1. Introduction
Feed can account for over 50% of production costs in an aquaculture system [
1]. Consequently, it is important to effectively convert feed into sellable products. Fish are among the most efficiently cultured animals in regard to feed conversion, but there is still a considerable amount of wasted nutrients associated with fish [
2,
3]. Recirculating aquaculture systems (RASs) improve water and space utilization over traditional pond-based systems, but traditional RASs do little to improve the nutrient use efficiency (NUE) of a system.
Biofloc technology (BFT) is a form of RAS that does not use traditional biofilters. BFT relies on the constant mixing of suspended solids in the water column. Solids in suspension in BFT-cultured water provide a surface area for heterotrophic and autotrophic bacterial growth. Most BFTs are operated where nitrogenous waste is primarily handled through mineralization by utilizing heterotrophic bacteria. Nitrogenous waste (primarily ammonia) is assimilated into microbial protein, converting N into a nontoxic form [
4,
5]. This technique is enhanced by increasing the C:N ratio of food, adding highly available carbon sources, or by lowering the protein content in feed [
6,
7]. BFT improves the feed conversion ratio (FCR) over clearwater systems, which enhances NUE of the system [
7]. The BFT system contains high concentrations of settleable solids that include microbial flocs, uneaten feed, and fecal waste.
Hydroponic vegetable production lends itself to integration into RASs, improving NUE. The integration of RASs with hydroponic vegetable production is commonly referred to as aquaponics [
8]. Aquaponic systems improve NUE, decrease water consumption, and improve water quality over conventional RASs [
9,
10,
11,
12]. Quillere et al. [
13] reported that 60% of applied nitrogen was recovered, with 28% being assimilated into plant biomass and 31% being assimilated into fish biomass when fish production was integrated with hydroponic tomato (
Solanum lycopersicum L) production.
Aquaponic research has primarily involved leafy greens [
9,
10,
11,
14,
15] and tomato [
16,
17,
18,
19,
20]. Savidov et al. [
21] evaluated 24 different plant species grown in an aquaponic system, demonstrating the variety of crops that can be grown aquaponically.
Little research has addressed integrating drip-irrigated plant production systems used in the greenhouse vegetable industry, with RASs. Soilless systems utilize highly porous substrates, allowing growers to manipulate nutrients in the root zone with frequent short irrigation cycles. Clogging of the micro-orifices associated with microirrigation with fish waste is a concern with aquaponics. High concentrations of settleable solids associated with BFT have had limited research on its integration with substrate-based growing systems. The purpose of this research is to evaluate the integration of BFT aquaculture effluent (AE) as a nutrient solution for greenhouse cherry tomato using a substrate-based hydroponic system.
2. Materials and Methods
Facilities used in this research consisted of 2 commercial-size greenhouses located at the E.W. Shell Fisheries Center, North Auburn Unit, approximately 10 km north of Auburn, Alabama, USA (32.649171° N, −85.486725° E). The fish culture system was housed in a 267.6 m
2 double-polyethylene covered greenhouse (9.1 × 29.2 m) with an east to west orientation and consisted of 2 rectangular tanks (1.2 × 3 × 26.8 m), each with an average volume of 100 m
3, operated as a biofloc system [
5]. A 1.9 m
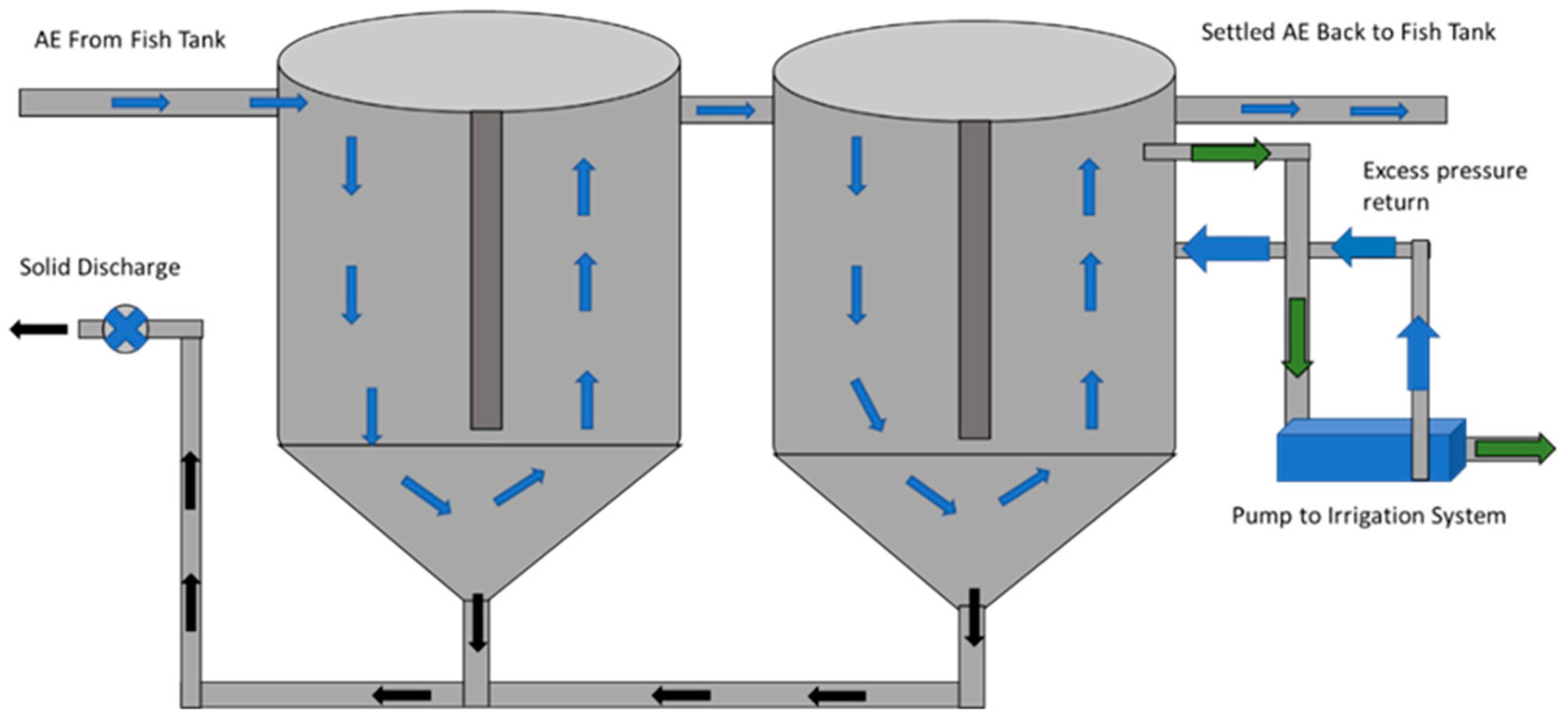
3 cone-bottomed clarifier (30% slope), adjacent to the greenhouse, was used to reduce suspended solids concentration from the system (
Figure 1). Water flowed through the clarifier at an approximate flow rate of 18.9 L∙min
−1 and entered a 1.1 m
3 cone-bottomed irrigation sump of a similar design before re-entering the fish production tank. Both vessels had an uninterrupted and constant flow of water, driven by airlift pumps. The clarifier and irrigation sump were flushed of collected solids twice daily.
The 267.5 m2 (9.1 × 29.2 m) plant greenhouse was covered with double-layered polyethylene sheeting with a north to south orientation. The plant greenhouse was outfitted for soilless vine crop production, with a steel cable trellis system running the length of the greenhouse and cables suspended approximately 2.1 m above the greenhouse floor. Two cables were suspended above each row, approximately 0.1 m from the row center. Each row was 1.5 m apart, and the plant-growing containers were spaced 40.6 cm apart within rows. Both greenhouses were equipped with environmental controls for year-round production.
For the purposes of this study, the south-facing tank was stocked with 3000 Nile tilapia (
Oreochromis niloticus; 157 g/fish), 40 fish/m
3, on 29 August 2012. Fish were hand-fed a 36% protein floating feed, with 6.0% crude fat and 3.5% crude fiber (Cargill
®, Franklinton, LA, USA), at 13% body weight/day twice daily (8:30 a.m. and 4:00 p.m.) for approximately 20 min. Calcium-hydroxide (Ca(OH)
2) was applied after each feeding to maintain a targeted pH of 6.8 to 7.0 [
8]. Oxygen was supplied by two 1118.5 W (1.5 horsepower) regenerative blowers feeding the air-diffusing tubing that was suspended around the walls of the fish tank. Dissolved oxygen and temperature of the fish culture water were recorded twice daily (YSI 550A, YSI Inc., Yellow Springs, OH, USA). Fish were harvested 150 days after stocking (24 January 2013). The water for fish production was sourced from a reservoir that is fed by the surrounding watershed.
To evaluate the yields of tomatoes grown with AE against conventionally grown plants, a commercially available hydroponic fertilizer, Bag Culture Tomato Special 3-13-29, (3.0% N; 13.0% P; 29.0% K; 0.1% B, Cu, and Mn; 0.34% Fe; 5.4% Mg; 0.01% Mo; 11.0% S; 0.045% Zn; Total Grow
TM, Winnsboro, LA, USA) and greenhouse-grade calcium nitrate (15.5N-0P-0K) were used for the control treatment. Plants were irrigated and fertilized at rates following recommendations by Hanna [
22] (
Table 1). The irrigation water source for CF-grown plants was supplied by the local municipal water source. The cherry tomato (
S.
lycopersicum var.
cerasiforme) cvs. Favorita and Goldita (Paramount Seeds, Stuart, FL, USA) were used. On 1 October 2012, 8-week-old tomato transplants were transplanted into 11 L Bato pots (Bato Plastics B.V. Zevenbergon, The Netherlands) filled with commercial grade perlite. Two tomatoes were planted in each pot, resulting in a plant density of 3.2 plants/m
2, and placed in the high wire trellis system previously described. Each pot served as a single experimental unit.
This study consisted of two treatments: aquaculture effluent (AE) and commercial fertilizer (CF). AE was pumped from the irrigation sump adjacent to the fish greenhouse. The tomato varieties were evaluated simultaneously but in separate experiments. Plants were arranged in a completely randomized design, with 10 replicates for both treatments of “Favorita”. “Goldita” had 9 replicates of AE-grown plants and 11 replicates of CF-grown plants. Tomato harvest began 61 days after transplanting (DAT), and fish harvest occurred at 117 DAT of the tomato crop. Tomato harvest continued daily until the termination of the study (157 DAT). Tomato fruits were harvested based on ripeness, with fruit color used as an indicator. Tissue samples were taken at the final harvest.
The irrigation sump was used to access clarified water for the drip irrigation system for the soilless culture of cherry tomatoes in the adjacent greenhouse. A 1118.5 W (1.5 horsepower) irrigation pump was plumbed to the previously described irrigation sump, where it drew AE water from ½ the depth of the tank and delivered pressurized settled water at 206.8 kPa (30 psi) to the tomato irrigation system (
Figure 1). The pressure was regulated by bleeding excess pressure back into the irrigation sump. Both treatments were delivered to plants using a clog-resistant pressure-compensated emitter (Bowsmith Nonstop Emitter, Bowsmith Inc. Exeter, CA, USA) at a flow rate of 3.785 L∙h
−1. Each container was outfitted with two emitters. Plants grown with AE received water directly from the irrigation sump. Plants grown with CF received water and fertilizer through 2 fertilizer injectors (Model DM11, Dosatron, Clearwater, FL, USA). This allowed separate, but simultaneous, injections of the hydroponic fertilizer blend and calcium nitrate. Solenoid valves responsible for delivering the respective treatments were wired in tangent so that both treatments were applied at the same time.
A three-liter sample of the fish culture water and AE from the irrigation sump was collected and analyzed once weekly to characterize total ammonia nitrogen (TAN), nitrate, nitrite, potassium, and orthophosphate of the water being used to irrigate the tomato plants receiving AE. Each sample was filtered using a 40-micron Whatman™ glass fiber filter (VWR International, Radnor, PA, USA). Standard curves were fit for TAN, nitrate–nitrogen, potassium, and orthophosphate on a GENESYS 20 visible spectrophotometer (Spectronic Unicam, Rochester, NY, USA). Nessler method 8038 (Hach Company, Loveland, CO, USA) was used to determine TAN; the ascorbic acid method 8048 (Hach Company, Loveland, CO, USA) was used to determine orthophosphate. Nitrate–nitrogen and potassium were analyzed using Cardy twin nitrate and potassium meters (Spectrum Technologies, Inc., Plainfield, IL, USA). Titration method 8329, using ethylenediamine–tetraacetic acid (Hach Company, Loveland, CO, USA), was used to determine calcium and magnesium. Total phosphorus and total nitrogen were determined through persulfate digestion [
23]. Digestates of nitrate and orthophosphate were determined using spectrophotometric screening and an ascorbic acid method [
23,
24,
25].
Plant tissue was collected at termination from the third leaf from the terminal shoot. Samples were analyzed at Brookside Laboratories (New Bremen, OH, USA) for N. Combustion analysis was used to determine total nitrogen [
26] using a Carlo Erba 1500 series analyzer (CE Elantech, Inc., Lakewood, NJ, USA). Minerals (P, K, Ca, Mg, S, B, Fe, Mn, Cu, Zn, and Al) were extracted using methods described by [
26] and analyzed with a Thermo 6500 duo ICP (Thermo Fisher Scientific, Inc., Waltham, MA, USA).
Settleable solids were determined for water contained in the fish tank and water returning to the tank from the irrigation sump using an adopted procedure of Standard Method 2540 F [
23]. Avliminech [
27] reported that floc particles became reanimated if left undisturbed for the 1-h recommended settling time in the procedure described in Standard Method 2450 due to gas bubbles forming. For purposes of this study, a 30-min period was used for settling. Suspended solids were measured according to Standard Method 2540 D [
23] using glass fiber filtration, followed by gravimetric analysis. The pH of AE of the samples was taken twice daily at approximately 9:00 a.m. and 4:00 p.m.
An analysis of variance was performed on all responses using a generalized linear model (PROC GLIMMIX in SAS version 9.4 SAS Institute, Cary, NC, USA). The experimental design for yield was a randomized design, with each variety analyzed separately. Due to a mistake at planting, the varieties were not randomized together; thus, no comparisons are made between varieties. The treatment design for yield was a two-way factorial of nutrient source and harvest date. Data recorded over harvest dates were analyzed as repeated measures using a heterogeneous compound symmetry covariance structure. Linear and quadratic trends were tested using qualitative/quantitative model regressions, and differences between fertility types least squares means were tested using F-tests. The experimental design for two yield totals (pre and post fish harvest) was a completely randomized design. Differences in yield between fertility types and between yield totals at fish harvest and termination were tested using F-tests. Where residual plots and a significant covariance test for homogeneity (H0 = homogeneity) indicated heterogeneous variance among treatments, a RANDOM statement with the GROUP option was used to correct heterogeneity.
Means comparisons for plant tissue were analyzed using a t-test (Proc TTest SAS, ver. 9.2, SAS Institute, Cary, NC.). If variances were equal, the pooled method was used to determine significance. If variances were unequal, the Satterthwaite method was used to determine significance.