Hydroponic Basil Production: Temperature Influences Volatile Organic Compound Profile, but Not Overall Consumer Preference
Abstract
:1. Introduction
2. Materials and Methods
2.1. Growing Environment
2.2. GC–MS Analysis
2.3. Sensory Analysis
2.4. Experimental Design and Statistical Analysis
3. Results
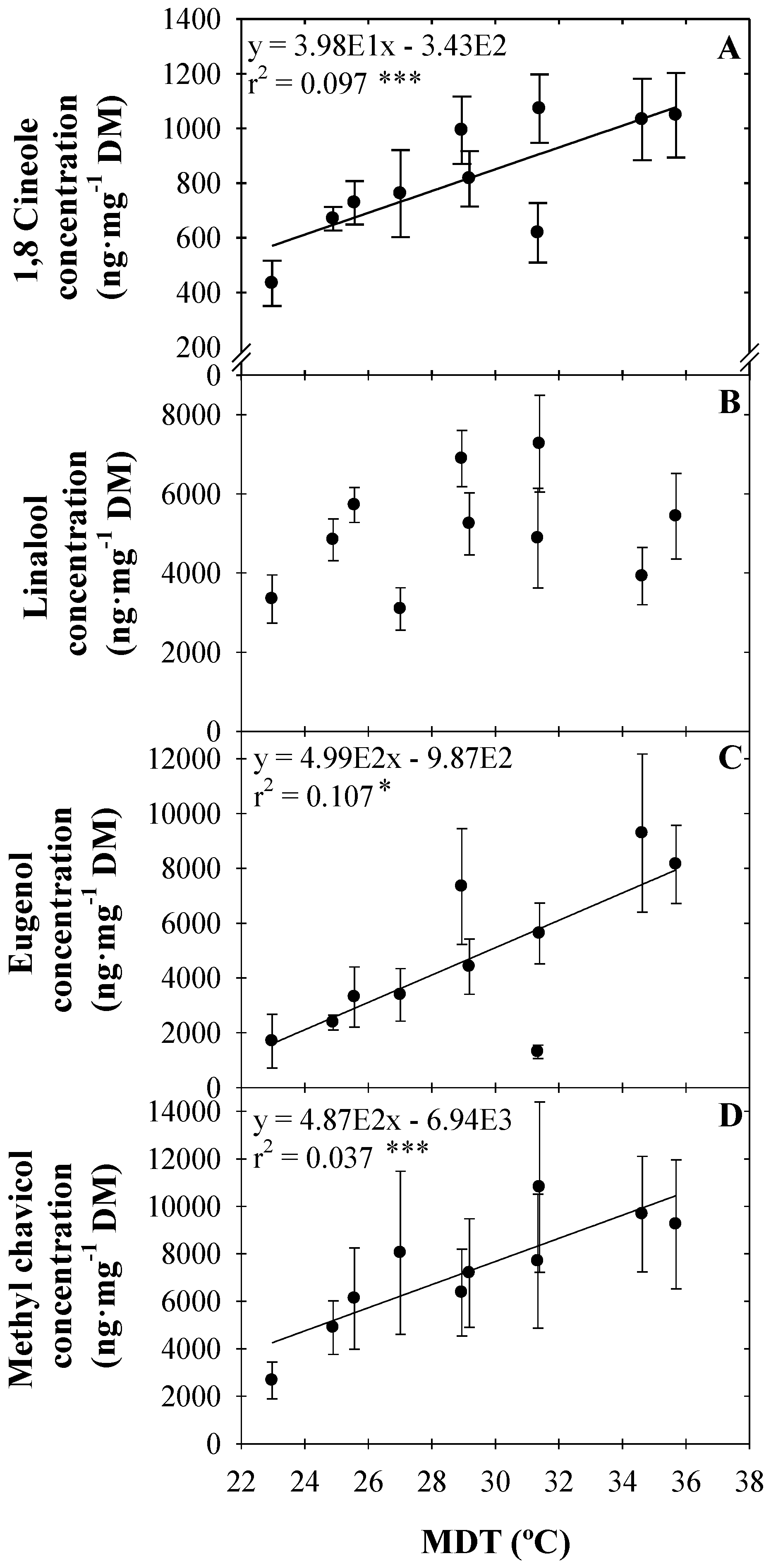
3.1. VOC Concentration
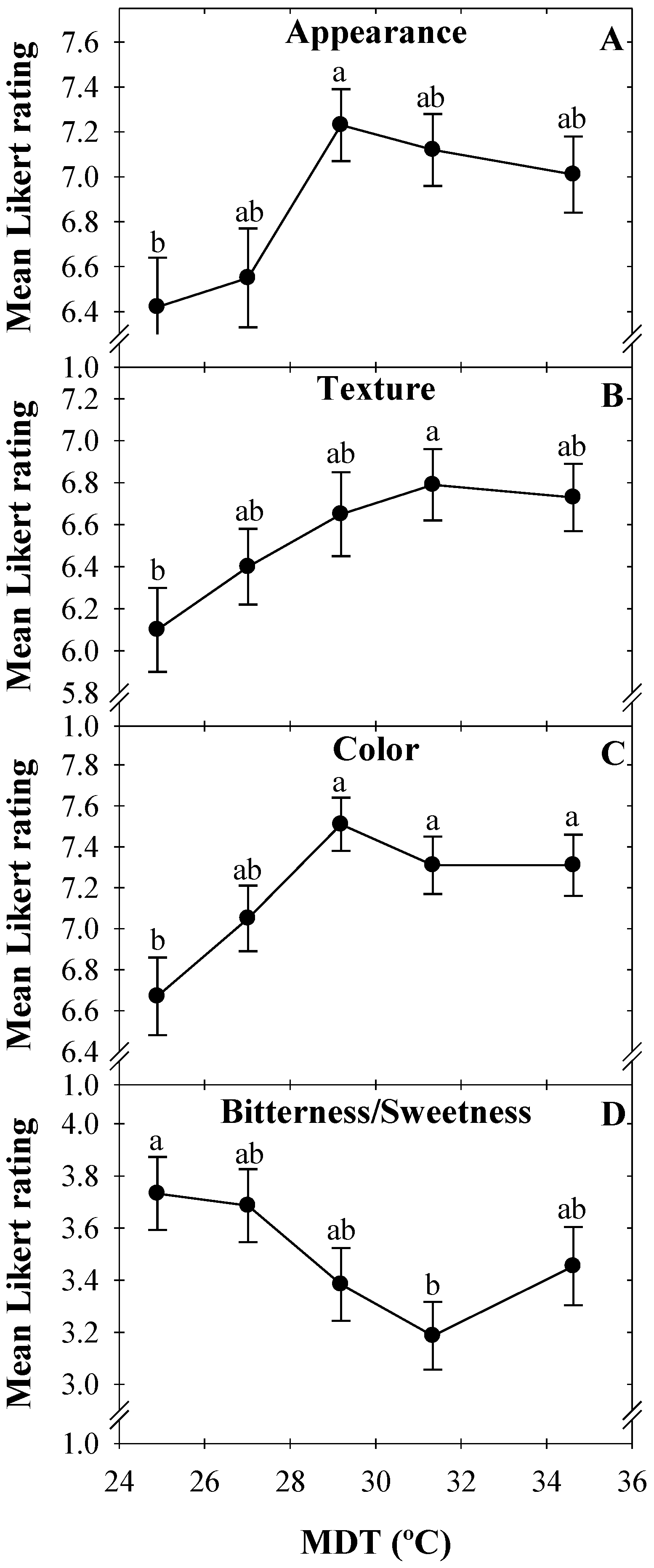
3.2. Consumer Preference
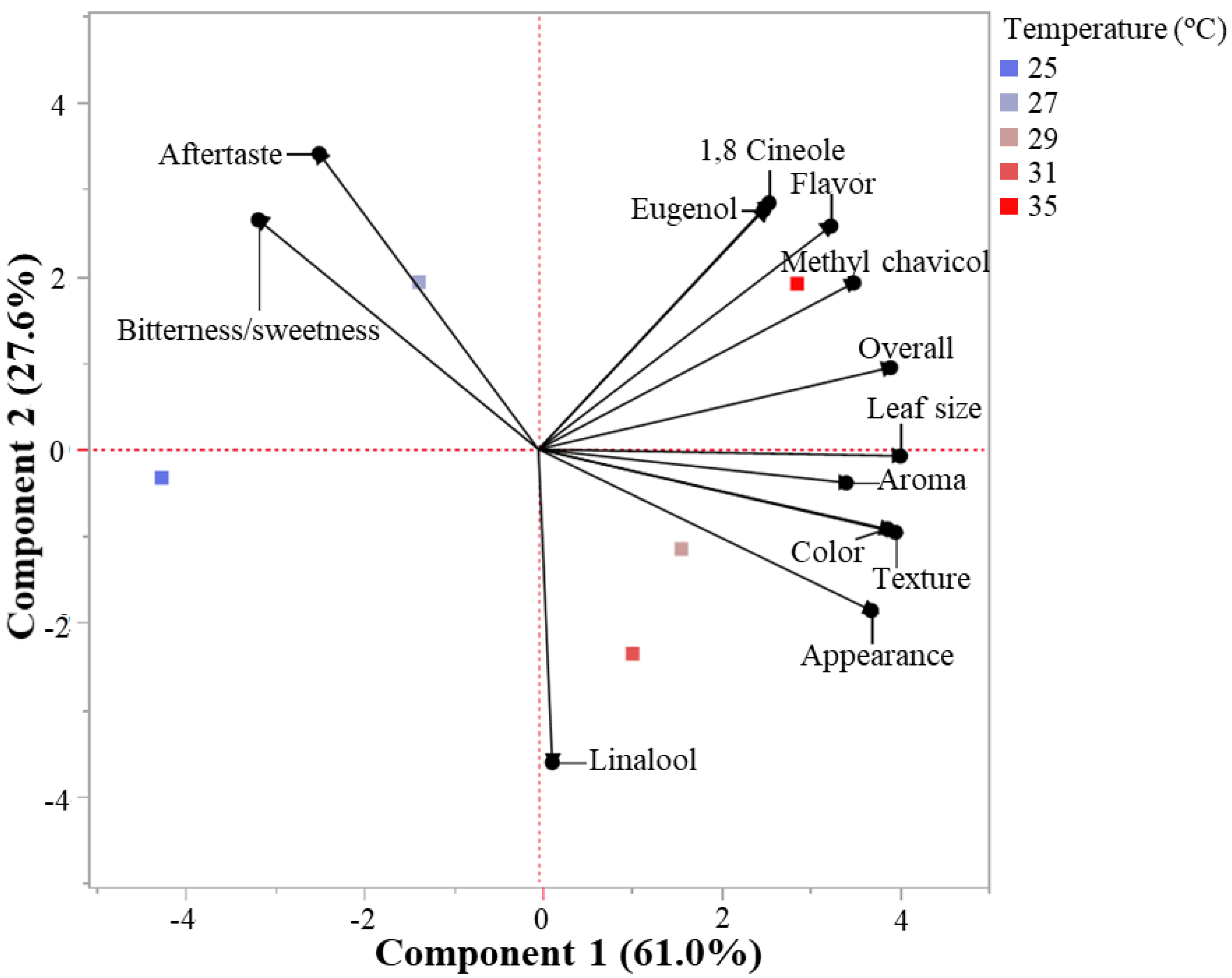
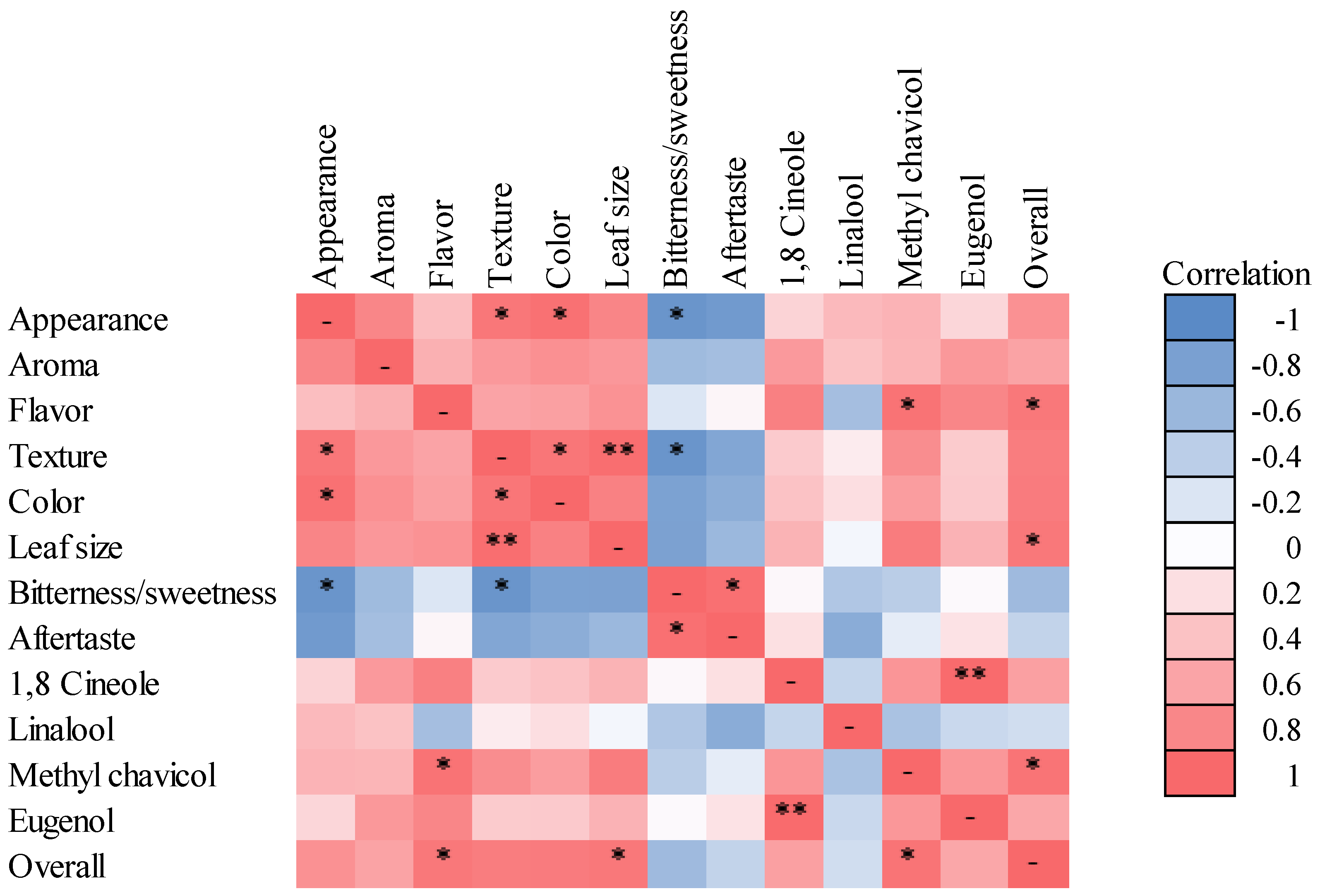
3.3. Comparison—Biplot and Correlations
4. Discussion
4.1. Volatile Organic Compound Concentration
4.2. Consumer Preferences and Correlations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Engler, N.; Krarti, M. Review of energy efficiency in controlled environment agriculture. Renew. Sustain. Energy Rev. 2021, 141, 110786. [Google Scholar] [CrossRef]
- Gomez, C.; Currey, C.J.; Dickson, R.W.; Kim, H.; Hernandez, R.; Sabeh, N.C.; Raudales, R.E.; Brumfield, R.G.; Laury-Shaw, A.; Wilke, A.K.; et al. Controlled environment food production for urban agriculture. HortScience 2019, 54, 1448–1458. [Google Scholar] [CrossRef]
- Kolodinsky, J.; Sitaker, M.; Chase, L.; Smith, D.; Wang, W. Food systems disruptions. J. Agric. Food Syst. Community Dev. 2020, 9, 5–8. [Google Scholar] [CrossRef]
- Walters, K.J.; Behe, B.K.; Currey, C.J.; Lopez, R.G. Historical, current, and future perspectives for controlled environment hydroponic food crop production in the United States. HortScience 2020, 55, 758–767. [Google Scholar] [CrossRef]
- Santos, A.; Chierice, G.; Alexander, K.; Riga, A.; Matthews, E. Characterization of the raw essential oil eugenol extracted from Syzygium aromaticum L. J. Therm. Anal. Calorim. 2009, 96, 821–825. [Google Scholar] [CrossRef]
- Simon, J.E.; Morales, M.R.; Phippen, W.B.; Vieira, R.F.; Hao, Z. Basil: A source of aroma compounds and a popular culinary and ornamental herb. Perspect. New Crop. New Uses 1999, 16, 499–505. [Google Scholar]
- Arena, E.; Guarrera, N.; Campisi, S.; Asmundo, C.N. Comparison of odour active compounds detected by gas-chromatography–olfactometry between hand-squeezed juices from different orange varieties. Food Chem. 2006, 98, 59–63. [Google Scholar] [CrossRef]
- De Vincenzi, M.; Silano, M.; de Vincenzi, A.; Maialetti, F.; Scazzocchio, B. Constituents of aromatic plants: Eucalyptol. Fitoterapia 2002, 73, 269–275. [Google Scholar] [CrossRef]
- Hälvä, S.; Craker, L.E.; Simon, J.E.; Charles, D.J. Growth and essential oil in dill, Anethum graveolens L., in response to temperature and photoperiod. J. Herbs Spices Med. Plants 1993, 1, 47–56. [Google Scholar] [CrossRef]
- Chang, X.; Alderson, P.; Wright, C. Effect of temperature integration on the growth and volatile oil content of basil (Ocimum basilicum L.). J. Hortic. Sci. Biotechnol. 2005, 80, 593–598. [Google Scholar] [CrossRef]
- Pogany, D.; Bell, C.L.; Kirch, E.R. Composition of oil of sweet basil (Ocimum basilicum L.) obtained from plants grown at different temperatures. Perfum. Essent. Oil Rec. 1968, 59, 858–865. [Google Scholar]
- Chang, X.; Alderson, P.G.; Hollowood, T.A.; Hewson, L.; Wright, C.J. Flavour and aroma of fresh basil are affected by temperature. J. Sci. Food Agric. 2007, 87, 1381–1385. [Google Scholar] [CrossRef]
- Walters, K.J.; Lopez, R.G.; Behe, B.K. Leveraging controlled-environment agriculture to increase key basil terpenoid and phenylpropanoid concentrations: The effects of radiation intensity and CO2 concentration on consumer preference. Front. Plant Sci. 2021, 11, 1641. [Google Scholar] [CrossRef] [PubMed]
- Walters, K.J.; Lopez, R.G. Modeling growth and development of hydroponically grown dill, parsley, and watercress in response to photosynthetic daily light integral and mean daily temperature. PLoS ONE 2021, 16, e0248662. [Google Scholar] [CrossRef]
- Schilmiller, A.; Shi, F.; Kim, J.; Charbonneau, A.L.; Holmes, D.; Jones, A.D.; Last, R.L. Mass spectrometry screening reveals widespread diversity in trichome specialized metabolites of tomato chromosomal substitution lines. Plant J. 2010, 62, 391–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, S.R.; Pearson, S.; Hadley, P. The effects of temperature, photoperiod and light integral on the time to flowering of pansy cv. Universal Violet (Viola × wittrockiana Gams.). Ann. Bot. 1997, 80, 107–112. [Google Scholar] [CrossRef] [Green Version]
- Karlsson, M.G.; Heins, R.D.; Erwin, J.E. Quantifying temperature controlled leaf unfolding rates in Lilium longiflorum Thunb. J. Am. Soc. Hortic. Sci. 1988, 113, 70–74. [Google Scholar]
- Pramuk, L.A.; Runkle, E.S. Modeling growth and development of celosia and impatiens in response to temperature and photosynthetic daily light integral. J. Am. Soc. Hortic. Sci. 2005, 130, 813–818. [Google Scholar] [CrossRef] [Green Version]
- Tollenaar, M.; Daynard, T.B.; Hunter, R.B. Effect of temperature on rate of leaf appearance and flowering date in maize. Crop. Sci. 1979, 19, 363–366. [Google Scholar] [CrossRef]
- Warner, R.M. Differential temperature sensitivity of flowering time and crop quality parameters of 20 seed-propagated petunia cultivars. HortScience 2020, 55, 362–367. [Google Scholar] [CrossRef]
- Cseke, L.J.; Kirakosyan, A.; Kaufman, P.B.; Warber, S.; Duke, J.A.; Brielmann, H.L. Natural Products from Plants; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Weisshaar, B.; Jenkins, G.I. Phenylpropanoid biosynthesis and its regulation. Curr. Opin. Plant Biol. 1998, 1, 251–257. [Google Scholar] [CrossRef]
- Leyva, A.; Jarillo, J.A.; Salinas, J.; Martinez-Zapater, J.M. Low temperature induces the accumulation of phenylalanine ammonia-lyase and chalcone synthase mRNAs of Arabidopsis thaliana in a light-dependent manner. Plant Physiol. 1995, 108, 39–46. [Google Scholar] [CrossRef]
- Iijima, Y.; Davidovich-Rikanati, R.; Fridman, E.; Gang, D.R.; Bar, E.; Lewinsohn, E.; Pichersky, E. The biochemical and molecular basis for the divergent patterns in the biosynthesis of terpenes and phenylpropenes in the peltate glands of three cultivars of basil. Plant Physiol. 2004, 136, 3724–3736. [Google Scholar] [CrossRef] [Green Version]
- Choi, N.E.; Han, J.H. How Flavor Works: The Science of Taste and Aroma; John Wiley & Sons: Chichester, UK, 2015. [Google Scholar]
- Carvalho, S.D.; Schwieterman, M.L.; Abrahan, C.E.; Colquhoun, T.A.; Folta, K.M. Light quality dependent changes in morphology, antioxidant capacity, and volatile production in sweet basil (Ocimum basilicum). Front. Plant Sci. 2016, 7, 1328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheen, L.Y.; Ou, Y.H.T.; Tsai, S.J. Flavor characteristic compounds found in the essential oil of Ocimum basilicum L. with sensory evaluation and statistical analysis. J. Agric. Food Chem. 1991, 39, 939–943. [Google Scholar] [CrossRef]
- Naim, M.; Nir, S.; Spielman, A.I.; Noble, A.C.; Peri, I.; Rodin, S.; Samuelov-Zubare, M. Hypothesis of receptor-dependent and receptor-independent mechanisms for bitter and sweet taste transduction: Implications for slow taste onset and lingering aftertaste. In Chemistry of Taste: Mechanisms, Behaviors, and Mimics; Given, P., Paredes, D., Eds.; American Chemical Society: Washington, DC, USA, 2002; pp. 2–17. [Google Scholar]
| Rep. | DLI | MDT °C | ||||
|---|---|---|---|---|---|---|
| Target | Actual | Target Air | Actual Air | Leaf | Solution | |
| 1 | 12 | 12.9 ± 1.1 | 23 | 23.0 ± 1.3 | 26.3 ± 1.3 | 21.7 ± 1.4 |
| 9 | 10.1 ± 1.0 | 23 | 23.0 ± 1.3 | - z | 21.5 ± 1.1 | |
| 7 | 7.4 ± 0.8 | 23 | 23.0 ± 1.3 | - | 21.4 ± 1.3 | |
| 12 | 11.1 ± 1.1 | 26 | 25.6 ± 0.6 | 27.7 ± 0.9 | 23.9 ± 0.7 | |
| 9 | 9.2 ± 1.0 | 26 | 25.6 ± 0.6 | - | 23.4 ± 0.7 | |
| 7 | 8.0 ± 0.8 | 26 | 25.6 ± 0.6 | - | 23.5 ± 0.7 | |
| 12 | 11.3 ± 1.2 | 29 | 28.9 ± 1.9 | 30.5 ± 0.8 | 26.0 ± 1.0 | |
| 9 | 8.9 ± 1.1 | 29 | 28.9 ± 1.9 | - | 25.9 ± 0.9 | |
| 7 | 6.6 ± 0.9 | 29 | 28.9 ± 1.9 | - | 25.7 ± 1.0 | |
| 12 | 13.2 ± 1.3 | 32 | 31.4 ± 0.7 | 35.7 ± 1.2 | 28.7 ± 0.8 | |
| 9 | 9.2 ± 1.0 | 32 | 31.4 ± 0.7 | - | 27.3 ± 0.8 | |
| 7 | 7.2 ± 0.7 | 32 | 31.4 ± 0.7 | - | 26.2 ± 0.8 | |
| 12 | 12.0 ± 1.3 | 35 | 35.7 ± 0.8 | 36.6 ± 0.8 | 30.3 ± 0.8 | |
| 9 | 8.5 ± 0.9 | 35 | 35.7 ± 0.8 | - | 29.6 ± 0.7 | |
| 7 | 5.5 ± 0.8 | 35 | 35.7 ± 0.8 | - | 29.2 ± 0.6 | |
| 2 | 12 | 12.4 ± 3.2 | 23 | 24.9 ± 2.2 | 26.5 ± 1.5 | 24.6 ± 1.7 |
| 9 | 9.3 ± 2.3 | 23 | 24.9 ± 2.2 | - | 22.9 ± 1.0 y | |
| 7 | 7.8 ± 2.0 | 23 | 24.9 ± 2.2 | - | 23.0 ± 1.2 | |
| 12 | 13.4 ± 3.5 | 26 | 27.0 ± 1.2 | 27.1 ± 2.0 | 26.4 ± 0.8 | |
| 9 | 9.9 ± 2.9 | 26 | 27.0 ± 1.2 | - | 25.3 ± 1.1 | |
| 7 | 7.1 ± 2.3 | 26 | 27.0 ± 1.2 | - | 24.9 ± 1.0 | |
| 12 | 14.0 ± 4.1 | 29 | 29.2 ± 0.6 | 30.2 ± 0.9 | 26.8 ± 0.6 | |
| 9 | 9.3 ± 2.9 | 29 | 29.2 ± 0.6 | - | 26.6 ± 0.8 | |
| 7 | 6.1 ± 2.1 | 29 | 29.2 ± 0.6 | - | 26.6 ± 0.6 | |
| 12 | 14.8 ± 3.8 | 32 | 31.3 ± 1.0 | 32.0 ± 1.8 | 28.9 ± 0.3 | |
| 9 | 9.8 ± 2.5 | 32 | 31.3 ± 1.0 | - | 28.7 ± 0.5 | |
| 7 | 7.3 ± 2.0 | 32 | 31.3 ± 1.0 | - | 28.6 ± 0.6 y | |
| 12 | - | 35 | 34.6 ± 1.8 | 33.6 ± 0.9 | 31.6 ± 1.3 | |
| 9 | 10.3 ± 2.7 | 35 | 34.6 ± 1.8 | - | 30.2 ± 0.7 | |
| 7 | 7.7 ± 2.4 | 35 | 34.6 ± 1.8 | - | 29.5 ± 0.8 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walters, K.J.; Lopez, R.G. Hydroponic Basil Production: Temperature Influences Volatile Organic Compound Profile, but Not Overall Consumer Preference. Horticulturae 2022, 8, 76. https://doi.org/10.3390/horticulturae8010076
Walters KJ, Lopez RG. Hydroponic Basil Production: Temperature Influences Volatile Organic Compound Profile, but Not Overall Consumer Preference. Horticulturae. 2022; 8(1):76. https://doi.org/10.3390/horticulturae8010076
Chicago/Turabian StyleWalters, Kellie J., and Roberto G. Lopez. 2022. "Hydroponic Basil Production: Temperature Influences Volatile Organic Compound Profile, but Not Overall Consumer Preference" Horticulturae 8, no. 1: 76. https://doi.org/10.3390/horticulturae8010076
APA StyleWalters, K. J., & Lopez, R. G. (2022). Hydroponic Basil Production: Temperature Influences Volatile Organic Compound Profile, but Not Overall Consumer Preference. Horticulturae, 8(1), 76. https://doi.org/10.3390/horticulturae8010076







