Seed Silhouettes as Geometric Objects: New Applications of Elliptic Fourier Transform to Seed Morphology
Abstract
1. Introduction
A Historical Anecdote
“I first consider—and then discard as inadequate—two commonly used representations of form. The first one makes the strong theoretical assumption that the empirical form can be idealized and replaced by a geometric figure.”
2. Geometric Properties of Seed Silhouettes
3. Quantification of Seed Shape by Comparison with Models
3.1. Quantification by Comparison with Known Geometric Figures as Models
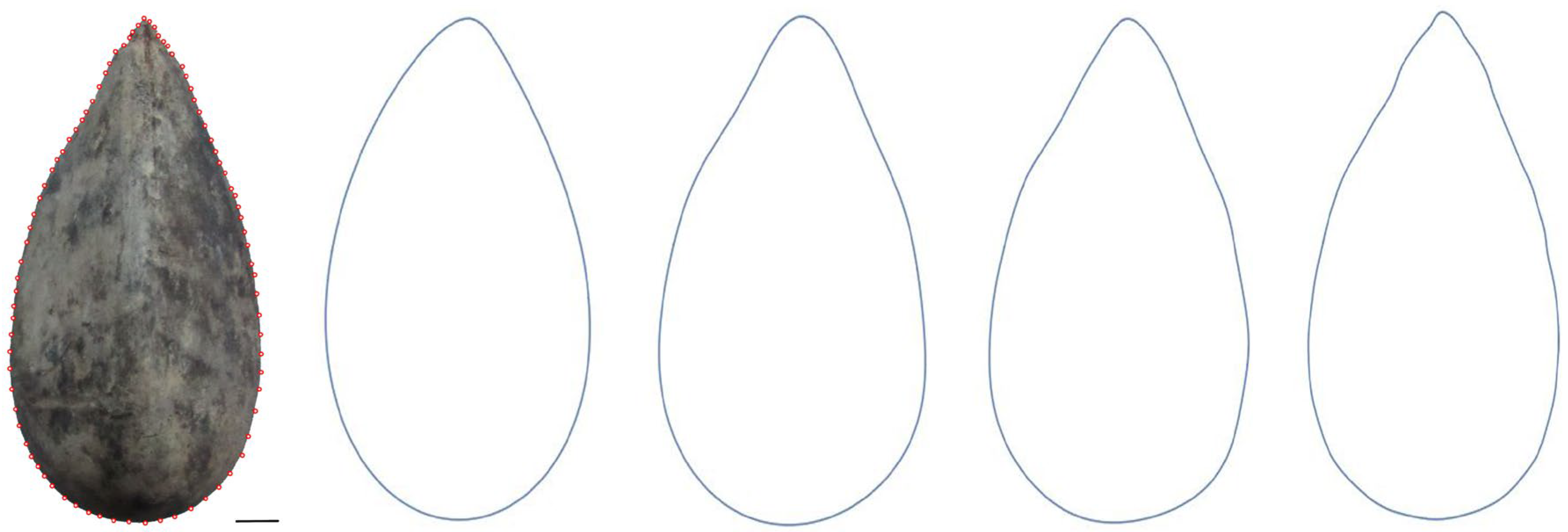
3.2. Closed Curves from Fourier Equations as Models
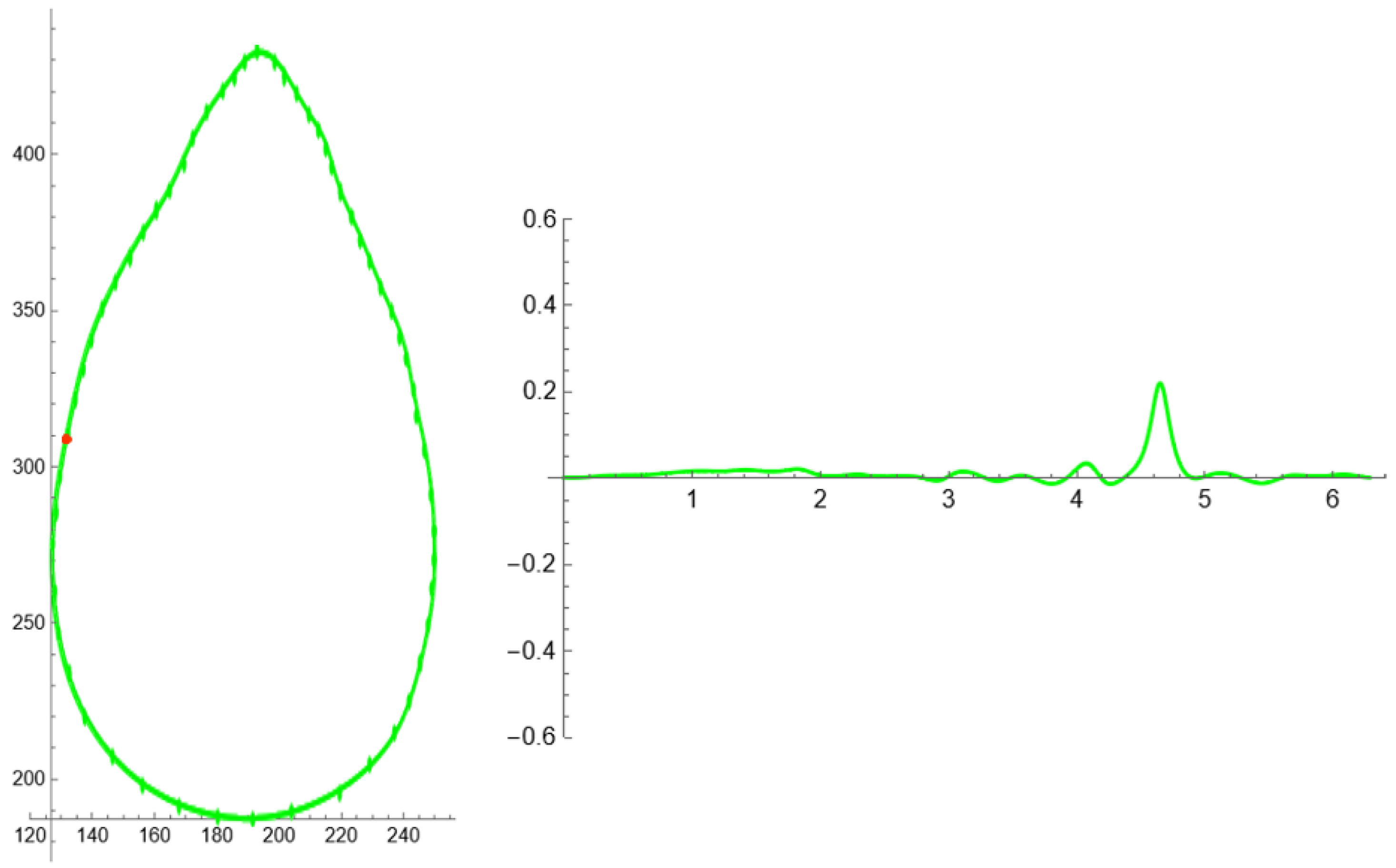
3.3. Curvature Analysis
4. Seed Shape Diversity Linked to Embryogeny and Fruit Development
4.1. Embryogeny
4.2. Variations in Seed Shape Related to Fruit Development
5. Variations in Seed Shape Related to Environmental Adaptations
6. Silene as a Model System for Seed Geometry
6.1. Fourier Analysis of Seeds: Calculation of Equations Corresponding to New Models
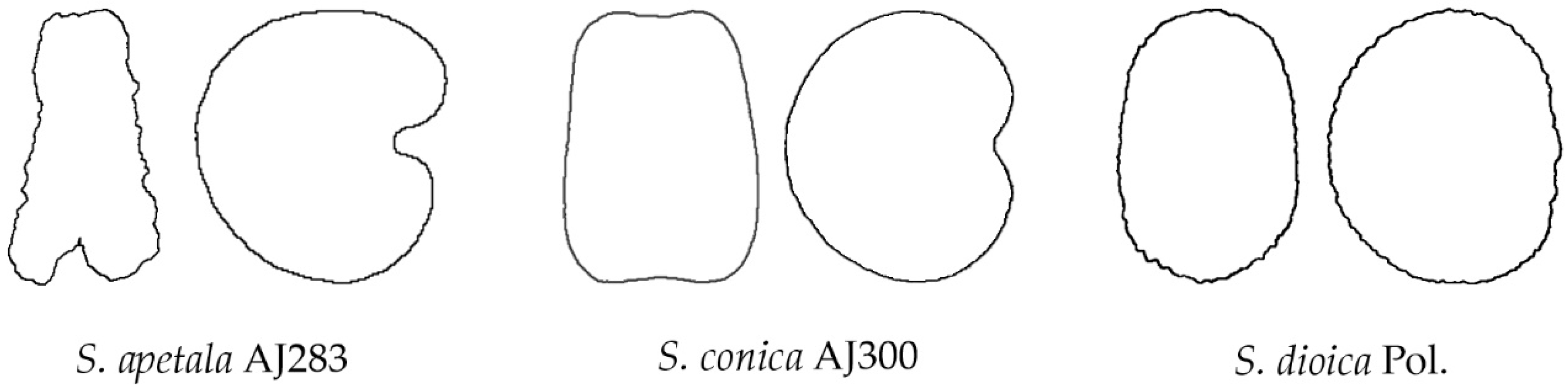
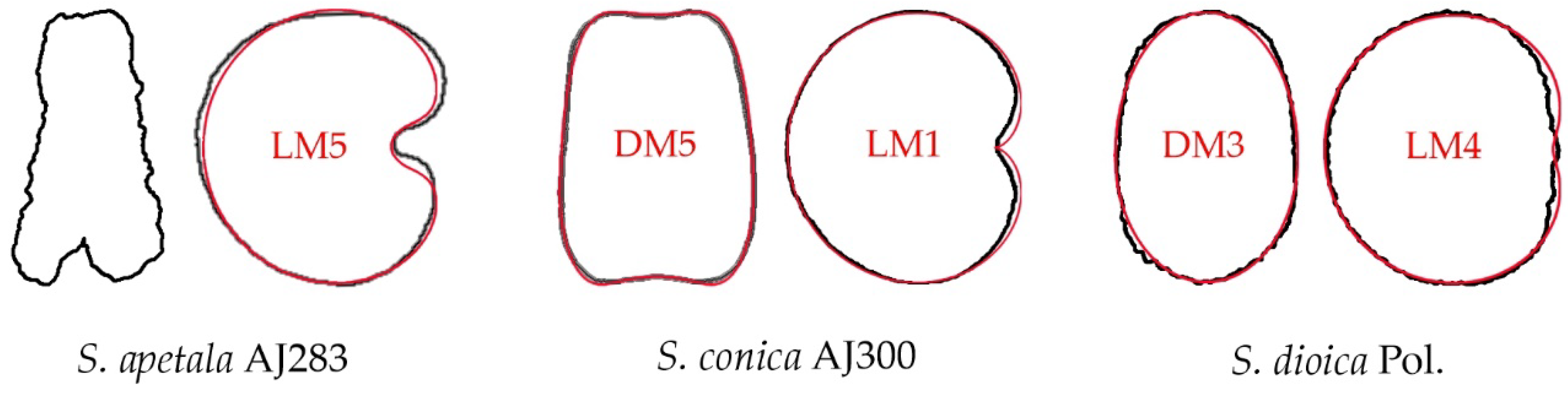
6.2. A Classification According to the Geometric Properties of the Seeds
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Harper, J.L.; Lovell, P.H.; Moore, K.G. The shapes and size of seeds. Ann. Rev. Ecol. Syst. 1970, 1, 327–356. [Google Scholar] [CrossRef]
- Moles, A.T.; Ackerly, D.D.; Tweddle, J.C.; Dickie, J.B.; Smith, R.; Leishman, M.R.; Mayfield, M.M.; Pitman, A.; Wood, J.T.; Westoby, M.; et al. Global patterns in seed size. Glob. Ecol. Biogeogr. 2007, 16, 109–116. [Google Scholar] [CrossRef]
- Ganhão, E.; Dias, L.S. Seed Volume Dataset—An ongoing inventory of seed size expressed by volume. Data 2019, 4, 61. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín Gómez, J.J.; Gutiérrez del Pozo, D.; Silva Dias, L. An angiosperm species dataset reveals relationships between seed size and two-dimensional shape. Horticulturae 2019, 5, 71. [Google Scholar] [CrossRef]
- Martín-Gómez, J.J.; Rodríguez-Lorenzo, J.L.; Juan, A.; Tocino, Á.; Janousek, B.; Cervantes, E. Seed morphological properties related to taxonomy in Silene species. Taxonomy 2022, 2, 298–323. [Google Scholar] [CrossRef]
- Martín-Gómez, J.J.; Rewicz, A.; Rodríguez-Lorenzo, J.L.; Janoušek, B.; Cervantes, E. Seed morphology in Silene based on geometric models. Plants 2020, 9, 1787. [Google Scholar] [CrossRef]
- Juan, A.; Martín-Gómez, J.J.; Rodríguez-Lorenzo, J.L.; Janoušek, B.; Cervantes, E. New techniques for seed shape description in Silene species. Taxonomy 2022, 2, 1–19. [Google Scholar] [CrossRef]
- Rodríguez-Lorenzo, J.L.; Martín-Gómez, J.J.; Tocino, Á.; Juan, A.; Janoušek, B.; Cervantes, E. New geometric models for shape quantification of the dorsal view in seeds of Silene species. Plants 2022, 11, 958. [Google Scholar] [CrossRef]
- Martín-Gómez, J.J.; Porceddu, M.; Bacchetta, G.; Cervantes, E. Seed morphology in species from the Silene mollissima aggregate (Caryophyllaceae) by comparison with geometric models. Plants 2022, 11, 901. [Google Scholar] [CrossRef]
- Stuppy, W.; Kesseler, R. Seeds: Time Capsules of Life; Papadakis Publishers: London, UK, 2006. [Google Scholar]
- Del Pozo, D.G.; Martín-Gómez, J.J.; Tocino, Á.; Cervantes, E. Seed geometry in the Arecaceae. Horticulturae 2020, 6, 64. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín-Gómez, J.J.; Gutiérrez Del Pozo, D.; Tocino, Á. Seed geometry in the Vitaceae. Plants 2021, 10, 1695. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, E.; Martín Gómez, J.J. Seed shape description and quantification by comparison with geometric models. Horticulturae 2019, 5, 60. [Google Scholar] [CrossRef]
- Hannachi, H.; Martín-Gómez, J.J.; Saadaoui, E.; Cervantes, E. Stone diversity in wild and cultivated olive trees (Olea europaea L.). Dendrobiology 2017, 77, 19–32. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín-Gómez, J.J. Seed shape quantification in the order Cucurbitales. Mod. Phytomorphol. 2018, 12, 1–13. [Google Scholar]
- Martín-Gómez, J.J.; Saadaoui, E.; Cervantes, E. Seed shape of castor bean (Ricinus communis L.) grown in different regions of Tunisia. J. Agric. Ecol. 2016, 8, 1–11. [Google Scholar]
- Saadaoui, E.; Martín-Gómez, J.J.; Bouazizi, R.; Ben Romdhane, C.; Grira, M.; Abdelkabir, S.; Khouja, M.L.; Cervantes, E. Phenotypic variability and seed yield of Jatropha curcas L. Introducen to Tunisia. Acta Bot. Mex. 2015, 110, 119–134. [Google Scholar] [CrossRef]
- Shamrov, I.I. Diversity and typification of ovules in flowering plants. Wulfenia 2018, 25, 81–109. [Google Scholar]
- Davis, G.L. Systematic Embryology of the Angiosperms; John Wiley &Sons, Inc.: Hoboken, NJ, USA; London, UK; Sydney, Australia, 1966; p. 528. [Google Scholar]
- Johri, B.M.; Ambegaokar, K.B.; Srivastava, P.S. Comparative Embryology of Angiosperms; Springer: Heidelberg, Germany, 1992; Volume 1. [Google Scholar]
- Martin, A.C. The comparative internal morphology of seeds. Am. Midl. Nat. 1946, 36, 513–660. [Google Scholar] [CrossRef]
- Read, D.W. From multivariate to qualitative measurement: Representation of shape. Hum. Evol. 1990, 5, 417–429. [Google Scholar] [CrossRef]
- Gray, A. Modern Differential Geometry of Curves and Surfaces with Mathematica; CRC Press: Boca Raton, FL, USA, 1998; pp. 163–165. [Google Scholar]
- Calero, E.; West, S.H.; Hinson, K. Water absorption of soybean seeds and associated causal factors. Crop Sci. 1981, 21, 926–933. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín Gómez, J.J.; Saadaoui, E. Updated methods for seed shape analysis. Scientifica 2016, 2016, 5691825. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, E.; Martin-Gómez, J.J.; Chan, P.K.; Gresshoff, P.M.; Tocino, A. Seed shape in model legumes: Approximation by a cardioid reveals differences in ethylene insensitive mutants of Lotus japonicus and Medicago truncatula. J. Plant Physiol. 2012, 169, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Saadaoui, E.; Martín-Gómez, J.J.; Cervantes, E. Intraspecific variability of seed morphology in Capparis spinosa L. Acta Biol. Crac. Ser. Bot. 2013, 55, 99–106. [Google Scholar] [CrossRef][Green Version]
- Saadaoui, E.; Martín-Gómez, J.J.; Tlili, E.N.; Khaldi, A.; Cervantes, E. Effect of climate in seed diversity of wild Tunisian Rhus tripartita (Ucria) Grande. J. Adv. Biol. Biotechnol. 2017, 13, 1–10. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín, J.J.; Ardanuy, R.; De Diego, J.G.; Tocino, Á. Modeling the Arabidopsis seed shape by a cardioid: Efficacy of the adjustment with a scale change with factor equal to the Golden Ratio and analysis of seed shape in ethylene mutants. J. Plant Physiol. 2010, 167, 408–410. [Google Scholar] [CrossRef]
- Martín Gómez, J.J.; Tocino, Á.; Ardanuy, R.; de Diego, J.G.; Cervantes, E. Dynamic analysis of Arabidopsis seed shape reveals differences in cellulose mutants. Acta Physiol. Plant. 2014, 36, 1585–1592. [Google Scholar] [CrossRef]
- Martín-Gómez, J.J.; Gutiérrez del Pozo, D.; Ucchesu, M.; Bacchetta, G.; Cabello Sáenz de Santamaría, F.; Tocino, Á.; Cervantes, E. Seed morphology in the Vitaceae based on geometric models. Agronomy 2020, 10, 739. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín-Gómez, J.J.; Espinosa-Roldán, F.E.; Muñoz-Organero, G.; Tocino, Á.; Cabello-Sáenz de Santamaría, F. Seed Morphology in Key Spanish Grapevine Cultivars. Agronomy 2021, 11, 734. [Google Scholar] [CrossRef]
- Martín-Gómez, J.J.; del Pozo, D.G.; Tocino, Á.; Cervantes, E. Geometric models for seed shape description and quantification in the Cactaceae. Plants 2021, 10, 2546. [Google Scholar] [CrossRef]
- Martín-Gómez, J.J.; Rewicz, A.; Goriewa-Duba, K.; Wiwart, M.; Tocino, Á.; Cervantes, E. Morphological description and classification of wheat kernels based on geometric models. Agronomy 2019, 9, 399. [Google Scholar] [CrossRef]
- Rohlf, F.J.; Archie, J.W. A comparison of Fourier methods for the description of wing shape in mosquitoes (Diptera: Culicidae). Syst. Zool. 1984, 3, 302–317. [Google Scholar] [CrossRef]
- Crampton, J.S. Elliptic Fourier shape analysis of fossil bivalves: Some practical considerations. Letahia 1995, 28, 179–186. [Google Scholar] [CrossRef]
- McLellan, T.; Endler, J.A. The relative success of some methods for measuring and describing the shape of complex objects. Syst. Biol. 1998, 47, 264–281. [Google Scholar] [CrossRef]
- Iwata, H.; Ebana, K.; Uga, Y.; Hayashi, T.; Jannink, J.L. Genome-wide association study of grain shape variation among Oryza sativa L. germplasms based on elliptic Fourier analysis. Mol. Breed. 2010, 25, 203–215. [Google Scholar] [CrossRef]
- Ohsawa, R.; Tsutsumi, T.; Uehara, H.; Namai, H.; Ninomiya, S. Quantitative evaluation of common buckwheat (Fagopyrum esculentum Moench) kernel shape by elliptic Fourier descriptor. Euphytica 1998, 101, 175–183. [Google Scholar] [CrossRef]
- Terral, J.; Tabard, E.; Bouby, L.; Ivorra, S.; Pastor, T.; Figueiral, I.; Picq, S.; Chevance, J.-B.; Jung, C.; Fabre, L.; et al. Evolution and history of grapevine (Vitis vinifera) under domestication: New morphometric perspectives to understand seed domestication syndrome and reveal origins of ancient European cultivars. Ann. Bot. 2010, 105, 443–455. [Google Scholar] [CrossRef]
- Mebatsion, K.K.; Paliwal, J.; Jayas, D.S. Evaluation of variations in the shape of grain types using principal components analysis of the elliptic Fourier descriptors. Comput. Electron. Agric. 2012, 80, 63–70. [Google Scholar] [CrossRef]
- Orrù, M.; Grillo, O.; Venora, G.; Bacchetta, G. Computer vision as a method complementary to molecular analysis: Grapevine cultivar seeds case study. C. R. Biol. 2012, 335, 602–615. [Google Scholar] [CrossRef]
- Orrù, M.; Grillo, O.; Lovicu, G.; Venora, G.; Bacchetta, G. Morphological characterisation of Vitis vinifera L. seeds by image analysis and comparison with archaeological remains. Veg. Hist. Archaeobot. 2013, 22, 231–242. [Google Scholar] [CrossRef]
- Kuhl, F.P.; Giardina, C.R. Elliptic Fourier features of a closed contour. Comput. Graph. Image Process. 1982, 18, 236–258. [Google Scholar] [CrossRef]
- Cervantes, E.; Martín-Gómez, J.J.; Espinosa-Roldán, F.E.; Muñoz-Organero, G.; Tocino, Á.; Cabello Sáenz de Santamaría, F. Seed apex curvature in key Spanish grapevine cultivars. Vitic. Data J. 2021, 3, e66478. [Google Scholar] [CrossRef]
- Noriega, A.; Cervantes, E.; Tocino, Á. Ethylene responses in Arabidopsis seedlings include the reduction of curvature values in the root cap. J. Plant Physiol. 2008, 165, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Tocino, A.; Cervantes, E. Curvature analysis reveals new functions for ethylene signalling pathway in shape determination of seed poles and root apices. Plant Signal. Behav. 2008, 3, 362–366. [Google Scholar] [CrossRef][Green Version]
- Noriega, A.; Cervantes, E.; Tocino, Á. Hydrogen peroxide treatment results in reduced curvature values in the Arabidopsis root apex. J. Plant Physiol. 2009, 166, 554–558. [Google Scholar] [CrossRef]
- Yamada, T.; Tobe, H.; Imaichi, R.; Kato, M. Developmental morphology of the ovules of Amborella trichopoda (Amborellaceae) and Chloranthus serratus (Chloranthaceae). Bot. J. Linn. Soc. 2001, 137, 277–290. [Google Scholar] [CrossRef][Green Version]
- Tobe, H. The embryology of angiosperms: Its broad application to the systematic and evolutionary study. Bot. Mag. 1989, 102, 351–367. [Google Scholar] [CrossRef]
- Simpson, M.G. Plant Systematics; Academic Press: Cambridge, MA, USA, 2019; pp. 567–582. [Google Scholar]
- Rudall, P.J. Evolution and patterning of the ovule in seed plants. Biol. Rev. 2021, 96, 943–960. [Google Scholar] [CrossRef]
- Bornand, A.D.V.; Beltramini, V.S. Caracterización morfológica de la semilla de Hibiscus cannabinus (Malvaceae) e influencia del tiempo de almacenamiento sobre la viabilidad. Lilloa 2021, 58, 51–62. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, Y.Y.; Liu, J.X. Embryology of Myosoton and Stellaria and its taxonomic significance (Caryophyllaceae). Phytotaxa 2017, 306, 124–134. [Google Scholar] [CrossRef]
- Dransfield, J.; Uhl, N.W.; Asmussen, C.B.; Baker, W.J.; Harley, M.M.; Lewis, C.E. Genera Palmarum—The Evolution and Classification of the Palms; Royal Botanic Gardens, Kew: Richmond, UK, 2008; pp. 36–37. [Google Scholar]
- Archibald, E.E.A. The development of the ovule and seed of jointed cactus (Opuntia aurantiaca Lindley). S. Afr. J. Sci. 1939, 36, 195–211. [Google Scholar]
- Kirkbride, J.H., Jr.; Gunn, C.R.; Dallwitz, M.J. 2000 Onwards. Family Guide for Fruits and Seeds: Descriptions, Illustrations, Identification, and Information Retrieval. Available online: Delta-intkey.com (accessed on 12 April 2021).
- Dias, L.S.; Dias, A.S. The relationships between shape and size of diaspores depends on being seeds or fruits. Horticulturae 2019, 5, 65. [Google Scholar] [CrossRef]
- López, J.; Devesa, J.A.; Ortega-Olivencia, A.; Ruiz, T. Production and morphology of fruit and seeds in Genisteae (Fabaceae) of south-west Spain. Bot. J. Linn. Soc. 2000, 132, 97–120. [Google Scholar] [CrossRef]
- Bawa, K.S.; Ingtv, T.; Revll, L.J.; Shivaprakash, K.N. Correlated evolution of flower size and seed number in flowering plants (monocotyledons). Ann. Bot. 2019, 123, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Stuppy, W.; Kesseler, R. Fruit: Edible, Inedible, Incredible; Papadakis Publisher: Newbury, UK; Berkshire: Winterbourne, UK, 2008; p. 264. [Google Scholar] [CrossRef]
- Weisstein, E.W. “Lune.” From MathWorld—A Wolfram Web Resource. Available online: https://mathworld.wolfram.com/Lune.html (accessed on 12 April 2021).
- Hufford, L. Seed morphology of Hydrangeaceae and its phylogenetic implications. Int. J. Plant Sci. 1995, 156, 555–580. Available online: https://www.jstor.org/stable/2475075 (accessed on 12 April 2021). [CrossRef]
- Diantina, S.; McGill, C.; Millner, J.; Nadarajan, J.; Pritchard, H.W.; Clavijo McCormick, A. Comparative seed morphology of Tropical and Temperate orchid species with different growth habits. Plants 2020, 9, 161. [Google Scholar] [CrossRef]
- Bregman, R. Forms of seed dispersal in Cactaceae. Acta Bot. Neerl. 1988, 37, 395–402. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Campbell, G.; Skillings, J.H. Nonparametric Stepwise Multiple Comparison Procedures. J. Am. Stat. Assoc. 1985, 80, 998–1003. [Google Scholar] [CrossRef]
- Sokal, R.R.; Braumann, C.A. Significance Tests for Coefficients of Variation and Variability Profiles. Syst. Zool. 1980, 29, 50. [Google Scholar] [CrossRef]




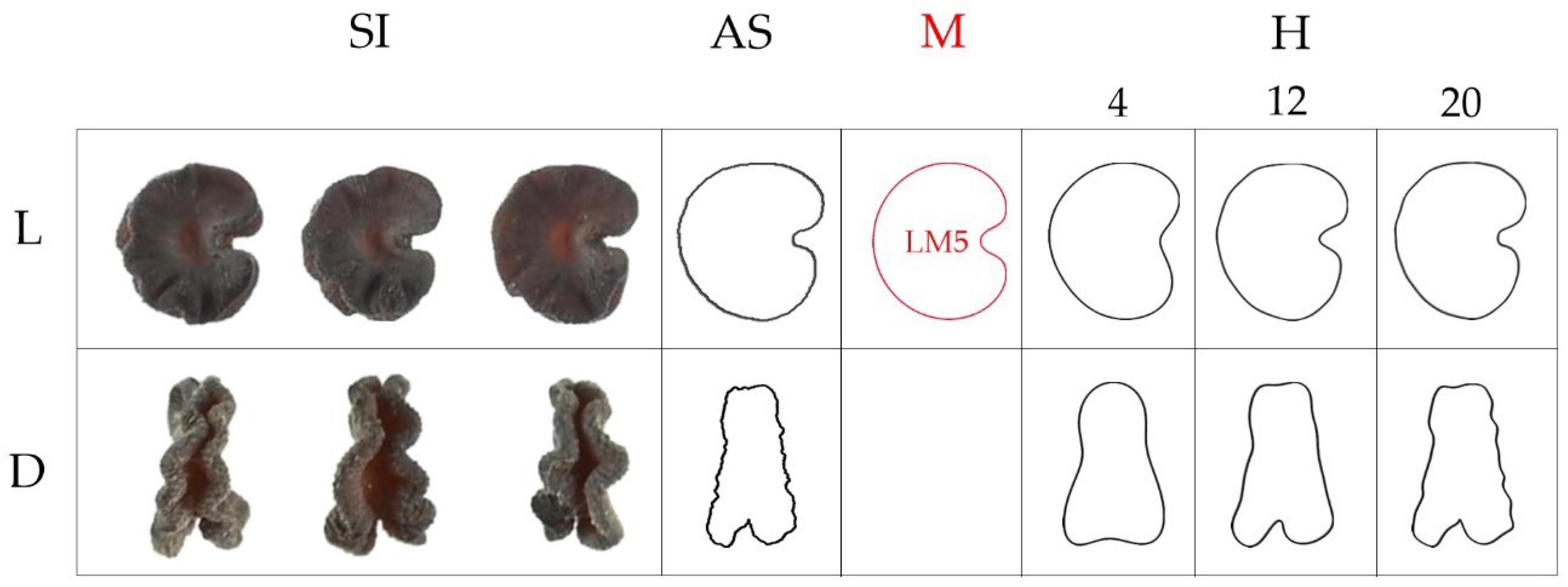
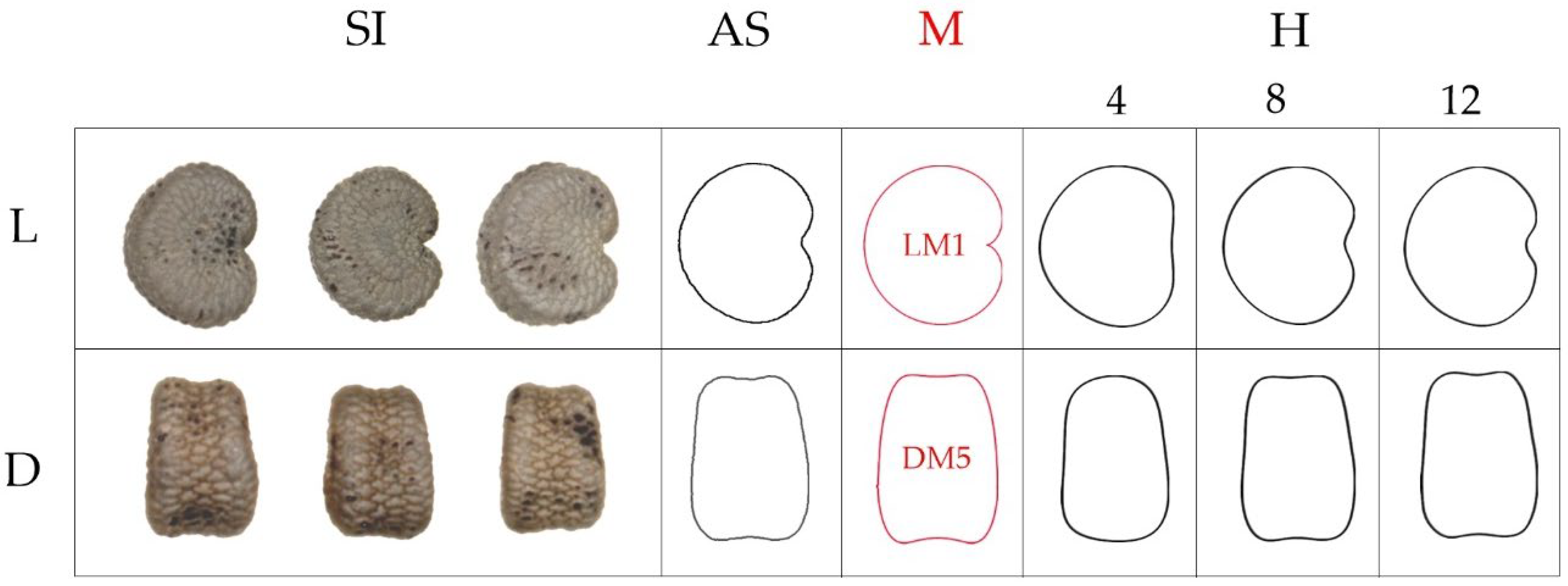

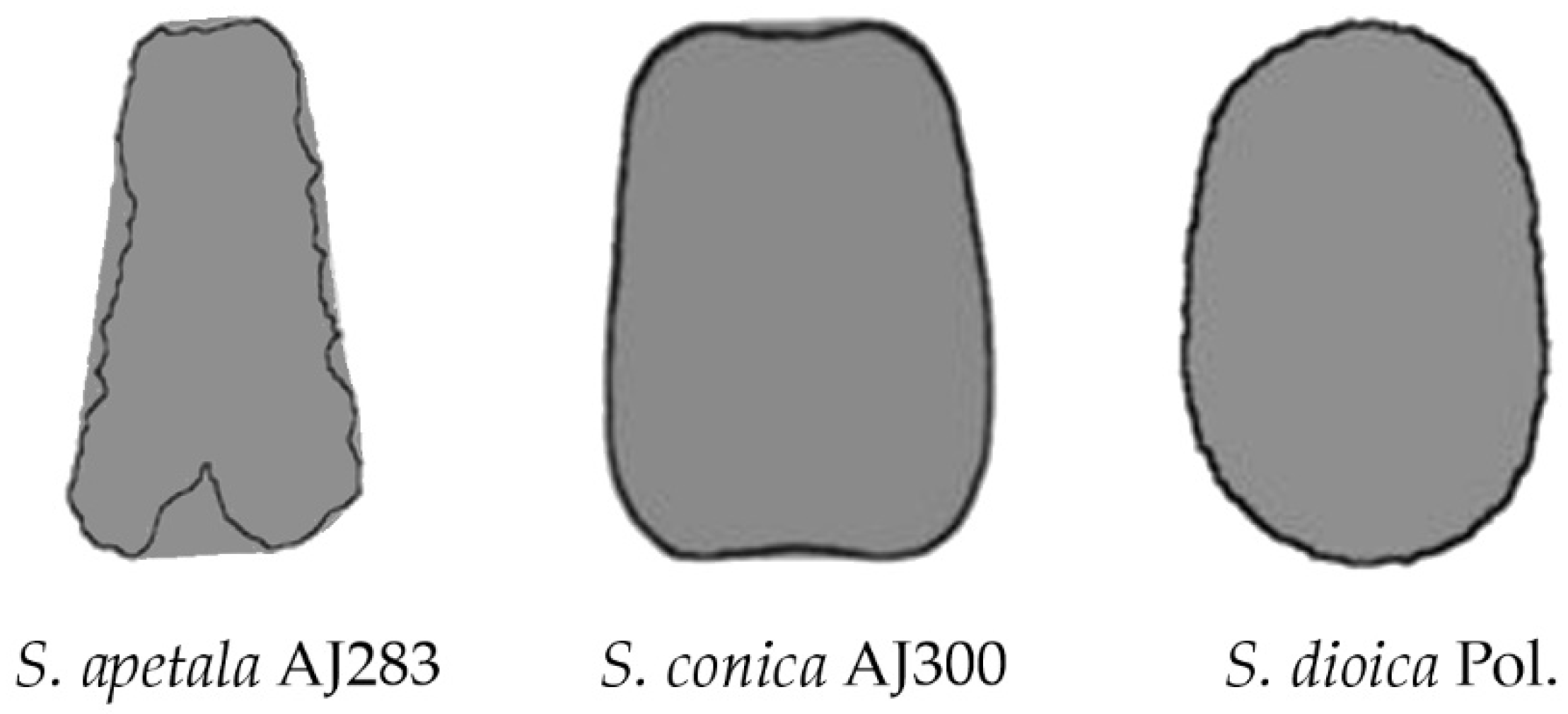

| N | Perimeter | Convex Hull Perimeter | Perimeter Ratio | Solidity | |
|---|---|---|---|---|---|
| S. apetala AJ283 | 3 | 1.01 b (6.02) | 0.86 a (6.02) | 0.852 a (0.76) | 0.834 a (1.78) |
| S. conica AJ300 | 3 | 0.88 a (2.78) | 0.85 a (2.96) | 0.967 c (0.31) | 0.972 b (0.06) |
| S. dioica Pol. | 3 | 1.13 c (1.90) | 1.03 b (1.10) | 0.914 b (1.54) | 0.968 b (0.32) |
| N | Perimeter | Convex Hull Perimeter | Perimeter Ratio | Solidity | |
|---|---|---|---|---|---|
| S. apetala AJ283 | 3 | 0.53 a (1.31) | 0.49 a (1.55) | 0.924 b (0.33) | 0.949 a (0.69) |
| S. conica AJ300 | 3 | 0.90 b (0.61) | 0.88 b (1.00) | 0.975 c (1.01) | 0.970 c (0.16) |
| S. dioica Pol. | 3 | 1.25 c (3.51) | 1.12 c (4.02) | 0.897 a (1.17) | 0.961 b (0.24) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cervantes, E.; Rodríguez-Lorenzo, J.L.; Gutiérrez del Pozo, D.; Martín-Gómez, J.J.; Janousek, B.; Tocino, Á.; Juan, A. Seed Silhouettes as Geometric Objects: New Applications of Elliptic Fourier Transform to Seed Morphology. Horticulturae 2022, 8, 974. https://doi.org/10.3390/horticulturae8100974
Cervantes E, Rodríguez-Lorenzo JL, Gutiérrez del Pozo D, Martín-Gómez JJ, Janousek B, Tocino Á, Juan A. Seed Silhouettes as Geometric Objects: New Applications of Elliptic Fourier Transform to Seed Morphology. Horticulturae. 2022; 8(10):974. https://doi.org/10.3390/horticulturae8100974
Chicago/Turabian StyleCervantes, Emilio, José Luis Rodríguez-Lorenzo, Diego Gutiérrez del Pozo, José Javier Martín-Gómez, Bohuslav Janousek, Ángel Tocino, and Ana Juan. 2022. "Seed Silhouettes as Geometric Objects: New Applications of Elliptic Fourier Transform to Seed Morphology" Horticulturae 8, no. 10: 974. https://doi.org/10.3390/horticulturae8100974
APA StyleCervantes, E., Rodríguez-Lorenzo, J. L., Gutiérrez del Pozo, D., Martín-Gómez, J. J., Janousek, B., Tocino, Á., & Juan, A. (2022). Seed Silhouettes as Geometric Objects: New Applications of Elliptic Fourier Transform to Seed Morphology. Horticulturae, 8(10), 974. https://doi.org/10.3390/horticulturae8100974









