Grapefruit Production in Open Hydroponics System
Abstract
1. Introduction
2. Materials and Methods
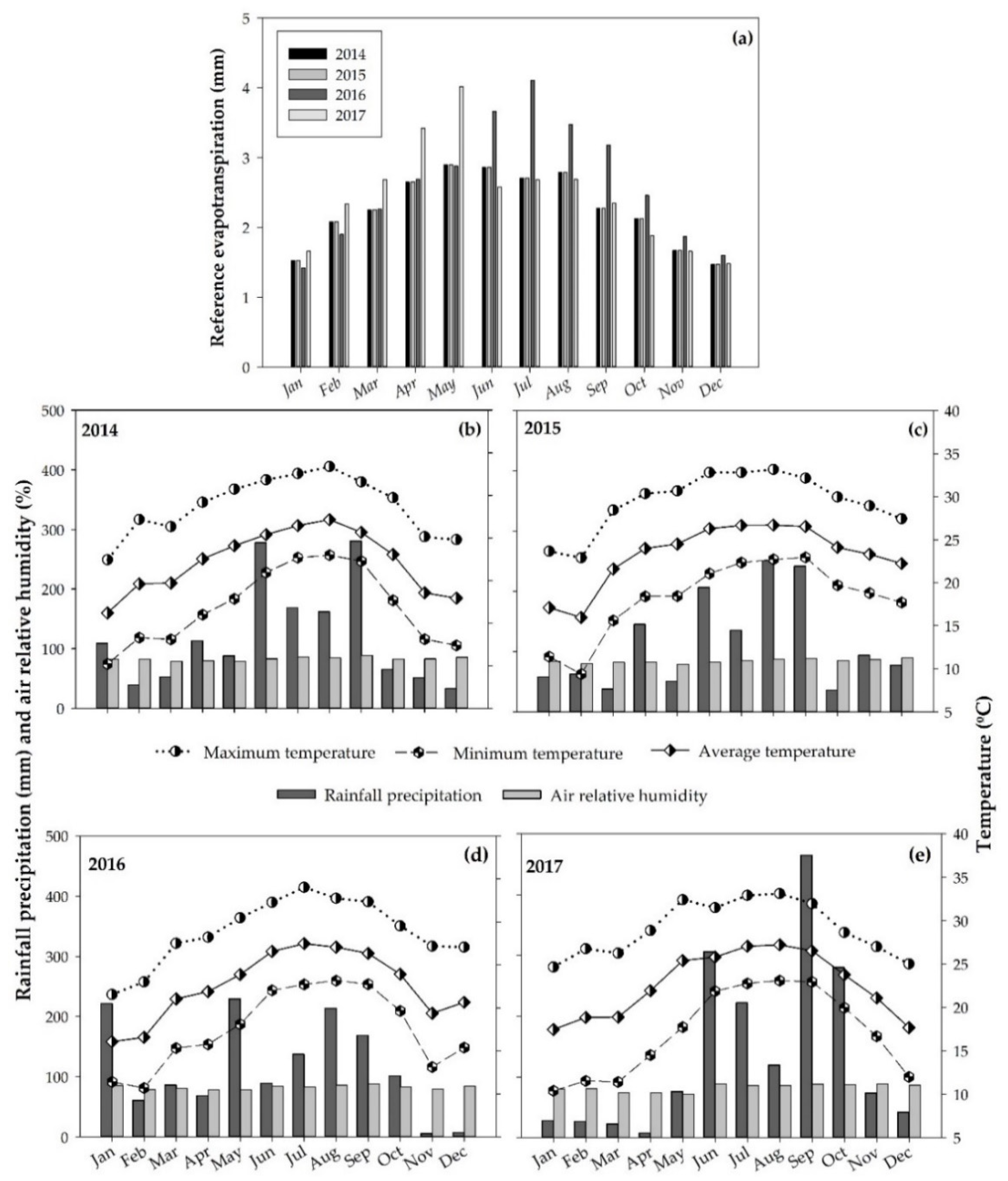
2.1. Experimental Area
2.2. Experimental Design and Treatments
- RR/SO_STD_dry_MS = “Ray Ruby” grapefruit (RR) on Sour Orange (SO) rootstock + standard spacing (STD, 3.8 × 7 m, 358 trees per ha) + 12N-1.31P-7.47K controlled-release fertilizer (CRF) applied in-ground + microsprinkler (MS) irrigation (one emitter per tree; blue microsprinklers—40.5 LPH at 138 kPa).
- RR/SO_HDS_fert_DD = “Ray Ruby” grapefruit on Sour Orange rootstock + HDS [(2.74 × 1.5 × 0.9 m) × 6.1 m, 953 trees per ha)] + 15N-2.6P-22.4K water-soluble fertilizer applied by fertigation + drip irrigation (four emitters per tree, installed on double rows; blue dripper—3.8 LPH at 138 kPa).
- RR/897_HDS_fert_MS = “Ray Ruby” grapefruit on US-897 rootstock + HDS + 15N-2.6P-22.4K applied by fertigation + microsprinkler irrigation (same as above).
- RR/897_HDS_fert_DD = “Ray Ruby” grapefruit on US-897 rootstock + HDS + 15N-2.6P-22.4K applied by fertigation + drip irrigation (same as above).
- RR/SO_HDS_fert_MS = “Ray Ruby” grapefruit on Sour Orange rootstock + HDS + 15N-2.6P-22.4K applied by fertigation + microsprinkler irrigation (same as above).
2.3. Treatments Application and Cultural Practices
2.4. Measurements
2.4.1. Concentration of CLas DNA in Plant Leaf Tissue
2.4.2. Tree Size
2.4.3. Foliar Nutrient Concentration
2.4.4. Number of Fruit, Fruit Size, and Fruit Yield
2.4.5. Fruit Quality
2.5. Statistical Analysis
3. Results and Discussion
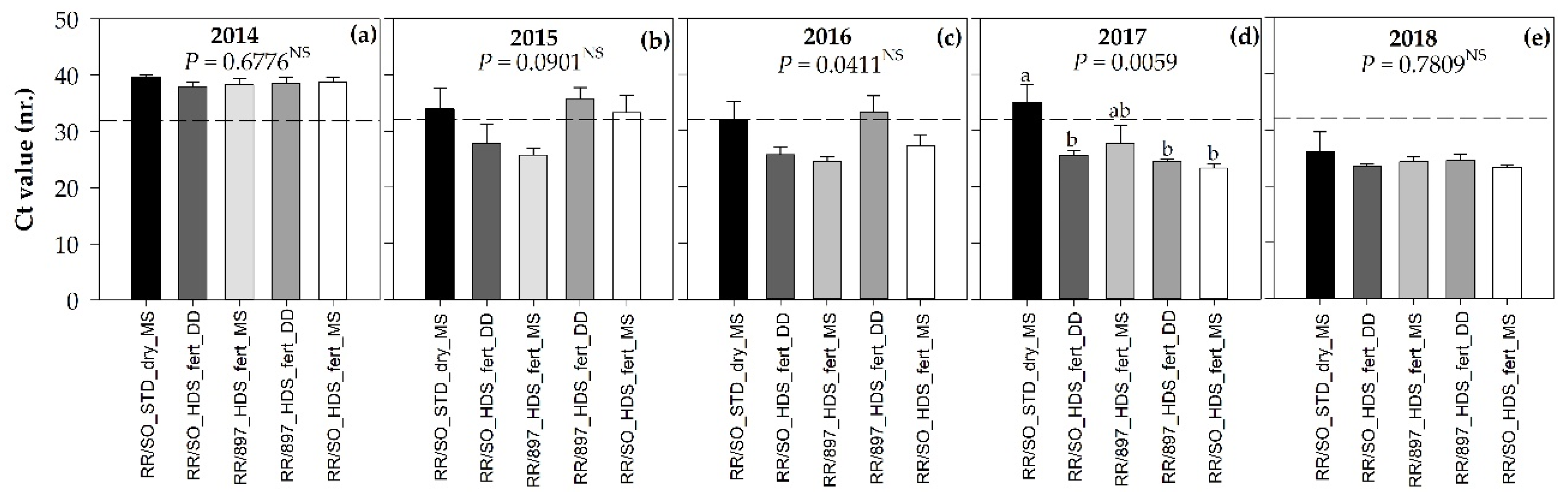
3.1. CLas Infection
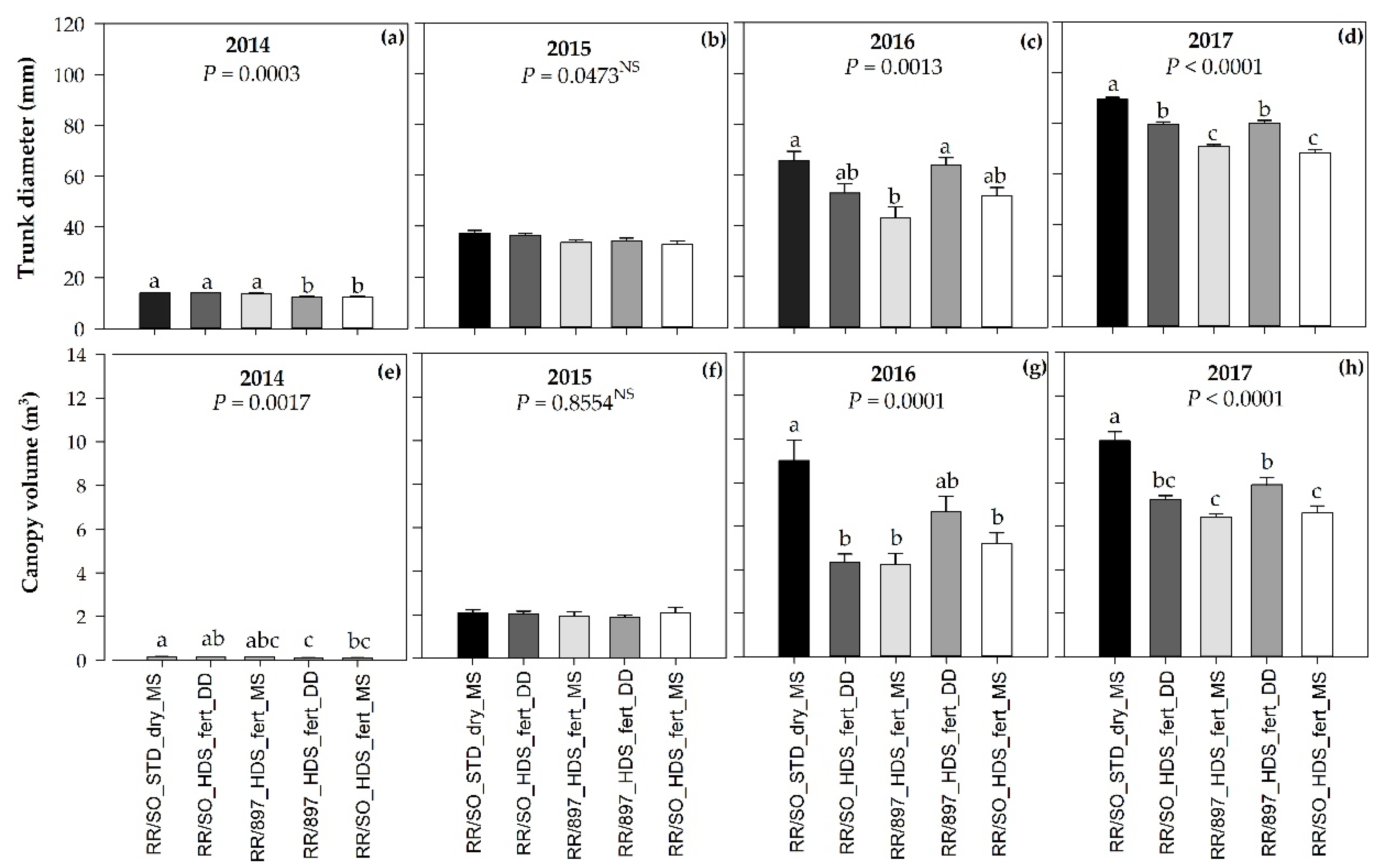
3.2. Tree Size
3.3. Foliar Nutrient Concentration
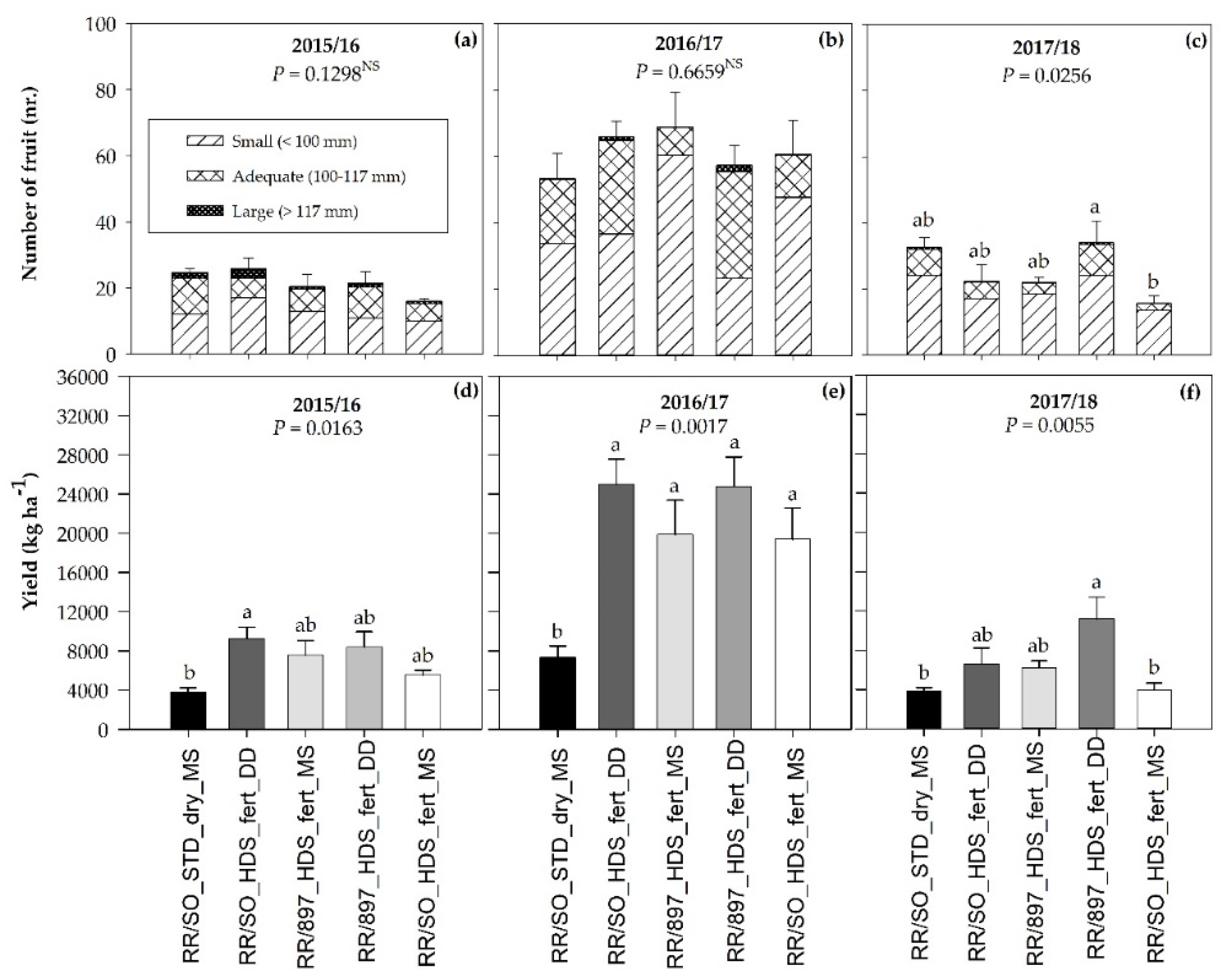
3.4. Number of Fruit, Fruit Size, and Fruit Yield
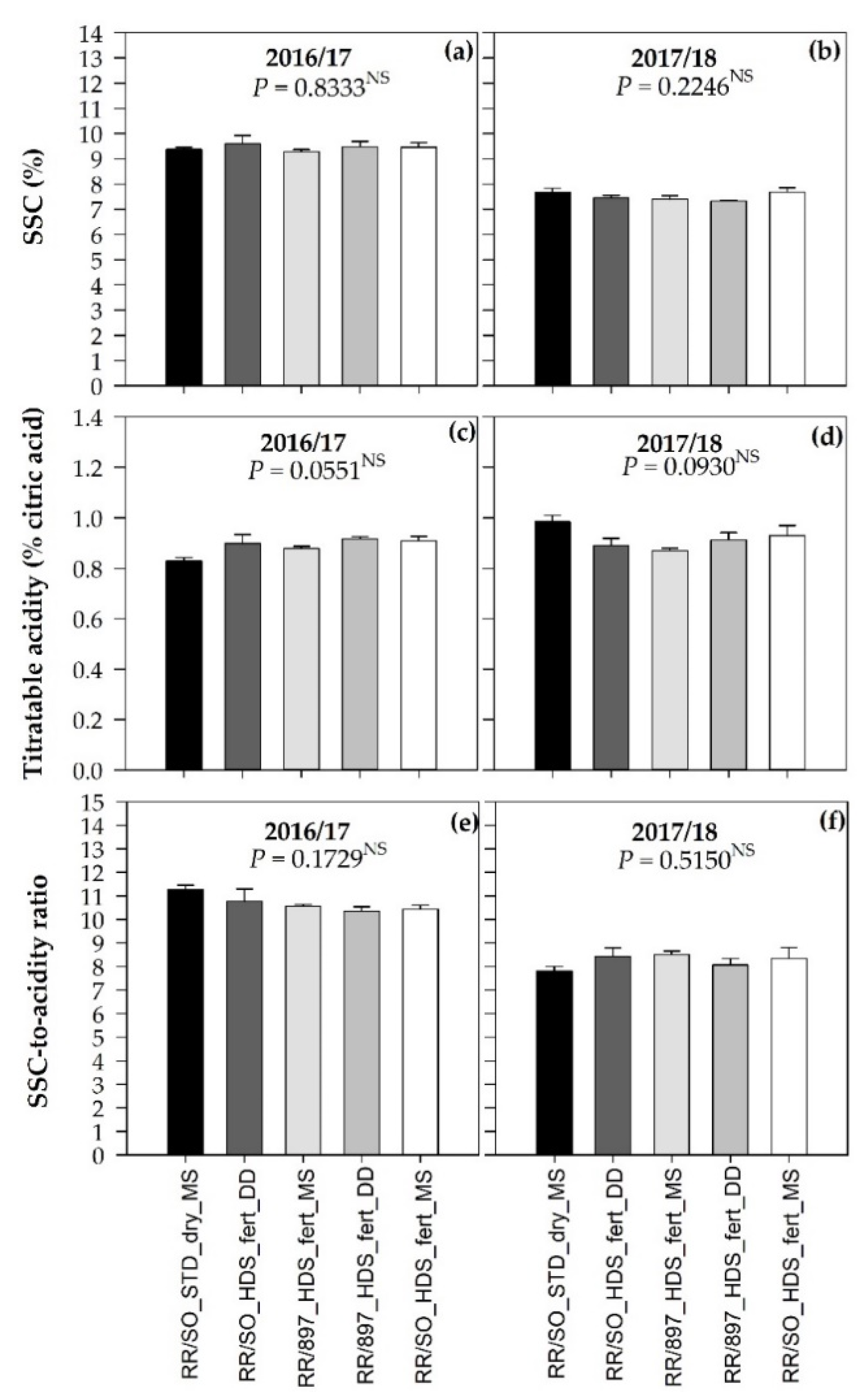
3.5. Fruit Quality
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- USDA. Florida citrus statistics 2018–2019. Florida Department of Agriculture and Consumer Services. National Agricultural Statistics Service. Available online: https://www.nass.usda.gov/Statistics_by_State/Florida/Publications/Citrus/Commercial_Citrus_Inventory/Commercial_Citrus_Inventory_Prelim/ccipr19.pdf (accessed on 4 May 2020).
- Morgan, K.T.; Wheaton, T.A.; William, S.C.; Parsons, L.R. Response of young and maturing citrus trees grown on a sandy soil to irrigation scheduling, nitrogen fertilizer rate, and nitrogen application method. HortScience 2009, 44, 145–150. [Google Scholar] [CrossRef]
- Kadyampakeni, D.M.; Morgan, K.T.; Schumann, A.W. Biomass, nutrient accumulation and tree size relationships for drip- and microsprinkler-irrigated orange trees. J. Plant Nutr. 2016, 39, 589–599. [Google Scholar] [CrossRef]
- Ferrarezi, R.S.; Rodriguez, K.; Sharp, D. How historical trends in Florida all-citrus production correlate with devastating hurricane and freeze events. Weather 2020, 75, 77–83. [Google Scholar] [CrossRef]
- Dala-Paula, B.M.; Plotto, A.; Bai, J.; Manthey, J.A.; Baldwin, E.A.; Ferrarezi, R.S.; Gloria, M.B.A. Effect of Huanglongbing or greening disease on orange juice quality, a review. Front. Plant Sci. 2019, 9, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Kadyampakeni, D.M.; Morgan, K.T.; Schumann, A.W.; Nkedi-Kizza, P. Effect of irrigation pattern and timing on root density of young citrus trees infected with Huanglongbing disease. HortTechnology 2014, 24, 209–221. [Google Scholar] [CrossRef]
- Farnsworth, D.; Grogan, K.A.; Van Bruggen, A.H.C.; Moss, C.B. The potential economic cost and response to greening in Florida citrus. Choices 2014, 29, 1–6. [Google Scholar]
- Kadyampakeni, D.M.; Morgan, K.T.; Schumann, A.W. Water and nutrient uptake in citrus open hydroponic systems. Proc. Fla. State Hort. Soc. 2013, 126, 58–61. [Google Scholar]
- Stover, E.W.; Castle, W.S.; Spyke, P. The citrus grove of the future and its implications for Huanglongbing management. Proc. Fla. State Hort. Soc. 2008, 121, 155–159. [Google Scholar]
- Schumann, A.; Hostler, K.; Waldo, L. Advanced Citrus Production Systems—Grove Designs for Higher Efficiencies. Available online: https://crec.ifas.ufl.edu/extension/trade_journals/2012/2012_Sept_advanced_citrus.pdf (accessed on 21 April 2020).
- Falivene, S. Open Hydroponics: Risks and Opportunities. Available online: http://www.arapahocitrus.com/files/OHS_Stage_Publications.pdf (accessed on 11 June 2020).
- Morgan, K.T.; Kadyampakeni, D.M. Open field hydroponics: Concept and application. In Advances in Citrus Nutrition; Srivastava, A.K., Ed.; Springer: Berlin, Germany, 2012; pp. 271–280. [Google Scholar]
- Morgan, K.T.; Zotarelli, L.; Dukes, M.D. Use of irrigation technologies for citrus trees in Florida. HortTechnology 2010, 20, 74–81. [Google Scholar] [CrossRef]
- Singerman, A.; Burani-Arouca, M.; Futch, S.H. The profitability of new citrus plantings in Florida in the era of Huanglongbing. HortScience 2018, 53, 1655–1663. [Google Scholar] [CrossRef]
- Dalal, R.P.S.; Sangwan, A.K.; Beniwal, B.S.; Sharma, S. Effect of planting density on canopy parameter, yield and water use efficiency of Kinnow mandarin. Indian J. Hort. 2013, 70, 587–590. [Google Scholar]
- Moreira, A.S.; Stuchi, E.S.; Silva, P.R.B.; Bassanezi, R.B.; Girardi, E.A.; Laranjeira, F.F. Could tree density play a role in managing Citrus Huanglongbing epidemics? Trop. Plant Pathol. 2019, 44, 268–274. [Google Scholar] [CrossRef]
- Phuyal, D.; Nogueira, T.A.R.; Jani, A.D.; Kadyampakeni, D.M.; Morgan, K.T.; Ferrarezi, R.S. ‘Ray Ruby’ grapefruit affected by Huanglongbing I. Planting density and soil nutrient management. HortScience 2020, in press. [Google Scholar]
- Castle, W.S.; Bowman, K.D.; Grosser, J.W.; Ferrarezi, R.S.; Futch, S.H.; Rogers, S. SP248. Florida Citrus Rootstock Selection Guide, 4th ed. 2019. Available online: https://edis.ifas.ufl.edu/pdffiles/HS/HS126000.pdf (accessed on 21 April 2020).
- Morgan, K.T.; Schumann, A.W.; Castle, W.S.; Stover, E.W.; Kadyampakeni, D.; Spyke, P.; Roka, F.M.; Muraro, R.; Morris, R.A. Citrus production systems to survive greening: Horticultural practices. Proc. Fla. State Hortic. Soc. 2009, 122, 114–121. [Google Scholar]
- USDA. Natural Resources Conservation Service. Web Soil Survey. 2020. Available online: https://websoilsurvey.sc.egov.usda.gov/App/WebSoilSurvey.aspx (accessed on 28 April 2020).
- Morgan, K.T.; Kadyampakeni, D.M. Nutrition of Florida Citrus Trees, 3rd ed. Available online: https://edis.ifas.ufl.edu/pdffiles/SS/SS47800.pdf (accessed on 14 June 2020).
- Folimonova, S.Y.; Robertson, C.J.; Garnsey, S.M.; Gowda, S.; Dawson, W.O. Examination of the responses of different genotypes of citrus to Huanglongbing (citrus greening) under different conditions. Phytopathology 2009, 99, 1346–1354. [Google Scholar] [CrossRef] [PubMed]
- Bowman, K.D.; Faulkner, L.; Kesinger, K. New citrus rootstocks released by USDA 2001–2010: Field performance and nursery characteristics. HortScience 2016, 51, 1208–1214. [Google Scholar] [CrossRef]
- FDACS Mobile Irrigation Labs. Available online: https://www.fdacs.gov/Water/Mobile-Irrigation-Labs#:~:text=Mobile%20Irrigation%20Labs%20(MILs)%20provide,efficiency%20of%20their%20water%20use.&text=These%20multi%2Dagency%2C%20partnership%2D,assisting%20agricultural%20producers%20throughout%20Florida (accessed on 24 June 2020).
- Li, W.; Hartung, J.S.; Levy, L. Quantitative real-time PCR for detection and identification of Candidatus Liberibacter species associated with citrus Huanglongbing. J. Microbiol. Methods 2006, 66, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, U.; Bowman, K.D. Tolerance of the trifoliate citrus hybrid US-897 (Citrus reticulata Blancox Poncirus trifoliata L. Raf.) to Huanglongbing. HortScience 2011, 46, 16–22. [Google Scholar] [CrossRef]
- Obreza, T.A.; Rouse, R.E. Fertilizer effects on early growth and yield of ‘Hamlin’ orange trees. HortScience 1993, 28, 111–114. [Google Scholar] [CrossRef]
- Morgan, K.T.; Kadyampakeni, D.M.; Zekri, M.; Shumann, A.W.; Vashisth, T.; Obreza, T.A. 2019–2020 Florida Citrus Production Guide: Nutrition Management for Citrus Trees. UF IFAS. University of Florida: Gainesville, 2019. Available online: https://edis.ifas.ufl.edu/pdffiles/CG/CG09100.pdf (accessed on 7 April 2020).
- Ferrarezi, R.S.; Qureshi, J.A.; Wright, A.L.; Ritenour, M.A.; Macan, N.P.F. Citrus production under screen as a strategy to protect grapefruit trees from Huanglongbing disease. Front. Plant Sci. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Wardowski, W.; Whigham, J.; Grierson, W.; Soule, J. Quality Tests for Florida Citrus. 1995. Available online: http://irrec.ifas.ufl.edu/postharvest/pdfs/Quality_Tests_for_FL_Citrus-SP_99.pdf (accessed on 7 April 2020).
- Gottwald, T.R.; Graham, J.H.; Irey, M.S.; McCollum, T.G.; Wood, B.W. Inconsequential effect of nutritional treatments on Huanglongbing control, fruit quality, bacterial titer and disease progress. Crop. Prot. 2012, 36, 73–82. [Google Scholar] [CrossRef]
- SAS Institute Inc. Base SAS® 9.4 Procedures Guide; SAS Institute Inc.: Cary, NC, USA, 2013. [Google Scholar]
- Shin, K.; Van Bruggen, A.H.C. Bradyrhizobium isolated from Huanglongbing (HLB) affected citrus trees reacts positively with primers for Candidatus Liberibacter asiaticus. Eur. J. Plant Pathol. 2018, 151, 291–306. [Google Scholar] [CrossRef]
- Morgan, K.T.; Rouse, R.E.; Ebel, R.C. Foliar applications of essential nutrients on growth and yield of ‘Valencia’ sweet orange infected with Huanglongbing. HortScience 2016, 51, 1482–1493. [Google Scholar] [CrossRef]
- Bove, J.M. Huanglongbing: A destructive, newly-emerging, century-old disease of citrus. J. Plant Pathol. 2006, 88, 7–37. [Google Scholar]
- Graham, J.H.; Johnson, E.G.; Gottwald, T.R.; Irey, M.S. Presymptomatic fibrous root decline in citrus trees caused by Huanglongbing and potential interaction with Phytophthora spp. Plant Dis. 2013, 97, 1195–1199. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, E.; Gonzalez, P.; Achor, D.; Albrigo, G. Anatomical distribution of abnormally high levels of starch in HLB-affected Valencia orange trees. Physiol. Mol. Plant P. 2009, 74, 76–83. [Google Scholar] [CrossRef]
- Achor, D.; Etxeberria, E.; Wang, N.; Folimonova, S.; Chung, K.; Albrigo, L. Sequence of anatomical symptom observations in citrus affected with Huanglongbing disease. Plant Pathol. J. 2010, 9, 56–64. [Google Scholar] [CrossRef]
- Folimonova, S.Y.; Achor, D.S. Early events of citrus greening (Huanglongbing) disease development at the ultrastructural level. Phytopathology 2010, 100, 949–958. [Google Scholar] [CrossRef]
- Handique, U.; Ebel, R.; Morgan, K.T. Influence of soil-applied fertilizer on greening development in new growth flushes of sweet orange. Proc. Fla. State. Hort. Soc. 2012, 125, 36–39. [Google Scholar]
- Vashisth, T.; Tang, L. Fruit Drop and HLB. Citrus Industry. Available online: http://citrusindustry.net/2018/09/12/fruit-drop-and-hlb/ (accessed on 27 April 2020).
- Stuchi, E.S.; Girardi, E.A.; Basssanezi, R.B.; Laranjeira, F.F. Yield and Huanglongbing progress at four tree spacings of sweet orange. In Proceedings of the International Citrus Congress, Londrina, Brazil, 18–23 September 2016; pp. S6–S299. [Google Scholar]
- Wheaton, T.A.; Castle, W.S.; Whitney, J.D.; Tucker, D.P.H. Performance of citrus scion cultivars and rootstock in a high-density planting. Hort. Sci. 1991, 26, 837–840. [Google Scholar] [CrossRef]
- Huang, R.F. Study on the effect of planting density on the growth and production of Ponkan mandarin. South China Fruit 1997, 26, 5–21. [Google Scholar]
- Zaman, Q.U.; Schumann, A.W. Performance of an ultrasonic tree volume measurement system in commercial citrus groves. Precis. Agric. 2005, 6, 467–480. [Google Scholar] [CrossRef]
- Kumar, D.; Ahmad, N.; Verma, M.K. Studies on high density planting in almond in Kashmir valley. Indian J. Hort. 2012, 69, 328–332. [Google Scholar]
- Acosta, D.F.R. Mitigation of Huanglongbing Effects on Grapefruit Trees Using Enhanced Nutritional Programs. Master’s Thesis, University of Florida, Gainesville, FL, USA, 2016. [Google Scholar]
- Li, J.; Li, L.; Pang, Z.; Kolbasov, V.G.; Ehsani, R.; Carter, E.W.; Wang, N. Developing citrus Huanglongbing (HLB) management strategies based on the severity of symptoms in HLB-endemic citrus-producing regions. Phytopathology 2019, 109, 582–592. [Google Scholar] [CrossRef]
- Gonzalez, P.; Reyes-De-Corcuera, J.; Etxeberria, E. Characterization of leaf starch from HLB-affected and unaffected-girdled citrus trees. Physiol. Mol. Plant Pathol. 2012, 79, 71–78. [Google Scholar] [CrossRef]
- Gottwald, T.R. Epidemiological understanding of citrus Huanglongbing. Annu. Rev. Phytopathol. 2010, 48, 119–139. [Google Scholar] [CrossRef]
- Mann, M.S.; Takkar, P.N. Antagonism of micronutrient cations on sweet orange leaves. Sci. Hortic. 1983, 20, 259–265. [Google Scholar] [CrossRef]
- Spann, T.M.; Danyluk, M.D. Effects of HLB Infection on Sweet Orange Fruit Size and Quality. Available online: https://crec.ifas.ufl.edu/extension/trade_journals/2010/2010_sept_effects_hlb.pdf (accessed on 27 April 2020).
- Hutton, R.J.; Broadbent, P.; Bevington, K. Viroid dwarfing for high density citrus planting. Hortic. Rev. 2000, 24, 277–317. [Google Scholar]
- Vidalakis, G.; Pagliaccia, D.; Bash, J.A.; Afunian, M.; Semancik, J.S. Citrus dwarfing viroid: Effects on tree size and scion performance specific to Poncirus trifoliate rootstock for high-density planting. Ann. Appl. Biol. 2011, 158, 204–217. [Google Scholar] [CrossRef]
- Weibel, F.P.; Alföldi, T. Improving the quality and shelf life of fruit from organic production systems. In Handbook of Organic Food Safety and Quality, 1st ed.; Cooper, J., Leifert, C., Niggli, U., Eds.; Woodhead Publishing: Sawston, Cambridge, UK, 2007; pp. 330–352. [Google Scholar]
- Hutton, R.J.; Loveys, B.R. A partial root zone drying irrigation strategy for citrus-effects on water use efficiency and fruit characteristics. Agric. Water Manag. 2011, 98, 1485–1496. [Google Scholar] [CrossRef]
- McCollum, G.; Bowman, K.D. Rootstock effects on fruit quality among ‘Ray Ruby’ grapefruit trees grown in the Indian River District of Florida. HortScience 2017, 52, 541–546. [Google Scholar] [CrossRef]
- USDA. United States Standards for Grades of Grapefruit Juice. 2012. Available online: https://www.ams.usda.gov/sites/default/files/media/Canned_Grapefruit_Juice_Standard%5B1%5D.pdf (accessed on 1 May 2020).
| Treatment | 2014 | 2015 | 2016 | 2017 | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|---|---|---|---|
| N (g kg−1) | P (g kg−1) | |||||||
| RR/SO_STD_dry_MS | 27.27 a | 26.72 a | 23.55 a | 27.13 a | 1.52 a | 1.40 a | 1.90 a | 1.82 a |
| RR/SO_HDS_fert_DD | 29.14 a | 26.41 a | 23.18 a | 25.56 a | 1.50 a | 1.33 a | 1.96 a | 1.90 a |
| RR/897_HDS_fert_MS | 30.18 a | 25.96 a | 22.12 a | 26.04 a | 1.56 a | 1.42 a | 1.92 a | 1.94 a |
| RR/897_HDS_fert_DD | 28.76 a | 27.72 a | 23.38 a | 25.22 a | 1.52 a | 1.37 a | 2.18 a | 1.80 a |
| RR/SO_HDS_fert_MS | 28.90 a | 29.02 a | 22.74 a | 25.54 a | 1.56 a | 1.38 a | 1.92 a | 1.86 a |
| Optimal range 1 | 25–27 | 1.2–1.6 | ||||||
| p-value | 0.0766 NS | 0.1211 NS | 0.1085 NS | 0.2223 NS | 0.8961 NS | 0.3297 NS | 0.2005 NS | 0.5107 NS |
| CV (%) | 5.12 | 6.96 | 3.79 | 5.15 | 7.43 | 4.91 | 10.22 | 7.31 |
| Ca (g kg−1) | Mg (g kg−1) | |||||||
| RR/SO_STD_dry_MS | 28.42 ab | 33.47 a | 36.35 a | 34.90 a | 3.55 ab | 2.69 a | 3.13 a | 2.98 a |
| RR/SO_HDS_fert_DD | 30.94 a | 32.71 a | 38.08 a | 34.34 a | 3.96 a | 2.74 a | 2.88 a | 2.98 a |
| RR/897_HDS_fert_MS | 30.46 a | 36.30 a | 37.28 a | 34.80 a | 3.04 bc | 2.73 a | 3.32 a | 3.16 a |
| RR/897_HDS_fert_DD | 27.16 bc | 32.02 a | 39.42 a | 32.26 a | 4.16 a | 2.74 a | 3.22 a | 2.84 a |
| RR/SO_HDS_fert_MS | 31.28 a | 34.06 a | 36.88 a | 33.98 a | 3.80 a | 2.82 a | 3.06 a | 2.90 a |
| Optimal range 1 | 30–49 | 3.0–4.9 | ||||||
| p-value | 0.0040 * | 0.1935 NS | 0.7953 NS | 0.3244 NS | 0.0003 ** | 0.1176 NS | 0.1590 NS | 0.4663 NS |
| CV (%) | 5.76 | 8.38 | 11.03 | 6.30 | 8.76 | 6.73 | 8.78 | 9.40 |
| K (g kg−1) | S (g kg−1) | |||||||
| RR/SO_STD_dry_MS | 19.30 a | 18.79 ab | 12.12 a | 14.40 a | 2.95 a | 3.57 a | 3.32 a | 3.25 a |
| RR/SO_HDS_fert_DD | 18.80 a | 19.29 ab | 13.10 a | 15.16 a | 2.72 a | 3.24 a | 3.02 a | 3.12 a |
| RR/897_HDS_fert_MS | 18.74 a | 16.47 bc | 8.38 bc | 13.90 a | 2.72 a | 3.40 a | 2.46 bc | 2.84 a |
| RR/897_HDS_fert_DD | 17.86 a | 21.04 a | 15.10 a | 15.18 a | 2.84 a | 3.51 a | 3.18 a | 3.10 a |
| RR/SO_HDS_fert_MS | 18.52 a | 19.64 ab | 11.52 ab | 14.18 a | 2.92 a | 3.48 a | 2.82 ab | 2.82 a |
| Optimal range 1 | 12–17 | -- | ||||||
| p-value | 0.7895 NS | 0.0225 * | 0.0004 ** | 0.1567 NS | 0.8453 NS | 0.3794 NS | 0.0011 * | 0.0418 NS |
| CV (%) | 9.63 | 10.27 | 15.79 | 6.53 | 14.58 | 7.96 | 9.68 | 7.92 |
| Treatment | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|
| B (mg kg−1) | ||||
| RR/SO_STD_dry_MS | 71.62 a | 67.76 a | 71.61 a | 72.62 a |
| RR/SO_HDS_fert_DD | 51.69 bc | 54.51 a | 67.19 a | 63.35 ab |
| RR/897_HDS_fert_MS | 59.79 ab | 53.18 a | 63.65 a | 61.07 ab |
| RR/897_HDS_fert_DD | 48.96 bc | 57.59 a | 70.40 a | 60.56 ab |
| RR/SO_HDS_fert_MS | 57.04 ab | 64.20 a | 71.10 a | 58.35 bc |
| Optimal range 1 | 60–120 | |||
| p-value | 0.0019 * | 0.2868 NS | 0.6589 NS | 0.0203 NS |
| CV (%) | 13.58 | 20.41 | 13.91 | 10.21 |
| Cu (mg kg−1) | ||||
| RR/SO_STD_dry_MS | 298.58 a | 49.60 a | 279.55 a | 136.551 a |
| RR/SO_HDS_fert_DD | 200.46 a | 44.38 a | 228.64 a | 126.20 a |
| RR/897_HDS_fert_MS | 211.04 a | 42.66 a | 231.68 a | 132.53 a |
| RR/897_HDS_fert_DD | 248.04 a | 47.73 a | 260.04 a | 165.42 a |
| RR/SO_HDS_fert_MS | 345.56 a | 43.76 a | 233.98 a | 116.59 a |
| Optimal range 1 | 5–16 | |||
| p-value | 0.3333 NS | 0.3045 NS | 0.0767 NS | 0.5715 NS |
| CV (%) | 47.36 | 12.55 | 12.76 | 35.08 |
| Fe (mg kg−1) | ||||
| RR/SO_STD_dry_MS | 73.92 a | 82.91 ab | 126.58 a | 66.95 a |
| RR/SO_HDS_fert_DD | 94.42 a | 71.34 c | 111.40 a | 69.08 a |
| RR/897_HDS_fert_MS | 82.47 a | 79.95 bc | 110.19 a | 64.47 a |
| RR/897_HDS_fert_DD | 94.56 a | 84.99 ab | 115.60 a | 63.88 a |
| RR/SO_HDS_fert_MS | 97.25 a | 89.49 a | 110.80 a | 64.11 a |
| Optimal range 1 | 60–120 | |||
| p-value | 0.0766 NS | 0.005 ** | 0.0977 NS | 0.5203 NS |
| CV (%) | 15.98 | 6.88 | 8.85 | 8.41 |
| Mn (mg kg−1) | ||||
| RR/SO_STD_dry_MS | 137.50 a | 35.29 a | 42.24 a | 70.62 a |
| RR/SO_HDS_fert_DD | 136.68 a | 31.44 a | 40.23 a | 69.12 a |
| RR/897_HDS_fert_MS | 132.80 a | 32.31 a | 26.97 a | 32.61 a |
| RR/897_HDS_fert_DD | 141.24 a | 35.81 a | 32.86 a | 54.06 a |
| RR/SO_HDS_fert_MS | 145.30 a | 39.60 a | 33.73 a | 47.68 a |
| Optimal range 1 | 25–100 | |||
| p-value | 0.8908 NS | 0.0952 NS | 0.5627 NS | 0.4129 NS |
| CV (%) | 14.61 | 13.68 | 44.60 | 58.52 |
| Zn (mg kg−1) | ||||
| RR/SO_STD_dry_MS | 72.65 a | 18.54 a | 27.37 a | 53.23 a |
| RR/SO_HDS_fert_DD | 70.18 a | 16.30 a | 25.36 a | 53.35 a |
| RR/897_HDS_fert_MS | 59.01 a | 16.73 a | 20.74 a | 24.86 a |
| RR/897_HDS_fert_DD | 71.12 a | 18.03 a | 25.51 a | 40.68 a |
| RR/SO_HDS_fert_MS | 63.54 a | 18.52 a | 22.93 a | 38.82 a |
| Optimal range 1 | 25–100 | |||
| p-value | 0.2682 NS | 0.0975 NS | 0.2839 NS | 0.6173 NS |
| CV (%) | 16.24 | 8.79 | 20.28 | 76.37 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrarezi, R.S.; Nogueira, T.A.R.; Jani, A.D.; Wright, A.L.; Ritenour, M.A.; Burton, R. Grapefruit Production in Open Hydroponics System. Horticulturae 2020, 6, 50. https://doi.org/10.3390/horticulturae6030050
Ferrarezi RS, Nogueira TAR, Jani AD, Wright AL, Ritenour MA, Burton R. Grapefruit Production in Open Hydroponics System. Horticulturae. 2020; 6(3):50. https://doi.org/10.3390/horticulturae6030050
Chicago/Turabian StyleFerrarezi, Rhuanito S., Thiago A. R. Nogueira, Arun D. Jani, Alan L. Wright, Mark A. Ritenour, and Randy Burton. 2020. "Grapefruit Production in Open Hydroponics System" Horticulturae 6, no. 3: 50. https://doi.org/10.3390/horticulturae6030050
APA StyleFerrarezi, R. S., Nogueira, T. A. R., Jani, A. D., Wright, A. L., Ritenour, M. A., & Burton, R. (2020). Grapefruit Production in Open Hydroponics System. Horticulturae, 6(3), 50. https://doi.org/10.3390/horticulturae6030050







