Postharvest Dips of Persimmon Fruit in Gibberellic Acid: An Efficient Treatment to Improve Storability and Reduce Alternaria Black Spot
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pathogen, Host and Commercial Treatments
2.2. Preharvest Treatments of Persimmon Trees with GA3
2.3. Postharvest Treatments of GA3
2.4. Comparison of Pre- and Postharvest GA3 Treatments in Commercial Orchards
2.5. Persimmon Fruit Firmness Index and ABS Severity after Storage
2.6. Color Evaluation of Fruits
2.7. Statistical Methods
3. Results and Discussion
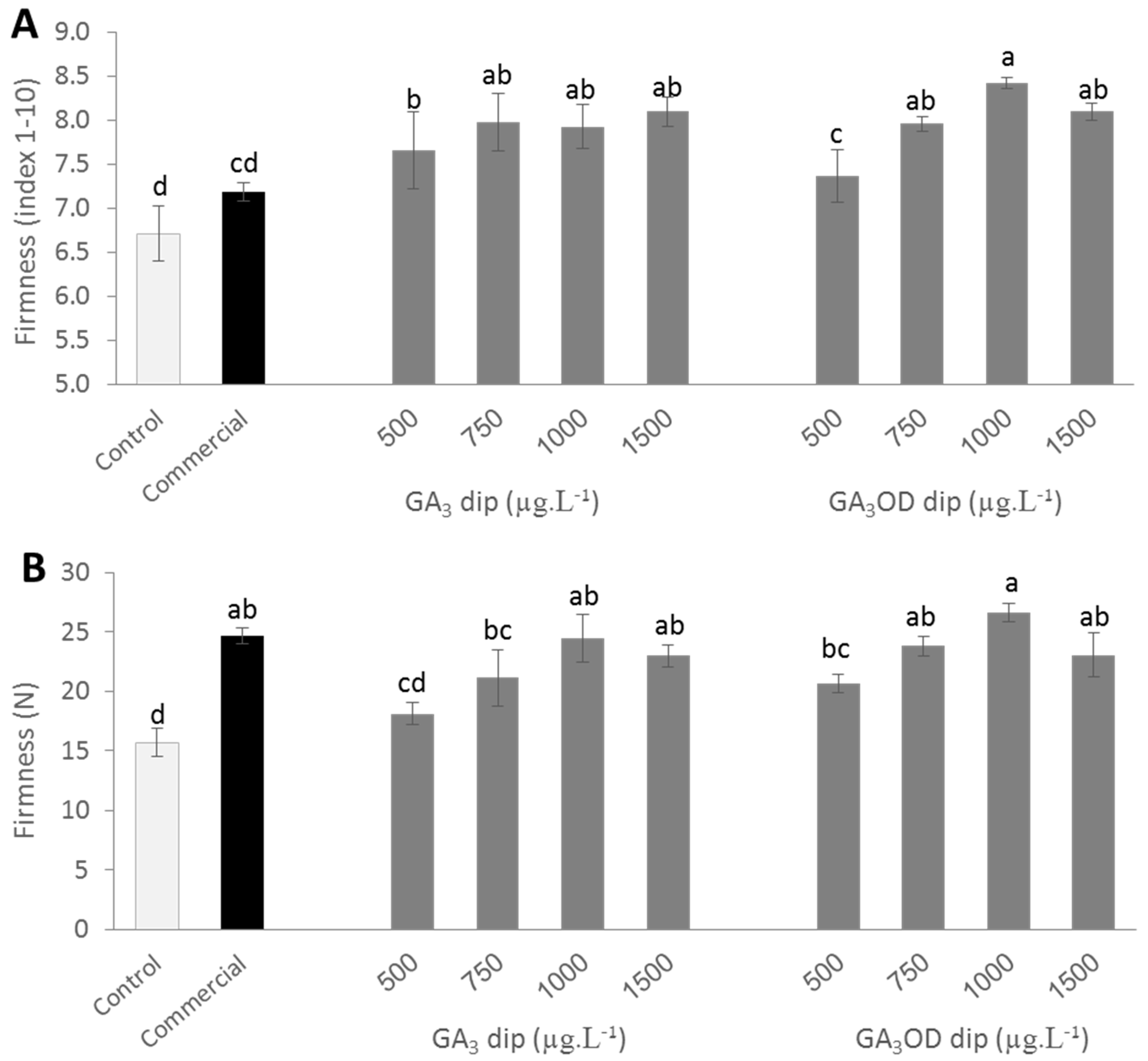
3.1. Effects of Pre- and Postharvest GA3 Treatments on the Firmness of Persimmon Fruit
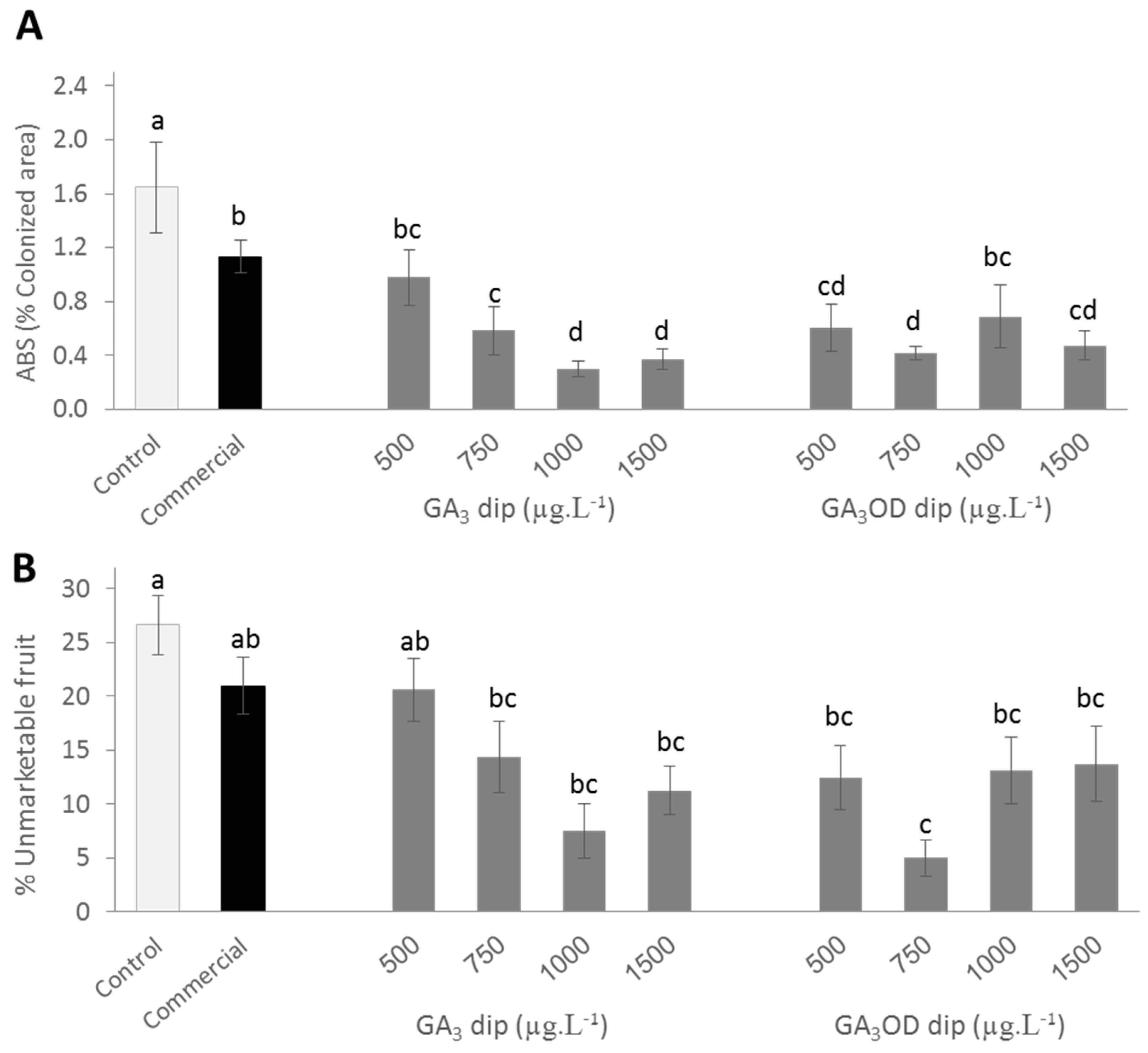
3.2. Effects of Pre and Postharvest Treatment with GA3 on ABS Development of Persimmon Fruit
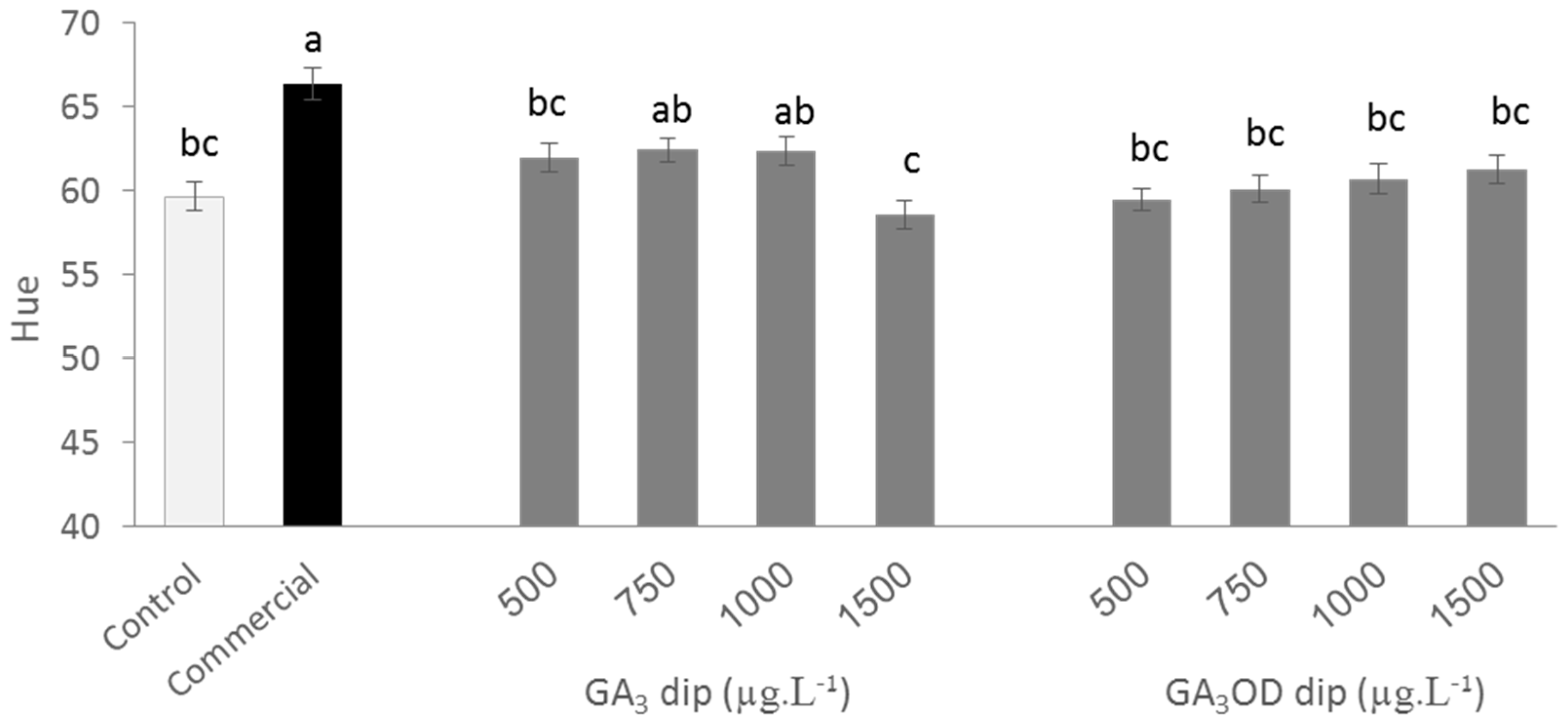
3.3. Effects of Pre and Postharvest Treatment with GA3 on Color Developmnet of Persimmon Fruit
3.4. Effect Postharvest GA3 Treatment under Commercial Conditions in Two Orchards
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ben-Arie, R.; Saks, Y.; Sonego, L.; Frank, A. Cell wall metabolism in gibberellin-treated persimmon fruits. Plant Growth Regul. 1996, 19, 25–33. [Google Scholar] [CrossRef]
- Ben-Arie, R.; Zhou, H.W.; Sonego, L.; Zutkhi, Y. Plant growth regulator effects on the storage and shelf-life of ‘Triumph’ persimmons. Acta Hortic. 1997, 436, 243–250. [Google Scholar] [CrossRef]
- Ben-Arie, R.; Bazak, H.; Blumenfeld, A. Gibberellin delays harvest and prolongs storage life of persimmon fruits. Acta Hortic. 1986, 179, 807–813. [Google Scholar] [CrossRef]
- Eshel, D.; Ben-Arie, R.; Dinoor, A.; Prusky, D. Resistance of gibberellin-treated persimmon fruit to Alternaria alternata arises from the reduced ability of the fungus to produce endo-1, 4-β-glucanase. Phytopathology 2000, 90, 1256–1262. [Google Scholar] [CrossRef] [PubMed]
- Prusky, D.; Ben-Arie, R.; Guelfat-Reich, S. Etiology and histology of Alternaria rot of persimmon fruits. Phytopathology 1981, 71, 1124–1128. [Google Scholar] [CrossRef]
- Prusky, D.; Eshel, D.; Kobiler, I.; Yakoby, N.; Beno-Moualem, D.; Ackerman, M.; Zuthji, Y.; Ben Arie, R. Postharvest chlorine treatments for the control of the persimmon black spot disease caused by Alternaria alternata. Postharvest Biol. Technol. 2001, 22, 271–277. [Google Scholar] [CrossRef]
- Biton, E.; Kobiler, I.; Feygenberg, O.; Yaari, M.; Kaplunov, T.; Ackerman, M.; Friedman, H.; Prusky, D. The mechanism of differential susceptibility to alternaria black spot, caused by Alternaria alternata, of stem- and bottom-end tissues of persimmon fruit. Postharvest Biol. Technol. 2014, 94, 74–81. [Google Scholar] [CrossRef]
- Kobiler, I.; Akerman, M.; Huberman, L.; Prusky, D. Integration of pre- and postharvest treatments for the control of black spot caused by Alternaria alternata in stored persimmon fruits. Postharvest Biol. Technol. 2010, 59, 166–171. [Google Scholar] [CrossRef]
- Winer, L.; Bendik, E. Effect of growth regulators on persimmon fruit storage and yield at the following year. Alon Hanotea 2012, 66, 49–51. (In Hebrew) [Google Scholar]
- Perez, A.; Ben Arie, R.; Dinoor, A.; Genizi, A.; Prusky, D. Prevention of Black spot disease in persimmon fruit by gibberellic acid and iprodione treatments. Phytopathology 1994, 85, 221–232. [Google Scholar] [CrossRef]
- Sudheeran, P.K.; Feygenberg, O.; Maurer, D.; Alkan, N. Improved cold tolerance of mango fruit with enhanced anthocyanin and flavonoid contents. Molecules 2018, 23, 1832. [Google Scholar] [CrossRef] [PubMed]
- Carpita, N.C.; Gibeaut, D.M. Structural models of primary cell walls in flowering plant: Consistency of molecular structure with the physical properties of the walls during growth. Plant J. 1993, 3, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Greve, L.C.; Labavitch, J.M. Cell wall metabolism in ripening fruit. Plant Physiol. 1991, 97, 1456–1461. [Google Scholar] [CrossRef] [PubMed]
- Kappel, F.; MacDonald, R.A. Gibberellic acid increases fruit firmness, fruit size, and delays maturity of ‘sweetheart’ sweet cherry. Am. Pomol. Soc. 2002, 56, 219–222. [Google Scholar]
- Jamal Uddin, A.F.M.; Hossan, M.J.; Islam, M.S.; Ahsan, M.K.; Mehraj, H. Strawberry growth and yield responses to gibberellic acid concentrations. J. Exp. Biosci. 2012, 3, 51–56. [Google Scholar]
- Lee, S.; Park, C.M. Modulation of reactive oxygen species by salicylic acid in Arabidopsis seed germination under high salinity. Plant Signal. Behav. 2010, 5, 1534–1536. [Google Scholar] [CrossRef] [PubMed]
- Junyu, H.; Yanfang, R.; Chen, C.; Jinping, L.; Houyu, L.; Yun, P. Defense responses of salicylic acid in mango fruit against postharvest anthracnose, caused by Colletotrichum gloeosporioides and its possible mechanism. J. Food Saf. 2016, 37, 1–10. [Google Scholar]
- Biton, E.; Kobiler, I.; Feygenberg, O.; Yaari, M.; Friedman, H.; Prusky, D. Control of alternaria black spot in persimmon fruit by a mixture of gibberellin and benzyl adenine, and its mode of action. Postharvest Biol. Technol. 2014, 94, 82–88. [Google Scholar] [CrossRef]
| Orchard | Fruit Firmness (N) | ABS (%) | ||||
|---|---|---|---|---|---|---|
| Control | Preharvest Spray 50 µg·L−1 | Postharvest Dip 1000 µg·L−1 | Control | Preharvest Spray 50 µg·L−1 | Postharvest Dip 1000 µg·L−1 | |
| Ein Carmel (2016) | 15.7 b z | 24.7 a | 24.5 a | 1.6 a | 1.1 b | 0.3 c |
| Bar Ness (2017) | 11.1 c | 16.6 a | 14.6 ab | 0.8 a | 0.2 b | 0.2 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maurer, D.; Feygenberg, O.; Tzoor, A.; Atzmon, G.; Glidai, S.; Prusky, D. Postharvest Dips of Persimmon Fruit in Gibberellic Acid: An Efficient Treatment to Improve Storability and Reduce Alternaria Black Spot. Horticulturae 2019, 5, 23. https://doi.org/10.3390/horticulturae5010023
Maurer D, Feygenberg O, Tzoor A, Atzmon G, Glidai S, Prusky D. Postharvest Dips of Persimmon Fruit in Gibberellic Acid: An Efficient Treatment to Improve Storability and Reduce Alternaria Black Spot. Horticulturae. 2019; 5(1):23. https://doi.org/10.3390/horticulturae5010023
Chicago/Turabian StyleMaurer, Dalia, Oleg Feygenberg, Alon Tzoor, Guy Atzmon, Shlomo Glidai, and Dov Prusky. 2019. "Postharvest Dips of Persimmon Fruit in Gibberellic Acid: An Efficient Treatment to Improve Storability and Reduce Alternaria Black Spot" Horticulturae 5, no. 1: 23. https://doi.org/10.3390/horticulturae5010023
APA StyleMaurer, D., Feygenberg, O., Tzoor, A., Atzmon, G., Glidai, S., & Prusky, D. (2019). Postharvest Dips of Persimmon Fruit in Gibberellic Acid: An Efficient Treatment to Improve Storability and Reduce Alternaria Black Spot. Horticulturae, 5(1), 23. https://doi.org/10.3390/horticulturae5010023





