Comparing Different Methods for Pruning Pitaya (Hylocereus undatus)
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Crop Management
2.2. Treatments and Experimental Design
2.3. Measured Parameters: Vegetative Growth, Flowering and Fruit Quality
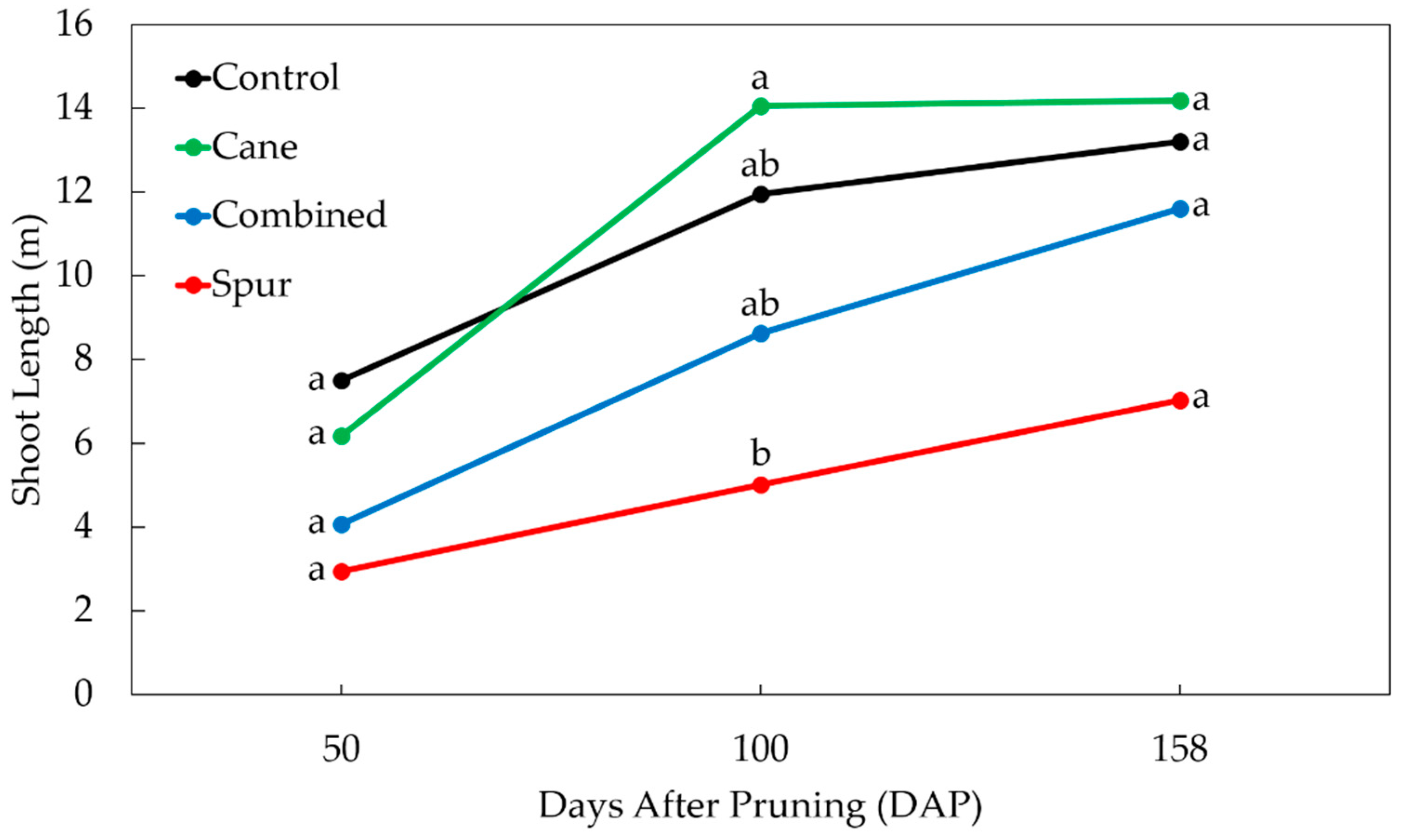
3. Results
3.1. Vegetative Growth
3.2. Flowering
3.3. Fruit Quality and Yield
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Angonese, M.; Motta, G.E.; Farias, N.S.; Molognoni, L.; Daguer, H.; Brugnerotto, P.; Costa, A.C.O.; Müller, C.M.O. Organic dragon fruits (Hylocereus undatus and Hylocereus polyrhizus) grown at the same edaphoclimatic conditions: Comparison of phenolic and organic acids profiles and antioxidant activities. LWT-Food Sci. Technol. 2021, 149, 111924. [Google Scholar] [CrossRef]
- Luu, T.T.H.; Le, T.L.; Huynh, N.; Quintela-Alonso, P. Dragon fruit: A review of health benefits and nutrients and its sustainable development under climate changes in Vietnam. Czech J. Food Sci. 2021, 39, 71–94. [Google Scholar] [CrossRef]
- Nerd, A.; Mizrahi, Y. Reproductive biology of cactus fruit crops. In Horticultural Reviews; Janick, J., Ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1996; Volume 18, pp. 321–346. [Google Scholar] [CrossRef]
- Weiss, J.; Nerd, A.; Mizrahi, Y. Flowering behavior and pollination requirements in climbing cacti with fruit crop potential. HortScience 1994, 29, 1487–1492. [Google Scholar] [CrossRef]
- Rabelo, J.M.; Cruz, M.C.M.; Alves, D.A.; Lima, J.E.; Reis, L.A.; Santos, N.C. Reproductive phenology of yellow pitaya in a high-altitude tropical region in Brazil. Acta Sci.-Agron. 2020, 42, e43335. [Google Scholar] [CrossRef]
- Le Bellec, F.; Vaillant, F.; Imbert, E. Pitahaya (Hylocereus spp.): A new fruit crop, a market with a future. Fruits 2006, 61, 237–250. [Google Scholar] [CrossRef]
- Costa, A.C.; Ramos, J.D.; Silva, F.O.R.; Duarte, M.H. Floração e frutificação em diferentes tipos de cladódios de pitaia vermelha em Lavras-MG. Rev. Bras. Frutic. 2014, 36, 279–284. [Google Scholar] [CrossRef]
- Jiang, Y.L.; Liao, Y.Y.; Lin, T.S.; Lee, C.L.; Yen, C.R.; Yang, W.J. The photoperiod-regulated bud formation of red pitaya (Hylocereus sp.). HortScience 2012, 47, 1063–1067. [Google Scholar] [CrossRef]
- Chu, Y.C.; Chang, J.C. Regulation of floral bud development and emergence by ambient temperature under a long-day photoperiod in white-fleshed pitaya (Hylocereus undatus). Sci. Hortic. 2020, 271, 109479. [Google Scholar] [CrossRef]
- Xiong, R.; Liu, C.; Xu, M.; Wei, S.S.; Huang, J.Q.; Tang, H. Transcriptomic analysis of flower induction for long-day pitaya by supplementary lighting in short-day winter season. BMC Genom. 2020, 21, 329. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, R.; Tollsten, L. An introduction to the scent of cacti. Flavour Frag. J. 1995, 10, 153–164. [Google Scholar] [CrossRef]
- Westwood, M.N. Temperate-Zone Pomology: Physiology and Culture, 3rd ed.; Timber Press: Portland, OR, USA, 1993. [Google Scholar]
- Gunasena, H.P.M.; Pushpakumara, D.K.N.G.; Kariyawasam, M. Dragon fruit: Hylocereus undatus (Haw.) Britton and Rose. In Underutilized Fruit Trees in Sri Lanka; Pushpakumara, D.K.N.G., Gunasena, H.P.M., Singh, V.P., Eds.; World Agroforestry Centre, South Asia Office: New Delhi, India, 2007; Volume 1, pp. 110–141. [Google Scholar]
- Papadakis, J. El Clima: Con Especial Referencia a los Climas de América Latina, Península Ibérica, Ex-Colonias Ibéricas y sus Potencialidades Agropecuarias; Albatros: Buenos Aires, Argentina, 1980. [Google Scholar]
- Nerd, A.; Sitrit, Y.; Kaushik, R.A.; Mizrahi, Y. High summer temperatures inhibit flowering in vine pitaya crops (Hylocereus spp.). Sci. Hortic. 2002, 96, 343–350. [Google Scholar] [CrossRef]
- Barbosa, J.C.; Maldonado Júnior, W. Experimentação Agronômica & AgroEstat: Sistema para Análises Estatísticas de Ensaios Agronômicos, 1st ed.; Multipress: Jaboticabal, Brazil, 2015. [Google Scholar]
- Crane, J.H.; Balerdi, C.F. Pitaya (Dragon Fruit) Growing in the Florida Home Landscape; Series of the Horticultural Sciences Department, UF/IFAS Extension (HS1068); The University of Florida: Gainesville, FL, USA, 2005; Available online: https://edis.ifas.ufl.edu/publication/HS303 (accessed on 10 June 2022).
- Sánchez, F.P. Evaluación de un Cultivo de Pitaya en la Pedanía de La Atalaya en Mazarrón; Universidad Politécnica de Cartagena: Cartagena, Spain, 2020; Available online: http://hdl.handle.net/10317/8525 (accessed on 30 May 2022).
- Strik, B.C.; Davis, A.J. Growing Kiwifruit: A Guide to Kiwiberries and Fuzzy Kiwifruit for Pacific Northwest Producers; Pacific Northwest Extension Publishing, Oregon State University: Corvallis, OR, USA, 2021; Available online: https://catalog.extension.oregonstate.edu/pnw507 (accessed on 20 June 2022).
- Marodin, F.A.; Souza, P.V.D.; Silveira, S.V.; Guasso, L.Z.; Lazarotto, M.; Sassi, A. Vegetative and productive behavior of kiwifruit ‘Elmwood’ submitted to pruning with different bud loading levels. Rev. Bras. Frutic. 2018, 40, e068. [Google Scholar] [CrossRef]
- Feitosa, C.A.M.; Mesquita, A.C.; Pavesi, A.; Ferreira, K.M.; Feitosa, C.V.M. Bud load management on table grape yield and quality—Cv. Sugrathirteen (Midnight Beauty®). Bragantia 2018, 77, 577–589. [Google Scholar] [CrossRef]
- Vaillant, F.; Perez, A.; Davila, I.; Dornier, M.; Reynes, M. Colorant and antioxidant properties of red-purple pitahaya (Hylocereus sp.). Fruits 2005, 60, 3–12. [Google Scholar] [CrossRef]
- Ochoa-Velasco, C.E.; García-Vidal, V.; Luna-Guevara, J.J.; Luna-Guevara, M.L.; Hernández-Carranza, P.; Guerrero-Beltrán, J.Á. Características antioxidantes, fisicoquímicas y microbiológicas de jugo fermentado y sin fermentar de tres variedades de pitahaya (Hylocereus spp). Sci. Agropecu. 2012, 3, 279–289. [Google Scholar] [CrossRef][Green Version]
- Warusavitharana, A.J.; Peiris, K.H.S.; Wickramatilake, K.M.D.G.; Ekanayake, A.T.; Hettiarachchi, H.A.D.S.; Bamunuarachchi, J. Performance of dragon fruit (Hylocerus undatus) in the low country wet zone (LCWZ) of Sri Lanka. Acta Hortic. 2017, 1178, 31–34. [Google Scholar] [CrossRef]
- Mercado-Silva, E.M. Pitaya—Hylocereus undatus (Haw). In Exotic Fruits; Rodrigues, S., Silva, E.O., Brito, E.S., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 339–349. [Google Scholar] [CrossRef]
- Chien, Y.C.; Chang, J.C. Net houses effects on microclimate, production, and plant protection of white-fleshed pitaya. HortScience 2019, 54, 692–700. [Google Scholar] [CrossRef]
- Goenaga, R.; Marrero, A.; Pérez, D. Yield and fruit quality traits of dragon fruit cultivars grown in Puerto Rico. HortTechnology 2020, 30, 803–808. [Google Scholar] [CrossRef]



| Treatments | Number of New Shoots | New Shoots Length | ||||
|---|---|---|---|---|---|---|
| Days after Pruning (DAP) | ||||||
| 50 | 100 | 158 | 50 | 100 | 158 | |
| Control | 2.0 a | 2.3 a | 2.4 a | 30.9 a | 42.9 a | 47.0 a |
| Cane | 1.8 a | 2.2 a | 2.3 a | 27.1 a | 53.1 a | 52.0 a |
| Combined | 1.1 a | 1.4 ab | 1.5 ab | 32.2 a | 52.4 a | 69.2 a |
| Spur | 1.0 a | 1.1 b | 1.1 b | 21.8 a | 48.5 a | 72.2 a |
| p | 0.0854 | 0.0281 | 0.0250 | 0.1391 | 0.2951 | 0.2863 |
| Treatments | Days after Pruning (DAP) | ||||
|---|---|---|---|---|---|
| 80 | 110 | 136 | 165 | Total | |
| Control | 0.5 a | 0.5 ab | 0.4 a | 0.1 a | 18.8 ab |
| Cane | 0.7 a | 0.6 a | 0.3 a | 0.1 a | 20.3 a |
| Combined | 0.2 ab | 0.5 ab | 0.1 a | 0.1 a | 11.0 b |
| Spur | 0.0 b | 0.1 b | 0.1 a | 0.0 a | 2.5 c |
| p | 0.0049 | 0.0447 | 0.1480 | 0.6158 | <0.0001 |
| Treatments | Fruits/Cladode | Weight (g) | Equatorial Diameter (mm) | Length (mm) | TSS (°Brix) |
|---|---|---|---|---|---|
| Control | 0.5 a | 472.5 a | 85.3 a | 120.9 a | 13.6 a |
| Cane | 0.7 a | 434.7 a | 85.6 a | 115.1 a | 13.7 a |
| Combined | 0.2 ab | 498.5 a | 87.8 a | 119.1 a | 13.9 a |
| Spur | 0.0 b | – | – | – | – |
| p | 0.0071 | 0.7283 | 0.9290 | 0.7292 | 0.5535 |
| Treatments | Fruits/Cladode | Weight (g) | Equatorial Diameter (mm) | Length (mm) | TSS (°Brix) |
|---|---|---|---|---|---|
| Control | 0.4 a | 408.1 a | 76.2 a | 125.1 a | 10.3 a |
| Cane | 0.3 a | 369.4 a | 73.6 a | 111.1 a | 11.2 a |
| Combined | 0.2 a | 417.1 a | 77.0 a | 106.5 a | 11.3 a |
| Spur | 0.2 a | 384.5 a | 77.8 a | 113.3 a | 12.4 a |
| p | 0.2382 | 0.8261 | 0.7828 | 0.4484 | 0.4184 |
| Treatments | Fruits/Cladode | Weight (g) | Equatorial Diameter (mm) | Length (mm) | TSS (°Brix) |
|---|---|---|---|---|---|
| Control | 0.2 a | 374.5 a | 75.4 a | 102.7 a | 16.2 a |
| Cane | 0.2 a | 326.0 a | 75.9 a | 95.0 a | 14.3 a |
| Combined | 0.3 a | 515.2 a | 87.6 a | 114.3 a | 14.9 a |
| Spur | 0.2 a | 400.4 a | 75.1 a | 102.6 a | 15.0 a |
| p | 0.2704 | 0.4526 | 0.4197 | 0.5902 | 0.6528 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arredondo, E.; Chiamolera, F.M.; Casas, M.; Cuevas, J. Comparing Different Methods for Pruning Pitaya (Hylocereus undatus). Horticulturae 2022, 8, 661. https://doi.org/10.3390/horticulturae8070661
Arredondo E, Chiamolera FM, Casas M, Cuevas J. Comparing Different Methods for Pruning Pitaya (Hylocereus undatus). Horticulturae. 2022; 8(7):661. https://doi.org/10.3390/horticulturae8070661
Chicago/Turabian StyleArredondo, Emilio, Fernando M. Chiamolera, Marina Casas, and Julián Cuevas. 2022. "Comparing Different Methods for Pruning Pitaya (Hylocereus undatus)" Horticulturae 8, no. 7: 661. https://doi.org/10.3390/horticulturae8070661
APA StyleArredondo, E., Chiamolera, F. M., Casas, M., & Cuevas, J. (2022). Comparing Different Methods for Pruning Pitaya (Hylocereus undatus). Horticulturae, 8(7), 661. https://doi.org/10.3390/horticulturae8070661







