Impact of Nitrate and Ammonium Ratios on Flowering and Asexual Reproduction in the Everbearing Strawberry Cultivar Fragaria × ananassa Albion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Plant Material Preparation
2.3. Plant Traits and Data Analysis
2.3.1. Inflorescences
2.3.2. Daughter Plant
2.3.3. Mother Plant Growth and Tissue Nutrient Analysis
2.3.4. Data Analysis
3. Results
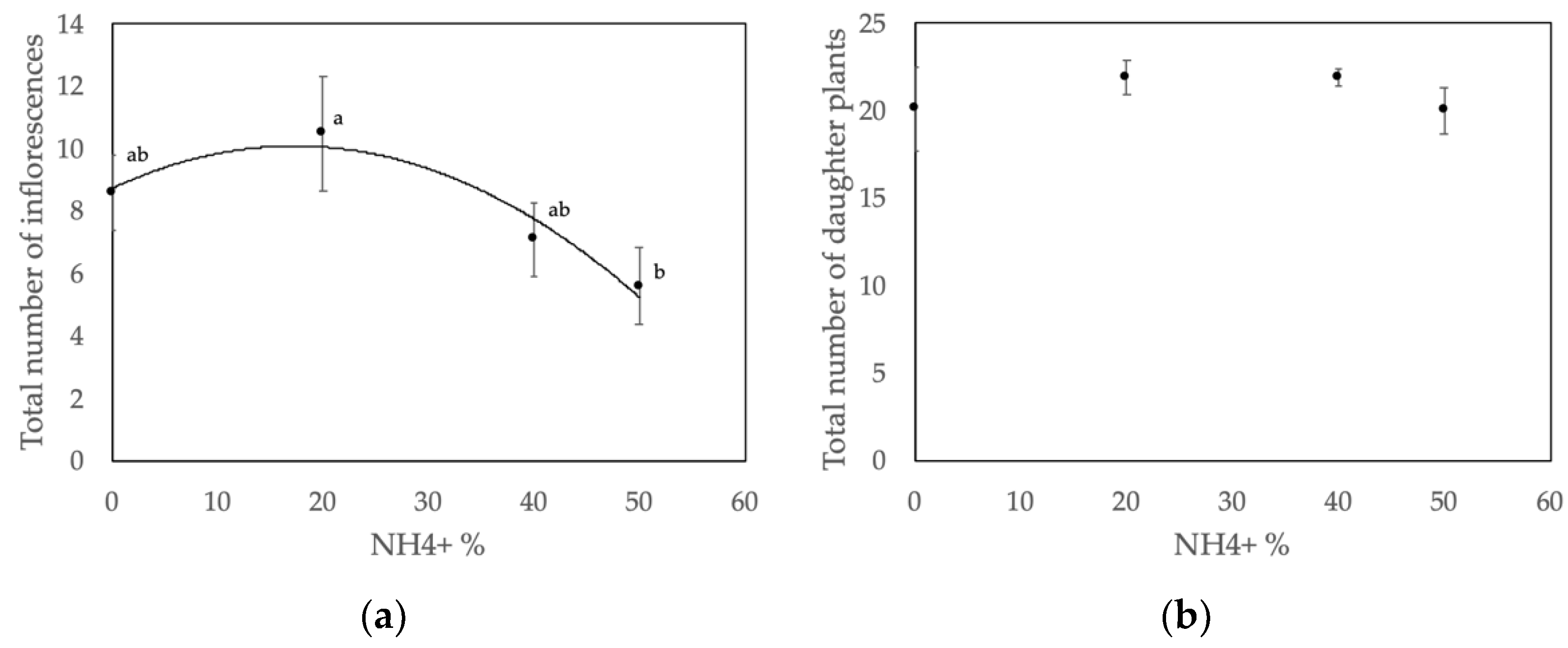
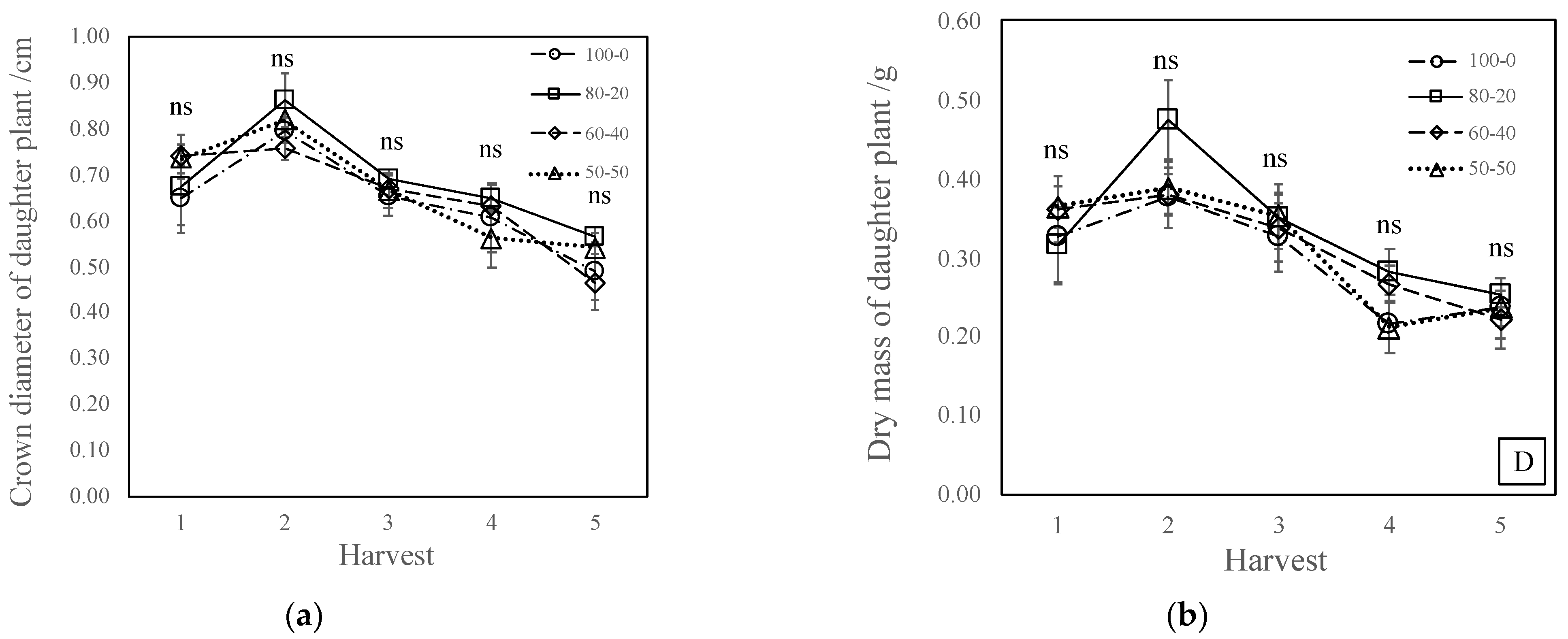
3.1. Inflorescence and Daughter Plant Production
3.2. Mother Plant Growth
4. Discussion
4.1. Nitrate–Ammonium Ratio Affects the Number of Inflorescences
4.2. Nitrate-to-Ammonium Ratio Has No Effect on Daughter Plant Production
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Statistics for Strawberries. USDA-NASS Report. 2019. Available online: https://www.nass.usda.gov/Statistics_by_Subject/result.php?C6E91360-DC42-3C4A-8D5C-1392239A0EAF§or=CROPS&group=FRUIT%20%26%20TREE%20NUTS&comm=STRAWBERRIES (accessed on 9 December 2021).
- 2014 Census of Horticultural Specialties. USDA Census of Agriculture. 2014. Available online: https://www.nass.usda.gov/Publications/AgCensus/2012/Online_Resources/Census_of_Horticulture_Specialties/hortic_1_026_028.pdf (accessed on 9 December 2021).
- Hancock, J.F. Strawberries; CAB International: Wallingford, UK, 1999; p. 237. [Google Scholar]
- Heide, O.M.; Stavang, J.A.; Sønsteby, A. Physiology and genetics of flowering in cultivated and wild strawberries—A review. J. Hort. Sci. Biotechnol. 2013, 88, 1–18. [Google Scholar] [CrossRef]
- Stewart, P.J.; Folta, K.M. A review of photoperiodic flowering research in strawberry (Fragaria spp.). Crit. Rev. Plant Sci. 2010, 29, 1–13. [Google Scholar] [CrossRef]
- Neri, D.; Baruzzi, G.; Massetani, F.; Faedi, W. Strawberry production in forced and protected culture in Europe as a response to climate change. Can. J. Plant Sci. 2012, 92, 1021–1036. [Google Scholar] [CrossRef]
- Castro, P.; Bushakra, J.M.; Stewart, P.; Weebadde, C.K.; Wang, D.; Hancock, J.F.; Finn, C.E.; Luby, J.J.; Lewers, K.S. Genetic mapping of day-neutrality in cultivated strawberry. Mol. Breed. 2015, 35, 79–94. [Google Scholar] [CrossRef]
- Arnade, C.; Kuchler, F. Measuring the impact of off-season imports. U.S. Department Agriculture Economic Research Service. 2015. Available online: http://www.ers.usda.gov/publications/err-economic-research-report/err197.aspx (accessed on 9 December 2021).
- Durner, E.F. Programmed Flowering of The F1 Long-day Strawberry Cultivar ‘Elan’ via Nitrogen and Daylength Manipulation. AIMS Agri. Food. 2016, 1, 4–19. [Google Scholar] [CrossRef]
- Durner, E.F. Conditioning alleviates reduced yield of smaller plugs of ‘Albion’ strawberry (Fragaria X ananassa Duch.) in off-season plasticulture production. J. Hort. Sci. Biotech. 2017, 93, 433–440. [Google Scholar] [CrossRef]
- Durner, E.F. Enhanced flowering of the F1 long-day strawberry cultivars “Tarpan” and “Gasana” with nitrogen and daylength management. AIMS Agri. Food. 2017, 2, 1–15. [Google Scholar] [CrossRef]
- Durner, E.F. Fall nitrogen enhances spring nitrogen enhanced flowering in the long-day strawberry cultivar ‘Elan’. AIMS Agri. Food. 2017, 2, 149–164. [Google Scholar] [CrossRef]
- Durner, E.F. Long-day and nitrogen conditioning of ‘Albion’ strawberry (Fragaria X ananassa Duch.) enhances off-season field production. J. Hort. Sci. Biotechnol. 2018, 93, 296–305. [Google Scholar] [CrossRef]
- Durner, E.F. Plant architecture of ‘Albion’ strawberry (Fragaria × ananassa Duch.) is influenced by photoperiod and nitrogen but not light source during conditioning. AIMS Agri. Food. 2018, 3, 246–265. [Google Scholar] [CrossRef]
- Wang, Y. High NO3-N to NH4-N Ratios Promote Growth and Flowering of a Hybrid Phalaenopsis Grown in Two Root Substrates. Hortscience 2008, 43, 350–353. [Google Scholar] [CrossRef] [Green Version]
- Shi, X.; Hernández, R.; Hoffmann, M. Timing of stolon removal alters daughter plant production and quality in the ever-bearing strawberry ‘Albion’. HortScience 2021, 56, 650–656. [Google Scholar] [CrossRef]
- Yamasaki, A.; Yoneyama, T.; Tanaka, F.; Tanaka, K.; Nakashima, N. Tracer studies on the allocation of carbon and nitrogen during flower induction of strawberry plants as affected by the nitrogen level. Acta Hortic. 2002, 567, 349–352. [Google Scholar] [CrossRef]
- Kronzucker, H.J.; Glass, A.D.M.; Siddiqi, M.Y. Inhibition of nitrate uptake by ammonium in barley. Analysis of component fluxes. Plant Physiol. 1999, 120, 283–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kronzucker, H.J.; Siddiqi, M.Y.; Glass, A.D.; Kirk, G.J. Nitrate– ammonium synergism in rice. A subcellular flux analysis. Plant Physiol. 1999, 119, 1041–1046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cerezo, M.; Tillard, P.; Filleur, S.; Muños, S.; Daniel-Vedele, F.; Gojon, A. Major alterations of the regulation of root NO3− uptake are associated with the mutation of Nrt2.1 and Nrt2.2 genes in Arabidopsis. Plant Physiol. 2001, 127, 262–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.L.; Tsay, T.F. Influence of differing nitrate and nitrogen availability on flowering control in Arabidopsis. J. Exp. Bot. 2017, 68, 2603–2609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Claussen, W.; Lenz, F. Effect of ammonium or nitrate nutrition on net photosynthesis, growth, and activity of the enzymes nitrate reductase and glutamine syntheses in blueberry, raspberry and strawberry. Plant Soil. 1999, 208, 95–102. [Google Scholar] [CrossRef]
- Cárdenas-Navarro, R.; López-Pérez, L.; Lobit, P.; Ruiz-Corro, R.; Castellanos-Monrales, V.V. Effects of nitrogen source on growth and development of strawberry plants. J. Plant Nutr. 2006, 29, 1699–1707. [Google Scholar] [CrossRef]
- Marino, D.; Moran, J.F. Can ammonium stress be positive for plant performance? Front. Plant Sci. 2019, 10, 1103. [Google Scholar] [CrossRef] [Green Version]
- Brown, T.; Wareign, P.F. The genetic control of the everbearing habit and three other characters in varieties of Fragaria vesca. Euphytica 1965, 14, 97–112. [Google Scholar] [CrossRef]
- Ito, H.; Saito, T. Studies on the flower formation in the strawberry plant. I. Effects of temperature and photoperiod on the flower formation. Tohoku J. Agri. Res. 1962, 13, 191–203. [Google Scholar]
- Remay, A.; Lalanne, D.; Thouroude, T.; Le Couvier, F.; Oyant, L.H.-S.; Foucher, F. A survey of flowering genes reveals the role of gibberellins in floral control in rose. Theor. Appl. Genet. 2009, 119, 767–781. [Google Scholar] [CrossRef] [PubMed]
- Koskela, E.A.; Mouhu, K.; Albani, M.C.; Kurokura, T.; Ranta-Nen, M.; Sargent, D.J.; Battey, N.H.; Coupland, G.; Elomaa, P.; Hytönen, T. Mutation in TERMINAL FLOWER1 reverses the photoperiodic requirement for flowering in the wild strawberry Fragaria Vesca. Plant Physiol. 2012, 159, 1043–1054. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konsin, M.; Voipio, I.; Palonen, P. Influence of photoperiod and duration of short-day treatment on vegetative growth and flowering of strawberry (Fragaria × ananassa Duch.). J. Hortic. Sci. Biotechnol. 2001, 76, 77–82. [Google Scholar] [CrossRef]
- Heide, O.M.; Sønsteby, A. Interactions of temperature and photoperiod in the control of flowering of latitudinal and altitudinal populations of wild strawberry (Fragaria vesca). Physiol. Plantarum. 2007, 130, 280–289. [Google Scholar] [CrossRef]
- Landrein, B.; Formosa-Jordan, P.; Malivert, A.; Schuster, C.; Melnyk, C.W.; Yang, W.; Turnbull, C.; Meyerowitz, E.M.; Locke, J.C.W.; Jönsson, H. Nitrate modulation of Arabidopsis shoot stem cells. Proc. Natl. Acad. Sci. USA 2018, 115, 1382–1387. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Chemical | Treatment | |||
|---|---|---|---|---|
| 100% NO3−: 0%NH4+ | 80%:20% | 60%:40% | 50%:50% | |
| KH2PO4 | 1 | 0 | 0 | 0 |
| (NH4)2SO4 | 0 | 0 | 0.4 | 1 |
| NH4H2PO4 | 0 | 1 | 1 | 1 |
| MgSO4 | 0.5 | 0.5 | 0.5 | 0.5 |
| KNO3 | 2 | 3 | 3 | 3 |
| NH4NO3 | 0 | 0.2 | 0.6 | 0 |
| Ca(NO3)2 | 2 | 0.8 | 0 | 0 |
| CaCl2 | 0 | 1.2 | 1.2 | 1.2 |
| Na2FeEDTA | 30 mg | |||
| MnSO4 | 0.01 | |||
| ZnSO4 | 0.0008 | |||
| CuSO4 | 0.0005 | |||
| (NH4)6Mo7O24 | 0.000085 | |||
| H3BO3 | 0.05 | |||
| Nutrients | Feed | Drain | ||||||
|---|---|---|---|---|---|---|---|---|
| 100%:0% | 80%:20% | 60%:40% | 50%:50% | 100%:0% | 80%:20% | 60%:40% | 50%:50% | |
| N-NO3− | 5.9 ± 0.24 | 4.6 ± 0.15 | 3.7 ± 0.19 | 2.6 ± 1.25 | 6.3 ± 0.70 | 5.1 ± 0.51 | 4.3 ± 0.61 | 2.9 ± 1.44 |
| N-NH4+ | 0.1 ± 0.03 | 1.3 ± 0.04 | 2.5 ± 0.05 | 3.0 ± 0.08 | 0.1 ± 0.04 | 0.9 ± 0.19 | 2.2 ± 0.09 | 2.9 ± 0.15 |
| P | 1.0 ± 0.07 | 1.0 ± 0.03 | 1.1 ± 0.03 | 1.1 ± 0.03 | 1.2 ± 0.04 | 1.2 ± 0.07 | 1.3 ± 0.20 | 1.3 ± 0.19 |
| K | 3.0 ± 0.11 | 2.9 ± 0.08 | 3.1 ± 0.14 | 2.6 ± 1.22 | 3.3 ± 0.43 | 3.1 ± 0.26 | 3.4 ± 0.53 | 2.8 ± 1.42 |
| Ca | 2.1 ± 0.1 | 2 ± 0.01 | 1.3 ± 0.02 | 1.3 ± 0.04 | 2.3 ± 0.23 | 2.4 ± 0.40 | 1.8 ± 0.59 | 1.8 ± 0.57 |
| Mg | 0.3 ± 0.02 | 0.3 ± 0.01 | 0.3 ± 0.02 | 0.3 ± 0.09 | 0.3 ± 0.01 | 0.3 ± 0.03 | 0.4 ± 0.02 | 0.3 ± 0.1 |
| S | 0.7 ± 0.04 | 0.7 ± 0.04 | 1.2 ± 0.06 | 1.8 ± 0.11 | 0.9 ± 0.12 | 0.8 ± 0.05 | 1.5 ± 0.23 | 2.2 ± 0.40 |
| pH | 6.6 ± 0.13 | 6.6 ± 0.07 | 6.5 ± 0.08 | 6.4 ± 0.09 | 6.4 ± 0.08 | 5.3 ± 1.10 | 5.1 ± 0.99 | 5.0 ± 0.99 |
| EC | 1.1 ± 0.03 | 1.3 ± 0.02 | 1.3 ± 0.04 | 1.3 ± 0.18 | 1.2 ± 0.14 | 1.4 ± 0.16 | 1.5 ± 0.26 | 1.4 ± 0.37 |
| Treatments | Nitrogen (N) | Phosphorus (P) | Potassium (K) | Calcium (Ca) | Sulphur (S) | Magnesium (Mg) |
|---|---|---|---|---|---|---|
| 100%:0% | 2.9 ± 0.23 | 0.59 ± 0.06 | 2.72 ± 0.59 | 1.71 ± 0.28 | 0.15 ± 0.00 | 0.27 ± 0.00 |
| 80%:20% | 2.9 ± 0.18 | 0.55 ± 0.04 | 2.68 ± 0.38 | 1.60 ± 0.14 | 0.18 ± 0.04 | 0.32 ± 0.01 |
| 60%:40% | 3.0 ± 0.08 | 0.49 ± 0.04 | 2.58 ± 0.06 | 0.94 ± 0.04 | 0.21 ± 0.06 | 0.28 ± 0.01 |
| 50%:50% | 3.2 ± 0.25 | 0.50 ± 0.01 | 2.73 ± 0.11 | 1.03 ± 0.37 | 0.27 ± 0.08 | 0.28 ± 0.05 |
| Treatments | Dry Mass (g) | Crown Diameter (cm) |
|---|---|---|
| 100%:0% | 22.81 ± 10.94 | 3.54 ± 1.35 |
| 80%:20% | 18.38 ± 8.61 | 3.30 ± 0.98 |
| 60%:40% | 16.63 ± 5.62 | 3.19 ± 1.03 |
| 50%:50% | 19.63 ± 6.07 | 3.31 ± 0.71 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, X.; Hernández, R.; Hoffmann, M. Impact of Nitrate and Ammonium Ratios on Flowering and Asexual Reproduction in the Everbearing Strawberry Cultivar Fragaria × ananassa Albion. Horticulturae 2021, 7, 571. https://doi.org/10.3390/horticulturae7120571
Shi X, Hernández R, Hoffmann M. Impact of Nitrate and Ammonium Ratios on Flowering and Asexual Reproduction in the Everbearing Strawberry Cultivar Fragaria × ananassa Albion. Horticulturae. 2021; 7(12):571. https://doi.org/10.3390/horticulturae7120571
Chicago/Turabian StyleShi, Xiaonan, Ricardo Hernández, and Mark Hoffmann. 2021. "Impact of Nitrate and Ammonium Ratios on Flowering and Asexual Reproduction in the Everbearing Strawberry Cultivar Fragaria × ananassa Albion" Horticulturae 7, no. 12: 571. https://doi.org/10.3390/horticulturae7120571
APA StyleShi, X., Hernández, R., & Hoffmann, M. (2021). Impact of Nitrate and Ammonium Ratios on Flowering and Asexual Reproduction in the Everbearing Strawberry Cultivar Fragaria × ananassa Albion. Horticulturae, 7(12), 571. https://doi.org/10.3390/horticulturae7120571





