Implementation of the Circular Economy Concept in Greenhouse Hydroponics for Ultimate Use of Water and Nutrients
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Set Up
2.2. Measurements
2.3. Calculations
2.4. Statistical Analysis
3. Results
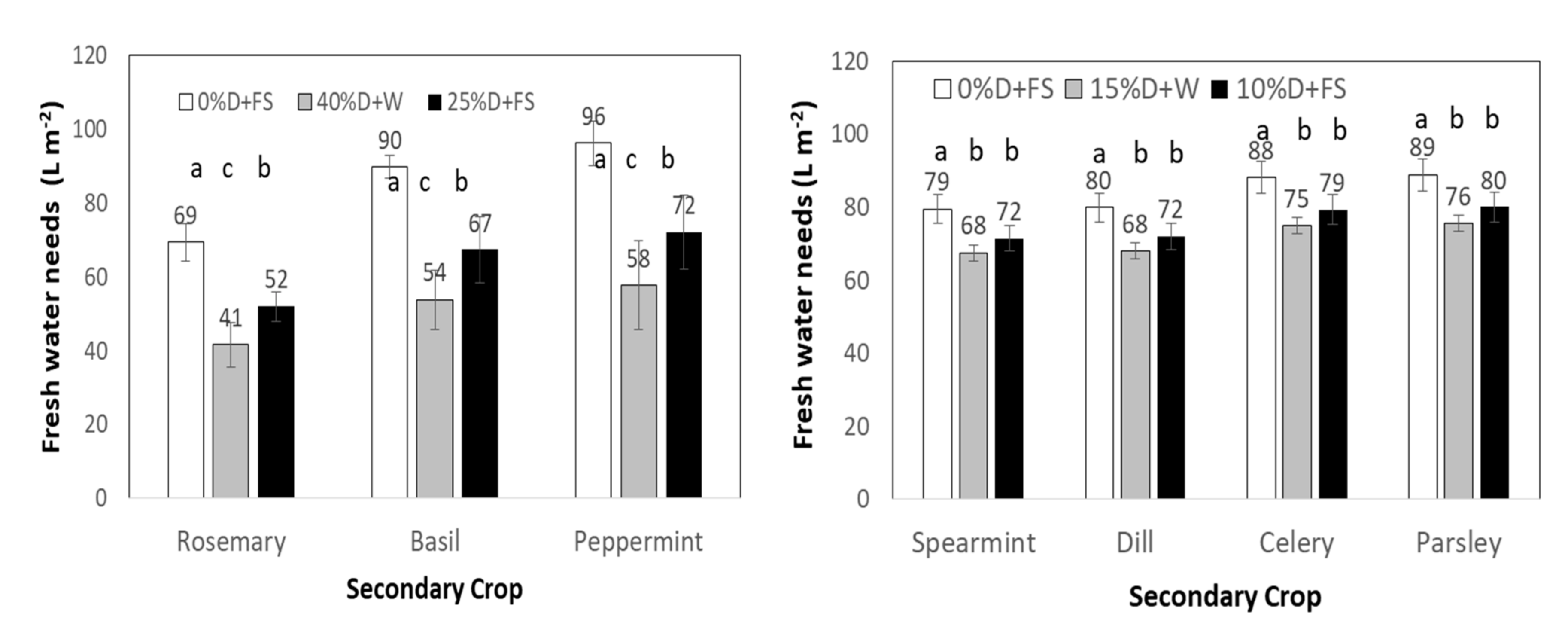
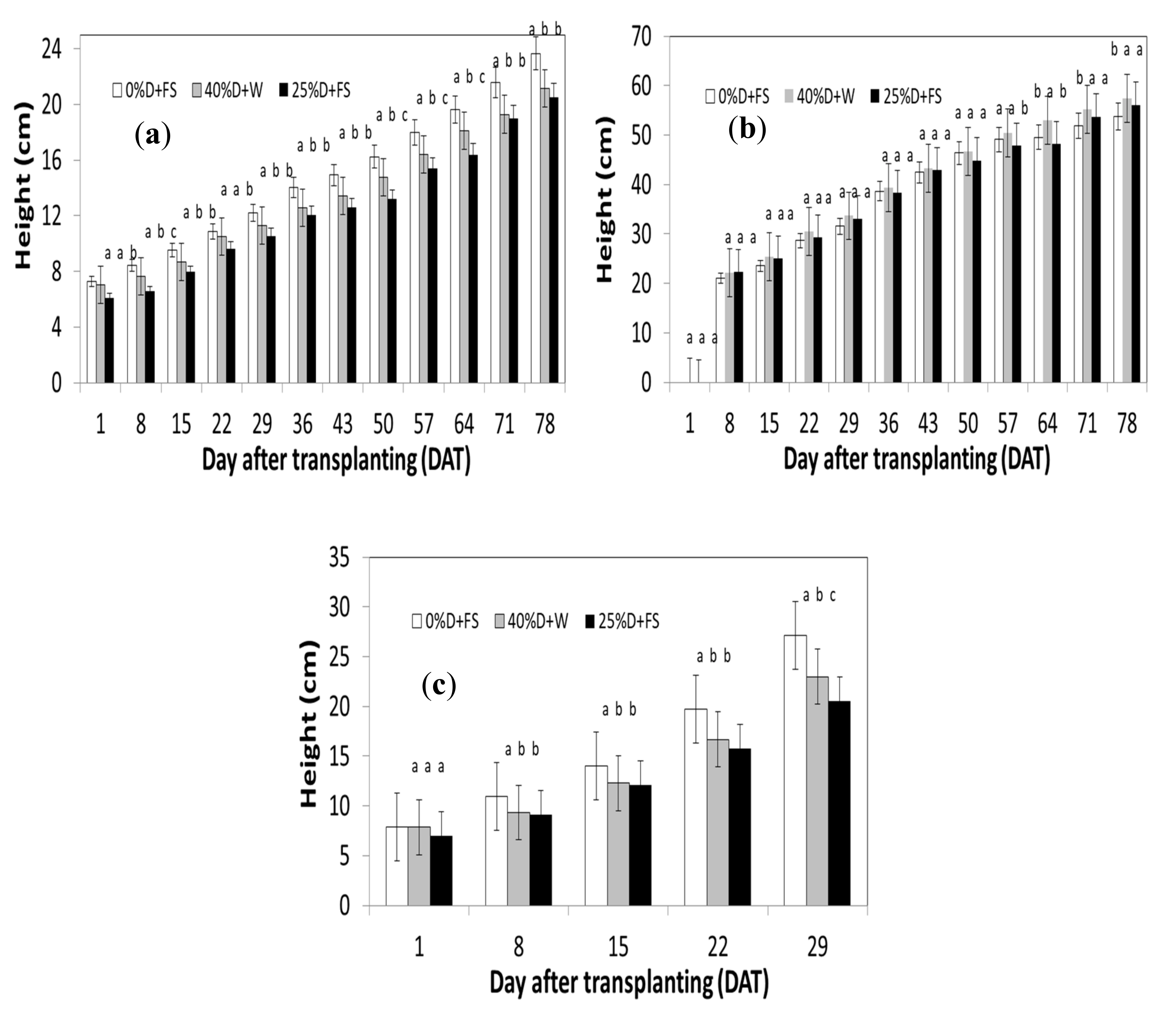
3.1. Water Consumption
3.2. Nutrient Solution Analysis
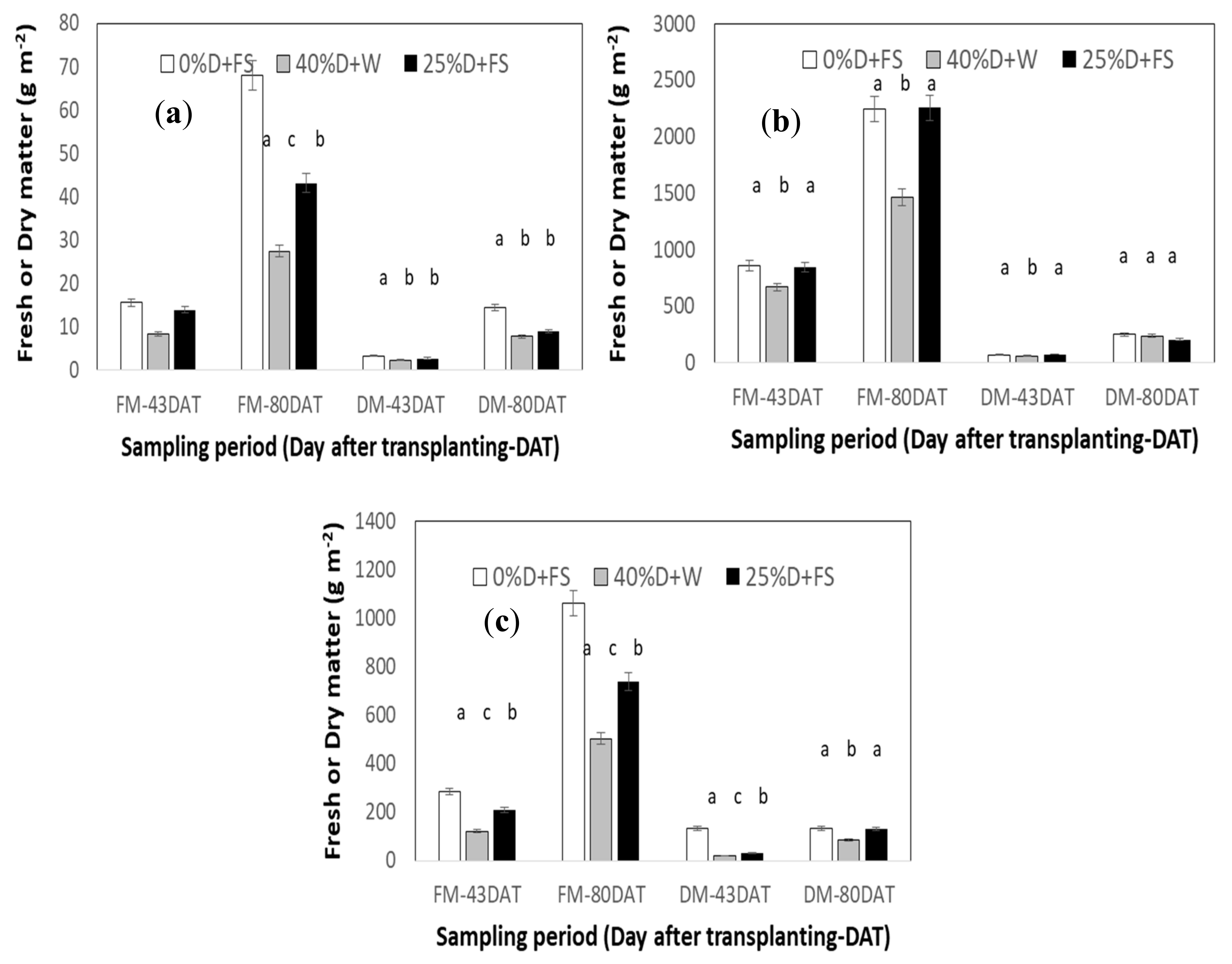
3.3. Yield
3.3.1. Secondary Crop Yield: 1st Period
3.3.2. Secondary Crops Yield: 2nd Period
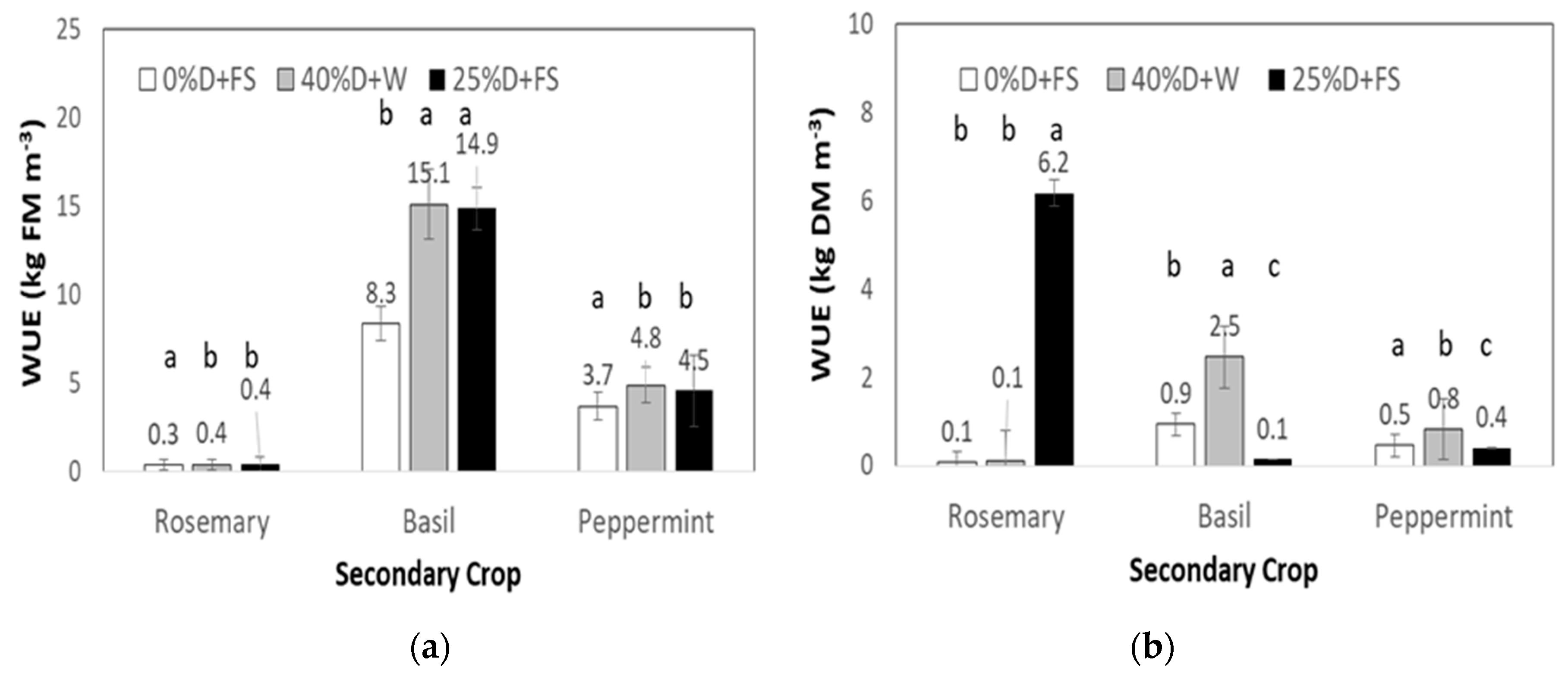
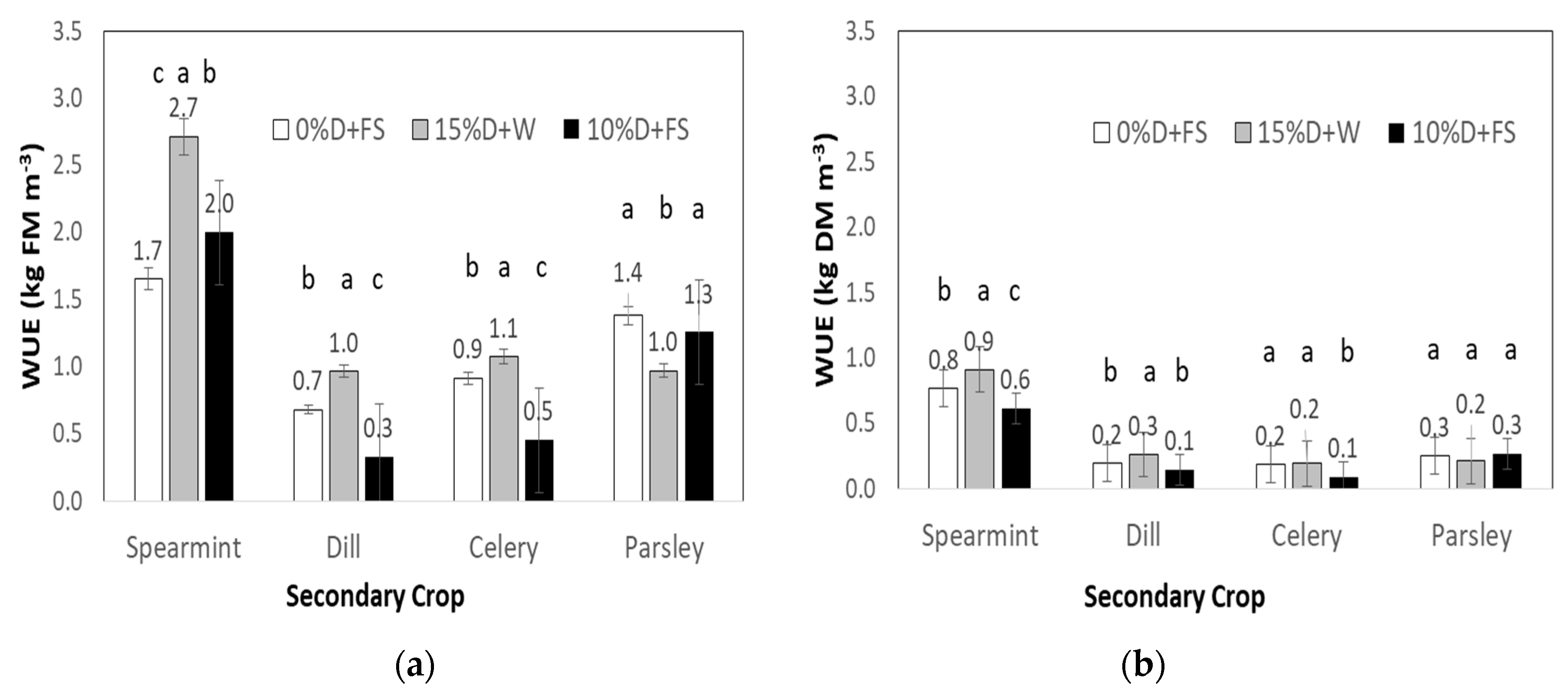
3.4. Water Use Efficiency
4. Discussion
4.1. Effect of Drainage Management on Secondary Crops Yield
4.2. Water Use Efficiency
4.3. Nutrient Use Efficiency
4.4. Implications to Circular Economy
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). The Future of Food and Agriculture-Alternative Pathways to 2050; FAOP: Rome, Italy, 2018; p. 224. [Google Scholar]
- Toop, T.A.; Ward, S.; Oldfield, T.; Hull, M.; Kirby, M.E.; Theodorou, M.K. AgroCycle-Developing a circular economy in agriculture. Energy Proc. 2017, 123, 76–80. [Google Scholar] [CrossRef]
- Duque-Acevedo, M.; Belmonte-Ureña, L.J.; Cortés-García, F.J.; Camacho-Ferre, F. Agricultural waste: Review of the evolution, approaches and perspectives on alternative uses. Glob. Ecol. Conserv. 2020, 22, e00902. [Google Scholar] [CrossRef]
- Verkerke, W. Circular Economy and Greenhouse Horticulture; Wageningen University & Research: Wageningen, The Netherlands, 2020. [Google Scholar]
- Katsoulas, N. EIP-AGRI Focus Group Circular Horticulture: Starting Paper. EIP-AGRI, 2017; p. 18. Available online: https://ec.europa.eu/eip/agriculture/en/publications/eip-agri-focus-group-circular-horticulture (accessed on 5 October 2020).
- Schulker, B.A.; Jackson, B.E.; Fonteno, W.C.; Heitman, J.L.; Albano, J.P. Comparison of water capture efficiency through two irrigation techniques of three common greenhouse soilless substrate components. Agronomy 2020, 10, 1389. [Google Scholar] [CrossRef]
- Varlagas, H.; Savvas, D.; Mouzakis, G.; Liotsos, C.; Karapanos, I.; Sigrimis, N. Modelling uptake of Na + and Cl-by tomato in closed-cycle cultivation systems as influenced by irrigation water salinity. Agric. Water Manag. 2010, 97, 1242–1250. [Google Scholar] [CrossRef]
- Kumar, R.R.; Cho, J.Y. Reuse of hydroponic waste solution. Environ. Sci. Pollut. Res. 2014, 21, 9569–9577. [Google Scholar] [CrossRef]
- García-Caparrós, P.; Chica, R.M.; Almansa, E.M.; Rull, A.; Rivas, L.A.; García-Buendía, A.; Barbero, F.J.; Lao, M.T. Comparisons of different lighting systems for horticultural seedling production aimed at energy saving. Sustainability 2018, 10, 3351. [Google Scholar] [CrossRef]
- Katsoulas, N.; Demmelbauer-Benitez, C.M.; Elvanidi, A.; Gourzoulidou, E.; Max, J.F.J. Reuse of cucumber drainage nutrient solution in secondary crops in greenhouses: Initial results. Acta Hortic. 2020. [Google Scholar]
- Salas, M.D.C.; Montero, J.L.; Diaz, J.G.; Berti, F.; Quintero, M.F.; Guzmán, M.; Orsini, F. Defining optimal strength of the nutrient solution for soilless cultivation of saffron in the Mediterranean. Agronomy 2020, 10, 1311. [Google Scholar] [CrossRef]
- Shannon, M.; Grieve, C. Tolerance of vegetable crops to salinity. Sci. Hortic. 1998, 78, 5–38. [Google Scholar] [CrossRef]
- Kjeldahl, J. A new method for the determination of nitrogen in organic matter. Z. Anal. Chem. 1883, 22, 366. [Google Scholar] [CrossRef]
- Katsoulas, N.; Savvas, D.; Kitta, E.; Bartzanas, T.; Kittas, C. Extension and evaluation of a model for automatic drainage solution management in tomato crops grown in semi-closed hydroponic systems. Comput. Electron. Agric. 2015, 113, 61–71. [Google Scholar] [CrossRef]
- IBM SPSS Statistics 21 Guide; IBM Corporation: Armonk, NY, USA, 2012.
- Trejo-Téllez, L.I.; Gómez-Merino, F.C. Nutrient solutions for hydroponic systems. In Hydroponics-A Standard Methodology for Plant Biological Researches; Asao, T., Ed.; InTech: Rijeka, Croatia, 2012; p. 12. [Google Scholar]
- Hejazi Mehrizi, M.; Shariatmadari, H.; Khoshgoftarmanesh, A.H.; Dehghani, F. Copper effects on growth, lipid peroxidation, and total phenolic content of rosemary leaves under salinity stress. J. Agric. Sci. Technol. 2012, 14, 205–212. [Google Scholar]
- Gharib, F.; Ghazi, S.; Aly, H.; El-Araby, M.; Moustafa, S. Effect of soil type and water content on rosemary growth and essential oil yield. Int. J. Sci. Eng. Res. 2016, 7, 183–189. [Google Scholar]
- Ushakova, S.; Kovaleva, N.; Gribovskaya, I.; Dolgushev, V.; Tikhomirova, N. Effect of NaCl concentration on productivity and mineral composition of Salicornia europaea as a potential crop for utilization NaCl in LSS. Adv. Space Res. 2005, 36, 1349–1353. [Google Scholar] [CrossRef]
- Flanigan, P.M.; Niemeyer, E.D. Effect of cultivar on phenolic levels, anthocyanin composition, and antioxidant properties in purple basil (Ocimum basilicum L.). Food Chem. 2014, 164, 518–526. [Google Scholar] [CrossRef]
- Faliagka, S.; Schmidt, L.; Lagner, F.; Elvanidi, A.; Papadopoulou, A.; Katsoulas, N.; Max, F.J. Effects of different glass covering materials on basil and mint quality. DGG-Proc. 2018, 8, 1–5. [Google Scholar] [CrossRef]
- Lei, Y.; Korpelainen, H.; Li, C. Physiological and biochemical responses to high Mn concentrations in two contrasting Populus cathayana populations. Chemosphere 2007, 68, 686–694. [Google Scholar] [CrossRef]
- Mahmoodabad, H.A.; Parchin, R.A.; Hokmalipoor, S.; Shaban, M. Changes in yield and growth of green Mint (Mentha spicata L.) Under foliar application of urea and soil application of vermicompost. Int. J. Adv. Biol. Biomed. Res. 2014, 2, 2072–2076. [Google Scholar]
- Saha, S.; Monroe, A.; Day, M.R. Growth, yield, plant quality and nutrition of basil (Ocimum basilicum L.) under soilless agricultural systems. Ann. Agric. Sci. 2016, 61, 181–186. [Google Scholar] [CrossRef]
- Muñoz, P.; Flores, J.; Antón, A.; Montero, J. Combination of greenhouse and open-field crop fertigation can increase sustainability of horticultural crops in the Mediterranean region. Acta Hortic. 2017, 627–634. [Google Scholar] [CrossRef]
- Ritzén, S.; Sandström, G.Ö. Barriers to the circular economy-Integration of perspectives and domains. Proc. CIRP 2017, 64, 7–12. [Google Scholar] [CrossRef]
| Sum L p−1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Period | Solution | Level | Crop | DAT56 | DAT80 | ||||
| 1st Period | 0%D+FS | 40%D+W | 25%D+FS | 0%D+FS | 40%D+W | 25%D+FS | |||
| Irrigation | Pr. | Cucumber | 55.58 ± 3.09 | 82.38 ± 4.07 | |||||
| Absorption | Pr. | Cucumber | 31.29 ± 1.84 | 51.23 ± 2.02 | |||||
| Irrigation | Sec. | Herbs | 43.01 ± 1.21 | 32.04 ± 1.25 | 46.95 ± 0.78 | 59.79 ± 1.12 | 49.47 ± 1.22 | 61.64 ± 0.80 | |
| Absorption | Sec. | Rosemary | 23.47 ± 0.72 | 14.13 ± 0.45 | 24.18 ± 0.79 | 32.27 ± 0.68 | 24.81 ± 0.43 | 34.76 ± 0.78 | |
| Absorption | Sec. | Basil | 19.69 ± 0.85 | 18.66 ± 0.50 | 30.14 ± 0.79 | 29.93 ± 0.74 | 28.49 ± 0.32 | 41.02 ± 0.76 | |
| Absorption | Sec. | Peppermint | 20.21 ± 0.99 | 15.63 ± 0.41 | 25.23 ± 0.92 | 32.06 ± 0.84 | 27.32 ± 0.43 | 36.79 ± 0.88 | |
| 2nd Period | 0%D+FS | 15%D+W | 10%D+FS | 0%D+FS | 15%D+W | 10%D+FS | |||
| Irrigation | Pr. | Tomato | 48.95 ± 0.12 | ||||||
| Absorption | Pr. | Tomato | 35.57 ± 0.63 | ||||||
| Irrigation | Sec. | Herbs | 45.76 ± 1.05 | 38.21 ± 1.12 | 45.53 ± 1.07 | ||||
| Absorption | Sec. | Spearmint | 82.33 ± 0.94 | 79.23 ± 0.96 | 86.60 ± 0.93 | ||||
| Absorption | Sec. | Dill | 72.31 ± 1.05 | 78.82 ± 1.06 | 80.13 ± 1.03 | ||||
| Absorption | Sec. | Celery | 79.08 ± 0.22 | 88.32 ± 0.28 | 90.38 ± 0.26 | ||||
| Absorption | Sec. | Parsley | 82.15 ± 0.48 | 87.92 ± 0.72 | 78.95 ± 1.21 | ||||
| Irrigation | Absorption | Drainage | ||||||
| Primary crop | Cucumber | Cucumber | ||||||
| Primary crop | NO3 (mmol L−1) | 9.85 ± 2.91 | 8.24 ± 2.29 | 12.50 ± 2.25 | ||||
| K (mmol L−1) | 3.10 ± 2.09 | 2.67 ± 0.46 | 3.83 ± 1.88 | |||||
| Ca (mmol L−1) | 6.08 ± 0.90 | 5.52 ± 0.75 | 6.98 ± 1.36 | |||||
| Na (mmol L−1) | 1.79 ± 0.13 | 1.77 ± 0.13 | 1.83 ± 0.43 | |||||
| Mg (mmol L−1) | 2.14 ± 0.33 | 2.01 ± 0.27 | 2.34 ± 0.85 | |||||
| Mn (μmol L−1) | 0.36 ± 0.001 | 0.36 ± 0.001 | 0.36 ± 0.001 | |||||
| Irrigation | Absorption | Drainage | ||||||
| Secondary crop | Rosemary | Basil | Peppermint | Rosemary | Basil | Peppermint | ||
| 0%D+FS | NO3 (mmol L−1) | 9.85 ± 2.91 | 9.14 ± 1.98 | 6.88 ± 2.25 | 5.80 ± 1.80 | 10.68 ± 2.81 | 12.82 ± 2.20 | 14.53 ± 2.54 |
| K (mmol L−1) | 3.10 ± 2.09 | 3.03 ± 0.74 | 3.02 ± 0.82 | 2.57 ± 0.46 | 3.20 ± 0.63 | 3.18 ± 0.36 | 3.72 ± 0.88 | |
| Ca (mmol L−1) | 6.08 ± 0.90 | 3.43 ± 0.62 | 4.66 ± 0.65 | 4.27 ± 0.77 | 9.18 ± 0.54 | 7.49 ± 0.96 | 8.16 ± 1.13 | |
| Na (mmol L−1) | 1.81 ± 0.22 | 1.66 ± 0.36 | 1.48 ± 0.57 | 1.44 ± 0.84 | 1.98 ± 0.90 | 2.13 ± 0.54 | 2.23 ± 0.42 | |
| Mg (mmol L−1) | 2.40 ± 0.42 | 1.82 ± 0.29 | 2.29 ± 0.47 | 2.01 ± 0.65 | 3.07 ± 0.30 | 2.50 ± 0.24 | 2.85 ± 0.76 | |
| Mn (μmol L−1) | 0.36 ± 0.001 | 0.36 ± 0.001 | 0.36 ± 0.001 | 0.34 ± 0.001 | 0.37 ± 0.001 | 0.36 ± 0.001 | 0.40 ± 0.001 | |
| 40%D+W | NO3 (mmol L−1) | 5.06 ± 2.29 | 2.82 ± 2.01 | 3.33 ± 2.75 | 3.47 ± 2.33 | 6.46 ± 2.76 | 7.18 ± 2.91 | 6.27 ± 2.53 |
| K (mmol L−1) | 1.53 ± 0.56 | 0.99 ± 0.81 | 1.32 ± 0.69 | 1.20 ± 0.71 | 1.77 ± 0.40 | 1.72 ± 0.77 | 1.69 ± 0.58 | |
| Ca (mmol L−1) | 4.32 ± 0.85 | 3.07 ± 0.95 | 3.67 ± 1.16 | 3.66 ± 1.43 | 4.66 ± 1.21 | 4.95 ± 1.81 | 4.35 ± 1.25 | |
| Na (mmol L−1) | 2.05 ± 0.28 | 2.01 ± 0.19 | 1.59 ± 0.47 | 1.72 ± 0.27 | 1.52 ± 0.02 | 2.58 ± 0.57 | 2.09 ± 0.26 | |
| Mg (mmol L−1) | 1.89 ± 0.53 | 1.87 ± 0.48 | 1.47 ± 0.31 | 1.36 ± 0.60 | 1.82 ± 0.13 | 2.37 ± 0.60 | 2.26 ± 0.25 | |
| Mn (μmol L−1) | 0.0093 ± 0.001 | 0.0010 ± 0.001 | 0.0012 ± 0.001 | 0.0011 ± 0.001 | 0.0364 ± 0.001 | 0.0415 ± 0.001 | 0.0478 ± 0.001 | |
| 25%D+FS | NO3 (mmol L−1) | 10.16 ± 2.19 | 10.05 ± 2.16 | 10.08 ± 2.06 | 10.16 ± 2.19 | 11.24 ± 1.61 | 13.59 ± 2.76 | 12.06 ± 2.71 |
| K (mmol L−1) | 3.18 ± 1.57 | 3.16 ± 0.95 | 3.11 ± 0.55 | 2.94 ± 0.61 | 3.14 ± 0.53 | 3.10 ± 0.46 | 2.80 ± 0.81 | |
| Ca (mmol L−1) | 6.11 ± 0.87 | 6.10 ± 1.06 | 6.10 ± 1.32 | 6.06 ± 1.46 | 6.64 ± 0.96 | 8.25 ± 1.15 | 6.81 ± 1.84 | |
| Na (mmol L−1) | 1.43 ± 0.36 | 1.20 ± 0.08 | 1.42 ± 0.27 | 1.20 ± 0.15 | 1.84 ± 0.42 | 2.13 ± 0.12 | 2.00 ± 0.67 | |
| Mg (mmol L−1) | 2.32 ± 0.68 | 2.31 ± 0.73 | 2.26 ± 0.91 | 2.30 ± 0.85 | 2.18 ± 0.29 | 2.92 ± 0.80 | 2.26 ± 0.93 | |
| Mn (μmol L−1) | 0.35 ± 0.001 | 0.35 ± 0.001 | 0.35 ± 0.001 | 0.35 ± 0.001 | 0.47 ± 0.001 | 0.38 ± 0.001 | 0.42 ± 0.001 |
| Crop | Number of Stems-DAT 54 | ||
|---|---|---|---|
| 0%D+FS | 5%D+W | 10%D+FS | |
| Spearmint | 34 ± 1.83 a z | 32 ± 2.55 a | 35 ± 2.25 a |
| Dill | 56 ± 9.58 a | 50 ± 8.48 b | 55 ± 6.41 a |
| Celery | 205 ± 36.18 a | 180 ± 28.79 b | 166 ± 42.93 c |
| Parsley | 29 ± 13.33 b | 34 ± 13.48 a | 30 ± 13.18 ab |
| Crop | 0% D+FS | 15%D+W | 10%D+FS |
|---|---|---|---|
| DAT28 | |||
| Spearmint | 45.41 ± 6.02 a z | 43.86 ± 5.83 a | 43.82 ± 7.56 a |
| Dill | 12.28 ± 9.15 a | 10.07 ± 7.33 b | 9.99 ± 6.83 b |
| Celery | 34.73 ± 10.08 c | 43.34 ± 9.47 a | 38.9 ± 9.96 b |
| Parsley | 49.96 ± 16.23 a | 34.73 ± 15.13 b | 33.92 ± 12.76 b |
| DAT56 | |||
| Spearmint | 45.93 ± 3.21 b | 53.02 ± 7.50 a | 47.50 ± 7.38 b |
| Dill | 9.82 ± 4.46 a | 7.43 ± 3.93 b | 9.57 ± 6.94 a |
| Celery | 50.50 ± 2.49 a | 33.80 ± 9.52 b | 31.59 ± 10.74 b |
| Parsley | 48.52 ± 5.68 a | 41.84 ± 17.46 b | 35.77 ± 14.59 c |
| Crop | FM (g m−2) | DM (g m−2) | ||||
|---|---|---|---|---|---|---|
| DAT28 | ||||||
| 0% D+FS | 15%D+W | 10%D+FS | 0%D+FS | 15%D+W | 10%D+FS | |
| Spearmint | 172.33 ± 4.91 a z | 159.00 ± 5.52 b | 175.83 ± 5.55 a | 64.00 ± 0.09 a | 69.83 ± 0.12 a | 70.67 ± 0.16 a |
| Dill | 63.04 ± 7.42 a | 55.36 ± 5.38 c | 59.99 ± 3.7 b | 5.85 ± 0.09 a | 3.76 ± 0.20 a | 4.40 ± 0.09 a |
| Celery | 42.83 ± 6.76 b | 50.50 ± 4.28 a | 40.10 ± 4.59 b | 5.73 ± 0.07 b | 7.71 ± 0.14 a | 5.53 ± 0.06 b |
| Parsley | 17.15 ± 2.33 b | 27.78 ± 2.83 a | 18.57 ± 2.53 b | 2.82 ± 0.33 b | 3.30 ± 0.40 a | 2.90 ± 0.48 b |
| DAT56 | ||||||
| 0% D+FS | 15%D+W | 10%D+FS | 0%D+FS | 15%D+W | 10%D+FS | |
| Spearmint | 463.17 ± 10.25 b | 549.83 ± 3.97 a | 428.38 ± 8.11 c | 182.67 ± 0.61 a | 184.00 ± 0.11 a | 131.67 ± 0.17 b |
| Dill | 162.65 ± 3.81 b | 196.58 ± 4.74 a | 70.98 ± 2.81 c | 46.67 ± 0.16 b | 53.32 ± 0.21 a | 30.62 ± 0.33 c |
| Celery | 230.00 ± 0.88 a | 239.29 ± 4.54 a | 106.95 ± 2.36 b | 48.26 ± 0.03 a | 42.97 ± 0.19 a | 21.47 ± 0.26 b |
| Parsley | 123.03 ± 3.00 a | 73.50 ± 2.16 c | 113.58 ± 2.95 b | 22.47 ± 0.32 a | 16.11 ± 0.22 b | 21.44 ± 0.19 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elvanidi, A.; Benitez Reascos, C.M.; Gourzoulidou, E.; Kunze, A.; Max, J.F.J.; Katsoulas, N. Implementation of the Circular Economy Concept in Greenhouse Hydroponics for Ultimate Use of Water and Nutrients. Horticulturae 2020, 6, 83. https://doi.org/10.3390/horticulturae6040083
Elvanidi A, Benitez Reascos CM, Gourzoulidou E, Kunze A, Max JFJ, Katsoulas N. Implementation of the Circular Economy Concept in Greenhouse Hydroponics for Ultimate Use of Water and Nutrients. Horticulturae. 2020; 6(4):83. https://doi.org/10.3390/horticulturae6040083
Chicago/Turabian StyleElvanidi, Angeliki, Cinthya Marilu Benitez Reascos, Elissavet Gourzoulidou, Alexander Kunze, Johannes F. J. Max, and Nikolaos Katsoulas. 2020. "Implementation of the Circular Economy Concept in Greenhouse Hydroponics for Ultimate Use of Water and Nutrients" Horticulturae 6, no. 4: 83. https://doi.org/10.3390/horticulturae6040083
APA StyleElvanidi, A., Benitez Reascos, C. M., Gourzoulidou, E., Kunze, A., Max, J. F. J., & Katsoulas, N. (2020). Implementation of the Circular Economy Concept in Greenhouse Hydroponics for Ultimate Use of Water and Nutrients. Horticulturae, 6(4), 83. https://doi.org/10.3390/horticulturae6040083






