Abstract
The feasibility of utilising spent mushroom substrates (SMSs) as a growing medium component for Pleurotus ostreatus cultivation was investigated. P. ostreatus was cultivated on traditional wheat straw (control) and wheat straw substrate supplemented with SMSs from Pholiota nameko (N-SMS), Hypsizygus marmoreus (M-SMS), and Hericium erinaceus (E-SMS) in varying supplementation rates (10%, 20%, and 30%). The yield, biological efficiency (BE), dry matter, and protein content of P. ostreatus fruiting bodies grown on ten substrates were evaluated. Significant differences in yield, BE, protein content, and dry matter of P. ostreatus were found among the studied substrates. The highest yield was recorded in 20% E-SMS (254.33 g), 20% N-SMS (253.43 g), and 10% E-SMS (251.67 g). The biological efficiency ranged from 66.48% (30% M-SMS) to 72.67% (20% E-SMS) and followed a similar trend to yield. The highest protein content was recorded in 30% M-SMS (29.93 g∙100 g dry weight−1). The highest dry matter of P. ostreatus was noticed in 30% of M-SMS (23.74 g) and 10% of M-SMS (23.06 g). Therefore, the spent mushroom substrates of Ph. nameko, H. marmoreus, and H. erinaceus could be used as a potential, low-cost sustainable alternative (10–30%) and as a renewable component of traditional growing media for P. ostreatus cultivation.
1. Introduction
Mushrooms have been used as food since ancient times and appreciated for their flavour and health-promoting properties [1,2,3]. They have also been considered as both functional food and a source of nutraceuticals [4,5,6,7]. Recently, besides as a nutritive and healthy food, mushrooms have been extensively utilised as a potential component in cosmetic industries [8,9]. In recent years, the production of mushrooms worldwide has been steadily increasing. The button mushroom, Agaricus bisporus, is the most commercially cultivated species in the world and covers 40% of the world’s mushroom production [10]. Lentinula and four other genera, namely Agaricus, Pleurotus, Auricularia, and Flammulina, account for 85% of the world’s total supply of cultivated edible mushrooms [11]. It is also estimated that the value of mushroom cultivation in the world has reached nearly USD 17 billion in 2021, and it is further expected to reach nearly USD 20.4 billion in 2026 [12].
Mushrooms are capable of degrading several lignocellulosic materials [13,14] and can be grown on biological, agricultural, or agro-industrial wastes [15,16,17,18]. Poppe [19] reported that there are about 200 kinds of waste in which edible mushrooms can be produced. Although in mushroom production wastes are converted into valuable food, at the same time, a large amount of organic residues is generated. Every kilogram of mushrooms produced results in 5–6 kg of by-product [20,21]. This by-product is known as spent mushroom substrate (SMS) and is often considered as farm waste [22,23]. As the mushroom industry is growing, the volume of SMS generated annually is also increasing. Recently, the global mushroom production has reached nearly 11.5 million tons [24], at the same time accumulating around 55 million tons of SMS (mushroom production multiplied by five times).
Considering the accumulating amount of SMS, mushroom production can be regarded as a non-sustainable agriculture activity. A review by Geissdoerfer et al. [25] defined a circular economy as a regenerative system in which generated wastes are minimised by closing or narrowing the loop. The possible utilisation of SMS in subsequent mushroom production is of great importance. In a review by Zied et al. [26], the need to re-use SMS considering environmental viability, which can create a transition towards a circular economy, and the call for scientific information in this regard were emphasised.
Improper handling of SMS may lead to various environmental hazards [27,28]. Due to the high nutritional value of SMS [29,30] and the environmental impact, more efficient use is urgent for its recycling [21,31]. There are many potential agricultural, horticultural, and industrial uses of SMS [21,23,31,32]. According to several authors [26,32,33,34], the effective utilisation of SMS has great potential in a circular economy.
The best and most economically viable method for the disposal of SMS is considered to be its re-utilisation in successive mushroom cultivation. Several studies have reported the use of SMS for mushroom re-cultivation [26,35,36,37,38,39,40,41].
Pleurotus species are popular and widely cultivated mushrooms throughout the world because of their simple, low-cost production technology and high biological efficiency [42,43]. The consumption of Pleurotus mushrooms is increasing due to their taste and nutritional and medicinal properties [4,44,45,46,47,48,49,50]. Furthermore, some of Pleurotus’s properties are widely used in many biotechnological and environmental applications [51,52,53,54].
Pleurotus ostreatus (also known as oyster mushroom or black oyster) is one among 40 Pleurotus species [55]. Pleurotus spp. are cultivated in a variety of substrates and conditions [42,43,50,56,57,58]. One of the most important aspects of Pleurotus cultivation is the rational choice of the substrate [59]. Pleurotus can be produced on a large variety of plant residues and agro-industrial by-products [13,60,61]. Growers typically select the most effective, economical, and locally available substrate materials for production [62,63,64,65]. The adaptation of Pleurotus to new wastes can provide a method of converting lignocellulosic materials into edible products with high nutritional value [50,56,66,67]. According to Adebayo and Martínez-Carrera [68], Grimm et al. [69], and Mahari et al. [70], SMS generated as a by-product of Pleurotus cultivation can be used in agriculture and energy conversion.
Suitable and efficient production of Pleurotus spp. is related to a number of factors, which may act individually or have interactive effects among them [42,43,71]. In many studies, remarkable variations have been observed in the nutritional contents of P. ostreatus grown on different substrates [72,73,74,75]. Generally, wheat straw is used as the main substrate for P. ostreatus commercial cultivation [13]; however, there is a growing interest in highly available low-cost alternative materials [76,77,78,79]. According to Ashrafi et al. [39], Jo et al. [80], Pardo-Giménez et al. [81], Wang et al. [82], Picornell et al. [83], Picornell-Buendía et al. [84], and Economou et al. [85], spent mushroom substrate can be an effective component of traditional growing media for the cultivation of oyster mushrooms.
The cultivation of P. ostreatus on straw-based substrates has been extensively studied [50,86], while scientific reports on utilising non-traditional substrate substitutes, for instance, sawdust [60], and/or the re-utilisation of SMS as a substrate substitute for P. ostreatus cultivation still remain in their infancy. Hence, knowledge on the possible utilisation of SMSs from different medicinal and industrial mushroom species in the cultivation of commercial P. ostreatus will be of great significance. With this background information, the present investigation was aimed to determine the feasibility of utilising the spent substrates of Pholiota nameko, Hypsizygus marmoreus, and Hericium erinaceus as components of growing media for the cultivation of P. ostreatus. The yield, biological efficiency, dry matter, and protein content of P. ostreatus fruiting bodies cultivated on SMS substituted traditional growing media and traditional wheat straw-based substrates were evaluated.
2. Materials and Methods
2.1. Fungal Strain
Pleurotus ostreatus mycelium was obtained from the fungal culture collection of the Department of Vegetable Crops, the Poznan University of Life Sciences (Poland). The mycelium was maintained on a malt extract agar medium (MEA) at 25 °C.
2.2. Substrate Preparation
The traditional straw-based substrate for P. ostreatus cultivation was prepared from wheat straw cut in an agricultural crusher (fraction 0.5–2.0 cm), constituting 96% straw, 3% wheat bran, and 1% gypsum (w/w, in terms of dry weight), which was considered as the control substrate in the study.
The substrate combinations tested in this study were prepared from the wheat straw-based substrate and spent mushroom substrates (SMSs) after the cultivation of Pholiota nameko (N-SMS), Hypsizygus marmoreus (M-SMS), and Hericium erinaceus (E-SMS). These three mushroom species were previously cultivated on a mixture of oak and beech sawdust (1:1 vol.) supplemented with 20% wheat bran, 5% corn flour, and 1% gypsum (w/w, in terms of dry weight).
The wheat straw-based substrate and spent mushroom substrates were mixed in varying rates (10%, 20%, and 30%) to obtain nine substrate combinations (based on SMS) and were compared to 100% wheat straw-based substrate. In the present investigation, ten substrates were evaluated for P. ostreatus cultivation (w/w, in terms of dry weight). The substrates tested in this study are given in Table 1.

Table 1.
Substrates used in the study for the cultivation of P. ostreatus.
All prepared substrates (w/w, in terms of dry weight) were dampened with tap water to a moisture content of 65%. The substrates were filled into polyethylene bags (17 × 33 cm) with a packing density of 1000 g per bag.
2.3. Spawn Preparation, Inoculation, and Incubation
The spawn preparation and/or multiplication was carried out according to the methodology described by Stamets [87]. The filled bags were pasteurised at 60 °C for 72 h. As soon as the substrate temperature reached room temperature, the substrates were inoculated with P. ostreatus spawn (mycelium on wheat grain) in the amount of 3% of substrate wet weight as described by Mleczek et al. [88]. The inoculated bags were later incubated at 25 °C and 85–90% air relative humidity (RH) under dark conditions until the substrates were completely covered with mycelium. Once the substrates were covered with mycelium, the bags were transferred to the cultivation room. In each bag, perforation (4 holes with a diameter of 1 cm) was made for fruiting bodies.
2.4. Cropping, Harvest, Determination of Biological Efficiency and Protein Content
Three bags were maintained in individual substrates facilitating three replicates. All bags were placed in a cultivation chamber, and the relative air humidity was maintained at 80–85% and temperature at 17 ± 1 °C. The cultivation was additionally lit with fluorescent light of 500 lx intensity for 10 h a day and aerated in such a way as to maintain CO2 concentration below 1000 ppm. Fruiting bodies were harvested successively as they matured, i.e., when the mushroom cap surfaces were flat to slightly up-rolled at the cap margins. Two flushes of P. ostreatus fruiting bodies were collected. The total duration of P. ostreatus was approximately six weeks (from incubation to the final harvest).
The fresh weight (i.e., yield g/bag) and dry weight of P. ostreatus fruiting bodies (g∙ 100 g fresh mushroom weight−1) were measured. At the end of the experiment, the recorded data were used to calculate biological efficiency (BE). Biological efficiency was calculated based on the formula suggested by Yang et al. [89]: BE (%) = weight of fresh mushrooms harvested per bag/weight of dry substrate per bag × 100.
The harvested fruiting bodies of P. ostreatus were initially dried at 40 °C for 24 h and later dried at 80 °C to obtain constant dry weight. The protein content in fruiting bodies was determined by the Kjeldahl thermal conductivity method. The conversion factor 4.38 was used to calculate the crude protein content [90].
2.5. Statistical Analyses
The experiment was carried out with ten substrates (treatments) in three replicates as a one-factor design, where different SMSs and their supplementation rates were studied. The data obtained from the experiment were subjected to a one-way ANOVA. The differences in means between substrates were later assessed using Duncan’s multiple range tests (DMRT) at a 95% confidence level (p < 0.05). The statistical analyses were conducted using STATISTICA 10.0 software for windows by TIBCO Software Inc. (StatSoft Inc., Tulsa, OK, USA).
3. Results
3.1. Yield
The yield of P. ostreatus was significantly influenced by the studied substrates (Figure 1). In the present investigation, the highest yield of P. ostreatus fruiting bodies was recorded in 20% E-SMS, 20% N-SMS, and 10% E-SMS (254.33 g, 253.43 g, and 251.67 g, respectively). There were no significant differences concerning yield performances among traditional wheat straw-based substrate (control), 10% N-SMS, 20% M-SMS, and 30% E-SMS, while the lowest yield of P. ostreatus was noticed in substrates with 30% M-SMS (232.70 g) and 30% N-SMS (232.93 g). Based on the yield performances among the studied substrates, it can be inferred that the higher supplementation rates (30%) of N-SMS and M-SMS resulted in lower yields.
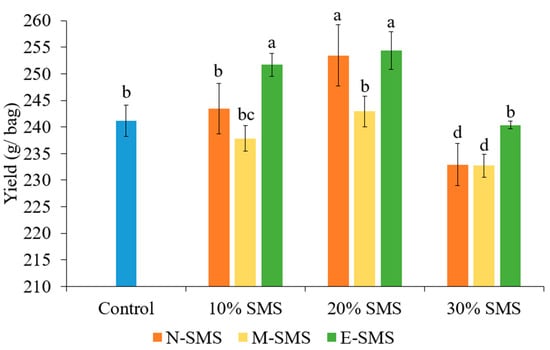
Figure 1.
The yield of P. ostreatus fruiting bodies in different substrates (mean ± SD, n = 3). Different letters above the bars indicate significant differences at p < 0.05.
3.2. Biological Efficiency
The biological efficiency (BE) of P. ostreatus fruiting bodies significantly differed among the studied substrates (Figure 2). The BE among the studied substrates ranged from 66.48% (30% M-SMS) to 72.67% (20% E-SMS) and followed a similar trend as yield. The highest BE was noticed in 20% E-SMS, 20% N-SMS, and 10% E-SMS (72.67%, 72.41%, and 71.9%, respectively), while the lowest BE was recorded in 30% M-SMS and 30% N-SMS (66.48% and 66.55%).
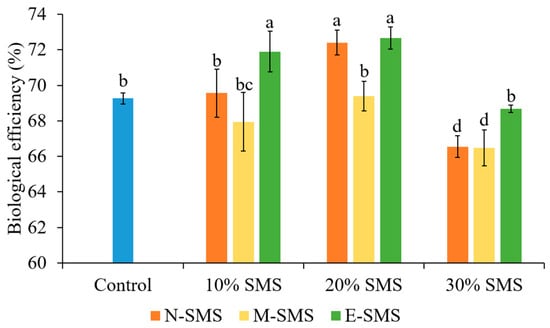
Figure 2.
Biological efficiency of P. ostreatus fruiting bodies in different substrates (mean ± SD, n = 3). Different letters above the bars indicate significant differences at p < 0.05.
3.3. Protein Content
In the present study, the protein content of P. ostreatus fruiting bodies significantly varied among the studied substrates (Figure 3). The highest amount of protein in fruiting bodies of P. ostreatus was recorded in the substrate supplemented with 30% M-SMS (29.93 g∙dry weight−1). This highest amount of protein was followed by 10% M-SMS (29.53 g∙100 g dry weight−1) and 30% E-SMS (29.43 g∙dry weight−1), which were observed to be similar. However, the lowest amount of protein content was observed in other substrates except for 30% N-SMS. The results of protein content suggest that the P. ostreatus cultivated on substrates supplemented with 30% N-SMS, M-SMS, and E-SMS as well as 10% M-SMS achieved a higher protein content than that of P. ostreatus fruiting bodies grown on 100% traditional wheat straw-based substrate (control).
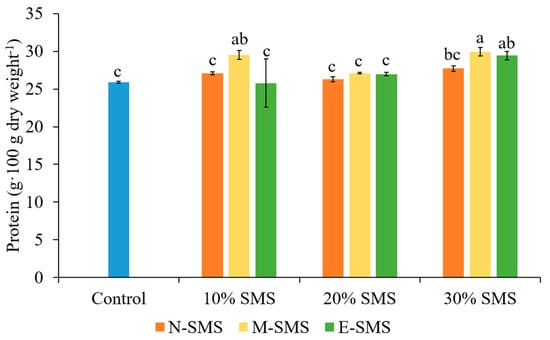
Figure 3.
The protein content of P. ostreatus fruiting bodies in different substrates (mean ± SD, n = 3). Different letters above the bars indicate significant differences at p < 0.05.
3.4. Dry Matter
The dry matter of P. ostreatus fruiting bodies significantly varied among the studied substrates (Figure 4). The highest dry matter of P. ostreatus was observed in mushrooms grown on substrates supplemented with 30% M-SMS (23.74 g) and 10% M-SMS (23.06 g). There were no significant differences among 100% wheat straw-based substrate (control) and all studied supplementation rates of E-SMS and 10% N-SMS, in which the lowest P. ostreatus dry weight was observed.
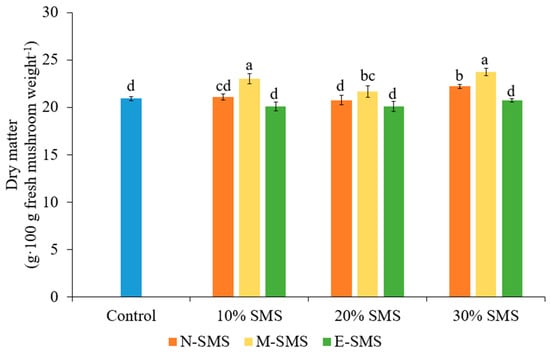
Figure 4.
Dry matter content of P. ostreatus in different substrates (mean ± SD, n = 3). Different letters above the bars indicate significant differences at p < 0.05.
Overall, the supplementation (10–30%) of N-SMS, M-SMS, and E-SMS to traditional wheat straw-based substrate resulted in achieving superior and/or equal yield, BE, protein content, and dry matter of P. ostreatus fruiting bodies when compared to 100% wheat straw-based substrate. The yield and BE of P. ostreatus were observed to be superior in 20% N-SMS, 10% E-SMS, and 20% E-SMS supplementation, while the highest protein content of P. ostreatus fruiting bodies was recorded in 30% M-SMS and superior dry matter was also noticed in 30% and 10% M-SMS supplemented substrates. In this context, the substrates that resulted in higher yields and BE had a lower protein content as well as dry matter.
4. Discussion
Pleurotus ostreatus can be produced on a variety of lignocellulosic substrates [42]. The rational choice of the substrate is one of the most important aspects in mushroom cultivation [59]. Recently, there is a growing interest in utilising easily available low-cost alternative materials for P. ostreatus cultivation [76,77,78,79]. Spent mushroom substrate can be a good component for P. ostreatus cultivation [80,81,82,83,84,85]. However, scientific evidence supporting the utilisation of spent substrates especially from other mushroom species in P. ostreatus cultivation is still in its infancy.
The main objective of the presented study was to determine the feasibility of utilising the spent substrates of three mushroom species, Pholiota nameko, Hypsizygus marmoreus, and Hericium erinaceus, as components of growing media for the cultivation of P. ostreatus. In the study, wheat straw-based substrate (control) and three spent mushroom substrates in three different supplementation rates (10%, 20%, and 30%) were used, in which the yield, biological efficiency, dry matter, and protein content of P. ostreatus fruiting bodies were evaluated. It has been reported that, in mushroom production, many factors might influence the yield and performance, such as substrate composition, moisture, and density, as well as temperature, relative humidity, and light intensity [71]. In this work, to eliminate the influence of such factors, substrate moisture, packing density, and growing conditions including temperature, relative humidity, lighting, and CO2 concentrations were maintained at the same level in all substrate combinations.
Significant differences in the yield of P. ostreatus among studied substrates were found in the present investigation. The yield was increased in the substrates supplemented with H. erinaceus (10% and 20%) and Ph. nameko (20%) spent substrates when compared to the control substrate and decreased as the supplementation rates of added SMS (N-SMS, M-SMS, and E-SMS) were higher (30%). The obtained results are in line with Wang et al. [82], who reported that 12–25% of added H. marmoreus spent substrate resulted in a better yield of P. ostreatus and the yield decreased when SMS supplementation was further continued. Siqueira et al. [91] evaluated the reuse of P. ostreatus spent substrate in P. ostreatus cultivation and noticed the best results on the substrate supplemented with lower supplementation rates, i.e., with 26% of SMS, while higher supplementation (45 and 64%) decreased the yield. Ashrafi et al. [39] also reported the highest yield for P. ostreatus in the substrate supplemented with a low concentration (20%) of P. ostreatus spent substrate.
The higher yield of P. ostreatus in the present study when compared to the control substrate was likely influenced by SMS supplementation, where N-SMS and E-SMS mixed with 80% of wheat straw-based substrate resulted in a higher yield than that of 100% wheat straw-based substrate. This superiority in the yield of P. ostreatus fruiting bodies was probably influenced by added SMS and previously cultivated mushroom species (Ph. nameko and H. erinaceus), which were used for P. ostreatus cultivation. As reported by Catal and Peksen [30], the composition of SMS, among others, mainly depends on the mushroom species produced and/or cultivated. According to Wang et al. [82], SMS contains easily available organic nitrogen, which can benefit mushroom growth. In the studies by Jo et al. [80], nitrogen content in the substrates supplemented with spent oyster mushroom substrate positively influenced the yield of P. ostreatus. Ritota and Manzi [43] and Jafarpour and Eghbalsaeed [92] also reported that a substrate supplemented with protein-rich materials can positively influence the mushroom yield.
The biological efficiency (BE) in the presented study ranged from 66.48% with 30% M-SMS to 72.67% with 20% E-SMS and followed a similar trend as yield. Among ten substrates concerning BE, the substrate with 20% E-SMS, 20% N-SMS, and 10% E-SMS showed the highest BE of 72.67%, 72.41%, and 71.90%, respectively.
The obtained BE values in the present study were found to be higher than those of Wang et al. [82], who recorded BE between 35.87% and 61.26% when P. ostreatus was cultivated on the various supplementations of the spent substrate from H. marmoreus, and Pardo-Giménez et al. [81] recorded BE from 28.7% to 48.9% for P. ostreatus grown on spent oyster mushroom substrate. However, Ashrafi et al. [39] reported a higher BE value (78.4–107.5%) for P. ostreatus grown on spent oyster mushroom substrate alone and in combination with sawdust and wheat bran. The highest BE value for P. ostreatus (185.38%) recorded in the literature among various lignocellulosic substrates was noticed by Economou et al. [85] on the SMS of P. ostreatus.
Picornell et al. [83] and Picornell Buendía et al. [84] reported the wide range of BE (2.77–62.48% and 0.2–65.5%, respectively) for P. ostreatus grown on substrates supplemented with different doses of P. ostreatus spent substrate. Siqueira et al. [91] studied the reuse of P. ostreatus spent substrate in varying supplementations and also noticed a wide range of BE from 3.85% to 42.86%, while in our study, a wider range of BE among the studied substrates was not noticed and the BE ranged from 66.48% to 72.67%.
In the studies of Ashrafi et al. [39], Pardo-Giménez et al. [81], Wang et al. [82], and Picornell Buendía et al. [84], the yield and BE on substrates with SMS supplementation were directly proportional to each other. Similar results were obtained in the present study. Based on the experimental results concerning yield and BE values, it can be inferred that the yield and BE followed a similar trend, where higher yield resulted in a higher BE value. SMS supplementation, especially 10% and 20% E-SMS and 20% N-SMS, resulted in superior yield and BE values.
The protein content of P. ostreatus fruiting bodies in the study was between 25.77 g∙100 g dry weight−1 and 29.93 g∙100 g dry weight−1. The content was found to be improved by substrate supplementation with SMS. As reported by Raman et al. [50], the protein content in P. ostreatus may depend on many factors. The significant influence of the kind of cultivation substrate on the protein content in P. ostreatus fruiting bodies was noticed by many researchers [72,74,93,94]. Hoa et al. [58] reported that the differences in the protein content of P. ostreatus fruiting bodies could be due to the varying nitrogen content of substrates. In the present study, the protein content was significantly influenced by the substrate composition and the best results were obtained for H. marmoreus spent substrate. The protein content of P. ostreatus was slightly similar to the range of Wang et al. [82], who reported protein content from 29.20 g to 34.39 g on substrate supplemented with H. marmoreus spent substrate, and Economou et al. [85] recorded 22.24 g on SMS of P. ostreatus. The content of protein obtained in this study was found to be higher than that of Pardo-Giménez et al. [81] and Siqueira et al. [91] on the spent oyster mushroom substrate (16.23 g–21.06 g and 17.02 g–19.08 g, respectively). As reported by Ritota and Manzi [43], the substrates with the highest yield also gave the highest protein content. In the present study, this tendency was not observed, where the substrates that had lower yield (30% N-SMS and M-SMS) than that of the control (100% wheat straw-based substrate) resulted in a higher amount of protein. Based on the obtained results, it can be inferred that the increase in protein content did not often result in a higher yield. A similar relationship between protein content and yield was reported by Gupta et al. [44].
The dry matter of P. ostreatus fruiting bodies in the present study was recorded between 20.08 g∙100 g fresh mushroom weight−1 and 23.74 g∙100 g fresh mushroom weight−1. The highest dry matter was noticed for the substrates supplemented with 10% and 30% of H. marmoreus spent substrate. The higher dry matter of P. ostreatus fruiting bodies also resulted in a higher amount of protein, which is in line with findings of Pardo-Giménez et al. [81], who reported that the higher dry matter of P. ostreatus fruiting bodies had superior protein content. However, in the present study, the substrates in which a higher dry matter of P. ostreatus was recorded (30% N-SMS and 30% M-SMS) had lower yields than that of the control substrate (wheat straw-based substrate). Similar findings were reported by Pardo-Giménez et al. [81], where the wheat straw-based substrate with the lowest dry matter had a higher yield of P. ostreatus fruiting bodies.
In the present investigation, the differences among yield, BE, protein content, and dry matter of P. ostreatus fruiting bodies among studied substrates were possibly influenced by the previously cultivated mushroom species [30]. In other words, SMSs obtained from different mushroom species (Ph. nameko, H. marmoreus, and H. erinaceus) greatly influenced the performances of P. ostreatus fruiting bodies. Overall, the outcomes of the present investigation demonstrated the suitability and potential utilisation of a designated agro-waste from mushroom enterprises (SMS) in successive P. ostreatus production. Such effective and immediate re-utilisation of SMS in successive mushroom production can achieve environmental sustainability [21], create a transition towards a circular economy [26], and can help to make mushroom production a zero-waste enterprise [95].
5. Conclusions
The spent substrates of three mushroom species, Ph. nameko, H. marmoreus, and H. erinaceus, could be used as a low-cost alternative and as renewable components of traditional growing media for the cultivation of P. ostreatus. In particular, considering the results of the present study, 10–30% of SMS supplementation has been found to be optimal. The utilisation of SMS could provide an economically acceptable, efficient, and sustainable production alternative, at the same time generating additional income for mushroom growers. Such effective and immediate utilisation of accumulating agro-waste from mushroom enterprises (SMS) can create a transition towards a circular economy.
Author Contributions
Conceptualization, J.L. and R.P.; methodology, J.L. and R.P.; validation, J.L., R.P. and A.J.; formal analysis, R.P.; investigation, J.L. and R.P.; resources, J.L., R.P. and A.J.; data curation, R.P. and J.L.; writing—original draft preparation, J.L. and R.P.; writing—review and editing, J.L., R.P. and A.J.; visualization, R.P.; supervision, J.L. and A.J. All authors have read and agreed to the published version of the manuscript.
Funding
The research was financed by the Department of Vegetable Crops, Faculty of Agronomy, Horticulture and Bioengineering, Poznan University of Life Sciences, Poland. The publication was co-financed from the European Union’s Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 751052.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author.
Acknowledgments
We sincerely acknowledge Marek Siwulski for providing P. ostreatus fungal strains and other material help during the study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Roncero-Ramos, I.; Delgado-Andrade, C. The beneficial role of edible mushrooms in human health. Curr. Opin. Food Sci. 2017, 14, 122–128. [Google Scholar] [CrossRef]
- Chaturvedi, V.K.; Agarwal, S.; Gupta, K.K.; Ramteke, P.W.; Singh, M.P. Medicinal mushroom: Boon for therapeutic applications. 3 Biotech 2018, 8, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Venturella, G.; Ferraro, V.; Cirlincione, F.; Gargano, M.L. Medicinal Mushrooms: Bioactive Compounds, Use, and Clinical Trials. Int. J. Mol. Sci. 2021, 22, 634. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-González, J.A.; Serna-Saldívar, S.O.; Gutiérrez-Uribe, J.A. Nutritional composition and nutraceutical properties of the Pleurotus fruiting bodies: Potential use as food ingredient. J. Food Compos. Anal. 2017, 58, 69–81. [Google Scholar] [CrossRef]
- Reis, F.S.; Martins, A.; Vasconcelos, M.H.; Morales, P.; Ferreira, I.C.F.R. Functional foods based on extracts or compounds derived from mushrooms. Trends Food Sci. Technol. 2017, 66, 48–62. [Google Scholar] [CrossRef]
- Ma, G.; Yang, W.; Zhao, L.; Pei, F.; Fang, D.; Hu, Q. A critical review on the health promoting effects of mushrooms nutraceuticals. Food Sci. Hum. Wellness. 2018, 7, 125–133. [Google Scholar] [CrossRef]
- Raghavendra, V.B.; Venkitasamy, C.; Pan, Z.; Nayak, C. Functional Foods from Mushroom. In Microbial Functional Foods and Nutraceuticals; Gupta, V.K., Treichel, H.V., Shapaval, V., Antonio de Oliveira, L., Tuohy, M.G., Eds.; Wiley: Hoboken, NJ, USA, 2017; pp. 65–91. [Google Scholar] [CrossRef]
- Taofiq, O.; González-Paramás, A.M.; Martins, A.; Barreiro, M.F.; Ferreira, I.C.F.R. Mushrooms extracts and compounds in cosmetics, cosmeceuticals and nutricosmetics—A review. Ind. Crops Prod. 2016, 90, 38–48. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Choi, M.H.; Li, J.; Yang, H.; Shin, H.J. Mushroom Cosmetics: The Present and Future. Cosmetics 2016, 3, 22. [Google Scholar] [CrossRef]
- Mushroom Cultivation Market by Type (Button Mushroom, Oyster Mushroom, Shiitake Mushroom, Other Types), By Phase, By Region (North America, Europe, Asia Pacific, South America, Rest of the World)—Global Forecast to 2025 Mushroom Cultivation Market by Type (Button Mushroom, Oyster Mushroom, Shiitake Mushroom, Other Types), By Phase, By Region (North America, Europe, Asia Pacific, South America, Rest of the World)—Global Forecast to 2025. Available online: https://www.researchandmarkets.com/reports/5018601/mushroom-cultivation-market-by-type-button (accessed on 20 July 2021).
- Royse, D.J.; Baars, J.; Tan, Q. Current Overview of Mushroom Production in the World. In Edible and Medicinal Mushrooms; Wiley: Hoboken, NJ, USA, 2017; pp. 5–13. [Google Scholar] [CrossRef]
- Market Data Forecast 2021, Mushroom Cultivation Market by Type (Button Mushroom, Shiitake Mushroom, Oyster Mushroom, and Others), By Region (North America, Europe, Asia Pacific, South America, Rest of the World)—Forecast to 2026. Available online: https://www.marketdataforecast.com/market-reports/mushroom-cultivation-market (accessed on 20 July 2021).
- Philippoussis, A.; Zervakis, G.; Diamantopoulou, P. Bioconversion of agricultural lignocellulosic wastes through the cultivation of the edible mushrooms Agrocybe aegerita, Volvariella volvacea and Pleurotus spp. World J. Microbiol. Biotechnol. 2001, 17, 191–200. [Google Scholar] [CrossRef]
- Sánchez, C. Lignocellulosic residues: Biodegradation and bioconversion by fungi. Biotechnol. Adv. 2009, 27, 185–194. [Google Scholar] [CrossRef]
- Philippoussis, A.N. Production of Mushrooms Using Agro-Industrial Residues as Substrates. In Biotechnology for Agro-Industrial Residues Utilisation; Springer: Dordrecht, The Netherlands, 2009; pp. 163–196. [Google Scholar] [CrossRef]
- Barshteyn, V.; Krupodorova, T. Utilization of agro-industrial waste by higher mushrooms: Modern view and trends. J. Microbiol. Biotechnol. Food Sci. 2016, 5, 563–577. [Google Scholar] [CrossRef] [Green Version]
- Kamthan, R.; Tiwari, I. Agricultural Wastes—Potential Substrates for Mushroom Cultivation. Eur. J. Exp. Biol. 2017, 7, 31. [Google Scholar] [CrossRef]
- Kumla, J.; Suwannarach, N.; Sujarit, K.; Penkhrue, W.; Kakumyan, P.; Jatuwong, K.; Vadthanarat, S.; Lumyong, S. Cultivation of Mushrooms and Their Lignocellulolytic Enzyme Production through the Utilization of Agro-Industrial Waste. Molecules 2020, 25, 2811. [Google Scholar] [CrossRef] [PubMed]
- Poppe, J. Use of agricultural waste materials in the cultivation of mushrooms. Mushroom Sci. 2000, 15, 3–22. [Google Scholar]
- Ma, Y.; Wang, Q.; Sun, X.; Wang, X.; Su, W.; Song, N. A Study on Recycling of Spent Mushroom Substrate to Prepare Chars and Activated Carbon. BioResources 2014, 9, 3939–3954. [Google Scholar] [CrossRef] [Green Version]
- Hanafi, F.H.; Rezania, S.; Taib, S.M.; Din, M.F.; Yamauchi, M.; Sakamoto, M.; Hara, H.; Park, J.; Ebrahimi, S.S. Environmentally sustainable applications of agro-based spent mushroom substrate (SMS): An overview. J. Mater. Cycles Waste Manag. 2018, 20, 1383–1396. [Google Scholar] [CrossRef]
- Phan, C.W.; Sabaratnam, V. Potential uses of spent mushroom substrate and its associated lignocellulosic enzymes. Appl. Microbiol. Biotechnol. 2012, 96, 863–873. [Google Scholar] [CrossRef]
- Rinker, D.L. Spent Mushroom Substrate Uses. In Edible and Medicinal Mushrooms; Diego, C.Z., Pardo-Giménez, A., Eds.; Wiley-Blackwell: West Sussex, UK, 2017; pp. 427–454. [Google Scholar] [CrossRef]
- Food and Agricultural Organization of the United Nations (FAO). FAO Statistics Division. 2019. Available online: http://www.fao.org/faostat/en/#data/QCL (accessed on 20 July 2021).
- Geissdoerfer, M.; Savaget, P.; Bocken, N.M.P.; Hultink, E.J. The Circular Economy—A new sustainability paradigm? J. Clean. Prod. 2017, 143, 757–768. [Google Scholar] [CrossRef] [Green Version]
- Zied, D.C.; Sánchez, J.E.; Noble, R.; Pardo-Giménez, A. Use of Spent Mushroom Substrate in New Mushroom Crops to Promote the Transition towards A Circular Economy. Agronomy 2020, 10, 1239. [Google Scholar] [CrossRef]
- Rao, J.R.; Nelson, D.; Lafferty, N.; Moore, J.E.; Millar, B.C.; Xu, J.; Watabe, M. Biohazards and ecotoxicological considerations of landspreading of spent compost wastes. Commun. Agric. Appl. Biol. Sci. 2003, 68, 885–892. [Google Scholar]
- Lou, Z.; Sun, Y.; Bian, S.; Ali Baig, S.; Hu, B.; Xu, X. Nutrient conservation during spent mushroom compost application using spent mushroom substrate derived biochar. Chemosphere 2017, 169, 23–31. [Google Scholar] [CrossRef]
- Kumbhar, A.; Gade, R.; Shitole, A.; Bandgar, M. Nutritional value of spent mushroom substrate of Agaricus bisporus. J. Mycopathol. Res. 2014, 52, 65–68. [Google Scholar]
- Catal, S.; Peksen, A. Physical, chemical and biological properties of spent mushroom substrates of different mushroom species. Acta Hortic. 2020, 1287, 353–360. [Google Scholar] [CrossRef]
- Antunes, F.; Marçal, S.; Taofiq, O.; Morais, A.; Freitas, A.C.; Ferreira, I.; Pintado, M. Valorization of Mushroom by-Products as a Source of Value-Added Compounds and Potential Applications. Molecules 2020, 25, 2672. [Google Scholar] [CrossRef] [PubMed]
- Grimm, D.; Wösten, H.A.B. Mushroom cultivation in the circular economy. Appl. Microbiol. Biotechnol. 2018, 102, 7795–7803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jasińska, A. Spent mushroom compost (SMC)—Retrieved added value product closing loop in agricultural production. Acta Agrar. Debr. 2018, 150, 185–202. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Rajan, S.; Sarsaiya, S.; Jain, S.K. Mushroom for the National Circular Economy. Int. J. Sci. 2020, 7, 58–66. [Google Scholar]
- Mamiro, D.P.; Royse, D.J.; Beelman, R.B. Yield, size, and mushroom solids content of Agaricus bisporus produced on non-composted substrate and spent mushroom compost. World J. Microbiol. Biotechnol. 2007, 23, 1289–1296. [Google Scholar] [CrossRef]
- Mamiro, D.P.; Royse, D.J. The influence of spawn type and strain on yield, size and mushroom solids content of Agaricus bisporus produced on non-composted and spent mushroom compost. Bioresour. Technol. 2008, 99, 3205–3212. [Google Scholar] [CrossRef]
- Gern, R.M.M.; Libardi Junior, N.; Patrício, G.N.; Wisbeck, E.; Chaves, M.B.; Furlan, S.A. Cultivation of Agaricus blazei on Pleurotus spp. spent substrate. Braz. Arch. Biol. Technol. 2010, 53, 939–944. [Google Scholar] [CrossRef] [Green Version]
- González Matute, R.; Figlas, D.; Curvetto, N. Agaricus blazei production on non-composted substrates based on sunflower seed hulls and spent oyster mushroom substrate. World J. Microbiol. Biotechnol. 2011, 27, 1331–1339. [Google Scholar] [CrossRef]
- Ashrafi, R.; Mian, M.H.; Rahman, M.; Jahiruddin, M. Recycling of Spent Mushroom Substrate for the Production of Oyster Mushroom. Res. Biotechnol. 2014, 5, 13–21. [Google Scholar]
- Noonsong, V.; Puttakun, N.; Tinsirisuk, M.; Seephueak, P. Recycling of spent Pleurotus compost for production of the Agrocybe cylindracea. Mycosphere 2016, 7, 36–43. [Google Scholar] [CrossRef]
- Zeng, X.; Han, F.; Ye, J.; Zhong, Y. Recycling spent Pleurotus eryngii substrate supplemented with Tenebrio molitor feces for cultivation of Agrocybe chaxingu. Int. J. Recycl. Org. Waste Agric. 2017, 6, 275–280. [Google Scholar] [CrossRef] [Green Version]
- Sánchez, C. Cultivation of Pleurotus ostreatus and other edible mushrooms. Appl. Microbiol. Biotechnol. 2010, 85, 1321–1337. [Google Scholar] [CrossRef] [PubMed]
- Ritota, M.; Manzi, P. Pleurotus spp. Cultivation on Different Agri-Food By-Products: Example of Biotechnological Application. Sustainability 2019, 11, 5049. [Google Scholar] [CrossRef] [Green Version]
- Patel, Y.; Naraian, R.; Singh, V.K. Medicinal properties of Pleurotus Species (Oyster Mushroom): A Review. WJFPB 2012, 3, 1–12. [Google Scholar]
- Deepalakshmi, K.; Mirunalini, S. Pleurotus ostreatus: An oyster mushroom with nutritional and medicinal properties. J. Biochem. Tech. 2014, 5, 718–726. [Google Scholar]
- Ahmed, M.; Abdullah, N.; Nuruddin, N.N. Yield and nutritional composition of oyster mushrooms: An alternative nutritional source for rural people. Sains Malays. 2016, 45, 1609–1615. [Google Scholar] [CrossRef] [Green Version]
- Piska, K.; Sułkowska-Ziaja, K.; Muszyńska, B. Edible mushroom Pleurotus ostreatus (Oyster mushroom)—Its dietary significance and biological activity. Acta Sci. Pol. Hort. Cult. 2017, 16, 151–161. [Google Scholar]
- Sałata, A.; Lemieszek, M.; Parzymies, M. The nutritional and health properties of an oyster mushroom (Pleurotus ostreatus (Jacq. Fr) P. Kumm.). Acta Sci. Pol. Hortorum Cultus 2018, 17, 185–197. [Google Scholar] [CrossRef]
- Waktola, G.; Temesgen, T. Pharmacological activities of Oyster mushroom (Pleurotus ostreatus). Novel Res. Microbiol. J. 2020, 4, 688–695. [Google Scholar] [CrossRef]
- Raman, J.; Jang, K.Y.; Oh, Y.L.; Oh, M.; Im, J.H.; Lakshmanan, H.; Sabaratnam, V. Cultivation and Nutritional Value of Prominent Pleurotus spp.: An Overview. Mycobiology 2021, 49, 1–14. [Google Scholar] [CrossRef]
- Cohen, L.; Persky, Y.; Hadar, R. Biotechnological applications and potential of wood-degrading mushrooms of the genus Pleurotus. Appl. Microbiol. Biotechnol. 2002, 58, 582–594. [Google Scholar] [CrossRef]
- Grabarczyk, M.; Mączka, W.; Wińska, K.; Uklańska-Pusz, C.M. Mushrooms of the Pleurotus genus—Properties and application. Biotechnol. Food Sci. 2019, 83, 13–30. [Google Scholar]
- Sekan, A.S.; Myronycheva, O.S.; Karlsson, O.; Gryganskyi, A.P.; Blume, Y.B. Green potential of Pleurotus spp. in biotechnology. PeerJ. 2019, 7, e6664. [Google Scholar] [CrossRef] [Green Version]
- Bhardwaj, K.; Sharma, A.; Tejwan, N.; Bhardwaj, S.; Bhardwaj, P.; Nepovimova, E.; Shami, A.; Kalia, A.; Kumar, A.; Abd-Elsalam, K.A.; et al. Pleurotus Macrofungi-Assisted Nanoparticle Synthesis and Its Potential Applications: A Review. J. Fungi 2020, 6, 351. [Google Scholar] [CrossRef] [PubMed]
- Kües, U.; Liu, Y. Fruiting body production in basidiomycetes. Appl. Microbiol. Biotechnol. 2000, 54, 141–152. [Google Scholar] [CrossRef]
- Patil, S.S.; Ahmed, S.A.; Telang, S.M.; Baig, M.M.V. The nutritional value of Pleurotus ostreatus (Jacq.:Fr.) Kumm cultivated on different lignocellulosic agro-wastes. Innov. Rom. Food Biotechnol. 2010, 7, 66–76. [Google Scholar]
- Carvalho, C.S.; Sales-Campos, C.; Andrade, C.N. Mushrooms of the Pleurotus Genus: A review of cultivation techniques. Interciencia 2010, 35, 177–182. [Google Scholar]
- Hoa, H.T.; Wang, C.L.; Wang, C.H. The Effects of Different Substrates on the Growth, Yield, and Nutritional Composition of Two Oyster Mushrooms (Pleurotus ostreatus and Pleurotus cystidiosus). Mycobiology 2015, 43, 423–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Besufekad, Y.; Mekonnen, A.; Girma, B.; Daniel, R.; Tassema, G.; Melkamu, J.; Asefa, M.; Fikiru, T.; Denboba, L. Selection of appropriate substrate for production of oyster mushroom (Pleurotus ostreatus). J. Yeast Fungal Res. 2020, 11, 15–25. [Google Scholar] [CrossRef]
- Obodai, M.; Cleland-Okine, J.; Vowotor, K.A. Comparative study on the growth and yield of Pleurotus ostreatus mushroom on different lignocellulosic by-products. J. Ind. Microbiol. Biotechnol. 2003, 30, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Estrada, A.E.; Royse, D.J. Yield, size and bacterial blotch resistance of Pleurotus eryngii grown on cottonseed hulls/oak sawdust supplemented with manganese, copper and whole ground soybean. Bioresour. Technol. 2007, 98, 1898–1906. [Google Scholar] [CrossRef] [PubMed]
- Dlamini, B.E.; Earnshaw, D.M.; Masarirambi, M.T. Growth and yield response of Oyster Mushroom (Pleurotus ostreatus) grown on different locally available substrates. Curr. Res. J. Biol. Sci. 2012, 4, 623–629. [Google Scholar]
- Mintesnot, B.; Ayalew, A.; Kebede, A. Evaluation of Biomass of Some Invasive Weed Species as Substrate for Oyster Mushroom (Pleurotus spp.) Cultivation. Pak. J. Biol. Sci. 2014, 17, 213–219. [Google Scholar] [CrossRef] [Green Version]
- Kinge, T.R.; Djidjou, T.A.M.; Nji, T.M.; Ache, N.A.; Mih, A.M. Effect of local substrates on the growth and yield of Pleurotus ostreatus K. in the North West Region. Cameroon Curr. Res. Environ. 2016, 6, 11–19. [Google Scholar] [CrossRef]
- Atila, F. Evaluation of suitability of various agro-wastes for productivity of Pleurotus djamor, Pleurotus citrinopileatus and Pleurotus eryngii mushrooms. J. Exp. Agric. Int. 2017, 17, 1–11. [Google Scholar] [CrossRef]
- Fernandes, Â.; Barros, L.; Martins, A.; Herbert, P.; Ferreira, I.C.F.R. Nutritional characterisation of Pleurotus ostreatus (Jacq. ex Fr.) P. Kumm. produced using paper scraps as substrate. Food Chem. 2015, 169, 396–400. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Zhang, Z.; Li, M.; Li, X.; Sun, Z. Yield, size, nutritional value, and antioxidant activity of oyster mushrooms grown on perilla stalks. Saudi J. Biol. Sci. 2017, 24, 347–354. [Google Scholar] [CrossRef] [Green Version]
- Adebayo, E.A.; Martínez-Carrera, D. Oyster mushrooms (Pleurotus) are useful for utilizing lignocellulosic biomass. Afr. J. Biotechnol. 2015, 14, 52–67. [Google Scholar] [CrossRef]
- Grimm, A.; Eilertsen, L.; Chen, F.; Huang, R.; Atterhem, L.; Xiong, S. Cultivation of Pleurotus ostreatus Mushroom on Substrates Made of Cellulose Fibre Rejects: Product Quality and Spent Substrate Fuel Properties. Waste Biomass Valor. 2021, 12, 4331–4340. [Google Scholar] [CrossRef]
- Mahari, W.A.; Peng, W.; Nam, W.L.; Yang, H.; Lee, X.Y.; Lee, Y.K.; Liew, R.K.; Ma, N.L.; Mohammad, A.; Sonne, C.; et al. A review on valorization of oyster mushroom and waste generated in the mushroom cultivation industry. J. Hazard. Mater. 2020, 400, 123156. [Google Scholar] [CrossRef]
- Bellettini, M.B.; Fiorda, F.A.; Maieves, H.A.; Teixeira, G.L.; Ávila, S.; Hornung, P.S.; Júnior, A.M.; Ribani, R.H. Factors affecting mushroom Pleurotus spp. Saudi J. Biol. Sci. 2019, 26, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Sakoda, A.; Suzuki, M. Biological efficiency and nutritional value of Pleurotus ostreatus cultivated on spent beer grain. Bioresour. Technol. 2001, 78, 293–300. [Google Scholar] [CrossRef]
- Bhattacharjya, D.K.; Paul, R.K.; Miah, M.N.; Ahmed, K.U. Comparative Study on Nutritional Composition of Oyster Mushroom (Pleurotus ostreatus Fr.) Cultivated on Different Sawdust Substrates. Biores. Commun. 2015, 1, 93–98. [Google Scholar]
- Garuba, T.; Abdukkareem, K.A.; Ibrahim, I.A.; Oyebamiji, O.I.; Shoyooye, O.A.; Ajibade, T.D. Influence of substrates on the nutritional quality of Pleurotus pulmonarius and Pleurotus ostreatus. Ceylon J. Sci. 2017, 46, 67. [Google Scholar] [CrossRef] [Green Version]
- Koutrotsios, G.; Danezis, G.P.; Georgiou, C.A.; Zervakis, G.I. Rare earth elements concentration in mushroom cultivation substrates affects the production process and fruit-bodies content of Pleurotus ostreatus and Cyclocybe cylindracea. J. Sci. Food Agric. 2018, 98, 5418–5427. [Google Scholar] [CrossRef] [PubMed]
- Masevhe, M.R.; Soundy, P.; Taylor, N.J. Alternative substrates for cultivating oyster mushrooms (Pleurotus ostreatus). S. Afr. J. Plant Soil. 2015, 33, 97–103. [Google Scholar] [CrossRef]
- Daba, A.S.; El Nakieb, F.; Hafez, E.E. The effect of Recycling Different Wastes (as a substrate) on Mushroom (Pleurotus ostreatus) Fruit Bodies, Morphologically, Genetically and its Metabolites. Biosci. Res. 2018, 15, 2286–2294. [Google Scholar]
- Girmay, Z.; Gorems, W.; Birhanu, G.; Zewdie, S. Growth and yield performance of Pleurotus ostreatus (Jacq. Fr.) Kumm (oyster mushroom) on different substrates. AMB Express 2016, 6, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Tesfay, T.; Godifey, T.; Mesfin, R.; Kalayu, G. Evaluation of waste paper for cultivation of oyster mushroom (Pleurotus ostreatus) with some added Supplementary Materials. AMB Express 2020, 10, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Jo, W.-S.; Kim, J.S.; Cho, D.H.; Park, S.D.; Jung, H.Y. Fruitbody Development of Pleurotus ostreatusvia Bottle Cultivation Using Recycled Substrate. Mycobiology 2008, 36, 157. [Google Scholar] [CrossRef] [Green Version]
- Pardo-Giménez, A.; Picornell Buendía, M.R.; de Juan Valero, J.A.; Pardo-González, J.E.; Cunha Zied, D. Cultivation of Pleurotus ostreatus using supplemented spent oyster mushroom substrate. Acta Hortic. 2012, 933, 267–272. [Google Scholar] [CrossRef]
- Wang, S.; Xu, F.; Li, Z.; Zhao, S.; Song, S.; Rong, C.; Geng, X.; Liu, Y. The spent mushroom substrates of Hypsizigus marmoreus can be an effective component for growing the oyster mushroom Pleurotus ostreatus. Sci. Hortic. 2015, 186, 217–222. [Google Scholar] [CrossRef]
- Picornell, M.R.; Pardo, A.; De Juan-Valerot, J.A. Reuse of degraded Pleurotus ostreatus substrate through supplementation with wheat bran and Calprozime® quantitative parameters. Agron. Colomb. 2015, 33, 261–270. [Google Scholar] [CrossRef]
- Picornell-Buendía, M.R.; Pardo-Gimenéz, A.; de Juan-Valero, J.A. Agronomic assessment of spent mushroom substrates for mushroom cultivation. Biotechnol. Agron. Soc. Environ. 2016, 20, 363–374. [Google Scholar]
- Economou, C.N.; Philippoussis, A.N.; Diamantopoulou, P.A. Spent mushroom substrate for a second cultivation cycle of Pleurotus mushrooms and dephenolization of agro-industrial wastewaters. FEMS Microbiol. Lett. 2020, 367, fnaa060. [Google Scholar] [CrossRef]
- Gąsecka, M.; Mleczek, M.; Siwulski, M.; Niedzielski, P. Phenolic composition and antioxidant properties of Pleurotus ostreatus and Pleurotus eryngii enriched with selenium and zinc. Eur. Food Res. Technol. 2015, 242, 723–732. [Google Scholar] [CrossRef] [Green Version]
- Stamets, P. Growing Gourmet and Medicinal Mushrooms, 3rd ed.; Ten Speed Press: Berkley, CA, USA, 2000. [Google Scholar]
- Mleczek, M.; Gąsecka, M.; Budka, A.; Niedzielski, P.; Siwulski, M.; Kalač, P.; Mleczek, P.; Rzymski, P. Changes in mineral composition of six strains of Pleurotus after substrate modifications with different share of nitrogen forms. Eur. Food Res. Technol. 2021, 247, 245–257. [Google Scholar] [CrossRef]
- Yang, W.; Guo, F.; Wan, Z. Yield and size of oyster mushroom grown on rice/wheat straw basal substrate supplemented with cotton seed hull. Saudi J. Biol. Sci. 2013, 20, 333–338. [Google Scholar] [CrossRef] [Green Version]
- Chang, S.T.; Miles, P.G. Edible Mushrooms and Their Cultivation; CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Siqueira, O.A.P.A.; Zanon, A.R.; Martins, O.G.; De Andrade, M.C.N. New substrates for the cultivation of Pleurotus ostreatus using exhausted compost. Afr. J. Agric. Res. 2016, 11, 2295–2301. [Google Scholar] [CrossRef] [Green Version]
- Jafarpour, M. High protein complementation with high fiber substrates for oyster mushroom cultures. Afr. J. Biotechnol. 2012, 11, 3284–3289. [Google Scholar] [CrossRef]
- Bonatti, M.; Karnopp, P.; Soares, H.; Furlan, S. Evaluation of Pleurotus ostreatus and Pleurotus sajor-caju nutritional characteristics when cultivated in different lignocellulosic wastes. Food Chem. 2004, 88, 425–428. [Google Scholar] [CrossRef]
- Jin, Z.; Li, Y.; Ren, J.; Qin, N. Yield, Nutritional Content, and Antioxidant Activity of Pleurotus ostreatuson Corncobs Supplemented with Herb Residues. Mycobiology 2018, 46, 24–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meera, P.; Kumaran, S.; Vasudeo, G. Making mushroom production process a zero waste enterprise. Int. J. Environ. Sci. 2014, 5, 236–242. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).