Impact of Ground Applied Micronutrients on Root Growth and Fruit Yield of Severely Huanglongbing-Affected Grapefruit Trees
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Canopy Volume
- π = 3.14
- TH = Canopy height
- ACR = Average canopy radius.
2.3. Root Growth Parameters
2.4. Visual HLB Ratings
2.5. Root Density
2.6. Leaf and Root CLas Ct Values
2.7. Fruit Yield
2.8. Experimental Design and Statistical Analysis
3. Results
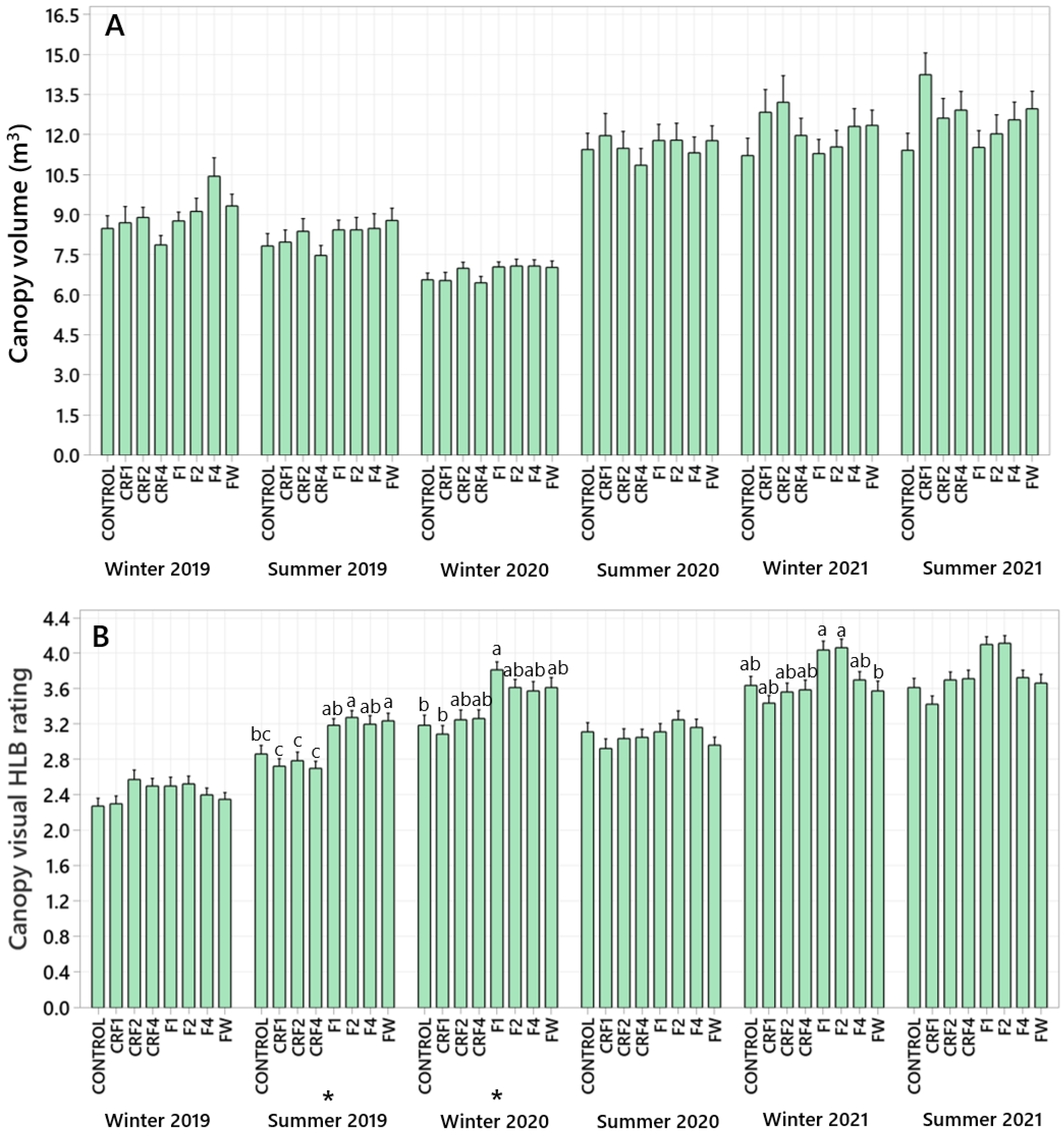
3.1. Canopy Volume
3.2. Visual HLB Symptoms
3.3. Root Count, Length, and Diameter
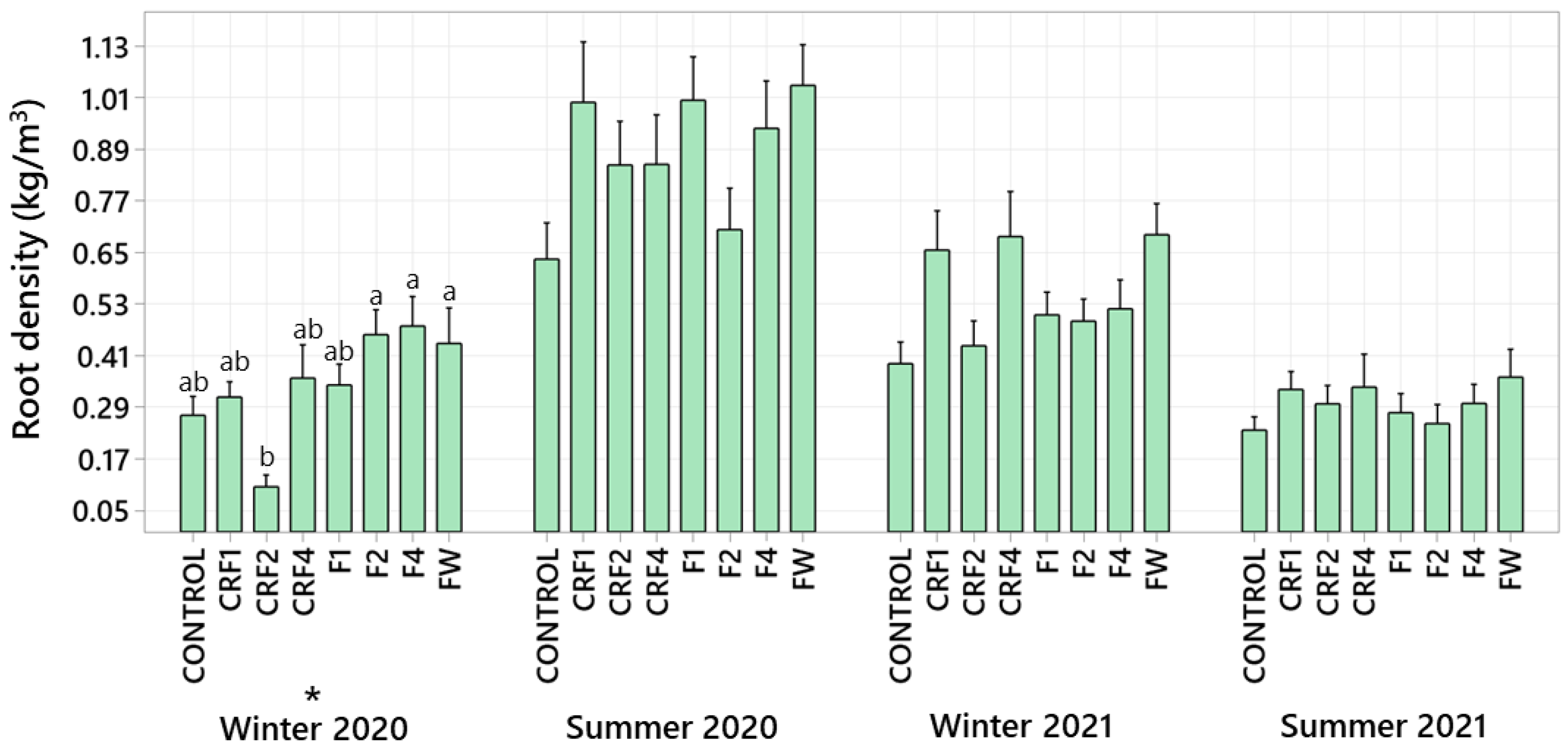
3.4. Root Density
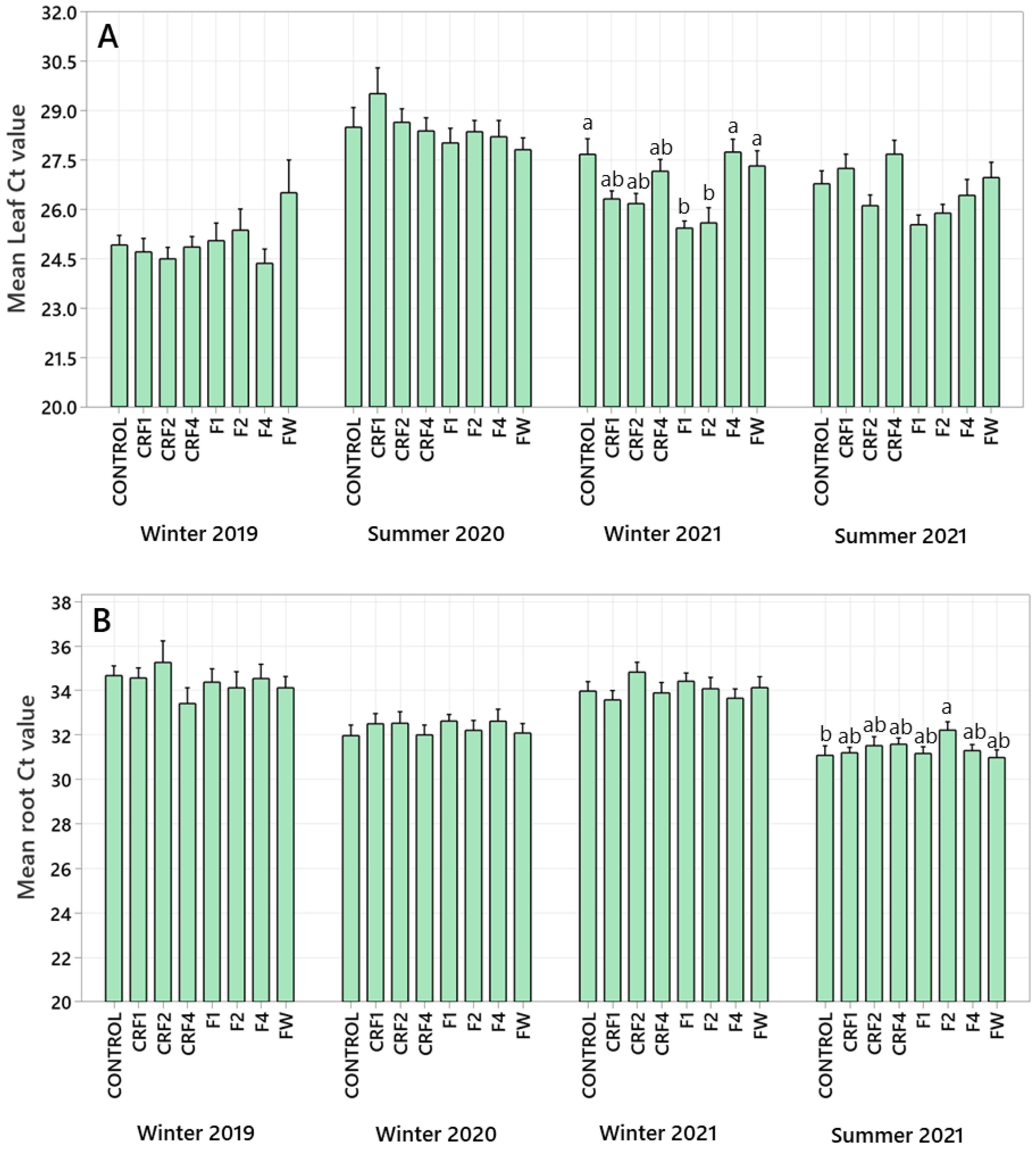
3.5. Root and Leaf CLas Ct Values
3.6. Fruit Yield
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grafton-Cardwell, E.E.; Stelinski, L.L.; Stansly, P.A. Biology and management of Asian citrus psyllid, vector of the huanglongbing pathogens. Annu. Rev. Entomol. 2013, 58. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.; Gottwald, T.; Setamou, M. Status of huanglongbing (HLB) outbreaks in Florida, California and Texas. Trop. Plant Pathol. 2020, 8, 31. [Google Scholar] [CrossRef]
- USDA. Florida Citrus Statistics 2020–2021; Florida Department of Agriculture and Consumer Services: Maitland, FL, USA, 2022; pp. 12–13. Available online: https://www.nass.usda.gov/Statistics_by_State/Florida/Publications/Citrus/Citrus_Statistics/2020-21/fcs2021b.pdf (accessed on 17 August 2022).
- Li, S.; Wu, F.; Duan, Y.; Singerman, A.; Guan, Z. Citrus greening: Management strategies and their economic impact. HortScience 2020, 55, 604–612. [Google Scholar] [CrossRef]
- Sinclair, W.B. The Grapefruit: Its Composition, Physiology & Products; University of California, Agriculture and Natural Resources: Oakland, CA, USA, 1972; Volume 4029. [Google Scholar]
- USDA. Florida Citrus Statistics 2019–2020; Florida Department of Agriculture and Consumer Services: Maitland, FL, USA, 2021; p. 36. Available online: https://www.nass.usda.gov/Statistics_by_State/Florida/Publications/Citrus/Citrus_Statistics/2019-20/fcs1920b.pdf (accessed on 17 August 2022).
- Folimonova, S.Y.; Robertson, C.J.; Garnsey, S.M.; Gowda, S.; Dawson, W.O. Examination of the responses of different genotypes of citrus to huanglongbing (Citrus Greening) under different conditions. Phytopathology 2009, 99, 1346–1354. [Google Scholar] [CrossRef]
- USDA. Florida Citrus Statistics 2019–2020; Florida Department of Agriculture and Consumer Services: Maitland, FL, USA, 2021; p. 21. Available online: https://www.nass.usda.gov/Statistics_by_State/Florida/Publications/Citrus/Citrus_Statistics/2019-20/fcs1920b.pdf (accessed on 17 August 2022).
- Johnson, E.G.; Graham, J.H.; Morgan, K.T. 2019–2020 Citrus Production Guide: Root Health Management; University of Florida, IFAS Cooperative Extension: Gainesville, FL, USA, 2019; Volume CMG15. [Google Scholar]
- Rossi, L.; Hallman, L.M.; Santiago, J.M. Citrus root and rhizosphere dynamics in the age of HLB. Annu. Plant Rev. Online 2021, 4, 605–624. [Google Scholar] [CrossRef]
- Louzada, E.S.; Vazquez, O.E.; Braswell, W.E.; Yanev, G.; Devanaboina, M.; Kunta, M. Distribution of ‘Candidatus Liberibacter asiaticus’ above and below ground in Texas citrus. Phytopathology 2016, 106, 702–709. [Google Scholar] [CrossRef]
- Kim, J.-S.; Sagaram, U.S.; Burns, J.K.; Li, J.-L.; Wang, N. Response of sweet orange (Citrus sinensis) to ‘Candidatus Liberibacter asiaticus’ infection: Microscopy and microarray analyses. Phytopathology 2009, 99, 50–57. [Google Scholar] [CrossRef]
- Zhang, X.-H.; Pizzo, N.; Abutineh, M.; Jin, X.-L.; Naylon, S.; Meredith, T.L.; West, L.; Harlin, J.M. Molecular and cellular analysis of orange plants infected with huanglongbing (citrus greening disease). Plant Growth Regul. 2020, 92, 333–343. [Google Scholar] [CrossRef]
- Koh, E.J.; Zhou, L.; Williams, D.S.; Park, J.; Ding, N.; Duan, Y.-P.; Kang, B.-H. Callose deposition in the phloem plasmodesmata and inhibition of phloem transport in citrus leaves infected with “Candidatus Liberibacter asiaticus”. Protoplasma 2012, 249, 687–697. [Google Scholar] [CrossRef]
- Cimò, G.; Lo Bianco, R.; Gonzalez, P.; Bandaranayake, W.; Etxeberria, E.; Syvertsen, J.P. Carbohydrate and nutritional responses to stem girdling and drought stress with respect to understanding symptoms of huanglongbing in citrus. HortScience 2013, 48, 920–928. [Google Scholar] [CrossRef]
- Pitino, M.; Sturgeon, K.; Dorado, C.; Cano, L.M.; Manthey, J.A.; Shatters, R.G.; Rossi, L. Quercus leaf extracts display curative effects against Candidatus Liberibacter asiaticus that restore leaf physiological parameters in HLB-affected citrus trees. Plant Physiol. Biochem. 2020, 148, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, E.; Gonzalez, P.; Achor, D.; Albrigo, G. Anatomical distribution of abnormally high levels of starch in HLB-affected Valencia orange trees. Physiol. Mol. Plant Pathol. 2009, 74, 76–83. [Google Scholar] [CrossRef]
- Raithore, S.; Dea, S.; Plotto, A.; Bai, J.; Manthey, J.; Narciso, J.; Irey, M.; Baldwin, D. Effect of blending huanglongbing (HLB) disease affected orange juice with juice from healthy orange on flavor quality. LWT—Food Sci. Technol. 2015, 62, 868–874. [Google Scholar] [CrossRef]
- Singerman, A.; Useche, P. Impact of Citrus Greening on Citrus Operations in Florida; University of Florida, IFAS Cooperative Extension, EDIS: Gainesville, FL, USA, 2018; Volume FE983. [Google Scholar]
- Dong, Z.; Low, W.; Srivastava, A.K.; Liu, X.-D.; Riaz, M.; Tan, Q.-L.; Sun, X.-C.; Hu, C.-X. Association between plant nutrients, the development of Huanglongbing and abnormal growth symptoms in navel orange. Plant Biol. 2021, 23, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Srivastava, A.K.; Liu, X.; Riaz, M.; Gao, Y.; Liang, X.; Tan, Q.; Sun, X.; Wu, S.; Hu, C. Interactions between nutrient and Huanglongbing pathogen in citrus: An overview and implications. Sci. Hortic. 2021, 290, 110511. [Google Scholar] [CrossRef]
- Shahzad, F.; Chun, C.; Schumann, A.; Vashisth, T. Nutrient Uptake in huanglongbing-affected sweet orange: Transcriptomic and physiological analysis. J. Am. Soc. Hortic. Sci. 2020, 145, 349–362. [Google Scholar] [CrossRef]
- Morgan, K.T.; Kadyampakeni, D.M.; Zekri, M.; Schumann, A.W.; Vashisth, T.; Vashisth, T.; Obreza, T.A.; Ferrarezi, R.S. 2020–2021 Florida Citrus Production Guide: Nutrition Management for Citrus Trees; University of Florida, IFAS Cooperative Extension EDIS: Gainesville, FL, USA, 2020. [Google Scholar]
- Zambon, F.T.; Kadyampakeni, D.M.; Grosser, J.W. Ground application of overdoses of manganese have a therapeutic effect on sweet orange trees infected with Candidatus Liberibacter asiaticus. Hortscience 2019, 54, 1077–1086. [Google Scholar] [CrossRef]
- Kwakye, S.; Kadyampakeni, D.M.; van Santen, E.; Vashisth, T.; Wright, A. Variable manganese rates influence the performance of huanglongbing-affected citrus trees in Florida. HortScience 2022, 57, 360–366. [Google Scholar] [CrossRef]
- Rouse, R.E.; Ozores-Hampton, M.; Roka, F.M.; Roberts, P. Rehabilitation of huanglongbing-affected citrus trees using severe pruning and enhanced Foliar nutritional treatments. HortScience 2017, 52, 972–978. [Google Scholar] [CrossRef]
- Phuyal, D.; Nogueira, T.A.R.; Jani, A.D.; Kadyampakeni, D.M.; Morgan, K.T.; Ferrarezi, R.S. ‘Ray Ruby’ grapefruit affected by huanglongbing II. planting density, soil, and foliar nutrient management. HortScience 2020, 55, 1420–1432. [Google Scholar] [CrossRef]
- Kadyampakeni, D.M. Interaction of soil boron application with leaf B concentration, root length density, and canopy size of citrus affected by Huanglongbing. J. Plant Nutr. 2020, 43, 186–193. [Google Scholar] [CrossRef]
- Nable, R.O.; Bañuelos, G.S.; Paull, J.G. Boron toxicity. Plant Soil 1997, 193, 181–198. [Google Scholar] [CrossRef]
- Kadyampakeni, D.M.; Chinyukwi, T. Are macronutrients and micronutrients therapeutic for restoring performance of trees affected by citrus greening? A discussion of current practices and future research opportunities. J. Plant Nutr. 2021, 44, 2949–2969. [Google Scholar] [CrossRef]
- Bauer, M.G.; Castle, W.S.; Boman, B.J.; Obreza, T.A. Field Guide to Soil Identification for Florida’s Citrus-Growing Regions; University of Florida, IFAS Coopertaive Extension: Gainesville, FL, USA, 2007; pp. 1–8. [Google Scholar]
- Driscoll, P.J. History of bedding citrus groves in the Indian River district to improve drainage. In Proceedings of the Florida State Horticultural Society, Sheraton World Resort, Orlando, FL, USA, 8–10 June 2003; pp. 201–204. [Google Scholar]
- Obreza, T.A.; Rouse, R.E. fertilizer effects on early growth and yield of ‘Hamlin’ orange trees. HortScience 1993, 28, 111–114. [Google Scholar] [CrossRef]
- Majdi, H. Root sampling methods—Applications and limitations of the minirhizotron technique. Plant Soil 1996, 185, 255–258. [Google Scholar] [CrossRef]
- Cheng, W.; Coleman, D.C.; Box, J.E. Measuring root turnover using the minirhizotron technique. Agric. Ecosyst. Environ. 1991, 34, 261–267. [Google Scholar] [CrossRef]
- DoVale, J.C.; Fritsche-Neto, R. Root Phenomics. In Phenomics: How Next-Generation Phenotyping Is Revolutionizing Plant Breeding; Fritsche-Neto, R., Borém, A., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 49–66. [Google Scholar]
- Varga, B.; Farkas, Z.; Varga-László, E.; Vida, G.; Veisz, O. Elevated atmospheric CO2 concentration influences the rooting habits of winter-wheat (Triticum aestivum L.) Varieties. Sustainability 2022, 14, 3304. [Google Scholar] [CrossRef]
- Gottwald, T.; Aubert, B.; Zhao, X.-Y. Preliminary analysis of Citrus greening (huanglungbin) epidemics in the People’s Republic of China and French Reunion Island. Phytopathology 1989, 79, 687–693. [Google Scholar] [CrossRef]
- Atkinson, D. Root characteristics: Why and what to measure. In Root Methods: A Handbook, Smit, A.L., Bengough, A.G., Engels, C., van Noordwijk, M., Pellerin, S., van de Geijn, S.C., Eds.; Springer: Berlin/Heidelberg, Germany, 2000; pp. 1–32. [Google Scholar]
- Johnson, E.G.; Wu, J.; Bright, D.B.; Graham, J.H. Association of ‘Candidatus Liberibacter asiaticus’ root infection, but not phloem plugging with root loss on huanglongbing-affected trees prior to appearance of foliar symptoms. Plant Pathol. 2014, 63, 290–298. [Google Scholar] [CrossRef]
- Wang, Z.; Yin, Y.; Hu, H.; Yuan, Q.; Peng, G.; Xia, Y. Development and application of molecular-based diagnosis for ‘Candidatus Liberibacter asiaticus’, the causal pathogen of citrus huanglongbing. Plant Pathol. 2006, 55, 99–104. [Google Scholar] [CrossRef]
- Trivedi, P.; Sagaram, U.S.; Kim, J.S.; Brlansky, R.H.; Rogers, M.E.; Stelinski, L.L.; Oswalt, C.; Wang, N. Quantification of viable Candidatus Liberibacter asiaticus in hosts using quantitative PCR with the aid of ethidium monoazide (EMA). Eur. J. Plant Pathol. 2009, 124, 553–563. [Google Scholar] [CrossRef]
- Team, R. Core. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2013. [Google Scholar]
- Huber, D.; Römheld, V.; Weinmann, M. Relationship between nutrition, plant diseases and pests. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed; Academic Press: Cambridge, MA, USA, 2011. [Google Scholar] [CrossRef]
- Razi, M.F.u.D.; Khan, I.A.; Jaskani, M.J. Citrus plant nutritional profile in relation to Huanglongbing prevalence in Pakistan. Pak. J. Agric. Sci. 2011, 48, 299–304. [Google Scholar]
- Graham, J.H.; Johnson, E.G.; Gottwald, T.R.; Irey, M.S. Presymptomatic Fibrous root decline in citrus trees caused by huanglongbing and potential interaction with Phytophthora spp. Plant Dis. 2013, 97, 1195–1199. [Google Scholar] [CrossRef] [PubMed]
- Atta, A.A.; Morgan, K.T.; Hamido, S.A.; Kadyampakeni, D.M. Effect of essential nutrients on roots growth and lifespan of huanglongbing-affected citrus trees. Plants 2020, 9, 483. [Google Scholar] [CrossRef] [PubMed]
- Atta, A.A.; Morgan, K.T.; Mahmoud, K.A. Split application of nutrients improve growth and yield of Huanglongbing-affected citrus trees. Soil Sci. Soc. Am. J. 2021, 85, 2040–2053. [Google Scholar] [CrossRef]
- McCollum, G.; Hilf, M.; Irey, M. Relationship between Ct values, HLB symptoms and CLas titer. J. Citrus Pathol. 2014, 1. [Google Scholar] [CrossRef]
- Gottwald, T.R.; Graham, J.H.; Irey, M.S.; McCollum, T.G.; Wood, B.W. Inconsequential effect of nutritional treatments on huanglongbing control, fruit quality, bacterial titer and disease progress. Crop Prot. 2012, 36, 73–82. [Google Scholar] [CrossRef]
- Da Silva, J.R.; de Alvarenga, F.V.; Boaretto, R.M.; Lopes, J.R.S.; Quaggio, J.A.; Filho, D.C.H.; Mattos, D. Following the effects of micronutrient supply in HLB-infected trees: Plant responses and ‘Candidatus Liberibacter asiaticus’ acquisition by the Asian citrus psyllid. Trop. Plant Pathol. 2020, 45, 597–610. [Google Scholar] [CrossRef]
- Bassanezi, R.B.; Primiano, I.V.; Vescove, H.V. Effect of enhanced nutritional programs and exogenous auxin spraying on huanglongbing severity, fruit drop, yield and economic profitability of orange orchards. Crop Prot. 2021, 145, 105609. [Google Scholar] [CrossRef]
- USDA. Disaster Assistance Tree Assistance Program—Florida Citrus Greening. 2021. Available online: https://www.fsa.usda.gov/Assets/USDA-FSA-Public/usdafiles/FactSheets/tap-florida-citrus-greening-factsheet.pdf (accessed on 17 August 2022).
| Rating | Description |
|---|---|
| 1 | No HLB symptoms |
| 2 | Minor HLB symptoms |
| 3 | Leaf drop present |
| 4 | Leaf drop present in most quadrants |
| 5 | Significant leaf/branch dieback |
| Treatment | Root Count | Root Length (mm) | Root Diameter (mm) |
|---|---|---|---|
| Control | 1.33 ± 1.33 | 2.63 ± 0.01 | 0.76 ± 0.76 |
| CRF1 | 51.00 ± 44.00 | 15.36 ± 2.11 | 1.41 ± 0.36 |
| CRF2 | 14.00 ± 7.09 | 20.84 ± 5.44 | 1.15 ± 0.14 |
| CRF4 | 1.00 ± 1.00 | 10.22 ± 5.40 | 0.36 ± 0.35 |
| F1 | 7.00 ± 6.02 | 9.36 ± 5.40 | 0.34 ± 0.33 |
| F2 | 3.33 ± 1.85 | 9.92 ± 2.40 | 0.97 ± 0.07 |
| F4 | 6.67 ± 2.02 | 12.53 ± 0.94 | 0.52 ± 0.26 |
| FW | 6.67 ± 3.75 | 14.62 ± 7.44 | 0.63 ± 0.31 |
| Treatment | Root Count | Root Length (mm) | Root Diameter (mm) |
|---|---|---|---|
| Control | 0.00 ± 0.00 b | 0.00 ± 0.00 | 0.00 ± 0.00 |
| CRF1 | 3.00 ± 1.15 ab | 19.60 ± 5.95 | 1.22 ± 0.06 |
| CRF2 | 8.66 ± 5.92 ab | 9.76 ± 5.17 | 0.38 ± 0.38 |
| CRF4 | 2.33 ± 1.85 ab | 9.77 ± 4.88 | 0.97 ± 0.09 |
| F1 | 1.00 ± 1.00 ab | 6.64 ± 6.64 | 0.41 ± 0.41 |
| F2 | 3.00 ± 2.51 ab | 22.13 ± 12.76 | 1.01 ± 0.57 |
| F4 | 14.33 ± 4.97 a | 11.01 ± 2.11 | 0.60 ± 0.30 |
| FW | 14.00 ± 3.21 a | 15.07 ± 1.05 | 0.76 ± 0.07 |
| Treatment | Root Count | Root Length (mm) | Root Diameter (mm) |
|---|---|---|---|
| Control | 5.66 ± 0.66 ab | 20.57 ± 0.81 | 1.70 ± 0.07 |
| CRF1 | 46.33 ± 19.47 a | 13.04 ± 6.05 | 4.49 ± 3.47 |
| CRF2 | 15.66 ± 8.95 ab | 8.31 ± 4.17 | 0.59 ± 0.29 |
| CRF4 | 2.00 ± 2.00 b | 8.81 ± 8.80 | 0.38 ± 0.38 |
| F1 | 5.33 ± 1.85 ab | 9.54 ± 2.67 | 0.58 ± 0.32 |
| F2 | 13.33 ± 10.03 ab | 6.22 ± 4.29 | 0.53 ± 0.27 |
| F4 | 10.00 ± 4.16 ab | 14.70 ± 3.48 | 0.67 ± 0.05 |
| FW | 20.00 ± 3.46 ab | 15.95 ± 1.83 | 0.84 ± 0.89 |
| Treatment | Root Count | Root Length (mm) | Root Diameter (mm) |
|---|---|---|---|
| Control | 1.00 ± 1.00 | 6.47 ± 6.47 | 0.31 ± 0.31 |
| CRF1 | 4.67 ± 3.28 | 10.74 ± 9.68 | 6.02 ± 5.48 |
| CRF2 | 2.33 ± 2.33 | 11.28 ± 11.27 | 0.00 ± 0.00 |
| CRF4 | 0.67 ± 0.66 | 10.58 ± 10.58 | 0.50 ± 0.49 |
| F1 | 4.00 ± 2.51 | 17.03 ± 5.65 | 1.09 ± 0.10 |
| F2 | 17.00 ± 13.20 | 6.80 ± 3.80 | 0.39 ± 0.19 |
| F4 | 2.67 ± 0.66 | 17.91 ± 2.82 | 0.85 ± 0.04 |
| FW | 9.00 ± 4.93 | 31.63 ± 14.67 | 0.90 ± 0.14 |
| Treatment | Fruit Weight in 2020 | Fruit Weight in 2021 |
|---|---|---|
| kg tree−1 | ||
| Control | 10.16 ± 1.25 | 4.36 ± 0.84 |
| CRF1 | 14.41 ± 1.91 | 4.37 ± 0.70 |
| CRF2 | 9.49 ± 1.26 | 3.65 ± 0.56 |
| CRF4 | 9.71 ± 1.09 | 3.63 ± 0.58 |
| F1 | 7.17 ± 0.89 | 3.03 ± 0.59 |
| F2 | 8.69 ± 1.51 | 3.06 ± 0.50 |
| F4 | 9.39 ± 1.20 | 5.08 ± 0.88 |
| FW | 11.26 ± 1.29 | 5.47 ± 0.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hallman, L.M.; Kadyampakeni, D.M.; Ferrarezi, R.S.; Wright, A.L.; Ritenour, M.A.; Johnson, E.G.; Rossi, L. Impact of Ground Applied Micronutrients on Root Growth and Fruit Yield of Severely Huanglongbing-Affected Grapefruit Trees. Horticulturae 2022, 8, 763. https://doi.org/10.3390/horticulturae8090763
Hallman LM, Kadyampakeni DM, Ferrarezi RS, Wright AL, Ritenour MA, Johnson EG, Rossi L. Impact of Ground Applied Micronutrients on Root Growth and Fruit Yield of Severely Huanglongbing-Affected Grapefruit Trees. Horticulturae. 2022; 8(9):763. https://doi.org/10.3390/horticulturae8090763
Chicago/Turabian StyleHallman, Lukas M., Davie M. Kadyampakeni, Rhuanito Soranz Ferrarezi, Alan L. Wright, Mark A. Ritenour, Evan G. Johnson, and Lorenzo Rossi. 2022. "Impact of Ground Applied Micronutrients on Root Growth and Fruit Yield of Severely Huanglongbing-Affected Grapefruit Trees" Horticulturae 8, no. 9: 763. https://doi.org/10.3390/horticulturae8090763
APA StyleHallman, L. M., Kadyampakeni, D. M., Ferrarezi, R. S., Wright, A. L., Ritenour, M. A., Johnson, E. G., & Rossi, L. (2022). Impact of Ground Applied Micronutrients on Root Growth and Fruit Yield of Severely Huanglongbing-Affected Grapefruit Trees. Horticulturae, 8(9), 763. https://doi.org/10.3390/horticulturae8090763










