Effects of Elevated Temperature and Potassium on Biomass and Quality of Dark Red ‘Lollo Rosso’ Lettuce
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Culture and Harvest
2.2. Flavonoid Analysis
2.3. Mineral Composition
2.4. Statistical Analysis
3. Results
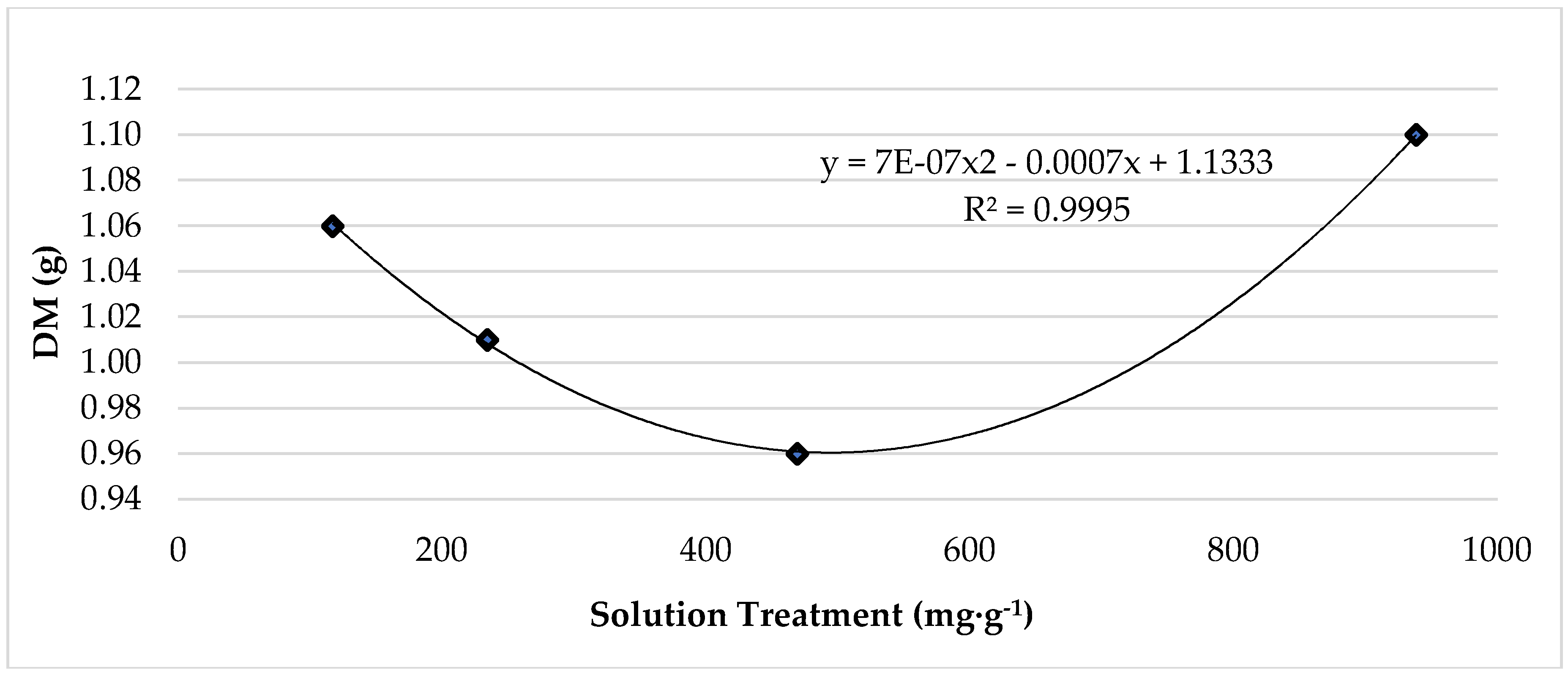
3.1. Temperature and Treatment Effect on Plant Growth
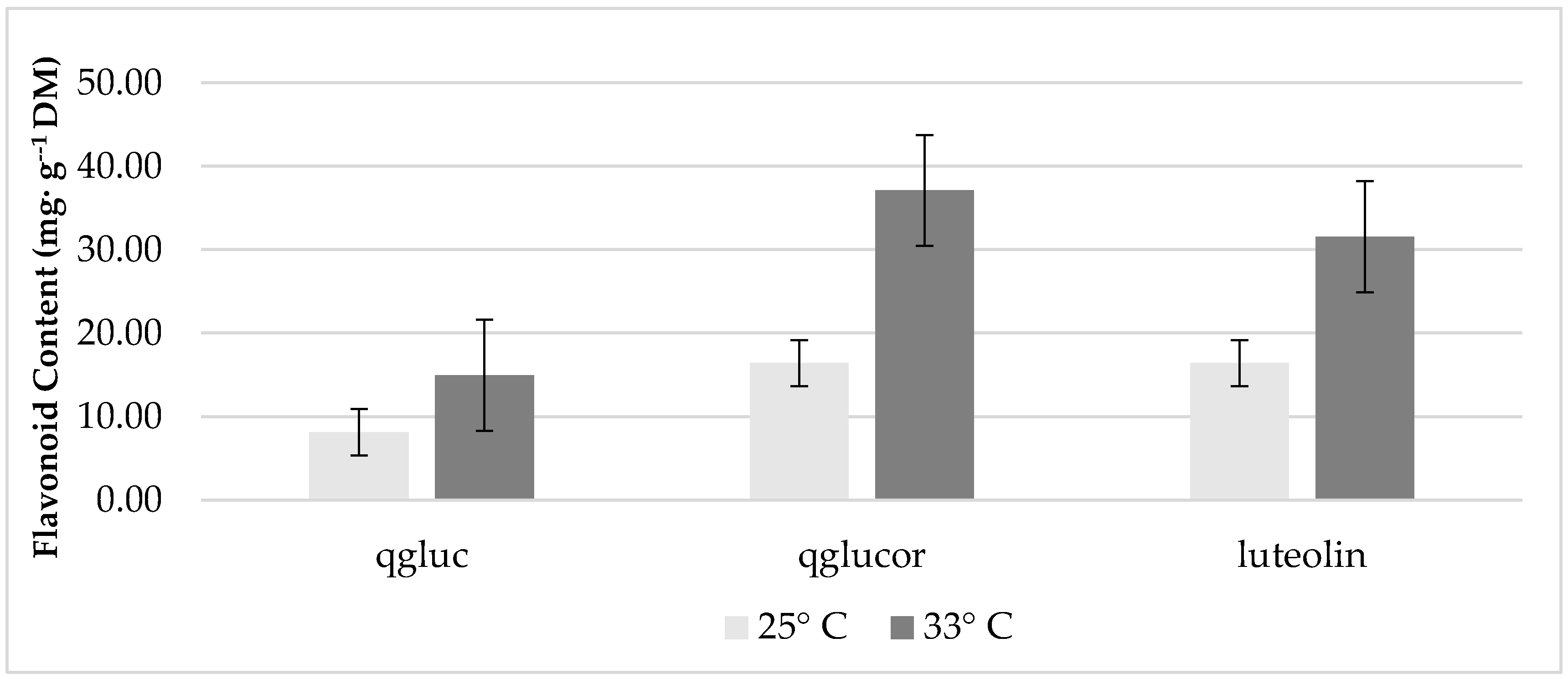
3.2. The Effect of Temperature and Treatment on Leaf Phenolics and Mineral Content
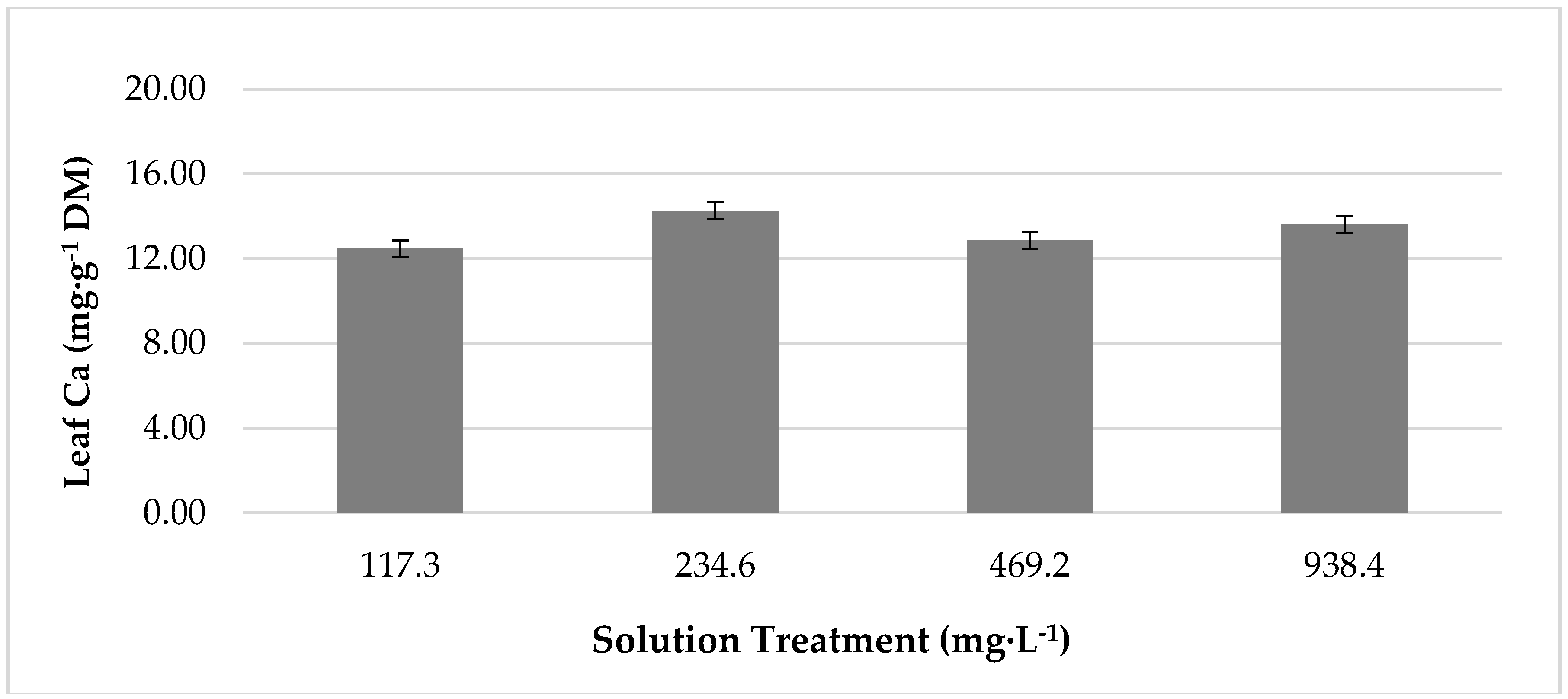
3.3. The Effect of Temperature and Treatment Interactions on Leaf Mineral Content
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Romani, A.; Pinelli, P.; Galardi, C.; Sani, G.; Cimato, A.; Heimler, D. Polyphenols in Greenhouse and Open-Air-Grown Lettuce. Food Chem. 2002, 79, 337–342. [Google Scholar] [CrossRef]
- Tsormpatsidis, E.; Henbest, R.G.C.; Davis, F.J.; Battey, N.H.; Hadley, P.; Wagstaffe, A. UV irradiance as a major influence on growth, development and secondary products of commercial importance in Lollo Rosso lettuce “Revolution” grown under polyethylene films. Environ. Exp. Bot. 2008, 63, 232–239. [Google Scholar] [CrossRef]
- Simonne, A.; Simonne, E.; Eitenmiller, R.; Coker, C.H. Bitterness and composition of lettuce varieties grown in the southeastern United States. Horttechnology 2002, 12, 721–726. [Google Scholar]
- Waycott, W. Photoperiodic Response of Genetically Diverse Lettuce Accessions. J. Am. Soc. Hortic. Sci. 1995, 120, 460–467. [Google Scholar]
- Jenni, S. Rib Discoloration: A Physiological Disorder Induced by Heat Stress in Crisphead Lettuce. HortScience 2005, 40, 2031–2035. [Google Scholar]
- Soundy, P.; Cantliffe, D.J.; Hochmuth, G.J.; Stoffella, P.J. Nutrient Requirements for Lettuce Transplants Using a Floatation Irrigation System II. Potassium. HortScience 2001, 36, 1071–1074. [Google Scholar]
- Masson, J.; Tremblay, N.; Gosselin, A. Nitrogen Fertilization and HPS Supplementary Lighting Influence Vegetable Transplant Production I. Transplant Growth. J. Am. Soc. Hortic. Sci. 1991, 116, 594–598. [Google Scholar]
- Fu, Y.; Li, H.; Yu, J.; Liu, H.; Cao, Z.; Manukovsky, N.S.; Liu, H. Interaction effects of light intensity and nitrogen concentration on growth, photosynthetic characteristics and quality of lettuce (Lactuca sativa L. Var. youmaicai). Sci. Hortic. (Amsterdam) 2017, 214, 51–57. [Google Scholar] [CrossRef]
- Petrazzini, L.L.; Souza, G.A.; Rodas, C.L.; Emrich, E.B.; Carvalho, J.G.; Souza, R.J. Nutritional deficiency in crisphead lettuce grown in hydroponics. Hortic. Bras. 2014, 32, 310–313. [Google Scholar] [CrossRef]
- Barickman, T.C.; Horgan, T.E.; Wheeler, J.R.; Sams, C.E. Elevated Levels of Potassium in Greenhouse-grown Red Romaine Lettuce Impacts Mineral Nutrient and Soluble Sugar Concentrations. HortScience 2016, 51, 504–509. [Google Scholar]
- Inthichack, P.; Nishimura, Y.; Fukumoto, Y. Effect of potassium sources and rates on plant growth, mineral absorption, and the incidence of tip burn in cabbage, celery, and lettuce. Hortic. Environ. Biotechnol. 2012, 53, 135–142. [Google Scholar] [CrossRef]
- Lester, G.E.; Jifon, J.L.; Makus, D.J. Impact of potassium nutrition on postharvest fruit quality: Melon (Cucumis melo L.) case study. Plant Soil 2010, 335, 117–131. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: San Diego, CA, USA, 2012. [Google Scholar]
- Hu, Y.; Schmidhalter, U. Drought and salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Cakmak, I. The role of potassium in alleviating detrimental effects of abiotic stresses in plants. J. Plant Nutr. Soil Sci. 2005, 168, 521–530. [Google Scholar] [CrossRef]
- Achilea, O. Alleviation of salinity—Induced stress in cash crops by multi-K (Potassium nitrate), five cases typifying the underlying pattern. Acta Hortic. 2002, 573, 43–48. [Google Scholar] [CrossRef]
- Tzortzakis, N.G. Potassium and calcium enrichment alleviate salinity-induced stress in hydroponically grown endives. Hortic. Sci. 2010, 37, 155–162. [Google Scholar] [CrossRef]
- Bres, W.; Weston, L. A Nutrient Accumulation and Tipburn in {NFT-grown} Lettuce at Several Potassium and {pH} Levels. HortScience 1992, 27, 790–792. [Google Scholar]
- Fallovo, C.; Rouphael, Y.; Cardarelli, M.; Rea, E.; Battistelli, A.; Colla, G. Yield and quality of leafy lettuce in response to nutrient solution composition and growing season. J. Food Agric. Environ. 2009, 7, 456–462. [Google Scholar]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Ca. Agric. Exp. Stn. Circ. 1950, 347, 1–32. [Google Scholar]
- Chen, H.J.; Inbaraj, B.S.; Chen, B.H. Determination of phenolic acids and flavonoids in Taraxacum formosanum kitam by liquid chromatography-tandem mass spectrometry coupled with a post-column derivatization technique. Int. J. Mol. Sci. 2012, 13, 260–285. [Google Scholar] [CrossRef] [PubMed]
- Barickman, T.C.; Kopsell, D.A.; Sams, C.E. Effect of selenium and sulfur fertilization on glucosinolate and isothiocyanates in Arabidopsis thaliana and rapid-cycling Brassica oleracea. Acta Hortic. 2014, 1040, 31–38. [Google Scholar] [CrossRef]
- Jenni, S.; Truco, M.J.; Michelmore, R.W. Quantitative trait loci associated with tipburn, heat stress-induced physiological disorders, and maturity traits in crisphead lettuce. Theor. Appl. Genet. 2013, 126, 3065–3079. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, V.M.; Soengas, P.; Alonso-Villaverde, V.; Sotelo, T.; Cartea, M.E.; Velasco, P. Effect of temperature stress on the early vegetative development of Brassica oleracea L. BMC Plant Biol. 2015, 15, 145. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.R. Effect of Temperature on the Growth and Development of Tomato Fruits. Ann. Bot. 2001, 88, 869–877. [Google Scholar] [CrossRef]
- Erickson, A.N.; Markhart, A.H. Flower Production, Fruit Set, and Physiology of Bell Pepper during Elevated Temperature and Vapor Pressure Deficit. J. Am. Soc. Hortic. Sci. 2001, 126, 697–702. [Google Scholar]
- Yuan, L.; Yuan, Y.; Liu, S.; Wang, J.; Zhu, S.; Chen, G.; Hou, J.; Wang, C. Influence of high temperature on photosynthesis, antioxidative capacity of chloroplast, and carbon assimilation among heat-tolerant and heat-susceptible genotypes of nonheading Chinese cabbage. HortScience 2017, 52, 1464–1470. [Google Scholar] [CrossRef]
- Gruda, N. Impact of environmental factors on product quality of greenhouse vegetables for fresh consumption. CRC Crit. Rev. Plant Sci. 2005, 24, 227–247. [Google Scholar] [CrossRef]
- Gent, M.P.N. Effect of temperature on composition of hydroponic lettuce. Acta Hortic. 2016, 1123, 95–100. [Google Scholar] [CrossRef]
- Lafta, A.; Turini, T.; Sandoya, G.V.; Mou, B. Field Evaluation of Green and Red Leaf Lettuce Genotypes in the Imperial, San Joaquin, and Salinas Valleys of California for Heat Tolerance and Extension of the Growing Seasons. HortScience 2017, 52, 40–48. [Google Scholar] [CrossRef]
- Leng, P.; Itamura, H.; Yamamura, H.; Deng, X.M. Anthocyanin accumulation in apple and peach shoots during cold acclimation. Sci. Hortic. (Amsterdam) 2000, 83, 43–50. [Google Scholar] [CrossRef]
- Christie, P.J.; Alfenito, M.R.; Walbot, V. Impact of low-temperature stress on general phenylpropanoid and anthocyanin pathways: Enhancement of transcript abundance and anthocyanin pigmentation in maize seedlings. Planta 1994, 194, 541–549. [Google Scholar] [CrossRef]
- Becker, C. Impact of Radiation, Temperature and Growth Stage on the Concentration of Flavonoid Glycosides and Caffeic Acid Derivatives in Red Leaf Lettuce. Ph.D. Thesis, Technische Universitat Berlin, Fakultat III-Prozesswissenschaften, Berlin, Germany, 2014. [Google Scholar] [CrossRef]
- Becker, C.; Klaering, H.P.; Kroh, L.W.; Krumbein, A. Cool-cultivated red leaf lettuce accumulates cyanidin-3-O-(6″-O-malonyl)-glucoside and caffeoylmalic acid. Food Chem. 2014, 146, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Becker, C. Flavonoids and phenolic acids in lettuce: How can we maximize their concentration? and why should we? Acta Hortic. 2016, 1142, 1–10. [Google Scholar] [CrossRef]
- Steyn, A.W.J.; Wand, S.J.E.; Holcroft, D.M.; Jacobs, G. Anthocyanins in Vegetative Tissues: A Proposed Unified Function in Photoprotection. New Phytol. 2016, 155, 349–361. [Google Scholar] [CrossRef]
- Oh, M.; Carey, E.E.; Rajashekar, C.B. Plant Physiology and Biochemistry Environmental stresses induce health-promoting phytochemicals in lettuce. Plant Physiol. Biochem. 2009, 47, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Marin, A.; Ferreres, F.; Barberá, G.G.; Gil, M.I. Weather variability influences color and phenolic content of pigmented baby leaf lettuces throughout the season. J. Agric. Food Chem. 2015, 63, 1673–1681. [Google Scholar] [CrossRef] [PubMed]
- Boo, H.-O.; Heo, B.-G.; Gorinstein, S.; Chon, S.-U. Positive effects of temperature and growth conditions on enzymatic and antioxidant status in lettuce plants. Plant Sci. 2011, 181, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Bunning, M.L.; Kendall, P.A.; Stone, M.B.; Stonaker, F.H.; Stushnoff, C. Effects of Seasonal Variation on Sensory Properties and Total Phenolic Content of 5 Lettuce Cultivars. J. Food Sci. 2010, 75, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Chiloane, T.S. Effect of Nutrient Concentration and Growing Seasons on Growth, Yield and Quality of Leafy Lettuce (Lactuca sativa L.) in a Hydroponic System. Ph.D. Thesis, University of Pretoria, Pretoria, South Africa, 2012; pp. 1–79. [Google Scholar]
| Temp (°C) | Leaf FM (g) | Leaf DM (g) | Leaf DM:FM a | Leaf Water (%) | Root FM (g) | Root DM (g) | Root DM:FM a | Root Water (%) |
|---|---|---|---|---|---|---|---|---|
| 25 | 12.12 a | 0.86 b | 0.07 b | 0.93 a | 10.11 a | 0.40 a | 0.04 a | 0.96 a |
| 33 | 12.47 a | 1.20 a | 0.10 a | 0.90 b | 3.22 b | 0.14 b | 0.05 a | 0.95 a |
| P-value b,c | ns | *** | *** | *** | *** | *** | ns | ns |
| Elemental Nutrient Concentrations a | ||||||||
|---|---|---|---|---|---|---|---|---|
| (mg·g−1 DM) | (μg·g−1 DM) | |||||||
| Temp | Mg | K | Ca | Fe | B | Mn | Zn | Mo |
| 25 | 4.11 a | 76.48 a | 14.25 a | 168.28 a | 32.44 a | 136.07 a | 25.49 a | 0.60 a |
| 33 | 3.32 b | 67.73 b | 12.34 b | 138.46 a | 31.44 a | 79.64 b | 28.17 a | 0.39 b |
| P-value b,c | *** | *** | ** | ns | ns | *** | ns | * |
| Elemental Nutrient Concentrations a | ||||
|---|---|---|---|---|
| (mg·g−1 DM) | (μg·g−1 DM) | |||
| Temperature (°C) | K Treatment (mg·L−1) | P | S | Cu |
| 25 | 117.3 | 8.50 a | 10.41 a | 2.59 a,b,c |
| 25 | 234.6 | 7.40 b | 10.61 a | 2.64 a,b,c |
| 25 | 469.2 | 7.69 b | 10.59 a | 3.82 a |
| 25 | 938.4 | 7.61 b | 10.02 a,b | 3.30 a,b |
| 33 | 117.3 | 6.20 c | 9.99 ab | 3.22 a,b |
| 33 | 234.6 | 6.54 c | 8.94 a,b | 1.36 b,c,d |
| 33 | 469.2 | 6.33 c | 8.82 a,b | 0.78 c,d |
| 33 | 938.4 | 6.33 c | 9.51 a,b | 0.37 d |
| P-value b,c | * | * | * | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sublett, W.L.; Barickman, T.C.; Sams, C.E. Effects of Elevated Temperature and Potassium on Biomass and Quality of Dark Red ‘Lollo Rosso’ Lettuce. Horticulturae 2018, 4, 11. https://doi.org/10.3390/horticulturae4020011
Sublett WL, Barickman TC, Sams CE. Effects of Elevated Temperature and Potassium on Biomass and Quality of Dark Red ‘Lollo Rosso’ Lettuce. Horticulturae. 2018; 4(2):11. https://doi.org/10.3390/horticulturae4020011
Chicago/Turabian StyleSublett, William L., T. Casey Barickman, and Carl E. Sams. 2018. "Effects of Elevated Temperature and Potassium on Biomass and Quality of Dark Red ‘Lollo Rosso’ Lettuce" Horticulturae 4, no. 2: 11. https://doi.org/10.3390/horticulturae4020011
APA StyleSublett, W. L., Barickman, T. C., & Sams, C. E. (2018). Effects of Elevated Temperature and Potassium on Biomass and Quality of Dark Red ‘Lollo Rosso’ Lettuce. Horticulturae, 4(2), 11. https://doi.org/10.3390/horticulturae4020011






