Deficit Irrigation and Arbuscular Mycorrhiza as a Water-Saving Strategy for Eggplant Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Growth Conditions
2.2. Experimental Design and Treatments
2.3. Estimation of Crop Water Requirement
2.4. Physiological Measurements and Sampling
2.5. Measurements of Crop Parameters
2.6. Plant and Soil Analysis
2.7. Statistical Analysis
3. Results
3.1. Crop Biomass
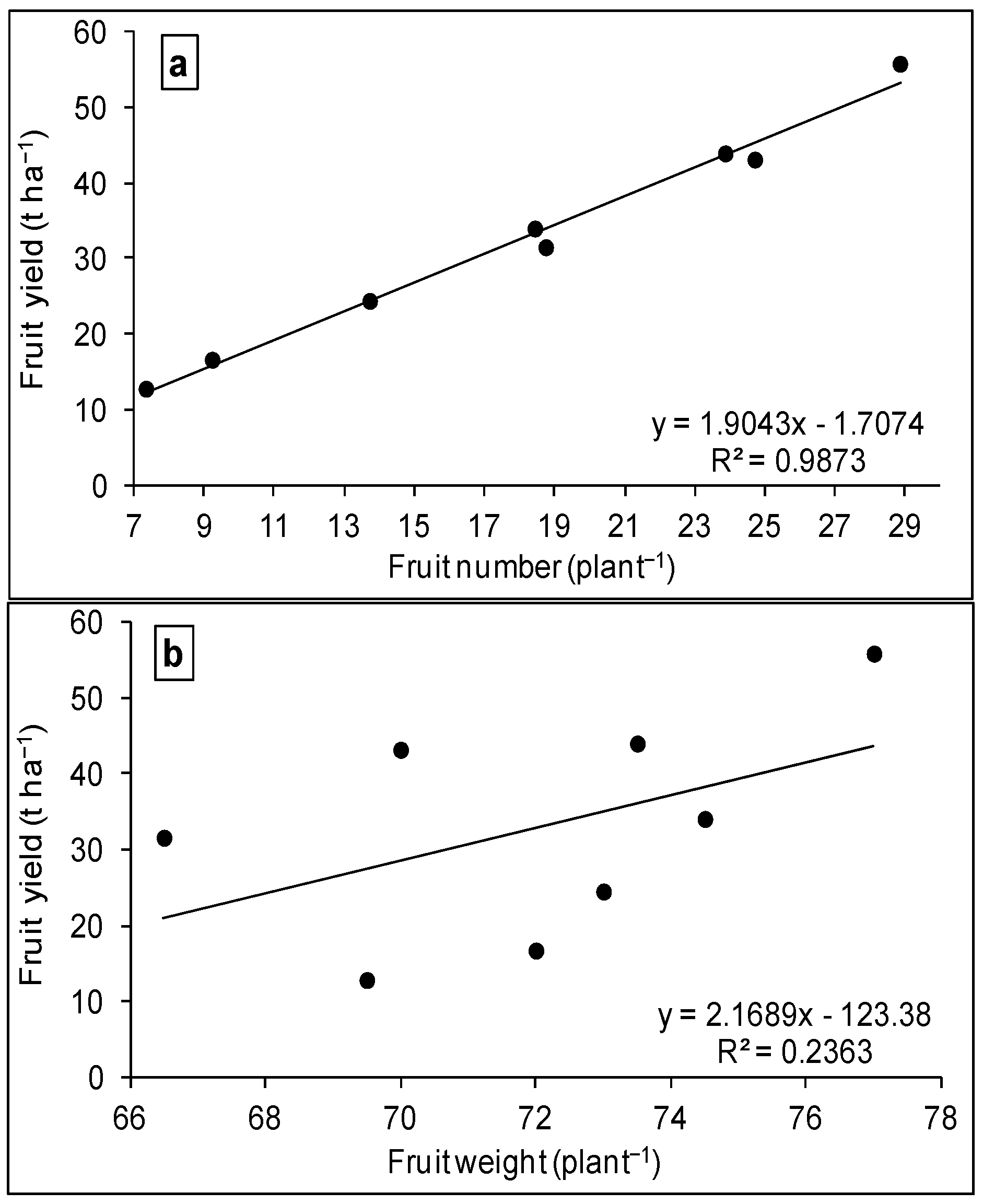
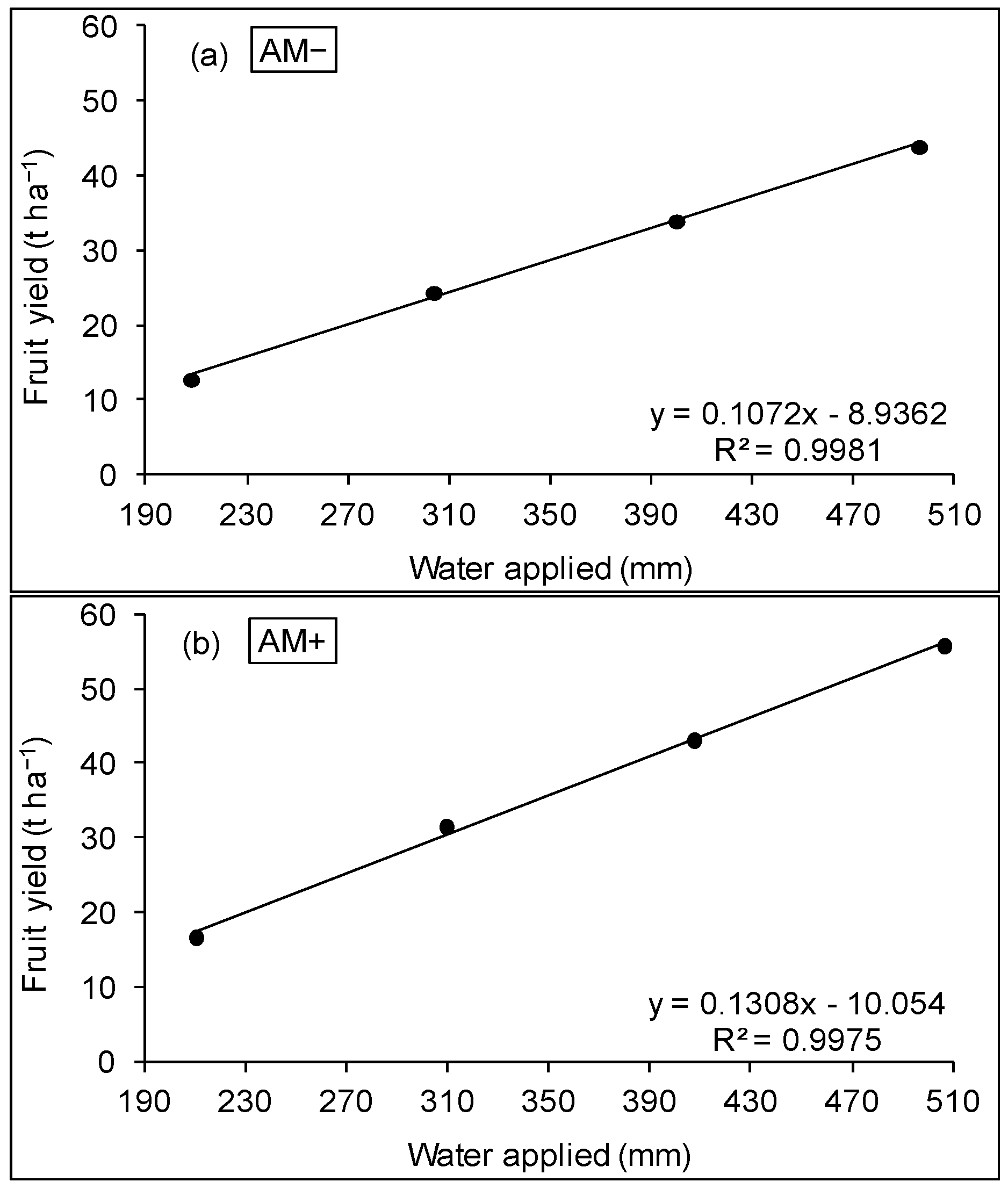
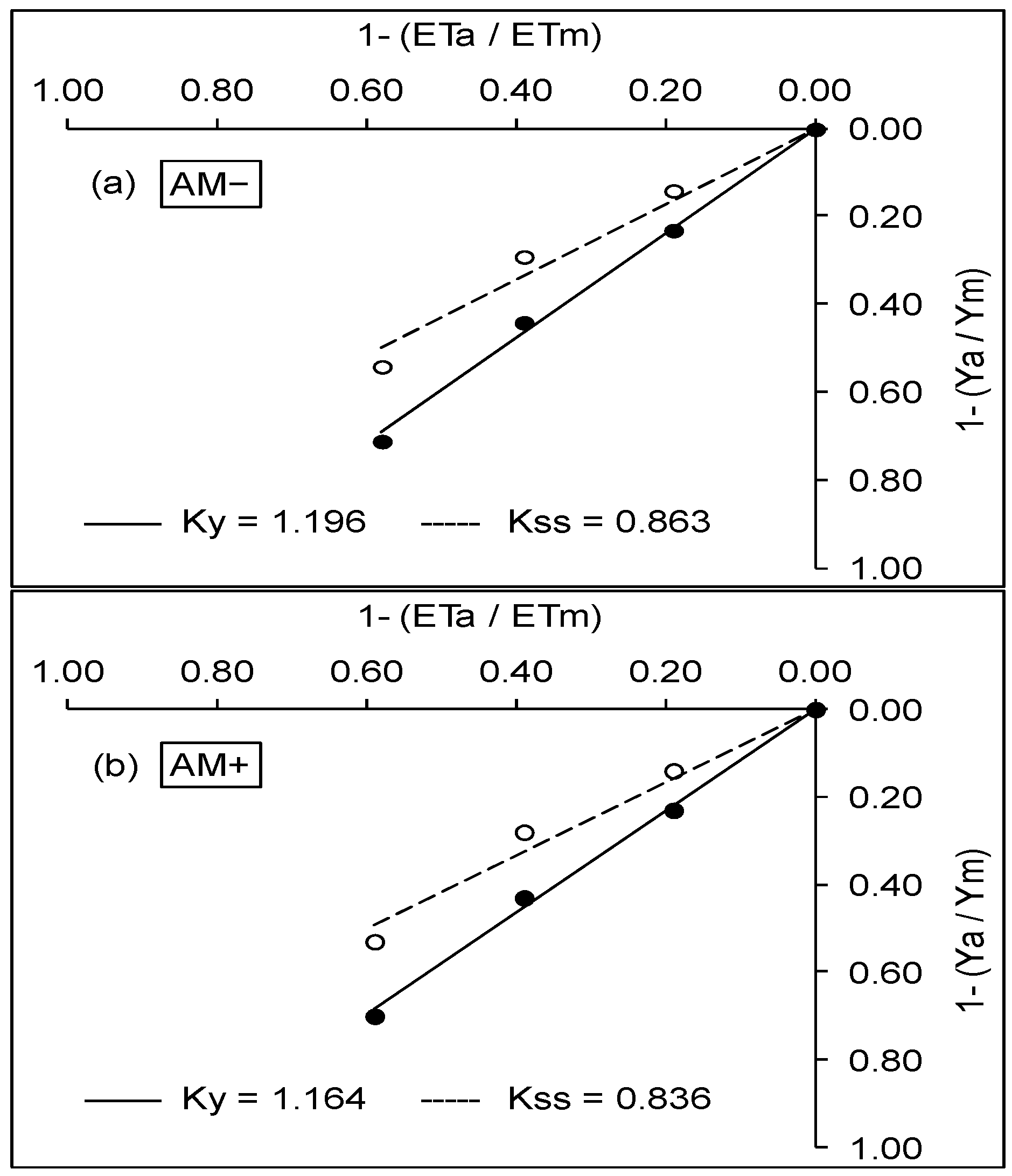
3.2. WUE and Production Function
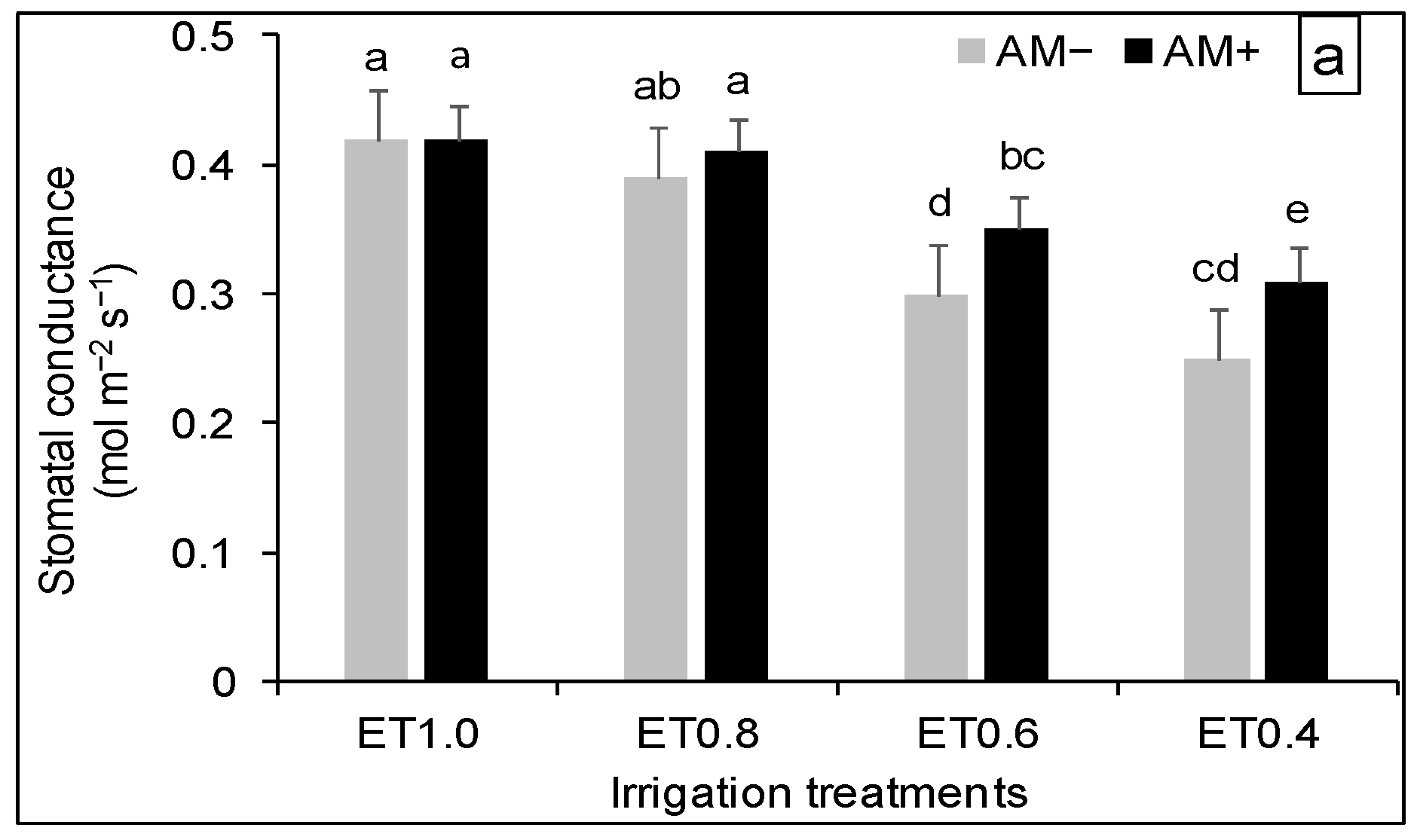
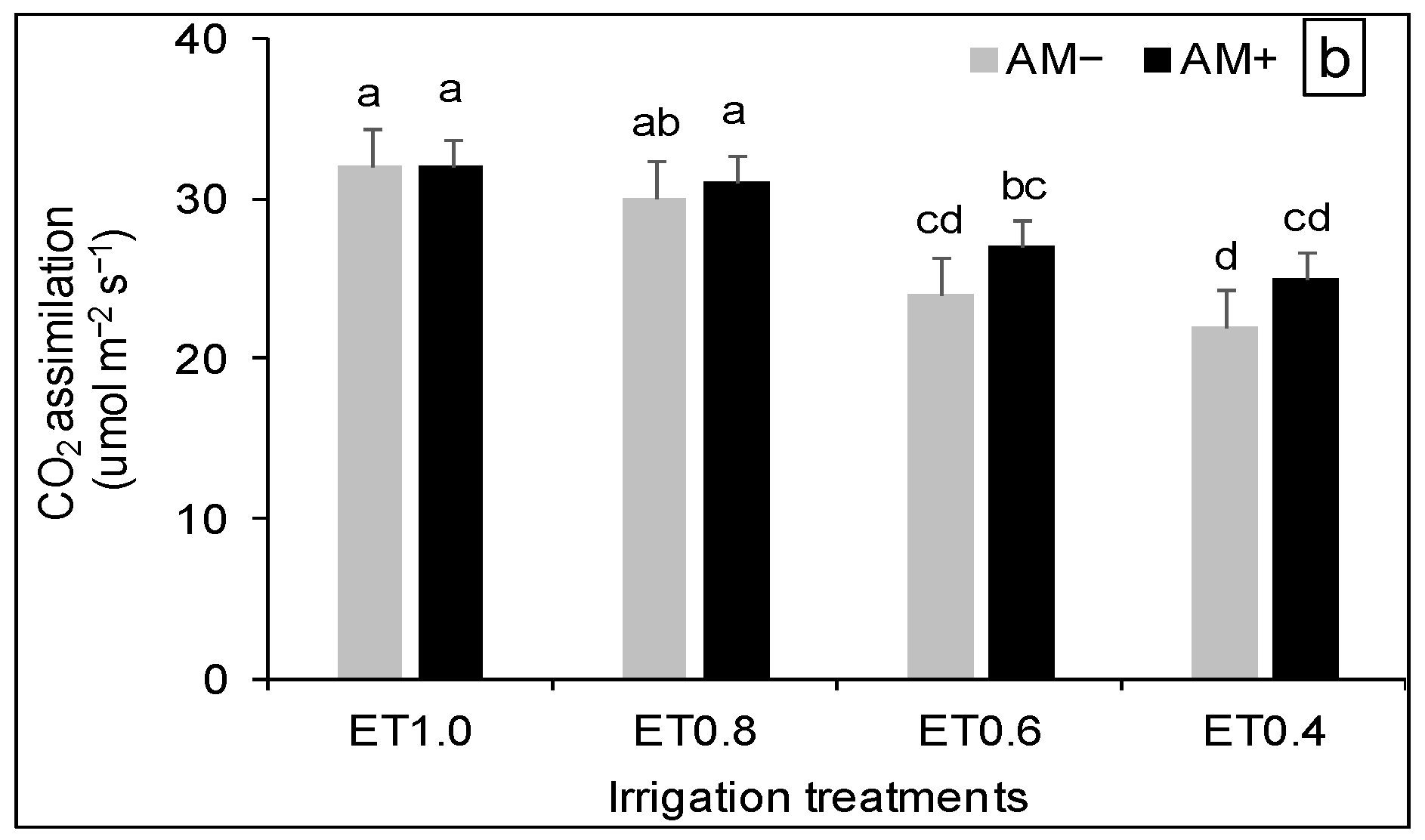
3.3. Plant Physiological Response
3.4. Plant N and P Uptake
3.5. Soil Fertility
4. Discussion
4.1. Yield Response and Plant Water Relations
4.2. Photosynthesis, Nutrient Status, and Soil OC
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AM | arbuscular mycorrhizal; |
| AMF | arbuscular mycorrhizal fungi; |
| AM+ | inoculated; |
| AM− | non-inoculated; |
| WUE | water use efficiency; |
| ET | evopotranspiration; |
| ET0 | evapotranspiration; |
| Tmax | average monthly maximum temperature; |
| Tmin | and average monthly minimum temperature; |
| Ym | reference maximum yield; |
| Ya 1 | actual yield; |
| Kss | biomass response factor; |
| Ky | yield response factor; |
| SS | total dry biomass; |
| gs | stomatal conductance; |
| Pn | photosynthetic rate; |
| Ft | total nutrient uptake; |
| F0 | total nutrient uptake under unfertilized treatment; |
| F | total amount of nutrient applied during the whole season; |
| DAT | days after transplantation; |
| HI | harvest index; |
| OC | organic carbon. |
References
- Elliott, J.; Deryng, D.; Müller, C.; Frieler, K.; Konzmann, M.; Gerten, D.; Glotter, M.; Flörke, M.; Wada, Y.; Best, N.; et al. Constraints and potentials of future irrigation water availability on agricultural production under climate change. Proc. Natl. Acad. Sci. USA 2014, 111, 3239–3244. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Lozano, J.M.; Porcel, R.; Azcón, R.; Bárzana, G.; Aroca, R. Contribution of arbuscular mycorrhizal symbiosis to plant drought tolerance: State of the art. In Responses to Drought Stress: From Morphological to Molecular Features; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 335–362. [Google Scholar]
- Costa, J.M.; Ortuño, M.F.; Chaves, M.M. Deficit irrigation as a strategy to save water: Physiology and potential application to horticulture. J. Integr. Plant Biol. 2007, 49, 1421–1434. [Google Scholar] [CrossRef]
- Badr, M.A.; El-Tohamy, W.A.; Zaghloul, A.M. Yield and water use efficiency of potato grown under different irrigation and nitrogen levels in an arid region. Agric. Water Manag. 2012, 110, 9–15. [Google Scholar] [CrossRef]
- Bardgett, R.D.; van der Putten, W.H. Belowground biodiversity and ecosystem functioning. Nature 2014, 515, 505–511. [Google Scholar] [CrossRef]
- Mohan, J.E.; Cowden, C.C.; Baas, P.; Dawadi, A.; Frankson, P.T.; Helmick, K.; Hughes, E.; Khan, S.; Lang, A.; Machmuller, M.; et al. Mycorrhizal fungi mediation of terrestrial ecosystem responses to global change: Mini-review. Fungal Ecol. 2014, 10, 3–19. [Google Scholar] [CrossRef]
- Badr, M.A.; Abou Hussein, S.D.; El-Tohamy, W.A.; Gruda, N. Efficiency of subsurface drip irrigation for potato production under different dry stress conditions. Healthy Plants 2010, 62, 63–70. [Google Scholar] [CrossRef]
- Badr, M.A.; El-Tohamy, W.A.; Hussein, S.D.A.; Gruda, N. Tomato yield, physiological response, water and nitrogen use efficiency under deficit and partial root zone drying irrigation in an arid region. J. Appl. Bot. Food Qual. 2018, 91, 332–340. [Google Scholar] [CrossRef]
- Augé, R. Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 2001, 11, 3–42. [Google Scholar] [CrossRef]
- Baum, C.; El-Tohamy, W.; Gruda, N. Increasing the productivity and product quality of vegetable crops using arbuscular mycorrhizal fungi: A review. Sci. Hortic. 2015, 187, 131–141. [Google Scholar] [CrossRef]
- Augé, R.M.; Toler, H.D.; Moore, J.L.; Cho, K.; Saxton, A.M. Comparing contributions of soil versus root colonization to variations in stomatal behavior and soil drying in mycorrhizal Sorghum bicolor and Cucurbita pepo. J. Plant Physiol. 2007, 164, 1289–1299. [Google Scholar] [CrossRef]
- Michalis, O.; Ioannides, I.M.; Ehaliotis, C. Mycorrhizal inoculation affects arbuscular mycorrhizal diversity in watermelon roots but leads to improved colonization and plant response under water stress only. Appl. Soil Ecol. 2013, 63, 112–119. [Google Scholar] [CrossRef]
- Aroca, R.; Vernieri, P.; Ruiz-Lozano, J.M. Mycorrhizal and non-mycorrhizal Lactuca sativa plants exhibit contrasting responses to exogenous ABA during drought stress and recovery. J. Exp. Bot. 2008, 59, 2029–2041. [Google Scholar] [CrossRef][Green Version]
- Kaschuk, G.; Kuyper, T.W.; Leffelaar, P.A.; Hungria, M.; Giller, K.E. Are the rates of photosynthesis stimulated by the carbon sink strength of rhizobial and arbuscular mycorrhizal symbioses? Soil Biol. Biochem. 2009, 41, 1233–1244. [Google Scholar] [CrossRef]
- Birhane, E.; Sterck, F.J.; Fetene, M.; Bongers, F.; Kuyper, T.W. Arbuscular mycorrhizal fungi enhance photosynthesis, water use efficiency, and growth of frankincense seedlings under pulsed water availability conditions. Oecologia 2012, 169, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Augé, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza 2015, 25, 13–24. [Google Scholar] [CrossRef]
- Lazcano, C.; Barrios-Masias, F.H.; Jackson, L.E. Arbuscular mycorrhizal effects on plant water relations and soil greenhouse gas emissions under changing moisture regimes. Soil Biol. Biochem. 2014, 74, 184–192. [Google Scholar] [CrossRef]
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C.; et al. Solutions for a cultivated planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef]
- Adesemoye, A.O.; Torbert, H.A.; Kloepper, J.W. Plant growth-promoting rhizobacteria allow reduced application rates of chemical fertilizers. Microb. Ecol. 2009, 58, 921–929. [Google Scholar] [CrossRef]
- Hungria, M.; Nogueira, M.A.; Araujo, R.S. Co-inoculation of soybeans and common beans with rhizobia and azospirilla: Strategies to improve sustainability. Biol. Fertil. Soils 2013, 49, 791–801. [Google Scholar] [CrossRef]
- Suriyagoda, L.D.B.; Ryan, M.H.; Renton, M.; Lambers, H. Plant responses to limited moisture and phosphorus availability: A meta-analysis. Adv. Agron. 2014, 124, 143–200. [Google Scholar]
- Neumann, E.; George, E. Colonisation with the arbuscular mycorrhizal fungus Glomus mosseae (Nicol. & Gerd.) enhanced phosphorus uptake from dry soil in Sorghum bicolor (L.). Plant Soil 2004, 261, 245–255. [Google Scholar]
- George, E.; Marschner, H.; Jakobsen, I. Role of arbuscular mycorrhizal fungi in uptake of phosphorus and nitrogen from soil. Crit. Rev. Biotechnol. 1995, 15, 257–270. [Google Scholar] [CrossRef]
- Hetrick, B.A.D.; Wilson, G.W.T.; Todd, T.C. Mycorrhizal response in wheat cultivars: Relationship to phosphorus. Can. J. Bot. 1996, 74, 19–25. [Google Scholar] [CrossRef]
- Subramanian, K.S.; Santhanakrishnan, P.; Balasubramanian, P. Responses of field grown tomato plants to arbuscular mycorrhizal fungal colonization under varying intensities of drought stress. Sci. Hortic. 2006, 107, 245–253. [Google Scholar] [CrossRef]
- Bowles, T.M.; Barrios-Masias, F.H.; Carlisle, E.A.; Cavagnaro, T.R.; Jackson, L.E. Effects of arbuscular mycorrhizae on tomato yield, nutrient uptake, water relations, and soil carbon dynamics under deficit irrigation in field conditions. Sci. Total Environ. 2016, 566, 1223–1234. [Google Scholar] [CrossRef]
- Gruda, N.; Bisbis, M.B.; Tanny, J. Impacts of protected vegetable cultivation on climate change and adaptation strategies for cleaner production—A review. J. Clean. Prod. 2019, 225, 324–339. [Google Scholar] [CrossRef]
- Gruda, N.; Bisbis, M.B.; Tanny, J. Influence of climate change on protected cultivation: Impacts and sustainable adaptation strategies—A review. J. Clean. Prod. 2019, 225, 481–495. [Google Scholar] [CrossRef]
- FAO. FAOSTAT. Food and Agriculture Organization of the United Nations. 2016. Available online: http://faostat.fao.org/statistics (accessed on 10 August 2020).
- Karam, F.; Sabiha, R.; Skaf, S.; Breidy, J.; Rouphael, Y.; Balendonck, J. Yield and water use of eggplants (Solanum Melongena L.) under full and deficit irrigation regimes. Agric. Water Manage. 2011, 98, 1307–1316. [Google Scholar] [CrossRef]
- Lovelli, S.; Perniola, M.; Ferrara, A.; Di Tommaso, T. Yield response factor to water (Ky) and water use efficiency of Carthamus tinctorius L. and Solanum melongena L. Agric. Water Manag. 2007, 92, 73–80. [Google Scholar] [CrossRef]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration. Guidelines for Computing Crop Water Requirements. FAO Irrigation and Drainage. Paper No. 56; FAO: Rome, Italy, 1998; p. 300. [Google Scholar]
- Stewart, J.I.; Cuenca, R.H.; Pruit, W.O.; Hagan, R.M.; Tosso, J. Determination and Utilization of Water Production Functions for Principal California Crops. W-67 California Contribution Project Report; University of California: Davis, CA, USA, 1977. [Google Scholar]
- Jones, J.B.J.; Case, V.W. Sampling, Handling and Analyzing Plant Tissue Samples; Westerman, R.L., Ed.; Soil Testing and Plant Analysis: Madison, WI, USA, 1991; pp. 289–427. [Google Scholar]
- Eaton, A.D.; Clesceri, L.S.; Greenberg, A.E. Standard Methods for the Examination of Water and Wastewater, 19th ed.; American Public Health Association: Washington, DC, USA, 1995. [Google Scholar]
- Wu, J.; Joergensen, R.G.; Pommerening, B.; Chaussod, R.; Brookes, P.C. Measurement of soil microbial biomass C by fumigation-extraction: An automated procedure. Soil Biol. Biochem. 1990, 22, 1167–1169. [Google Scholar] [CrossRef]
- Bathke, G.R.; Gassel, D.K.; Hargrove, W.L.; Porter, P.M. Modification of soil physical properties and root growth response. Soil Sci. 1992, 154, 316–329. [Google Scholar] [CrossRef]
- Masle, J.; Passiour, J.B. The effect of soil strength on the growth of young wheat plants. Aust. J. Plant Physiol. 1987, 14, 643–656. [Google Scholar] [CrossRef]
- Bryla, D.R.; Koide, R.T. Regulation of reproduction in wild and cultivated Lycopersicon esculentum Mill. by vesicular-arbuscular mycorrhizal infection. Oecologia 1990, 84, 74–81. [Google Scholar] [CrossRef]
- Poulton, J.L.; Bryla, D.; Koide, R.T.; Stephenson, A.G. Mycorrhizal infection and high soil phosphorus improve vegetative growth and the female and male functions in tomato. New Phytol. 2002, 154, 255–264. [Google Scholar] [CrossRef]
- Conversa, G.; Lazzizera, C.; Bonasia, A.; Elia, A. Yield and phosphorus uptake of a processing tomato crop grown at different phosphorus levels in a calcareous soil as affected by mycorrhizal inoculation under field conditions. Biol. Fertil. Soils 2013, 49, 691–703. [Google Scholar] [CrossRef]
- Aujla, M.S.; Thind, H.S.; Buttar, G.S. Fruit yield and water use efficiency of eggplant (Solanum melongema L.) as influenced by different quantities of nitrogen and water applied through drip and furrow irrigation. Sci. Hortic. 2007, 112, 142–148. [Google Scholar] [CrossRef]
- Colak, Y.B.; Yazar, A.; Çolak, I.; Akça, H.; Duraktekin, G. Evaluation of crop water stress index (CWSI) for eggplant under varying irrigation regimes using surface and subsurface drip systems. Agric. Sci. Procedia 2015, 4, 372–382. [Google Scholar] [CrossRef]
- Kothari, S.K.; Marschner, H.; George, E. Effect of VA fungi and rhizosphere organisms root and shoot morphology, growth and water relations in maize. New Phytol. 1990, 116, 303–311. [Google Scholar] [CrossRef]
- Augé, R.M.; Duan, X.; Ebel, R.C.; Stodola, A.J.W. Nonhydraulic signaling of soil drying in mycorrhizal maize. Planta 1994, 193, 74–82. [Google Scholar] [CrossRef]
- Barrios-Masias, F.H.; Knipfer, T.; McElrone, A. Differential responses of grapevine rootstocks to water stress are associated with adjustments in fine root hydraulic physiology and suberization. J. Exp. Bot. 2015, 66, 6069–6078. [Google Scholar] [CrossRef]
- Cavagnaro, T.R.; Jackson, L.E.; Six, J.; Ferris, H.; Goyal, S.; Asami, D.; Scow, K.M. Arbuscular mycorrhizas, microbial communities, nutrient availability, and soil aggregates in organic tomato production. Plant Soil 2006, 282, 209–225. [Google Scholar] [CrossRef]
- Patanè, C.; Tringali, S.; Sortinob, O. Effects of deficit irrigation on biomass, yield, water productivity and fruit quality of processing tomato under semi-arid Mediterranean climate conditions. Sci. Hortic. 2011, 129, 590–596. [Google Scholar] [CrossRef]
- Cheng, W. Priming effect: Its functional relationships with microbial turnover, Rhizosphere evapotranspiration, and C–N budgets. Soil Biol. Biochem. 2009, 41, 1795–1801. [Google Scholar] [CrossRef]
- Richards, R.A. Selectable traits to increase crop photosynthesis and yield of grain crops. J. Exp. Bot. 2000, 51, 447–458. [Google Scholar] [CrossRef]
- Chitarra, W.; Pagliarani, C.; Maserti, B.; Lumini, E.; Siciliano, I.; Cascone, P.; Schubert, A.; Gambino, G.; Balestrini, R.; Guerrieri, E. Insights on the Impact of Arbuscular Mycorrhizal Symbiosis on Tomato Tolerance to Water Stress. Plant Physiol. 2016, 171, 1009–1023. [Google Scholar] [CrossRef]
- Duan, X.; Neuman, D.; Reiber, J.; Green, C.; Saxton, A.; Augé, R. Mycorrhizal influence on hydraulic and hormonal factors implicated in the control of stomatal conductance during drought. J. Exp. Bot. 1996, 47, 1541–1550. [Google Scholar] [CrossRef]
- Subramanian, K.S.; Charest, C. Acquisition of N by external hyphae of an arbuscular mycorrhizal fungus and its impact on physiological responses in maize under drought-stressed and well-watered conditions. Mycorrhiza 1999, 9, 69–75. [Google Scholar] [CrossRef]
- Tei, F.; Benincasa, P.; Guiducci, M. Critical nitrogen concentration in processing tomato. Eur. J. Agron. 2002, 18, 45–55. [Google Scholar] [CrossRef]
- Liu, C.; Ravnskov, S.; Liu, F.; Rubaek, G.H. Arbuscular mycorrhizal fungi alleviate abiotic stresses in potato plants caused by low phosphorus and deficit irrigation/partial root-zone drying. J. Agric. Sci. 2018, 156, 46–58. [Google Scholar] [CrossRef]
- Cramer, M.; Hawkins, H.; Verboom, G. The importance of nutritional regulation of plant water flux. Oecologia 2009, 161, 15–24. [Google Scholar] [CrossRef]
- Javaid, A. Arbuscular mycorrhizal mediated nutrition in plants. J. Plant Nutr. 2009, 32, 1595–1916. [Google Scholar] [CrossRef]
| Month | Tmax (°C) | Tmin (°C) | Relative Humidity (%) | Rain Fall (mm) | ET0 (mm d−1) | Wind Speed (km h−1) |
|---|---|---|---|---|---|---|
| 2017 | ||||||
| March | 23.6 | 10.2 | 46 | 7.4 | 3.8 | 11.67 |
| April | 28.3 | 12.5 | 37 | 3.9 | 4.7 | 11.30 |
| May | 32.2 | 15.9 | 38 | 4.5 | 5.9 | 10.56 |
| June | 34.5 | 18.7 | 42 | 0 | 6.7 | 10.56 |
| 2018 | ||||||
| March | 23.8 | 10.1 | 45 | 6.2 | 3.7 | 11.85 |
| April | 28.3 | 12.6 | 36 | 2.4 | 4.7 | 11.65 |
| May | 32.4 | 15.9 | 38 | 3.6 | 5.9 | 10.56 |
| June | 36.2 | 18.7 | 43 | 0 | 6.7 | 10.19 |
| Year | AM z | Irrigation Levels | ||||
|---|---|---|---|---|---|---|
| ET1.0 | ET0.8 | ET0.6 | ET0.4 | Mean | ||
| 2017 | Fruit yield (t ha−1) | |||||
| AM− | 42.75 b,y | 32.82 c | 23.75 d | 12.32 e | 27.91 B | |
| AM+ | 54.25 a | 41.97 b | 30.52 c | 15.94 e | 35.67 A | |
| Mean | 48.50 A | 37.40 B | 27.14 C | 14.13 D | ||
| Total dry biomass (t ha−1) | ||||||
| AM− | 7.35 c | 6.28 d | 5.02 e | 3.34 g | 5.54 B | |
| AM+ | 9.33 a | 8.03 b | 6.68 d | 4.32 f | 7.09 A | |
| Mean | 8.34 A | 7.16 B | 5.94 C | 3.83 D | ||
| Fruit number (g plant−1) | ||||||
| AM− | 23.64 b | 17. 69 c | 14.10 d | 7.33 e | 15.69 B | |
| AM+ | 28.46 a | 23.90 b | 18.97 c | 9.09 e | 20.10 A | |
| Mean | 26.05 A | 20.80 B | 16.53 C | 8.21 D | ||
| Fruit weight (g plant−1) | ||||||
| AM− | 72.33 a,b | 74.20 a,b | 67.39 a,b | 67.20 a,b | 70.28 A | |
| AM+ | 76.26 a | 70.23 a,b | 64.35 b | 70.18 a,b | 70.25 A | |
| Mean | 74.29 A | 72.22 A | 65.87 B | 68.69 B | ||
| 2018 | Fruit yield (t ha−1) | |||||
| AM− | 45.12 b | 35.18 c | 25.12 d | 13.32 e | 29.69 B | |
| AM+ | 57.32 a | 44.27 b | 32.54 c | 17.38 e | 37.88 A | |
| Mean | 51.22 A | 40.23 B | 28.83 C | 15.35 D | ||
| Total dry biomass (t ha−1) | ||||||
| AM− | 7.76 b,c | 6.73 d | 5.50 e | 3.61 g | 5.90 B | |
| AM+ | 9.86 a | 8.47 b | 7.12 c,d | 4.71 f | 7.54 A | |
| Mean | 8.81 A | 7.60 B | 6.31 C | 4.16 D | ||
| Fruit number (g plant−1) | ||||||
| AM− | 24.08 b | 19.18 c | 13.36 d | 7.36 e | 16.00 B | |
| AM+ | 29.23 a | 25.51 b | 18.52 c | 9.39 e | 20.66 A | |
| Mean | 26.66 A | 22.53 B | 15.94 C | 8.38 D | ||
| Fruit weight (g plant−1) | ||||||
| AM− | 74.94 a,b | 73.37 a,b | 75.22 a,b | 72.35 a,b | 73.97 A | |
| AM+ | 78.44 a | 69.41 b | 70.27 b | 74.07 a,b | 73.05 A | |
| Mean | 76.69 A | 71.39 B | 72.75 B | 73.21 B | ||
| Year | AM z | Irrigation Levels | ||||
|---|---|---|---|---|---|---|
| ET1.0 | ET0.8 | ET0.6 | ET0.4 | Mean | ||
| 2017 | AM− | 86 b,y | 82 b | 78 b | 59 c | 76 B |
| AM+ | 109 a | 105 a | 100 a | 77 b | 98 A | |
| Mean | 98 A | 93 A | 89 A | 68 B | ||
| 2018 | AM− | 89b b | 86 b | 81 b | 63 c | 80 B |
| AM+ | 113 a | 108 a | 105 a | 83 b | 102 A | |
| Mean | 101 A | 97 A | 93 A | 73 B | ||
| Year | Treatment z | N Uptake (kg ha−1) | N Recovery % | P Uptake (kg ha−1) | P Recovery % | N:P Ratio | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Fruit | Shoot | Total | Fruit | Shoot | Total | |||||
| 2017 | ET1.0 AM− | 75 b,y | 69 c | 144 c | 44 | 12.0 c | 9.8 c | 21.8 c | 16 | 6.6 |
| AM+ | 95 a | 87 a | 183 a | 57 | 17.4 a | 15.0 a | 32.3 a | 24 | 5.7 | |
| ET0.8 AM− | 63 c | 60 d | 123 d | 37 | 10.0 d | 7.9 d | 17.9 d | 12 | 6.9 | |
| AM+ | 80 b | 77 b | 157 b | 48 | 14.6 b | 12.3 b | 26.9 b | 20 | 5.8 | |
| ET0.6 AM− | 47 d | 48 e | 95 e | 28 | 7.0 e | 6.3 e | 13.3 e | 9 | 7.1 | |
| AM+ | 60 c | 66 c,d | 126 d | 38 | 9.8 d | 8.9 c | 18.7 c,d | 13 | 6.7 | |
| ET0.4 AM− | 30 f | 31 f | 61 g | 16 | 4.2 f | 4.1 f | 8.3 f | 4 | 7.4 | |
| AM+ | 38 e | 43 e | 82 f | 23 | 6.3 e | 5.8 e | 12.1 e | 8 | 6.7 | |
| 2018 | ET1.0 AM− | 78 b | 73 c | 150 c | 46 | 12.6 c | 10.4 d | 23.0 c | 17 | 6.5 |
| AM+ | 99 a | 92 a | 191 a | 60 | 18.3 a | 15.8 a | 34.2 a | 26 | 5.6 | |
| ET0.8 AM− | 67 c | 64 c | 132 d | 40 | 10.7 d | 9.2 c | 19.9 d | 14 | 6.6 | |
| AM+ | 85 b | 81 b | 166 b | 51 | 15.4 b | 13.9 b | 29.3 b | 22 | 5.7 | |
| ET0.6 AM− | 50 d | 51 d | 101 e | 30 | 7.4 e | 6.4 d | 13.8 e | 9 | 7.3 | |
| AM+ | 64 c | 70 c | 135 d | 41 | 10.5 d | 10.3 c | 21.7 c,d | 15 | 6.5 | |
| ET0.4 AM− | 32 f | 34 e | 66 f | 18 | 4.5 f | 4.2 e | 8.7 f | 5 | 7.6 | |
| AM+ | 42 e | 47 d | 89 e | 26 | 6.8 e | 6.9 d | 13.7 e | 9 | 6.5 | |
| Year | Treatments z | Organic C (%) | Available Nutrients (mg kg−1 soil) | ||
|---|---|---|---|---|---|
| N | P | K | |||
| 2017 | AM− | 0.27 a,y | 15 a | 10 a | 40 a |
| AM+ | 0.34 b | 18 b | 13 b | 46 b | |
| 2018 | AM− | 0.29 a | 19 a | 10 a | 42 a |
| AM+ | 0.38 b | 24 b | 15 b | 49 b | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badr, M.A.; El-Tohamy, W.A.; Abou-Hussein, S.D.; Gruda, N.S. Deficit Irrigation and Arbuscular Mycorrhiza as a Water-Saving Strategy for Eggplant Production. Horticulturae 2020, 6, 45. https://doi.org/10.3390/horticulturae6030045
Badr MA, El-Tohamy WA, Abou-Hussein SD, Gruda NS. Deficit Irrigation and Arbuscular Mycorrhiza as a Water-Saving Strategy for Eggplant Production. Horticulturae. 2020; 6(3):45. https://doi.org/10.3390/horticulturae6030045
Chicago/Turabian StyleBadr, M. A., W. A. El-Tohamy, S. D. Abou-Hussein, and N. S. Gruda. 2020. "Deficit Irrigation and Arbuscular Mycorrhiza as a Water-Saving Strategy for Eggplant Production" Horticulturae 6, no. 3: 45. https://doi.org/10.3390/horticulturae6030045
APA StyleBadr, M. A., El-Tohamy, W. A., Abou-Hussein, S. D., & Gruda, N. S. (2020). Deficit Irrigation and Arbuscular Mycorrhiza as a Water-Saving Strategy for Eggplant Production. Horticulturae, 6(3), 45. https://doi.org/10.3390/horticulturae6030045






