Trichoderma spp. and Mulching Films Differentially Boost Qualitative and Quantitative Aspects of Greenhouse Lettuce under Diverse N Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setting, Design, and Plant Material
2.2. Plant Management, Nitrogen Fertilization, and Trichoderma Application
2.3. Plant Growth Parameters, Marketable Yield, SPAD index, Leaf Colorimetry, and Nitrate Determination
2.4. Chlorophyllous Pigments, Carotenoids and Bioactive Molecules Analysis
2.5. Antioxidant Capacity Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Effect of N Fertilization Dose, Trichoderma Application, and Mulching on Marketable Yield and Growth Parameters
3.2. Effect of N Fertilization Dose, Trichoderma Application, and Mulching Films on Leaf Colorimetry and SPAD Index
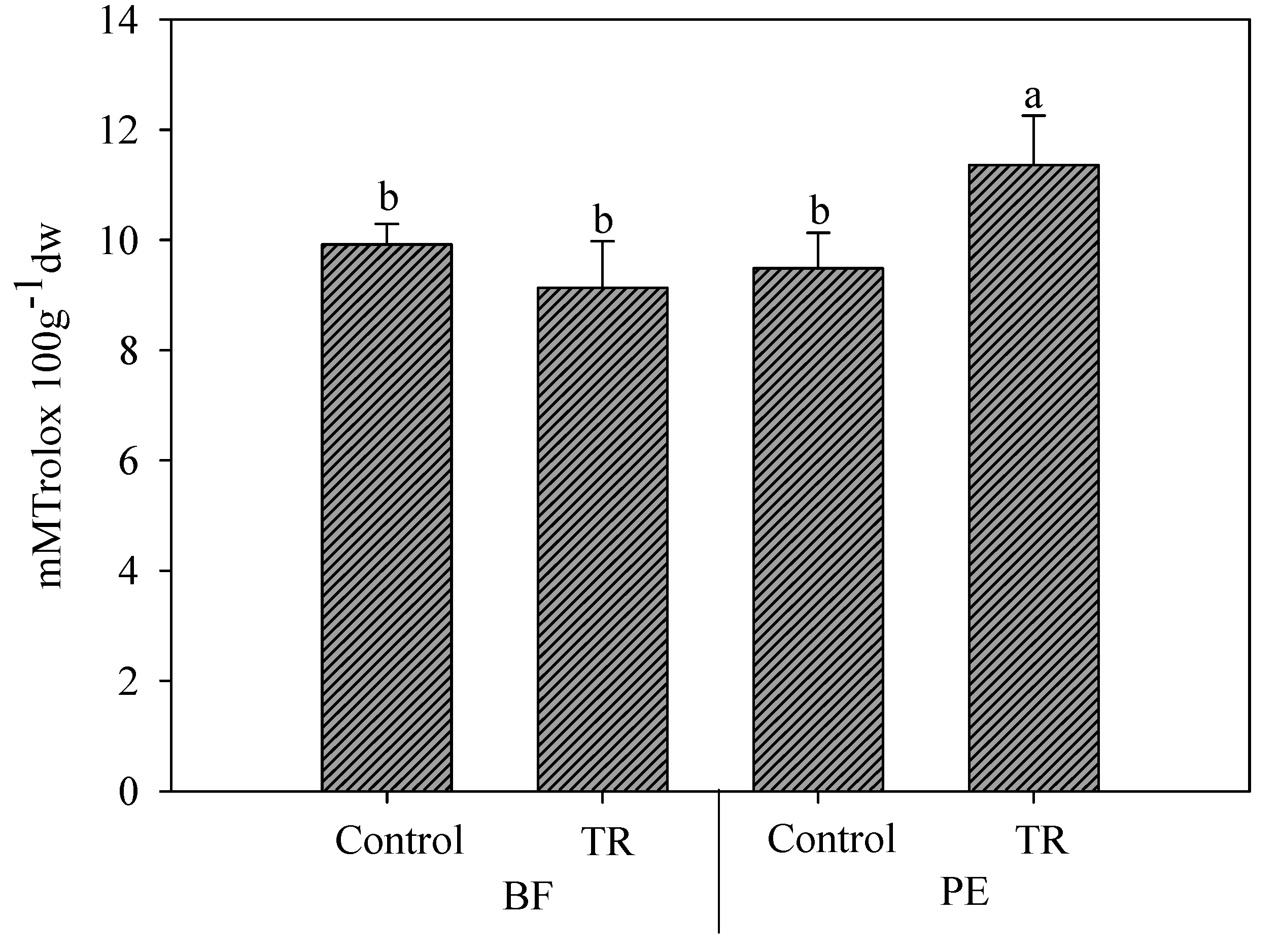
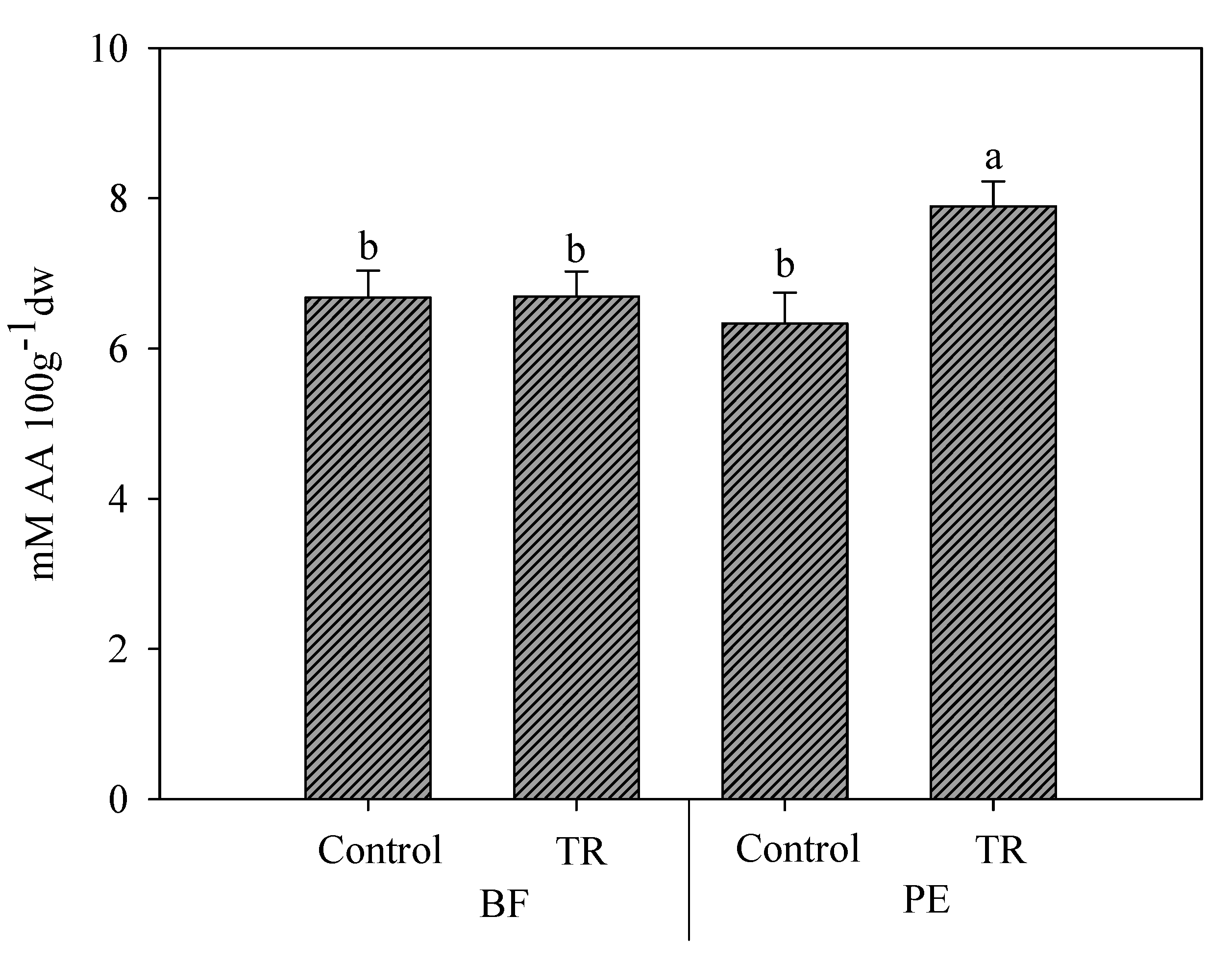
3.3. Effect of N Fertilization Dose, Trichoderma Application, and Mulching Films on Antioxidant Capacity and Bioactive Content
3.4. Effect of N Fertilization Dose, Trichoderma Application, and Mulching Films on Biochemical Parameters and Nitrate Content
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SPAD | Soil Plant Analysis Development |
| LAA | Lipophilic Antioxidant Activity |
| HAA | Hydrophilic Antioxidant Activity |
| TAA | Total Ascorbic Acid |
References
- FAO. The Future of Food and Agriculture–Alternative Pathways to 2050; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018. [Google Scholar]
- Di Mola, I.; Cozzolino, E.; Ottaiano, L.; Duri, L.G.; Riccardi, R.; Spigno, P.; Mori, M. The effect of novel biodegradable films on agronomic performance of zucchini squash grown under open-field and greenhouse conditions. Aust. J. Crop. Sci. 2019, 13, 1810. [Google Scholar]
- Kasirajan, S.; Ngouajio, M. Polyethylene and biodegradable mulches for agricultural applications: A review. Agron. Sustain. Dev. 2012, 32, 501–529. [Google Scholar] [CrossRef]
- Kyrikou, I.; Briassoulis, D. Biodegradation of agricultural plastic films: A critical review. J. Polym. Env. 2007, 15, 125–150. [Google Scholar] [CrossRef]
- Mormile, P.; Rippa, M.; Petti, L.; Immirzi, M.; Malinconico, M.B.; Lahoz, E.; Morra, L. Improvement of soil solarization through a hybrid system simulating a solar hot water panel. J. Adv. Agric. Technol. 2016, 3, 226–230. [Google Scholar] [CrossRef]
- Mormile, P.; Capasso, R.; Rippa, M.; Petti, L. Light filtering by innovative plastic films for mulching and soil solarization: State of the art. Acta Horticulturae 2013, 1015, 113–121. [Google Scholar] [CrossRef]
- Schettini, E.; De Salvador, F.R.; Scarascia Mugnozza, G.; Vox, G. Radiometric properties of photoselective and photoluminescent greenhouse plastic films and their effects on peach and cherry tree growth. J. Hortic. Sci. Biothec. 2011, 86, 79–83. [Google Scholar] [CrossRef]
- Rippa, M.; Yan, C.; Petti, L.; Mormile, P. A simple method for saving water in agriculture based on the use of photo-selective mulch film. Available online: https://ijaer.in/2018files/ijaer_04__03.pdf (accessed on 3 September 2020).
- Di Mola, I.; Ottaiano, L.; Cozzolino, E.; Senatore, M.; Giordano, M.; El-Nakhel, C.; Mori, M. Plant-based biostimulants influence the agronomical, physiological, and qualitative responses of baby rocket leaves under diverse nitrogen conditions. Plants 2019, 8, 522. [Google Scholar] [CrossRef]
- Cavaiuolo, M.; Ferrante, A. Nitrates and glucosinolates as strong determinants of the nutritional quality in rocket leafy salads. Nutrients 2014, 14, 1519–1538. [Google Scholar] [CrossRef]
- Alberici, A.; Quattrini, E.; Penati, M.; Martinetti, L.; Gallina, P.M.; Ferrante, A.; Schiavi, M. Effect of the reduction of nutrient solution concentration on leafy vegetables quality grown in floating system. Acta Horticulturae 2008, 801, 1167–1176. [Google Scholar] [CrossRef]
- Wang, Z.; Li, S. Effects of nitrogen and phosphorus fertilization on plant growth and nitrate accumulation in vegetables. J. Plant. Nutr. 2004, 27, 539–556. [Google Scholar] [CrossRef]
- Colla, G.; Kim, H.J.; Kyriacou, M.C.; Rouphael, Y. Nitrate in fruits and vegetables. Sci. Hortic. 2018, 237, 231–238. [Google Scholar] [CrossRef]
- Lassaletta, L.; Billen, G.; Grizzetti, B.; Anglade, J.; Garnier, J. 50 year trends in nitrogen use efficiency of world cropping systems: The relationship between yield and nitrogen input to cropland. Env. Res. Lett. 2014, 9, 105011. [Google Scholar] [CrossRef]
- Sutton, M.A.; Bleeker, A.; Howard, C.M.; Bekunda, M.; Grizzetti, B.; de Vries, W.; van Grinsven, H.J.M.; Abrol, Y.P.; Adhya, T.K.; Billen, G.; et al. Our Nutrient World: The challenge to produce more food and energy with less pollution. Cent. Ecol. Hydrol. 2013, 8, 95–108. [Google Scholar]
- Di Mola, I.; Cozzolino, E.; Ottaiano, L.; Nocerino, S.; Rouphael, Y.; Colla, G.; Mori, M. Nitrogen Use and Uptake Efficiency and Crop Performance of Baby Spinach (Spinacia oleracea L.) and Lamb’s Lettuce (Valerianella locusta L.) Grown under Variable Sub-Optimal N Regimes Combined with Plant-Based Biostimulant Application. Agronomy 2020, 10, 278. [Google Scholar] [CrossRef]
- López-Bucio, J.; Pelagio-Flores, R.; Herrera-Estrella, A. Trichoderma as biostimulant: Exploiting the multilevelproperties of a plant beneficial fungus. Sci. Hortic. 2015, 196, 109–123. [Google Scholar]
- Woo, S.; Ruocco, M.; Vinale, F.; Nigro, M.; Marra, R.; Lombardi, N.; Pascale, A.; Lanzuise, S.; Manganiello, G.; Lorito, M. Trichoderma-based products and their widespread use in agriculture. Open Mycol. J. 2014, 8. [Google Scholar] [CrossRef]
- Carillo, P.; Woo, S.L.; Comite, E.; El-Nakhel, C.; Rouphael, Y.; Fusco, G.M.; Vinale, F. Application of Trichoderma harzianum, 6-pentyl-α-pyrone and plant biopolymer formulations modulate plant metabolism and fruit quality of plum tomatoes. Plants 2020, 9, 771. [Google Scholar] [CrossRef]
- Caruso, G.; El-Nakhel, C.; Rouphael, Y.; Comite, E.; Lombardi, N.; Cuciniello, A.; Woo, S.L. Diplotaxis tenuifolia (L.) DC. Yield and Quality as Influenced by Cropping Season, Protein Hydrolysates, and Trichoderma Applications. Plants 2020, 9, 697. [Google Scholar] [CrossRef]
- Rouphael, Y.; Carillo, P.; Colla, G.; Fiorentino, N.; Sabatino, L.; El-Nakhel, C.; Cozzolino, E. Appraisal of Combined Applications of Trichoderma virens and a Biopolymer-Based Biostimulant on Lettuce Agronomical, Physiological, and Qualitative Properties under Variable N Regimes. Agronomy 2020, 10, 196. [Google Scholar] [CrossRef]
- Fiorentino, N.; Ventorino, V.; Woo, S.L.; Pepe, O.; De Rosa, A.; Gioia, L.; Romano, I.; Lombardi, N.; Napolitano, M.; Colla, G.; et al. Trichoderma-based biostimulants modulate rhizosphere microbial populations and improve N uptake efficiency, yield, and nutritional quality of leafy vegetables. Front. Plant. Sci. 2018, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Colla, G.; Rouphael, Y.; Di Mattia, E.; El-Nakhel, C.; Cardarelli, M. Co-inoculation of Glomus intraradices and Trichoderma atroviride acts as a biostimulant to promote growth, yield and nutrient uptake of vegetable crops. J. Sci. Food Agric. 2015, 95, 1706–1715. [Google Scholar] [CrossRef] [PubMed]
- Lichtenhaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Kampfenkel, K.; Montagu, M.V.; Inzè, D. Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Annu. Rev. Biochem. 1995, 225, 165–167. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Fogliano, V.; Verde, V.; Randazzo, G.; Ritieni, A. Methods for measuring antioxidant activity and its application to monitoring the antioxidant capacity of wines. J. Agric. Food Chem. 1999, 47, 1035–1040. [Google Scholar] [CrossRef]
- Pellegrini, N.; Re, R.; Yang, M.; Rice-Evans, C. Screening of dietary carotenoids and carotenoid-rich-fruit extracts for antioxidant activities applying 2,2- Azinobis (3-ethylenebenzothiazoline-6-sulfonic acid radical cation decoloration assay. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 379–384. [Google Scholar]
- Chang, Y.C.; Chang, Y.C.; Baker, R.; Kleifeld, O.; Chet, I. Increased growth of plants in the presence of the biological control agent Trichoderma harzianum. Plant. Dis. 1986, 70, 145–148. [Google Scholar] [CrossRef]
- Guerrini, S.; Yan, C.; Malinconico, M.; Mormile, P. Agronomical overview of mulch film systems. In Polymers for Agri-Food Applications; Springer: Cham, Switzerland, 2019; pp. 241–264. [Google Scholar]
- Bonanomi, G.; Chirico, G.B.; Palladino, M.; Gaglione, S.A.; Crispo, D.G.; Lazzaro, U.; Rippa, M. Combined application of photo-selective mulching films and beneficial microbes affects crop yield and irrigation water productivity in intensive farming systems. Agric. Water Manag. 2017, 184, 104–113. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; Rouphael, Y. Towards a new definition of quality for fresh fruits and vegetables. Sci. Hortic. 2018, 234, 463–469. [Google Scholar] [CrossRef]
- Khanam, U.K.S.; Oba, S.; Yanase, E.; Murakami, Y. Phenolic acids, flavonoids and total antioxidant capacity of selected leafy vegetables. J. Fun. Foods 2012, 4, 979–987. [Google Scholar] [CrossRef]
- Di Mola, I.; Cozzolino, E.; Ottainao, L.; Giordano, M.; Rouphael, Y.; Colla, G.; Mori, M. Effect of Vegetal- and Seaweed Extract-based Biostimulants on Agronomical and Leaf Quality Traits of Greenhouse Baby Lettuce under Four Regimes of Nitrogen Fertilization. Agronomy 2019, 10, 571. [Google Scholar] [CrossRef]
- Wang, Z.H.; Li, S.X.; Malhi, S. Effects of fertilization and other agronomic measures on nutritional quality of crops. J. Sci. Food Agric. 2008, 88, 7–23. [Google Scholar] [CrossRef]
- Lombardi, N.; Salzano, A.M.; Troise, A.D.; Scaloni, A.; Vitaglione, P.; Vinale, F.; Lanzuise, S. Effect of trichoderma bioactive metabolite treatments on the production, quality and protein profile of strawberry fruits. J. Agric. Food Chem. 2020, 68, 7246–7258. [Google Scholar] [CrossRef]
- Petrisor, C.; Paica, A.; Constantinescu, F. Influence of abiotic factors on in vitro growth of Trichoderma strains. Proc. Rom. Acad. Ser. B 2016, 18, 11–14. [Google Scholar]
- Singh, A.; Shahid, M.; Srivastava, M.; Pandey, S.; Sharma, A.; Kumar, V. Optimal physical parameters for growth of Trichoderma species at varying pH, temperature and agitation. Virol. Mycol. 2014, 3, 127–134. [Google Scholar]
- European Community. Reg. n° 1258 of 2 December 2011. Off. J. Eur. Union 2011, L 320, 15–17. [Google Scholar]
- Li, R.X.; Cai, F.; Pang, G.; Shen, Q.R.; Li, R.; Chen, W. Solubilisation of phosphate and micronutrients by Trichoderma harzianum and its relationship with the promotion of tomato plant growth. PLoS ONE 2015, 10, e0130081. [Google Scholar] [CrossRef]
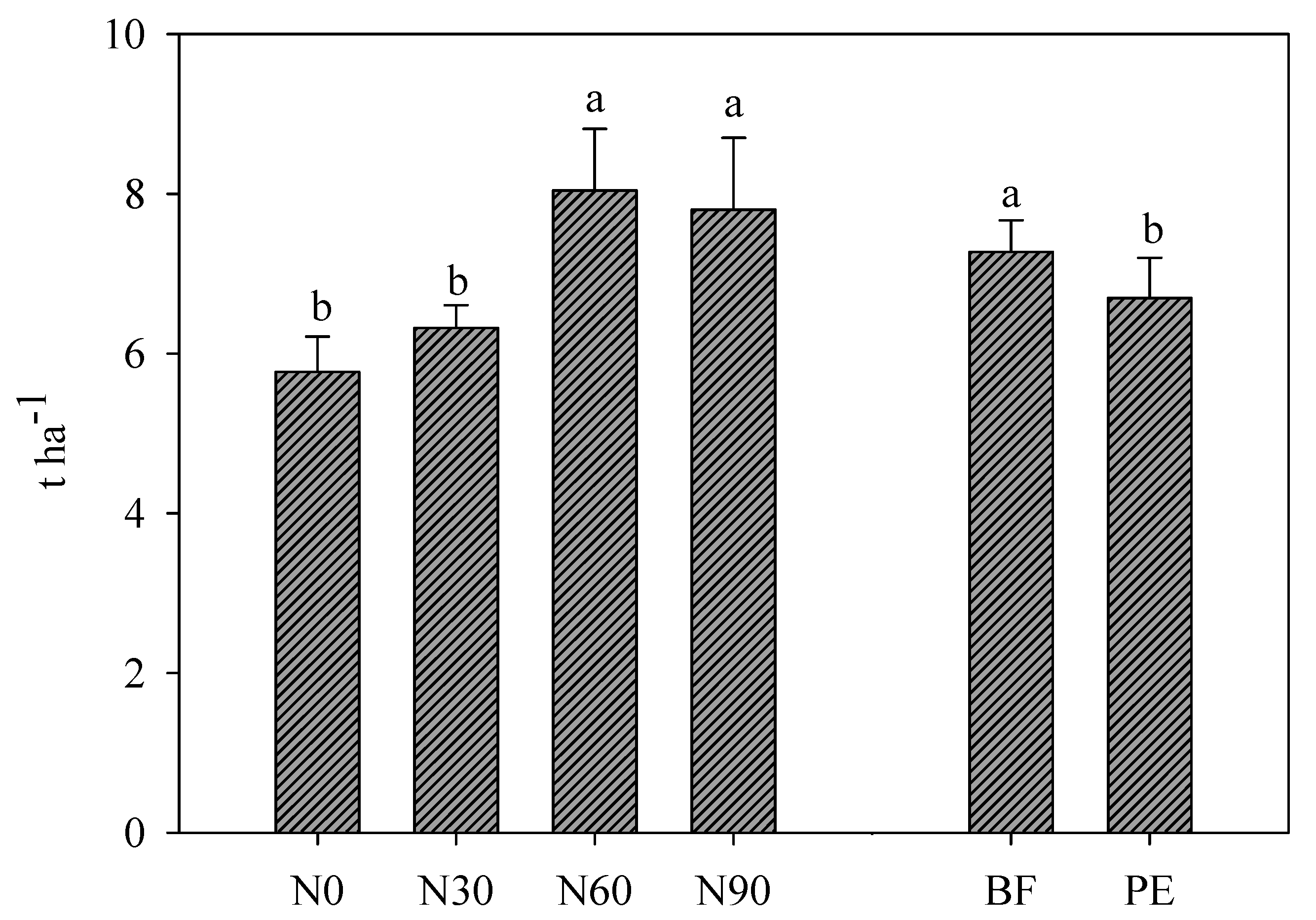
| Treatments | Head | ||
|---|---|---|---|
| Fresh Weight | Diameter | Number of Leaves | |
| (g Plant-1) | (cm) | (No. Plant−1) | |
| Mulching | |||
| BF | 290.83 a | 25.22 a | 47.75 a |
| PE | 267.90 b | 23.89 b | 43.54 b |
| Fertilization | |||
| N0 | 230.80 c | 23.15 b | 41.50 c |
| N30 | 252.79 b | 23.86 b | 44.42 b |
| N60 | 321.74 a | 25.82 a | 48.83 a |
| N90 | 312.12 a | 25.39 a | 47.83 a |
| Trichoderma | |||
| Control | 281.65 | 24.30 | 46.04 |
| TR | 277.07 | 24.81 | 45.25 |
| Significance | |||
| Mulching (M) | ** | * | * |
| Fertilization (F) | ** | ** | * |
| Trichoderma (TR) | NS | NS | NS |
| M × F | NS | NS | NS |
| M × TR | NS | NS | NS |
| F × TR | NS | NS | NS |
| M × F × TR | NS | NS | NS |
| Treatments | SPAD | L * | a * | b * |
|---|---|---|---|---|
| Mulching | ||||
| BF | 38.62 a | 49.89 | −11.11 | 34.56 |
| PE | 37.79 b | 48.95 | −11.66 | 36.07 |
| Fertilization | ||||
| N0 | 36.36 c | 48.02 c | −10.69 a | 36.84 |
| N30 | 38.04 b | 49.15 bc | −11.42 ab | 35.73 |
| N60 | 39.50 a | 50.68 a | −11.61 bc | 34.88 |
| N90 | 38.93 a | 49.83 ab | −11.82 c | 33.81 |
| Trichoderma | ||||
| Control | 38.04 | 49.55 | −11.44 | 34.90 |
| TR | 38.38 | 49.28 | −11.31 | 35.73 |
| Significance | ||||
| Mulching (M) | ** | NS | NS | NS |
| Fertilization (F) | ** | * | * | NS |
| Trichoderma (TR) | NS | NS | NS | NS |
| M × F | NS | NS | NS | NS |
| M × TR | NS | NS | NS | NS |
| F × TR | NS | NS | NS | NS |
| M × F × TR | NS | NS | NS | NS |
| Treatments | LAA | HAA | Phenols | TAA |
|---|---|---|---|---|
| (mM Trolox 100g−1 dw) | (mM AA 100g−1 dw) | (mg gallic acid g−1 dw) | (mg g−1 fw) | |
| Mulching | ||||
| BF | 9.52 | 6.69 | 1.552 | 19.59 a |
| PE | 10.42 | 7.16 | 1.674 | 18.16 b |
| Fertilization | ||||
| N0 | 11.75 a | 7.68 a | 1.723 a | 21.98 a |
| N30 | 10.70 ab | 7.62 a | 1.704 ab | 20.76 b |
| N60 | 8.97 ab | 6.14 b | 1.576 bc | 18.30 c |
| N90 | 8.48 b | 6.24 b | 1.448 c | 14.47 d |
| Trichoderma | ||||
| Control | 9.70 | 6.51 b | 1.676 | 18.30 b |
| TR | 10.24 | 7.33 a | 1.549 | 19.45 a |
| Significance | ||||
| Mulching (M) | NS | NS | NS | * |
| Fertilization (F) | * | ** | * | ** |
| Trichoderma (T) | NS | * | NS | * |
| M × F | NS | NS | NS | NS |
| M × T | * | * | NS | NS |
| F × T | NS | NS | NS | NS |
| M × F × T | NS | NS | NS | NS |
| Treatments | Chlorophyll a | Chlorophyll b | Total Chlorophyll | Carotenoids | Nitrate |
|---|---|---|---|---|---|
| (mg g−1 fw) | (mg g−1 fw) | (mg g−1 fw) | (µg g−1 fw) | (mg g−1 fw) | |
| Mulching | |||||
| BF | 0.552 | 0.224 | 0.776 b | 293 | 1570.7 |
| PE | 0.599 | 0.242 | 0.841 a | 309 | 1526.7 |
| Fertilization | |||||
| N0 | 0.487 b | 0.187 b | 0.674 b | 264 b | 1163.8 c |
| N30 | 0.591 a | 0.246 a | 0.838 a | 301 ab | 1479.9 b |
| N60 | 0.606 a | 0.246 a | 0.852 a | 316 ab | 1794.3 a |
| N90 | 0.618 a | 0.252 a | 0.871 a | 324 a | 1756.7 a |
| Trichoderma | |||||
| Control | 0.581 | 0.217 b | 0.798 | 311 | 1483.9 b |
| TR | 0.570 | 0.249 a | 0.819 | 292 | 1613.5 a |
| Significance | |||||
| Mulching | NS | NS | * | NS | NS |
| Fertilization | * | * | * | * | ** |
| Trichoderma | NS | * | NS | NS | * |
| M × F | NS | NS | NS | NS | NS |
| M × T | NS | NS | NS | NS | NS |
| F × T | NS | NS | NS | NS | NS |
| M × F × T | NS | NS | NS | NS | NS |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Mola, I.; Ottaiano, L.; Cozzolino, E.; Senatore, M.; Sacco, A.; El-Nakhel, C.; Rouphael, Y.; Mori, M. Trichoderma spp. and Mulching Films Differentially Boost Qualitative and Quantitative Aspects of Greenhouse Lettuce under Diverse N Conditions. Horticulturae 2020, 6, 55. https://doi.org/10.3390/horticulturae6030055
Di Mola I, Ottaiano L, Cozzolino E, Senatore M, Sacco A, El-Nakhel C, Rouphael Y, Mori M. Trichoderma spp. and Mulching Films Differentially Boost Qualitative and Quantitative Aspects of Greenhouse Lettuce under Diverse N Conditions. Horticulturae. 2020; 6(3):55. https://doi.org/10.3390/horticulturae6030055
Chicago/Turabian StyleDi Mola, Ida, Lucia Ottaiano, Eugenio Cozzolino, Mauro Senatore, Adriana Sacco, Christophe El-Nakhel, Youssef Rouphael, and Mauro Mori. 2020. "Trichoderma spp. and Mulching Films Differentially Boost Qualitative and Quantitative Aspects of Greenhouse Lettuce under Diverse N Conditions" Horticulturae 6, no. 3: 55. https://doi.org/10.3390/horticulturae6030055
APA StyleDi Mola, I., Ottaiano, L., Cozzolino, E., Senatore, M., Sacco, A., El-Nakhel, C., Rouphael, Y., & Mori, M. (2020). Trichoderma spp. and Mulching Films Differentially Boost Qualitative and Quantitative Aspects of Greenhouse Lettuce under Diverse N Conditions. Horticulturae, 6(3), 55. https://doi.org/10.3390/horticulturae6030055









