1. Introduction
Sales of vegetable bedding plants for home gardening increased from USD 107 million in 2019 to USD 146 million in 2020 [
1]. Although vegetable gardening has steadily gained popularity in recent years [
2], interest in gardening surged in 2020 as quarantine measures of the COVID-19 pandemic drove people to spend more time at home. An industry survey reported that independent garden centers grew more vegetables and herbs in 2020 than they did in the two years prior (2018 and 2019), out-competing the production of ornamental annuals and bedding plants due to a significant increase in consumer demand [
3]. Furthermore, national sales for garden retailers more than doubled in 2020 due to the heightened interest in gardening activities during the pandemic [
4]. Over 80% of new gardeners are expected to continue to support this growing market trend [
5]. These findings illustrate a significant opportunity for the horticulture industry to continue supplying plant products for gardening enthusiasts.
Vegetable gardening has been primarily an outdoor activity using raised or in-ground beds [
6]. However, increasing urbanization and immigration to cities have promoted the ”urban gardening” trend, where consumers engage in small-scale edible plant production in residential spaces [
7]. In urban sites that lack accessible outdoor garden space, consumers typically grow plants in containers placed on balconies or patios, or they use hydroponic systems indoors, enabling year-round gardening. Despite the increase in popularity, small-scale vegetable gardening has received limited research attention compared with commercial-scale plant production. With the increasing interest in urban gardening, research is needed to identify compact, high-yielding vegetables that will thrive in space-limited indoor and outdoor environments.
Although salad greens and herbs are the most popular crop types for indoor gardening, interest in growing tomato (
Solanum lycopersicum L.) plants indoors is increasing. Tomato is the second most-consumed vegetable in the United States (US), and the most popular outdoor gardening vegetable, cultivated in 86% of residential food gardens [
8,
9,
10]. However, the management and environmental requirements of tomato plants can make production challenging for novice gardeners, as they generally have longer production cycles, higher nutritional demands, and require more light than salad greens and herbs [
7].
Tomato cultivars commonly used for gardening have been bred for outdoor growing, and, thus, their performance in indoor environments is largely unknown. Breeding priorities for most fruiting vegetables often include high yields (fruit number or fresh mass) and consistent product quality under biotic and abiotic outdoor stresses [
11]. Researchers have proposed that breeding priorities for indoor plant production should include rapid, compact growth and high yields under light-limited conditions [
11,
12]. However, considering that motivations for consumers who engage in gardening activities often center on the appreciation of growing plants, access to fresh produce, and the perceived health benefits [
13], key characteristics of compact vegetables that cater to indoor gardeners may differ from those used for commercial plant production.
Based on recent posts in popular online communities, indoor gardeners seem to prefer compact, resilient plants with attractive morphologies and flavorful fruit. In addition, a lack of susceptibility to physiological disorders like intumescence, common in tomato plants grown in environments that lack ultraviolet (UV) radiation (100 to 400 nm), is likely to be important to ensure consumer success and repeat purchases of plant material. Intumescence causes hypertrophic lesions that sometimes senesce and produce necrotic spots on the leaves or stems of susceptible plants [
14]; therefore, tomato cultivars that are susceptible to intumescence could potentially lead to negative gardening experiences.
The objective of this study was to characterize the growth and productivity of various compact tomato cultivars grown in two environments. A greenhouse was used to simulate patio or backyard growing conditions under sunlight, characterized by having a fluctuating environment throughout the day and during the growing season. An indoor environment was used to simulate a residential space characterized by having a constant daily light integral (DLI), cool-to-moderate temperature, and moderate relative humidity (RH). We also aimed to screen for intumescence injury, as levels of susceptibility were expected to vary in the different tomato cultivars. We hypothesized that growth and productivity would be higher in the greenhouse, as plants would be exposed to environmental conditions that promote active growth. We also hypothesized that intumescence would be more severe and affect more cultivars grown indoors compared to those in the greenhouse, primarily due to the lack of UV radiation indoors.
2. Materials and Methods
Two experimental runs were conducted in this study. In the first run, seeds of nine tomato cultivars were sown on 15 January 2020. In the second run, seeds of 20 tomato cultivars were sown on 6 October 2020.
Table 1 lists all cultivars evaluated in this study, with the corresponding abbreviations used hereafter. This includes all available cultivars from commercial breeders at the time the study was conducted. All seeds were sown into individual partial plug trays (55 mL individual cell volume) divided into 5 × 5 cell sections filled with horticultural grade substrate composed of (
v/
v) 79% to 87% peat moss, 10% to 14% perlite, and 3% to 7% vermiculite (Pro-Mix BX general purpose; Premier Tech Horticulture; Quakertown, PA, USA). Seedlings were propagated in a passively ventilated polycarbonate greenhouse with retractable shade curtains in Gainesville, Florida (30° N latitude). In the first experimental run, seedlings were grown under supplemental lighting delivered by 430 watt high-pressure sodium lamps (P.L. Light Systems; Beamsville, ON, USA) used for 12 h·d
–1 (0700 to 1900 HR). Seedlings in both experimental runs were fertigated as needed with a complete fertilizer solution (Peter’s Professional 15-5-15; ICL Specialty Fertilizer; Summerville, SC, USA) providing (mg·L
–1) 100 nitrogen (N), 15 phosphorus (P), 83 potassium (K), 13 magnesium (Mg), 33 calcium (Ca), 0.50 iron (Fe), 0.25 manganese (Mn), 0.12 boron (B), 0.12 copper (Cu), 0.05 molybdenum (Mo), and 0.25 zinc (Zn). A weather station (Watchdog 2400 Mini Station; Spectrum Technologies; Aurora, IL, USA) and datalogger (HOBO Micro Station; Onset; Bourne, MA, USA) were used in the first and second experimental runs, respectively, to record ambient temperature, RH (second run only), and DLI. In the first experimental run, the average daily temperature and DLI during propagation were (mean ± standard deviation) 20 ± 2 °C and 12 ± 5 mol·m
–2·d
–1, respectively. In the second experimental run, average daily temperature, RH, and DLI during propagation were 24 ± 3 °C, 87 ± 12%, and 12 ± 4 mol·m
–2·d
–1, respectively.
Seedlings were transplanted on 18 February 2020 and 11 November 2020 in the first and second experimental runs, respectively. Sixteen uniform seedlings of all cultivars were selected to be used in each experimental run, and were planted as one seedling per 8 inch diameter (20.3 cm) ‘azalea’ plastic container (3.1 L) (BWI; Nash, TX, USA) and filled with the same substrate described above. A 2.5 cm layer of parboiled rice hulls (Sungro; Agawam, MA, USA) was applied to the substrate surface of each container to minimize complications with fungus gnats [
15]. Periodical applications of biological control products such as NemaShield
® were used as preventative measures for pest control.
After transplanting, eight plants of each cultivar were moved to one of the two environments and each replicate plant was considered an experimental unit. In the greenhouse, the experiment was arranged as a completely randomized design. Plants in the first experimental run were randomly placed on four 1.8 m wide × 7.6 m long metallic benches located in a passively ventilated polycarbonate-glazed greenhouse with unit heaters. In the second experimental run, plants were randomly placed on four 1.8 m wide × 4.6 m long metallic benches located in another polycarbonate-glazed greenhouse with unit heaters and pad-and-fan evaporative cooling. In both experimental runs, plants in the greenhouse were spaced 46 cm apart and fertigated as needed with drippers using the same fertilizer solution previously described, providing 150 mg·L
–1 N. A single datalogger (HOBO Micro Station; Onset) was used in the first experimental run, placed at above-canopy height in a central bench within the experimental area. In the second experimental run, temperature and RH probes (HMP60-L; Campbell Scientific; Logan, UT, USA) and quantum sensors (SQ512; Apogee Instruments Inc.; Logan, UT, USA) interfaced to a data logger (CR1000 with AM16/32B multiplexer; Campbell Scientific) were placed at above-canopy height in the center of each bench. Measurements were recorded at 60 min intervals. In the first and second experimental run, respectively, average daily temperature, RH, and DLI recorded in the greenhouse were 24 ± 5 °C and 22 ± 3 °C, 61 ± 19% and 68 ± 16%, and 14 ± 2 mol·m
–2·d
–1 and 12 ± 4 mol·m
–2·d
–1, respectively. The day and night temperature and DLI for each experimental run are shown in
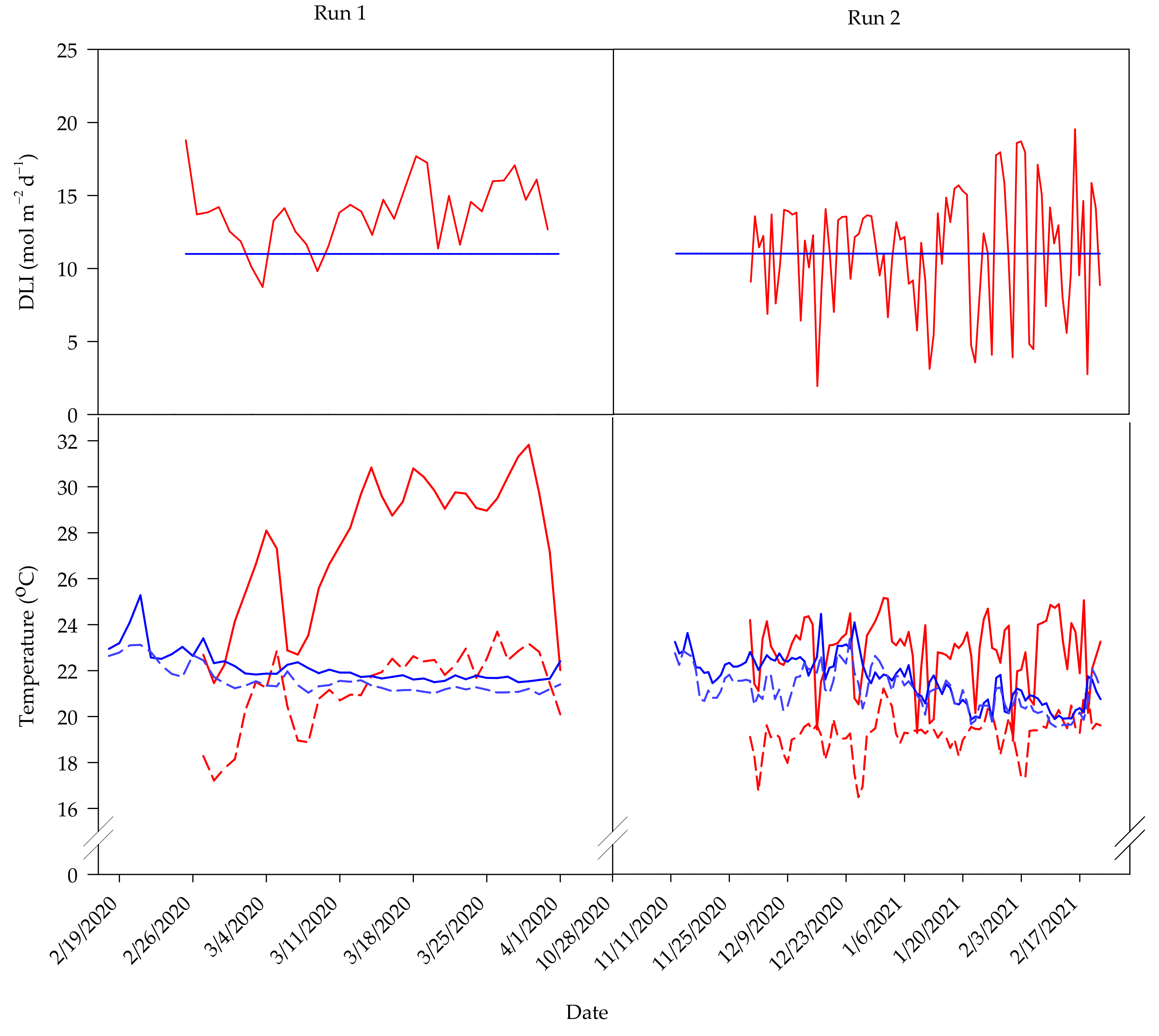
Figure 1.
The indoors experiment was arranged as a randomized complete block design with four blocks, each containing two replicate plants per cultivar spaced 30 cm apart. Due to the different number of plants grown in both experimental runs, one and two 12 m2 air-conditioned growth rooms were used in the first and second runs, respectively. Each room had two opposite shelving units. Each shelving unit had an upper and a lower compartment (91.4 cm height × 91.4 cm width × 365.8 cm length) lined with insulation foam at the bottom. In the first run, each compartment within a shelving unit was regarded as a block. In the second run, two compartments within a shelving unit (upper and lower) were regarded as a block.
Four broadband white LED fixtures (RAY66 PhysioSpec Indoor TM; Fluence Bioengineering; Austin, TX, USA) were placed in each compartment to provide a DLI of 11 mol·m–2·d–1 (220 µmol·m–2·s–1; 14 h·d–1 photoperiod from 0900 to 2300 HR), which was selected based on preliminary studies evaluating the growth and yield of compact vegetables under different DLIs. Before starting the experiments, a lightmap was generated using a spectroradiometer (SS-110; Apogee Instruments Inc.) placed at mid-canopy height (46 cm from the compartment surface), with every fixture turned on to account for light pollution. To achieve the target photosynthetic photon flux density (PPFD), the light output was controlled with dimmers (Solunar; Fluence Bioengineering) connected to a backup battery (BE425M-LM; APCAPC, West Kingston, RI, USA).
Growth rooms were kept at a constant ambient temperature of 22 ± 2 °C. Near-canopy air temperature and RH were monitored hourly with shielded dataloggers (Elitech RS-4HC; Milpitas, CA, USA) placed in the middle of each compartment. In the first and second experimental runs, respectively, the average daily air temperature and RH recorded indoors were 20 ± 1 °C and 20 ± 2 °C, and 57 ± 20% and 65 ± 13%. The day and night temperature and DLI for each experimental run are shown in
Figure 1.
In both experimental runs, indoor-grown plants were fertigated as needed with the same fertilizer solution described above, providing 150 mg·L–1 N. In the first run, plants were hand watered. In the second run, drip irrigation connected to a fertilizer injector system was used as needed. Once flowering started in both experimental runs, plants were hand-pollinated using a vibrating wand every other day from 1100 to 1300 HR.
In both experimental runs, shoot height, longest width (from the endpoints of the two most distal leaves), and perpendicular width were recorded five weeks after transplanting to calculate growth index using the formula π × h × r
2, where h is shoot height and r is calculated by multiplying ½ times the mean of two leaf widths. This was immediately followed by a visual assessment for the development of intumescences using a subjective 1 to 6 scale based on Eguchi et al. [
16], where 1 = no intumescence injury; 2 = 1 to 10% of the plant affected, minimal isolated intumescence on terminal leaves; 3 = 11 to 50% of the plant affected, dense intumescence on the terminal leaflet with pronounced topical necrotic spotting; 4 = 51 to 75% of the plant affected, pronounced upward leaf curling, prolific top leaf surface necrosis; 5 = 76 to 100% of the plant affected, full senescence of leaflets; and 6 = complete abscission/senescence.
The first experimental run was prematurely terminated on 2 April 2020 (six weeks after transplanting) due to a university closure caused by the COVID-19 pandemic. Therefore, days to first harvest, total number of fruit, and total fruit fresh mass data were only recorded for plants grown during the second experimental run. The date of the first harvest was recorded when at least one fruit per plant was fully mature. After that initial harvest, the number of fruit and fruit fresh mass were recorded weekly starting on 21 December 2020. In addition, during the experimental termination on 23 February 2021 (15 weeks after transplanting), the number and fresh mass of mature and immature fruit were recorded for all plants to calculate the total number of fruit and total fruit fresh mass, respectively. Shoots were severed at the substrate surface and shoot dry mass was recorded after drying in a forced-air oven at 70 °C for 4 d. Canopy density was calculated by dividing the shoot dry mass by plant height.
Non-destructive growth data for the nine cultivars grown in both experimental runs were pooled, as the variances between experiments were not different and the statistical interactions between cultivar and experimental run were not significant (
p ≥ 0.05). Intumescence severity for each experimental run is presented separately (n = 8) for the indoor environment only because of the significant experimental run × cultivar interaction (
p ≤ 0.05). Intumescence data is only presented for indoor-grown plants because of the lack of variability in greenhouse-grown plants. Generalized linear mixed model procedures implemented in SAS
® PROC GLIMMIX [
17] were used to determine differences among cultivars within each growing environment. A paired
t-test was then used to assess differences between plants of the same cultivar grown in the greenhouse compared to indoors for all variables, except for intumescence severity. Quantitative, continuous response variables (growth index, canopy density, and total fruit fresh mass) were treated as normally distributed, whereas count variables (days to first harvest, total number of fruits, and intumescence severity) were modeled through the Poisson distribution. A negative binomial transformation was used when the underlying assumption for the Poisson distribution (variance = mean) was not valid (= overdispersion) for a specific variable. Canopy density was modeled through a lognormal distribution. For each variable, the experiment and environment × cultivar interactions were calculated using the LSMEANS statement, which were then compared using the SLICEDIFF option. Cultivars were compared within each environment and environments were compared within each cultivar. No adjustments for multiplicity were made to the calculated
p values, as this was an exploratory/screening experiment [
18,
19]. A linear regression was applied to the quantitative response of total fruit yield and increasing growth index. To evaluate the price of cherry tomatoes per plant, the corresponding average retail price (USD 3.80/lb, adapted from the USDA [
20]) was applied to the second
y-axis of the linear regression. Regressions were analyzed using the R statistical analysis software [
21].
3. Results and Discussion
Overall, greenhouse-grown plants were larger, had a lower canopy density, and yielded more fruit than those grown indoors (
Table 2). These trends are likely attributed to differences in average DLI, daily temperature, and RH between the two environments. Large differences in daily environmental fluctuations could also explain these trends (
Figure 1). For example, plants in the greenhouse were grown under solar DLIs that ranged from 9 to 18 mol·m
–2·d
–1, with average daily temperatures of 19 to 27 °C during the two experimental runs. In contrast, plants indoors were continuously grown under a DLI of 11 mol·m
–2·d
–1 from sole-source lighting with an ambient temperature of 20 ± 2 °C. Greenhouse plants were also exposed to larger differences between day and night temperatures (DIF) than those grown indoors. Further, greenhouse plants received a changing spectral composition throughout the day, as far-red radiation from sunlight tends to be higher in the early morning and late afternoon than during solar noon [
22]. In contrast, indoor plants were grown under broadband white LED fixtures with a fixed spectrum that provided 19% blue, 41% green, and 40% red light and lacked far-red radiation. As shown by Xiong et al. [
23], applying end-of-day far-red radiation and providing a positive temperature DIF increases stem elongation of some vegetable plants. Further, various studies have shown that tomato plants benefit from DIF treatments that enable the translocation of carbohydrates to fruits [
24]. The combined effects of providing a slightly higher DLI, warmer average temperatures, positive DIF, and far-red radiation from sunlight likely contributed to the larger plant size and higher yield measured in the greenhouse plants compared to the indoor plants.
Except for cultivars CY, HBTO, HBTR, MT, MI, and SSJ, growth index was 82% to 402% higher in plants grown in the greenhouse compared to those grown indoors (
Table 2). In the greenhouse, growth index was highest for TZ (0.13 m
3) and LB (0.13 m
3) and lowest for MT (0.006 m
3). Indoors, growth index was highest for LB (0.046 m
3), SSG (0.034 m
3), SSJ (0.036 m
3), and SSJO (0.034 m
3), and lowest for FA (0.005 m
3) and MI (0.006 m
3). Interestingly, growth index was similar for CY, HBTO, HBTR, MT, MI, and SSJ in both environments. These similarities could be attributed to their true compact growth habit, suggesting that growth of these cultivars was maximized under the environmental conditions indoors.
Canopy density, which incorporates shoot dry mass and height, was 31% to 56% lower in plants grown in the greenhouse than indoors for CR, HBTO, HBTR, LB, MT, FA, MI, RE, and SSJO (
Table 2). In the greenhouse, RF, RR, SSG, SSJO, and YC had the largest canopy density, while CR, HBTO, HBTR, MT, FA, MI, and RE had the lowest canopy density. Indoors, MO, RF, SSJ, and SSJO had the highest canopy density, while CR, CY, HBTO, FA, and MI had the lowest canopy density. Plants with a denser canopy often have more foliage per unit area, which tends to be associated with greater plant quality and aesthetic appeal. For example, denser canopies are typically preferred by ornamental and bedding plant consumers [
25,
26]. Based on our findings, indoor gardening can result in a higher canopy density than gardening under sunlight.
There were generally no differences in the number of days to harvest between plants grown in the greenhouse and those indoors (
Table 2). CY, HBTO, HBTR, MT, FA, and MI were the fastest cultivars to produce fruit in the greenhouse, with their first harvest occurring from 47 to 53 d after transplanting. All other cultivars had similar days to first harvest, ranging from 54 to 62 d. Indoors, FA and MI were the first cultivars to produce fruit (55 d after transplanting). All other cultivars grown indoors generally had a similar number of days to first harvest, ranging from 55 to 63 d. Except for TZ, all tomatoes grown in this study were determinate cultivars, which are known to have a bush-like growth habit where shoot elongation stops after a certain point and the stem terminates with a fruit cluster [
26]. Determinate tomato plants are generally categorized into three harvesting periods: early (50 to 60 d), midseason (70 to 80 d), and late (85 to 90 d) harvest [
27]. All plants grown in this study fell under the early harvest category, which is likely a desirable trait for home gardeners who are often eager to start the harvesting phase.
In general, the total number of fruits (mature and immature fruits) was 27% to 107% higher in the greenhouse than indoors (
Table 2). The highest total number of fruits were produced by SSG in the greenhouse (200 fruits) and MT and SSG indoors (98 and 97 fruits, respectively). In both environments, HBTR and FA produced the lowest total number of fruits (39 and 34 fruits in the greenhouse, and 48 and 38 fruit indoors, respectively). Interestingly, MT and MI produced more fruits indoors than in the greenhouse. This discrepancy is likely attributed to the fact that both cultivars were over-irrigated in the greenhouse, as the irrigation frequency was adjusted to prevent general wilting of all plants. As most other cultivars were larger and produced more fruit, these small plants were over-irrigated throughout the trial, which could have hindered their growth and productivity. However, our results could also suggest that for truly compact cultivars like MT and MI, indoor conditions may be better suited to maximizing yield compared to greenhouse conditions.
The total fruit fresh mass (mature and immature fruits) was 45% to 142% higher in the greenhouse than indoors for half of the tomato cultivars (LB, RF, RR, SNS, SNY, SSG, SSJ, SSJO, TZ, and YC) grown in this study (
Table 2). In the greenhouse, LB (1021 g), SSG (1238 g), and TZ (1216 g) produced the highest total fruit fresh mass, which was also highest in LB (563 g), SSG (616 g), TZ (508 g), RF (511 g), S (519 g), and YC (541 g) grown indoors. However, CR, CY, HBTO, HBTR, MT, MO, FA, MI, RE, and S produced a similar total fruit fresh mass in both environments, which tended to be lower than that from other cultivars.
As previously mentioned, plants in the greenhouse were exposed to changing solar DLI, whereas those indoors were exposed to a consistent DLI (
Figure 1). This variability partly explains the differences in fruit yield between plants in the two environments, as tomato yield has been shown to increase with total intercepted radiation [
28,
29]. For example, Cockshull et al. [
29] showed that when shade was applied to greenhouse-grown tomatoes for two weeks during the growing cycle, fruit yield was lower compared to that of unshaded plants. In addition, as plants in our study were larger and produced more fruit in the greenhouse than indoors, they were also fertigated more frequently. A greater availability of water and nutrients plausibly contributed to the larger fruit yield of plants grown in the greenhouse compared to those grown indoors.
Overall, our findings suggest that consumers interested in home gardening are likely to harvest more fruits from compact tomatoes grown under sunlight than those grown indoors using LEDs. Although the more compact cultivars (CR, CY, HBTO, HBTR, MT, FA, MI, and RE) yielded the least fruit (146 to 282 g in the greenhouse and 192 to 285 g indoors), they are likely suitable for indoor gardening purposes, particularly considering that home gardeners are likely to be satisfied by harvesting ≥ 10 tomato fruits per plant (J. Thompson, pers. comm.). On average, these compact cultivars produced between 34 to 64 fruits in the greenhouse, and 38 to 98 fruits indoors. However, the highest-yielding cultivars in both environments were generally the largest plants (
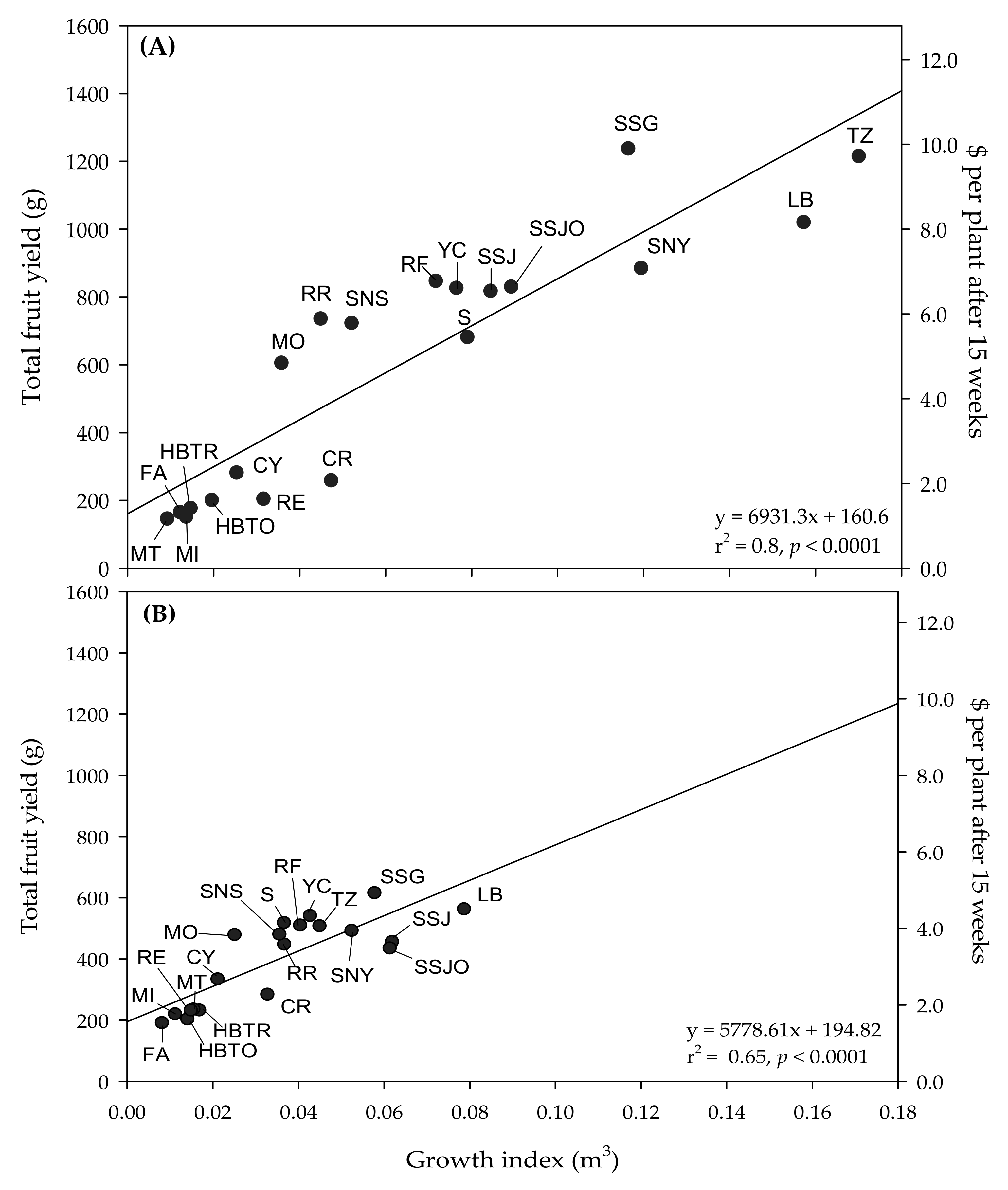
Figure 2), suggesting that to maximize yield, home growers should produce the largest plant that can fit within their desired growing space.
The cultivars LB, RF, SNY, and YC were affected by intumescence indoors, but symptoms for the disorder were more severe in the first compared to the second experimental run (
Table 3). Although the disorder was not severe, symptoms appeared on both upper and lower leaf surfaces. Intumescence has been reported to affect plants grown in greenhouses covered with glass or other glazing materials that block most solar UV [
30]. However, the greenhouse-grown plants in our study were not affected by intumescence. It is likely that the UV transmittance from the polycarbonate glazing [
31] was sufficient to minimize the disorder for susceptible cultivars, as UV has been shown to mitigate intumescence on tomato seedlings [
32,
33].
The variance in intumescence between the two experimental runs could be attributed to differences in plant density, as more plants were grown in the first compared to the second experimental run (four vs. three plants/m
2, respectively). The higher density in the same growing area likely increased RH around plant canopies due to increased transpiration from a larger number of plants [
34]. Based on our observations, tomato plants tended to grow out of this disorder as they grew bigger and fruits matured (data not shown). Similar findings were reported by Mohmmed et al. [
35], who showed that as pepper (
Capsicum annuum L.) plants grew, new leaves were not affected by intumescence. Although effective, providing UV radiation may not be a practical suppression strategy for indoor gardening applications. One potential solution for home gardeners to reduce the severity of this disorder could be to maintain moderate-to-low RH around plant canopies by providing a good source of airflow [
34,
36,
37]. Alternatively, home growers could use cultivars that are not susceptible to intumescence (
Table 3). Although intumescence may not always affect yield, the disorder can decrease the aesthetic quality of plants, which can negatively affect gardening experiences. As indicated by others, an unhealthy-appearing plant may cause a decrease or cessation of gardening activities [
6].
3.1. Implications for the Urban Gardening Market Segment
Others have shown that consumers prefer healthy plants with numerous branches and a dense canopy, qualities that often increase gardening satisfaction and ensure repeat purchases of plant material [
38]. Consequently, cultivars susceptible to intumescence are not recommended until research-based recommendations adaptable to small-scale indoor gardening are made available (
Table 3). All cultivars grown in this study are suitable for outdoor gardening under sunlight. However, the plants of LB, SSG, SSJ, SSJO, and TZ are likely too large for indoor gardening in small growing spaces.
Although no recommendations currently exist for compact vegetables to be sold for home gardening, high-quality ornamental bedding plants are characterized as having many flowers, compact size, numerous branches, and high biomass [
39,
40]. Furthermore, high-quality vegetable transplants are uniform in size and have well-developed leaves and roots, straight stems, and thick, deep-green leaves [
27]. Commercial ornamental or bedding plant growers tend to consider tall plants as being of low quality, as they increase shipping costs and are prone to falling over during shipping and handling [
41]. Additionally, growers are able to fit more plants on shipping racks when they are compact, potentially increasing revenue [
42]. Based on these established criteria of quality for ornamental and bedding plant growers, small (CR, CY, FA, HBTO, HBTR, MI, and MT) or mid-sized (MO, RR, S, SNS, RF, and YC) cultivars that are not prone to falling over and are uniform in size are good candidates for commercial plant production. However, recommendations may differ based on consumer preferences for compact size and acceptable fruit yield.
3.2. Implications for the Commercial Vertical Farming Industry
Our study provides baseline information about indoor plant production of compact tomato cultivars, which could potentially be grown commercially by the vertical farming industry. Compact plants offer advantages for commercial growers, as they help maximize space-use efficiency [
12,
43]. However,
Figure 2 shows that plants from larger cultivars typically yield more fruit. Although compact tomato plants would expectedly yield less than the typical indeterminate cultivar used for commercial greenhouse cultivation, producing a larger number of small plants at a high density could compensate for the reduction in yield per plant for indoor vertical farming applications [
43]. Regardless of environment, CR, CY, FA, HBTO, HBTR, MI, MT, and RE had a small growth index (
Table 2 and
Figure 2), which could make them adequate cultivars for vertical farming applications, considering that compact plants are desirable for these space-limited environments [
12]. However, these cultivars would only produce USD 1.50–2.70 of fruit per plant grown indoors and USD 1.20–2.50 in the greenhouse after a 15 week production period (from transplant to harvest). Instead, mid-sized varieties (MO, RR, S, SNS, RF, and YC) could produce up to USD 4.30 per plant indoors and USD 6.80 in the greenhouse, whereas larger varieties (TZ, SNY, SSG, SSJ, SSJO, and LB) could produce up to USD 4.90 per plant indoors and up to USD 9.70 per plant in the greenhouse, as indicated in
Figure 2. Based on these calculations, decisions for the commercial production of compact tomato plants should consider the economic implication of maximizing space-use efficiency with compact plants that likely have a limited yield potential compared to larger cultivars that will yield more fruit.
Further, compared with leafy greens, which are standard crops for vertical farming applications due to their compact size and rapid crop cycling, tomato fruits can take a long time to be harvested [
12]. Leafy greens typically have a three-to-five week production cycle, depending on species and cultivar. Further, hydroponic lettuce is typically sold for USD 5.00 to 10.00/lb [
42]. With the recent availability of compact tomato cultivars, there is potential for vertical farming applications to diversify their operation. However, based on results for plant size and fruit yield, MO, RR, S, SNS, RF, and YC are the only likely cultivars evaluated in our study that could be suitable for commercial production (
Table 2 and
Table 3), as they can still be considered small plants, but produced a greater yield than other compact varieties (CR, CY, FA, HBTO, HBTR, MI, and MT). Further studies need to be conducted to identify methods that can maximize growth, fruit yield, and fruit quality for commercial vertical farming applications where light, temperature, and fertility are carefully monitored and controlled to maximize profits. Further, breeders interested in identifying cultivars to be grown for commercial indoor production should focus on selecting early harvesting plants that produce numerous high-quality fruits [
11].