.1. Inhibitory and Stimulatory Factors of Seed Germination in the Genus Rosa
- Scarification requirements: Some R. canina seeds may require scarification, which is the mechanical treatment applied to the seed coat, to break dormancy. Scarification methods include nicking or sanding the seed coat or subjecting it to hot water or acid treatment within chemical treatments [21,49].
- Light requirements: Some R. canina seeds have light sensitivity and require exposure to light so that germination occurs. Seeds that require light will have low germination rates in darkness, so light can act as a germination stimulant for Rosa [49].
- Environmental factors: R. canina seeds may have specific environmental requirements for germination, such as moisture, temperature, and oxygen availability. If these conditions are not optimal, germination rates can be reduced [50].
- Stratification: Many R. canina seeds require a period of cold stratification to break dormancy. Cold stratification involves subjecting the seeds to a period of cold, typically around 4 °C, for a certain duration. This mimics the natural conditions that the seeds would experience during the winter and helps overcome dormancy [51].
- Scarification: Some R. canina seeds have hard seed coats that can inhibit germination. Scarification techniques, such as mechanical scarification (e.g., nicking or scratching the seed coat) or chemical scarification (e.g., acid treatment), can help break the seed coat and enhance germination [52].
- Light exposure: While some R. canina seeds require light for germination, others may germinate better in darkness. Understanding the light requirements of specific seed lots can help optimize germination conditions accordingly [53].
- Moisture and temperature: Providing the seeds with adequate moisture and maintaining a suitable temperature range can promote germination. R. canina seeds generally require a moist environment, but excessive moisture can lead to fungal or bacterial issues. Optimal temperatures for germination typically range from 15 °C to 25 °C [54].
- Seed quality and age: Using high-quality, fresh seeds can improve germination rates. Older seeds may have reduced viability and lower germination rates [50].
3.2. Physical/Mechanical Treatments
- Harvesting time/period: For best seed quality, rose hips should be harvested when the fruits are mature, ripe, but firm [58]. Despite showing dormancy in the first year after fruition, the seeds typically germinate in the second year [59]. However, different harvesting periods have an impact on germination, with a previously documented 13–60% difference in germination frequency, with the best interval for harvesting rose hip being from late September until the beginning of October [60].
- Storage of seeds: In the study of Hoşafçı et al. [30], which aimed to investigate the effect of gibberellic acid (GA3) treatments on the germination of dog rose seeds, it was discovered that even the seed storage type can influence the germination rate. The three pre-treatments used—(1) seeds kept at 4 °C; (2) seeds in fruit kept at 4 °C; and (3) seeds in fruit kept at room temperature (25 °C)—had considerable effects on the germination of dog rose seeds in the field experiment. The lowest (all-over mean) rate of germination (22.5%) was obtained for seeds kept in the refrigerator. The highest (all-over mean) rate of germination (39.1%) was obtained for seeds kept inside fruits at room temperature [30].
- Scarification: While most vegetable seeds germinate readily upon exposure to normally favorable environmental conditions, many seed plants that are vegetatively (asexually) propagated fail to germinate readily because of physical or physiologically imposed dormancy. Physical dormancy is due to structural limitations for germination, such as hard, impervious seed coats. Under natural conditions, weathering for a number of years weakens the seed coat. Certain seeds have a tough husk that can be artificially worn or weakened to render the seed coat permeable to gases and water by a process known as scarification [61]. Scarification is a process that includes modifying, weakening, or opening a seed’s covering in order to promote germination. Scarification frequently involves mechanical, thermal, and chemical processes. Many plant species have seeds that are frequently resistant to water and gases, which delay or prevent germination [48].
- Gamma ray irradiation: To remain competitive in the flower industry, breeders are constantly striving for fresh and innovative varieties. Low seed germination rates continue to be a significant issue in conventional rose breeding projects. To improve seed vigor and productivity and to improve the sprouting and emergence of buds that are carried out by seed coating, mutagenic agents, such as ionizing radiation, may be utilized. Ionizing radiation treatments must be tailored for each unique cultivar since the effects of radiation on seed vigor are typically genotype-dependent [64]. In a study by Giovannini et al. [64], hybrid tea rose seeds were exposed to gamma rays (0, 50, 100, and 200 Gy) in order to establish a radiation regimen for boosting seed germination. Rosa hybrid commercial cultivars’ six different crossings’ seeds were used to collect data on germination capacity and radio-tolerance. The final germination percentage and germination energy of the seeds, regardless of the cross, were not significantly affected by the range of gamma ray doses evaluated. These findings contrast with research done on seeds of various species, including Vigna unguiculata L. [65], Citrus jambhiri Lush. [66], and Withania somnifera L. [67], in which a gradual decline in seed germination and seedling vigor from lower doses to higher doses in given treatments of gamma rays was found. Since there are other findings that show that ionizing radiation has positive impacts on seed vigor, this is a contentious topic.
- Medium: Anderson and Byrne [68] tested the influence of stratification media and genotype over the germination process. They stratified fresh rose seeds from different hybrids for 10–12 weeks at 2.8 °C in milled sphagnum moss, sand, perlite, vermiculite, and moist filter paper. The achenes germinated the best on moss. When placed on moist milled sphagnum moss or agar and stratified for 8 weeks at 2.8 °C, once again, the best germination was on moss. The germination of the genotypes varied greatly, ranging from 0.7 to 37.1% [64]. According to Carter [69], rose seed germinated more effectively after stratification in damp peat moss or sand as opposed to moist vermiculite. The effectiveness of sphagnum moss as a stratification medium was demonstrated in their experiment. The outcomes provided more proof that the medium is essential for seed germination. The smallest rate of germination was encouraged by stratification on filter paper, while the best germination was obtained with sphagnum moss. In terms of stimulating germination, perlite fell somewhere between sphagnum moss and the other two media (sand and vermiculite). Sphagnum moss performed better or on par with the other stratification media across all genotypes examined. In contrast to Yambe et al.’s [70] findings for R. multiflora, leaching of WOB-28 rose seed for three or more days significantly reduced germination. Such a divergence may have been caused by changes in leaching technique, genotypic effects, or leaching water temperature.
- Stratification: Aches treated with warm and cold stratification were used to examine the effects of temperature and water stress. Freshly harvested achenes are devoid of any physical, morphological, or morphophysiological dormancy since the pericarps are permeable and the embryo is completely formed. Despite softening or even completely removing the pericarp, the germination percentage remained low (5%), while completely removing the testa greatly increased germination (39%), indicating the potential existence of germination inhibitors in the testa [52]. According to Zhou et al. [52], dry storage for 68 weeks followed by cold stratification for 16 or 24 weeks resulted in maximum germination (72–79%) in R. multibracteata. The most popular method for removing rose seed dormancy is chilling, as most species’ achenes will eventually germinate if cooled for an extended length of time. For certain species, cold stratification periods equivalent to one field winter are sufficient. Only if the temperature of warm incubation was too high did the interruption of chilling with warm incubation result in subsequent dormancy induction. The dormancy of the seeds did not change when they were kept below this “compensating” temperature, and they may accrue the effects of chilling despite warm breaks [71].
- Temperature: Temperature is an important environmental component that inhibits seed germination. A hallmark of rose (R. canina L.) seedlings is the physical and physiological dormancy, which is often disrupted during warm weather, followed by cold stratification. When prepared seeds were subjected to a temperature of 20 °C, secondary dormancy was produced. A study by Pawowski [72] sought to discover and functionally describe the proteins linked to rose seed dormancy management. Using 2-D electrophoresis, proteins from primary dormant, following warm and cold stratification (nondormant), and secondary dormant seeds were examined. Mass spectrometry was used to determine which proteins were abundant in different ways.
- Tetrazolium staining is typically used to assess the quality of rose seeds due to the vast variety of dormancy-breaking needs within each species [73]. The first step is to soak the achenes in water for 24 h. The pericarp is broken open by applying firm pressure with a knife. After the testa has been scratched or snipped at the cotyledon end, the seed is submerged for 6 h at room temperature in 1% tetrazolium chloride. The testa is slit along the side, and the embryo, which fills the seed cavity, is squeezed or teased out for evaluation [74]. The excised embryo method may also be used, although it has little advantage over tetrazolium staining [73]. For purposes of determining fill and chalcid infestation levels, x-radiography is suitable [74]. X-radiography is helpful for assessing the amount of fill and chalcid infestation [74].
3.3. Chemical Treatments
- Sulphuric acid: To boost ingestions and break physical dormancy, sulphuric treatment of the rose pericarp is advised rather than scarification. However, due to the uneven thickness of the rose pericarp, the application of sulphuric acid necessitates substantially more stabilization. This kind of scarification technique has been employed in certain research on roses, although it often only applies to a small number of cross-combinations or particular species [52,63,74]. Zhou et al. [48] treated seeds with sulphuric acid for 2, 4, and 6 h and observed no germination. The overall rate of germination was 30%. The treated and untreated samples did not differ significantly from one another. The longer the sulphuric acid treatment, the smaller the rate obtained. It appears that even during this brief course of treatment, sulfuric acid could harm the embryo [63].
- Nitric acid: Younis et al. [75] tried to break achenes dormancy by using nitric acid. Nitric acid is a nitrogen oxoacid that has a role as a protic solvent and reagent. As is the case with sulphuric acid, it was used in order to break the seed’s pericarp. Nitric acid (65%) with different time exposures (30, 60, and 90 s) was tested. Although it was expected, no significant effect of the acid was observed [78].
3.4. Biological Treatments
- Microorganism Treatment: Effective microorganisms (EM) are used to facilitate seed germination [79]. Microorganism: Klebsiella oxytoca C1036 is a strain that has been identified to act against the soft-rot pathogen in Tabacco [80]. Lee et al. [55] used this strain to enhance germination. The seeds were immersed in a suspension of the strain for 1 and 48 h. The germination rate rose to 50% with longer immersion. Treated seeds germinated twice as well as those of non-treated seeds. Other useful microorganisms should be investigated in further studies [55].
- Macerating enzymes: R. multiflora Thunb. achenes were treated for 36 h with 1% Driselase, a macerating enzyme, which significantly enhanced the germination percentage. When the achenes were exposed to the enzyme for a longer time, the seeds germinated more quickly. In comparison to Driselase, treatment with Cellulase Onozuka increased seed germination at a lower dosage. Pure pectinase and cellulase preparations had outcomes that were comparable to those of the mentioned enzyme treatments. Pectinase treatment was more effective than cellulase treatment. These enzymes probably made the pericarp’s suture less rigid, which caused the pericarp to split [82].
- Genetic make-up of the seed: One important factor contributing to poor germination is the genetic make-up of the seed. This is the reason many breeders maintain thorough records of seed set and germination in order to select the genitors that produce the greatest number of offspring. Techniques for treating seeds, such as harvesting, stratification, scarification, and leaching, are essential to maximizing germination [57].
- Break seed dormancy under in vitro conditions: In the study by Hajyzadeh [57], it was aimed to break the seed dormancy of rose hip under in vitro conditions by applying multiple strategies in an efficient manner. The seeds were given various doses of GA3, mechanically scarified, stratified on agar-solidified MS medium containing GA3 alone or in combinations with the suggested treatments, and then given a controlled physiological treatment that involved alternately giving the seeds warm/chilling and cold/dark treatments for 21 days, followed by 18 days of warm/light treatments.
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rehder, A. Bibliography of Cultivated Trees and Shrubs: Hardy in the Cooler Temperate Regions of the Northern Hemisphere; Arnold Arboretum of Harvard University: Jamaica Plain, MA, USA, 1949. [Google Scholar] [CrossRef]
- Wissemann, V. Conventional taxonomy of wild roses. In Encyclopedia of Rosa Science, 1st ed.; Roberts, A., Debener, T., Gudin, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; pp. 111–117. [Google Scholar]
- Wissemann, V. Hybridization and the evolution of the nrITS spacer region. Plant genome, biodiversity and evolution. Part A Phanerogams 2003, 1, 57–71. [Google Scholar]
- Guimaraes, R.; Barros, L.; Carvalho, A.M.; Ferreira, I.C. Studies on chemical constituents and bioactivity of Rosa micrantha: An alternative antioxidants source for food, pharmaceutical, or cosmetic applications. J. Agric. Food Chem. 2010, 58, 6277–6284. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, C.; Roufogalis, B.D.; Müller-Ladner, U.; Chrubasik, S.A. Systematic review on the Rosa canina effect and efficacy profiles. Phytother. Res. 2008, 22, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Strålsjö, L.; Alklint, C.; Olsson, M.E.; Sjöholm, I. Total folate content and retention in rosehips (Rosa ssp.) after drying. J. Agric. Food Chem. 2003, 51, 4291–4295. [Google Scholar] [CrossRef] [PubMed]
- Larsen, E.; Kharazmi, A.; Christensen, L.P.; Christensen, S.B. An antiinflammatory galactolipid from rose hip (Rosa canina) that inhibits chemotaxis of human peripheral blood neutrophils in vitro. J. Nat. Prod. 2003, 66, 994–995. [Google Scholar] [CrossRef] [PubMed]
- Rahnavard, A.; Asadian, G.; Pourshamsian, K.; Taghavi, M. Assessing genetic variation of dog rose (Rosa canina L.) in Caspian climate. Biosci. Biotechnol. Res. Asia 2014, 10, 119–125. [Google Scholar] [CrossRef]
- Grant, V. Plant speciation. New Phytol. 1971, 161, 8–11. [Google Scholar] [CrossRef]
- Wissemann, V. Molekulargenetische und morphologisch-anatomische Untersuchungen zur Evolution und Genomzusammensetzung von Wildrosen der Sektion Caninae (DC.) Ser. E. Schweiz. Verlagsbuchhandlung 2000, 122, 357–429. [Google Scholar]
- Özkan, G.; Sagdiç, O.; Baydar, N.G.; Baydar, H. Antioxidant and antibacterial activities of Rosa damascena flower extracts. Food Sci. Technol. 2004, 10, 277–281. [Google Scholar] [CrossRef]
- Szentmihályi, K.; Vinkler, P.; Lakatos, B.; Illés, V.; Then, M. Rose hip (Rosa canina L.) oil obtained from waste hip seeds by different extraction methods. Bioresour. Technol. 2002, 82, 195–201. [Google Scholar] [CrossRef]
- Fan, C.; Pacier, C.; Martirosyan, D.M. Rose hip (Rosa canina L): A functional food perspective. Funct. Foods Health Dis. 2014, 4, 493–509. [Google Scholar] [CrossRef]
- Rosu, C.M.; Manzu, C.; Olteanu, Z.; Oprica, L.; Oprea, A.; Ciornea, E.; Zamfirache, M.M. Several fruit characteristics of Rosa sp. genotypes from the Northeastern region of Romania. Not. Bot. Horti Agrobot. Cluj-Napoca 2011, 39, 203–208. [Google Scholar] [CrossRef]
- Ercisli, S. Chemical composition of fruits in some rose (Rosa spp.) species. Food Chem. 2007, 104, 1379–1384. [Google Scholar] [CrossRef]
- Leus, L.; Van Laere, K.; De Riek, J.; Van Huylenbroeck, J. Rose. In Ornamental Crops; Van Huylenbroeck, J., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 736–738. [Google Scholar] [CrossRef]
- Gudin, S.; Arene, L.; Chavagnat, A.; Bulart, C. Influence of endocarp thickness on rose achene germination: Genetic and environmental factors. HortScience 1990, 25, 786–788. [Google Scholar] [CrossRef]
- Basu, S.; Zandi, P.; Cetzal-Ix, W.; Sengupta, R. The genus Rosa: An aristocrat from the plant family with class, color and fragrance. Iran. Soc. Environ. Newsl. 2014, 1–9. [Google Scholar]
- Wagner, S.; Ardelean, M.; Sestras, R.; Ghidra, V.; Pamfil, D.; Roman, G.; Florincescu, A.; Coredea, M.; Catana, C.; Budiu, V. Achievements in rose breeding at Cluj-Napoca, Romania, in the last thirty years. Biotechnol. Biotechnol. Equip. 2000, 14, 37–41. [Google Scholar] [CrossRef]
- Izadi, Z.; Zarei, H.; Alizadeh, M. Effect of time, cultivar and rootstock on success of rose propagation through stenting technique. Am. J. Plant Sci. 2014, 5, 1644–1650. [Google Scholar] [CrossRef]
- Wagner, Ş. Trandafirul-de la Mit la Mileniul Trei: O Carte Pentru Toţi Iubitorii Rozelor; Wagner, S., Ed.; Cluj-Napoca, Romania, 2002. [Google Scholar]
- Ambros, E.V.; Vasilyeva, O.Y.; Novikova, T.I. Effects of in vitro propagation on ontogeny of Rosa canina L. micropropagated plants as a promising rootstock for ornamental roses. Plant Cell Biotechnol. Mol. Biol. 2016, 17, 72–78. [Google Scholar]
- Vasilyeva, O.Y. Reproduction systems of representatives of the genus Rosa L. under conditions of continental climate. Contemp. Probl. Ecol. 2009, 2, 361–368. [Google Scholar] [CrossRef]
- VonAbrams, G.J.; Hand, M.E. Seed dormancy in Rosa as a function of climate. Am. J. Bot. 1956, 43, 7–12. [Google Scholar] [CrossRef]
- Bewley, J.D. Seed germination and dormancy. Plant Cell 1997, 9, 1055–1066. [Google Scholar] [CrossRef]
- Finch-Savage, W.E.; Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 2006, 1716, 501–523. [Google Scholar] [CrossRef] [PubMed]
- Gudin, S. Seed propagation. In Ornamental Crops; Reference Module in Life Sciences; Elsevier: Amsterdam, The Netherlands, 2017; pp. 719–767. [Google Scholar]
- Pipino, L.; Scariot, V.; Gaggero, L.; Mansuino, A.; Van Labeke, M.C.; Giovannini, A. Enhancing seed germination in hybrid tea roses. Propag. Ornam. Plants 2011, 11, 111–118. [Google Scholar]
- Khosh-Khui, M.; Teixeira da Silva, J.A. In vitro culture of the Rosa species. In Floriculture, Ornamental and Plant Biotechnology; Advances and Topical Issues, 1st ed.; Jaime, A., da Silva, T., Eds.; Global Science Books: East Sussex, UK, 2016; Volume 2, pp. 513–526. [Google Scholar]
- Hoşafçı, H.; Arslan, N.; Sarıhan, E.O. Propagation of dog roses (Rosa canina L.) by seed. Acta Hortic. 2004, 690, 159–164. [Google Scholar] [CrossRef]
- Werlemark, G.; Carlson-Nilsson, U.; Uggla, M.; Nybom, H. Effects of temperature treatments on seedling emergence in dogroses, Rosa sect. Caninae (L). Acta Agric. Scand. 1995, 45, 278–282. [Google Scholar] [CrossRef]
- Hornero-Mendez, D.; Mınguez-Mosquera, M.I. Carotenoid pigments in Rosa mosqueta hips, an alternative carotenoid source for foods. J. Agric. Chem. 2000, 48, 825–828. [Google Scholar] [CrossRef]
- Uggla, M. Domestication of Wild Roses for Fruit Production. Ph.D. Thesis, Swedish University of Agricultural Sciences, Alnarp, UK, 2004. [Google Scholar]
- Pati, P.K.; Rath, S.P.; Sharma, M.; Sood, A.; Ahuja, P.S. In vitro propagation of rose—A review. Biotechnol. Adv. 2006, 24, 94–114. [Google Scholar] [CrossRef] [PubMed]
- Tincker, M.A.; Wisley, M.A. Rose seeds: Their after-ripening and germination. J. R. Hortic. Soc. 1935, 60, 399–417. [Google Scholar]
- Xu, B.M.; Zhang, Z.M.; Zhang, H.J. Germination and dormancy of Rosa seed. Seed 1993, 63, 5–9. [Google Scholar]
- Bo, J.; Huiru, D.; Xiaohan, Y. Shortening hybridization breeding cycle of rose—A study on mechanisms controlling achene dormancy. Acta Hortic. 1995, 404, 40–47. [Google Scholar] [CrossRef]
- Jackson, G.A.D.; Blundell, J.B. Germination in Rosa. J. Hortic. Sci. 1963, 38, 310–320. [Google Scholar] [CrossRef]
- Densmore, R.; Zasada, J.C. Germination requirements of Alaskan Rosa acicularis. Can. Field-Nat. 1977, 91, 58–62. [Google Scholar]
- Svejda, F. Water uptake of rose achenes. Can. J. Plant Sci. 1972, 52, 1043–1047. [Google Scholar] [CrossRef]
- Bhanuprakash, K.; Tejaswini, Y.; Yogeesha, H.S.; Naik, L.B. Effect of scarification and gibberellic acid on breaking dormancy of rose seeds. Seed Res. 2004, 32, 105–107. [Google Scholar]
- Svejda, F.J.; Poapst, P.A. Effects of different after-ripening treatments on germination and endogenous growth inhibitors in Rosa rugosa. Can. J. Plant Sci 1972, 52, 1049–1058. [Google Scholar] [CrossRef]
- Gudin, S. Breeding/Overview. In Encyclopedia of Rose Science; Roberts, A., Debener, T., Gudin, S., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: Oxford, UK, 2003. [Google Scholar]
- Stewart, R.N.; Semeniuk, P. The effect of the interaction of temperature with after-ripening requirement and compensating temperature on germination of seed of five species of Rosa. Am. J. Bot. 1965, 52, 755–760. [Google Scholar] [CrossRef]
- Semeniuk, P.; Stewart, R.N. Effect of temperature and duration of after-ripening period on germination of Rosa nutkana seeds. Proc. Am. Soc. Hortic. Sci. 1966, 89, 689. [Google Scholar]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography, and, Evolution of Dormancy and Germination; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. A classification system for seed dormancy. Seed Sci. Res. 2004, 14, 1–6. [Google Scholar] [CrossRef]
- Zhou, Z.Q.; Bao, W.K.; Wu, N. Dormancy and germination in Rosa multibracteata Hemsl. EH Wilson. Sci. Hortic. 2009, 119, 434–441. [Google Scholar] [CrossRef]
- Bonny, L. Seed Germination Test Methods Used for Australian Tree Species at Coffs Harbour Research Centre; Technical Paper; New South Wales Forestry Commission: Coffs Harbour, NSW, Australia, 1987. [Google Scholar]
- McDonald, M.B. Seed quality assessment. Seed Sci. Res. 1998, 8, 265–276. [Google Scholar] [CrossRef]
- Alp, S.; Çelik, F.; Türkoglu, N.; Karagöz, S. The effects of different warm stratification periods on the seed germination of some Rosa taxa. AJB 2009, 8, 5838–5841. [Google Scholar] [CrossRef]
- Zhou, Z.; Bao, W.; Wu, N. Effects of scarification, stratification and chemical treatments on the germination of Rosa soulieana Crépin achenes. In Floriculture and Ornamental Biotechnology; da Silva, T., Ed.; Global Science Books: London, UK, 2009; pp. 75–80. [Google Scholar]
- Woolley, J.T.; Stoller, E.W. Light penetration and light-induced seed germination in soil. Plant Physiol. 1978, 61, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Roberts, E.H. Temperature and seed germination. In Symposia of the Society for Experimental Biology; Company of Biologists Ltd.: Cambridge, MA, USA, 1988; Volume 42, pp. 109–132. [Google Scholar]
- Lee, J.Y.; Kim, Y.C.; Han, T.H.; Kim, S.T.; Gi, G.Y. Study on increasing rose seed germination. Acta Hortic. 2010, 855, 183. [Google Scholar] [CrossRef]
- Morpeth, D.R.; Hall, A.M. Microbial enhancement of seed germination in Rosa corymbifera ‘Laxa’. Seed Sci. Res. 2000, 10, 489–494. [Google Scholar] [CrossRef]
- Hajyzadeh, M. Application of multiple strategies to efficiently break seed dormancy of permanently odd pentaploid rose hip (Rosa canina L.) under in vitro conditions. Int. J. Agric. Environ. Food Sci. 2022, 6, 471–479. [Google Scholar] [CrossRef]
- Tipi, E. Kusburnu fidan üretim teknikleri ve üretim hedefleri. In Proceedings of the Kusburnu Sempozyumu, Gümüshane, Bildiri Kitabı, Gümüşhane, Turkey, 5–6 September 1996; pp. 127–133. [Google Scholar]
- Yılmaz, S. Kusburnu bitkisinin erozyon kontrolundeki yeri ve onemi. In Proceedings of the Kusburnu Sempozyumu, Gümüşhane, Bildiri Kitabı, Gümüşhane, Turkey, 5–6 September 1996; pp. 167–168. [Google Scholar]
- Rowley, G.D. Germination in Rosa canina. Am. Rose Annu. 1956, 41, 70–73. [Google Scholar]
- Ercişli, S. Farklı uygulamaların kusburnu tohumlarının cimlenmesi üzerine etkisi. II. In Proceedings of the Ulusal Fidancılık Sempozyumu, Bademli, Odemis, Turkey, 25–29 September 2000. [Google Scholar]
- Noble, R.E. Effects of UV-irradiation on seed germination. Sci. Total Environ. 2022, 299, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, J.; Ki, G.; Kim, S.; Han, T. Improvement of seed germination in Rosa rugosa. Korean J. Hortic. Sci. Technol. 2011, 29, 352–357. [Google Scholar]
- Giovannini, A.; Scariot, V.; Caser, M.; Buttafava, A.; Mansuino, A.; Ghione, G.G.; Balestrazzi, A. Mutation breeding using gamma rays to increase seed germination in Rosa hybrida. Acta Hortic. 2015, 1087, 373–378. [Google Scholar] [CrossRef]
- Bind, D.; Dwivedi, V.K. Effect of mutagenesis on germination, plant survival and pollen sterility in M1 generation of in cowpea (Vigna unguiculata (L.) Walp). Ind. J. Agric. Res. 2014, 48, 398–401. [Google Scholar] [CrossRef]
- Sharma, L.K.; Kaushal, M.; Gill, M.I.S.; Bali, S.K. Germination and survival of Citrus jambhiri seeds and epicotyls after treating with different mutagens under in vitro conditions. Middle-East J. Sci. Res. 2013, 16, 250–255. [Google Scholar]
- Bhosale, R.S.; More, A.D. Effect of gamma radiation on seed germination, seedling height and seedling injury in Withania somnifera (L.) Dunal. Int. J. Life Sci. 2014, 2, 226–228. [Google Scholar]
- Anderson, N.; Byrne, D.H. Methods for Rosa germination. Acta Hortic. 2007, 751, 503–507. [Google Scholar] [CrossRef]
- Carter, A.R. Rose rootstocks-performance and propagation from seed. Comb. Proc. Int. Plant Propagators Soc. 1968, 19, 172–180. [Google Scholar]
- Yambe, Y.; Hori, Y.; Takeno, K. Levels of endogenous abscisic acid in rose achenes and leaching with activated charcoal to improve seed germination. J. Jpn. Soc. Hortic. Sci 1992, 61, 383–387. [Google Scholar] [CrossRef][Green Version]
- Meyer, S.E. Rosa L. The Woody Plant Seed Manual; USDA Forest Service Agriculture Handbook; USDA Forest Service: Washington, DC, USA, 2008; Volume 727, pp. 974–980.
- Pawłowski, T.A.; Bujarska-Borkowska, B.; Suszka, J.; Tylkowski, T.; Chmielarz, P.; Klupczyńska, E.A.; Staszak, A.M. Temperature regulation of primary and secondary seed dormancy in Rosa canina L.: Findings from proteomic analysis. Int. J. Mol. Sci. 2020, 21, 7008. [Google Scholar] [CrossRef]
- Gill, J.D.; Pogge, F.L. Rosa L., rose. In Seeds of Woody Plants in the United States; Schopmeyer CS, Tech. Coordinator; USDA Agriculture Handbook 450; USDA Forest Service: Washington, DC, USA, 1974; pp. 732–737. [Google Scholar]
- Belcher, E. Handbook on Seeds of Browse-Shrubs and Forbs; Technical Publication R8-8; USDA Forest Service, Southern Region: Atlanta, GA, USA, 1985; p. 246.
- Younis, A.; Riaz, A.; Ahmed, R.; Raza, A. Effect of hot water, sulphuric acid and nitric acid on the germination of rose seeds. Acta Hortic. 2007, 755, 105–108. [Google Scholar] [CrossRef]
- Roberts, L.; Shardlow, A.D. Practical aspects of the acid treatment of rose seed [Rosa dumetorum ‘Laxa’, dormancy breaking]. Plant Propagator 1979, 25, 13–14. [Google Scholar]
- Cullum, F.J.; Bradley, S.J.; Williams, M.E. Improved germination of Rosa corymbifera ‘Laxa’ seed using a compost activator. Comb. Proc.—Int. Plant Propagators Soc. 1990, 40, 244–250. [Google Scholar]
- Svejda, F. Hybrid Rugosa Rose’ martin Frobisher’. Can. J. Plant Sci. 1969, 49, 100. [Google Scholar] [CrossRef]
- Taylor, A.G.; Harman, G.E. Concepts and technologies of selected seed treatments. Annu. Rev. Phytopathol. 1990, 28, 321–339. [Google Scholar] [CrossRef]
- Park, M.R.; Kim, Y.C.; Lee, S.W.; Kim, I.S. Identification of an ISR-related metabolite produced by rhizobacterium Klebsiella oxytoca C1036 active against soft-rot disease pathogen in tobacco. Pest Manag. Sci. 2009, 65, 1114–1117. [Google Scholar] [CrossRef] [PubMed]
- Kazaz, S.; Erbas, S.; Baydar, H. Breaking seed dormancy in oil rose (Rosa damascena Mill.) by microbial inoculation. AJB 2010, 9, 6503–6508. [Google Scholar]
- Yambe, Y.; Takeno, K. Improvement of rose achene germination by treatment with macerating enzymes. HortScience 1992, 27, 1018–1020. [Google Scholar] [CrossRef]




| Nutrient | Value per 100 g | References |
|---|---|---|
| Water | 58.66 g * | [13] |
| Energy | 162 kcal * | [13] |
| Protein | 1.6 g * | [13] |
| 0.36 g | [14] | |
| Total lipid | 0.34 g * | [13] |
| Ash | 1.18 g * | [13] |
| Carbohydrate | 38.22 g * | [13] |
| Fiber | 24.1 g * | [13] |
| Sugars, total | 2.58 g * | [13] |
| 13.28 g | [14] | |
| Minerals | ||
| Calcium | 169 mg * | [13] |
| 944 mg | [11] | |
| Iron | 1.06 mg * | [13] |
| 1.1 mg | [11] | |
| Magnesium | 69 mg * | [13] |
| 122.6 mg | [11] | |
| Phosphorus | 61 mg * | [13] |
| 122.4 mg | [11] | |
| Sodium | 4 mg * | [13] |
| 15.8 mg | [11] | |
| Zinc | 0.25 mg * | [13] |
| 1.3 mg | [11] | |
| Copper | 0.113 mg * | [13] |
| 0.4 mg | [11] | |
| Manganese | 1.02 mg * | [13] |
| 5.9 mg | [11] | |
| Potassium | 1025 mg | [11] |
| Vitamins | ||
| Vitamin C | 426 mg * | [13] |
| 411.0 mg | [11] | |
| 643.38 mg | [14] | |
| Vitamin B-6 | 0.076 mg * | [13] |
| Vitamin A, IU | 4345 IU * | [13] |
| Vitamin E (alpha tocopherol) | 5.84 mg * | [13] |
| 34.20 µg/g | [11] | |
| Tocopherol beta | 0.05 mg * | [13] |
| Tocopherol gamma | 1.34 mg * | [13] |
| Tocopherol delta | 0.14 mg * | [13] |
| Vitamin K | 25.9 µg * | [13] |
| Carotene alfa | 31 µg * | [13] |
| Carotene beta | 2350 µg * | [13] |
| 2.60 µg/g | [11] | |
| Cryptoxanthin, beta | 483 µg * | [13] |
| Thiamin | 0.016 mg * | [13] |
| Riboflavin | 0.166 mg * | [13] |
| Niacin | 1.3 mg * | [13] |
| Lycopene | 6800 µg * | [13] |
| 390 mg/kg DW | [14] | |
| Lutein + Zeaxanthin | 2001 µg * | [13] |
| Pantothenic acid | 0.8 mg * | [13] |
| Method, Procedures and Technics | Details | Germination | Reference |
|---|---|---|---|
| Scarification | Grinding (1, 5, 10 min) | 30% | [55] |
| Scarification | H2SO4 97% (1, 5, 10 min) | 30% | [55] |
| Scarification | H2SO4 50% (30 and 60 s) | >30% | [52] |
| Scarification | H2SO4 (2, 4, 6 h) | No germination | [48] |
| Scarification | NaOCl | 65.9% | [28] |
| Scarification | Fully removed testa | 39% | [48] |
| Microorganism | Inoculation in Klebsiella oxytoca C1036 | 50% | [55] |
| Microorganism | Inoculation natural microflora | 3% | [56] |
| Microorganism | Inoculation natural microflora + GarottaTM | 95% | [56] |
| Stratification | 8 weeks at 2.8 °C on moss | 37.1% | [54] |
| Stratification | Dry storage 68 w + cold stratification 16–24 w | 72–79% | [48] |
| Stratification | 11 w warm stratification + cold stratification | 13–18% | [51] |
| In vitro | GA3 + manual scarification + agar medium + warm/chilling—cold/dark 21 days | 80–85% | [57] |
| GA3 | 2000 ppm GA3 for 12 h (greenhouse) | 74% | [30] |
| GA3 | 300 ppm GA3 for 24 h (Green house) | 24.7% | [30] |
| GA3 | Chilled seeds + 200 ppm GA3 for 6 h | 11.7% | [30] |
| GA3 | Unchilled seeds + 400 GA3 for 12 h | 52% | [30] |
| GA3 | Stratification + pre-sowing GA3 | 77.6 | [28] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoian-Dod, R.L.; Dan, C.; Morar, I.M.; Sestras, A.F.; Truta, A.M.; Roman, G.; Sestras, R.E. Seed Germination within Genus Rosa: The Complexity of the Process and Influencing Factors. Horticulturae 2023, 9, 914. https://doi.org/10.3390/horticulturae9080914
Stoian-Dod RL, Dan C, Morar IM, Sestras AF, Truta AM, Roman G, Sestras RE. Seed Germination within Genus Rosa: The Complexity of the Process and Influencing Factors. Horticulturae. 2023; 9(8):914. https://doi.org/10.3390/horticulturae9080914
Chicago/Turabian StyleStoian-Dod, Roxana L., Catalina Dan, Irina M. Morar, Adriana F. Sestras, Alina M. Truta, Gabriela Roman, and Radu E. Sestras. 2023. "Seed Germination within Genus Rosa: The Complexity of the Process and Influencing Factors" Horticulturae 9, no. 8: 914. https://doi.org/10.3390/horticulturae9080914
APA StyleStoian-Dod, R. L., Dan, C., Morar, I. M., Sestras, A. F., Truta, A. M., Roman, G., & Sestras, R. E. (2023). Seed Germination within Genus Rosa: The Complexity of the Process and Influencing Factors. Horticulturae, 9(8), 914. https://doi.org/10.3390/horticulturae9080914











