Short-Term Effects of Cover Crop Species and Termination Methods on Soil pH and Key Enzymatic Activities (β-Glucosidase, Phosphatase and Urease Activities) in a Citrus Orchard (Eureka Lemons)
Abstract
1. Introduction
2. Materials and Methods
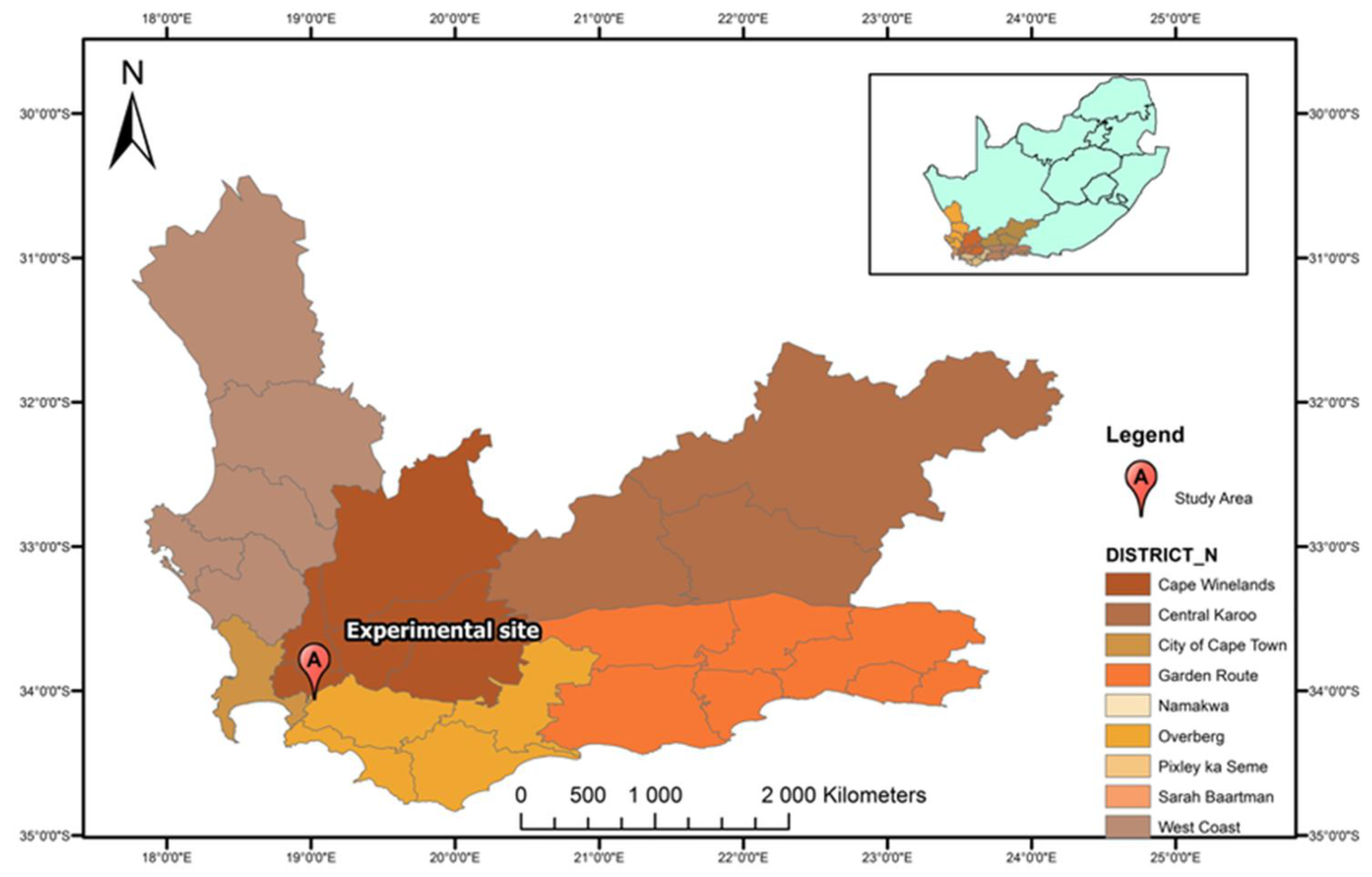
2.1. Study Location
2.2. Factorial Experimental Design
2.3. The Establishment of a Selection of Three Cover Crop Species
2.4. Seed Sowing
2.5. Termination
2.6. Data Collection
Soil Chemical Analysis
2.7. Soil Enzyme Activity Assay
2.8. Soil Carbon, Dry Weight and Soil Moisture Measurements
2.9. Statistical Analyses
3. Results and Discussion
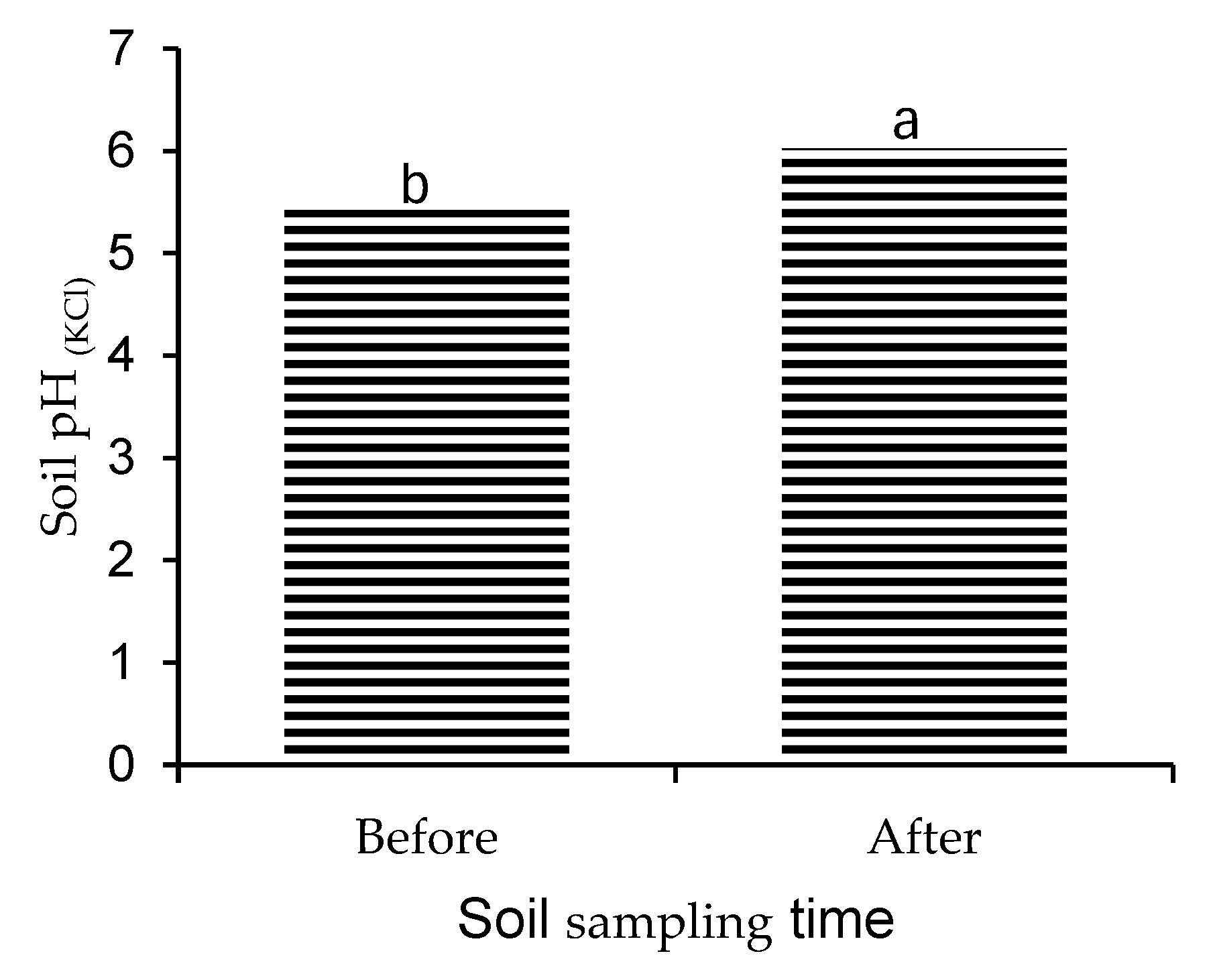
3.1. Influence of Cover Crops on Soil pH After One Year
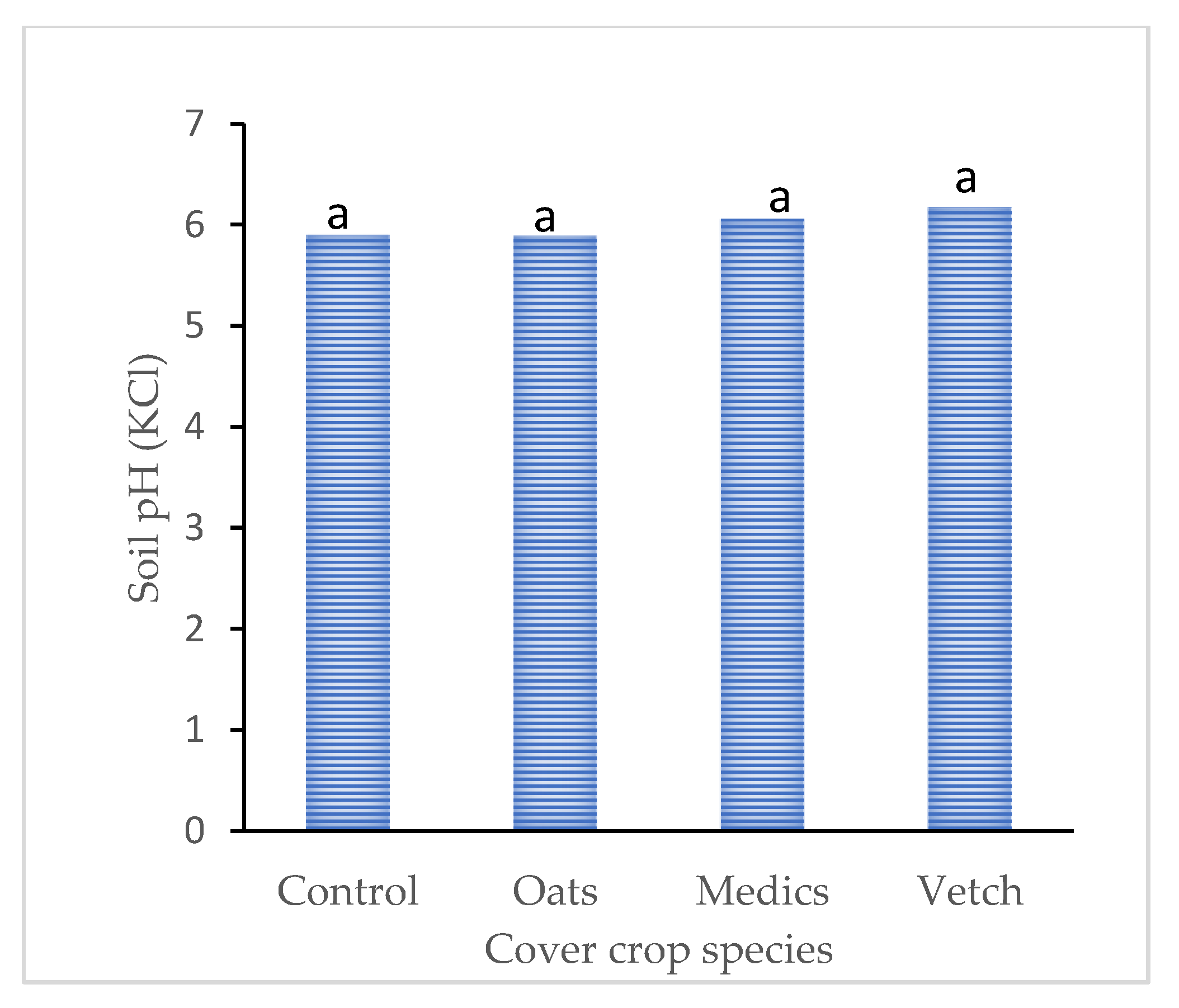
3.2. Minimal Response of Soil pH After Planting Different Cover Crop Species
3.3. Soil β-Glucosidase, Phosphatase and Urease Activities After One Year of Planting Cover Crops
3.4. Soil Urease, β-Glucosidase and Phosphatase Activities as Affected by Cover Crop Species
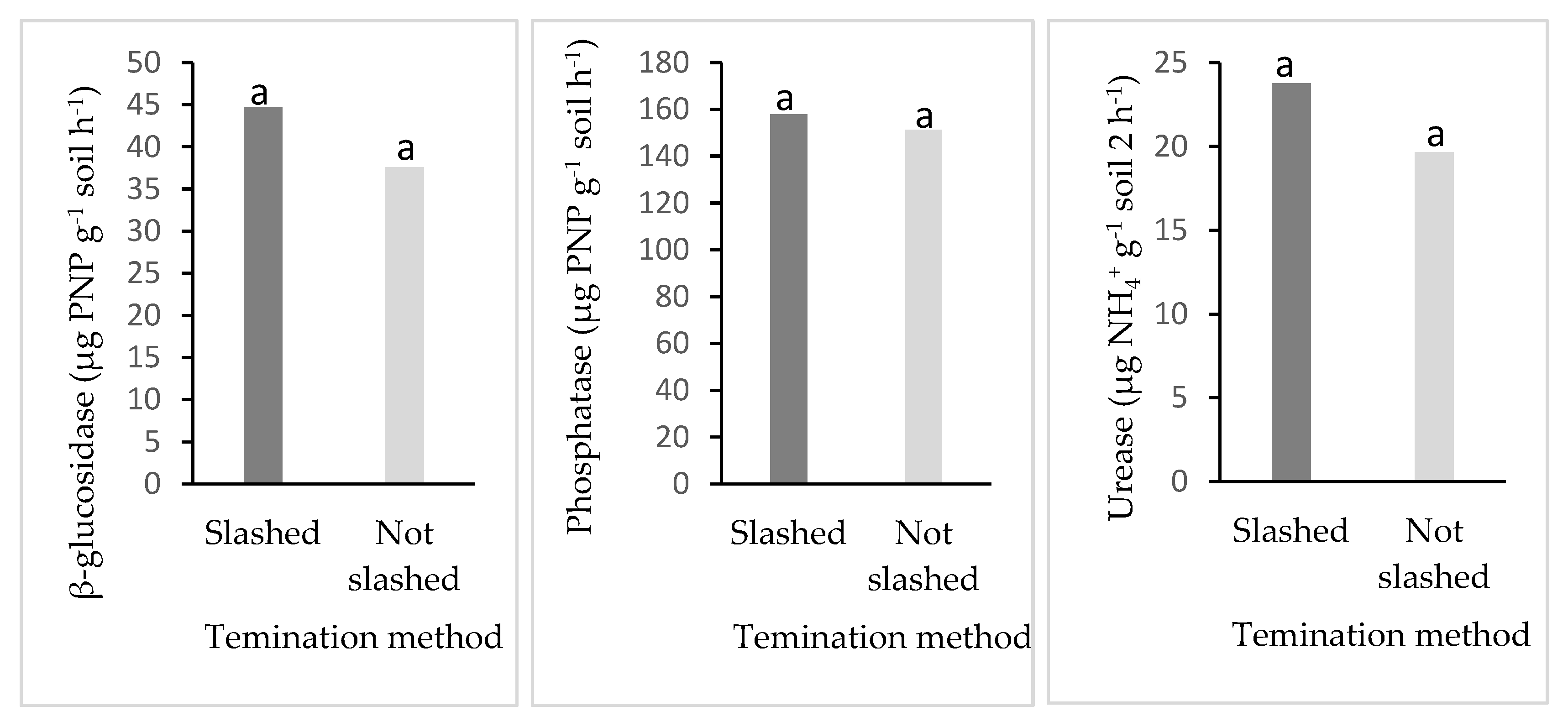
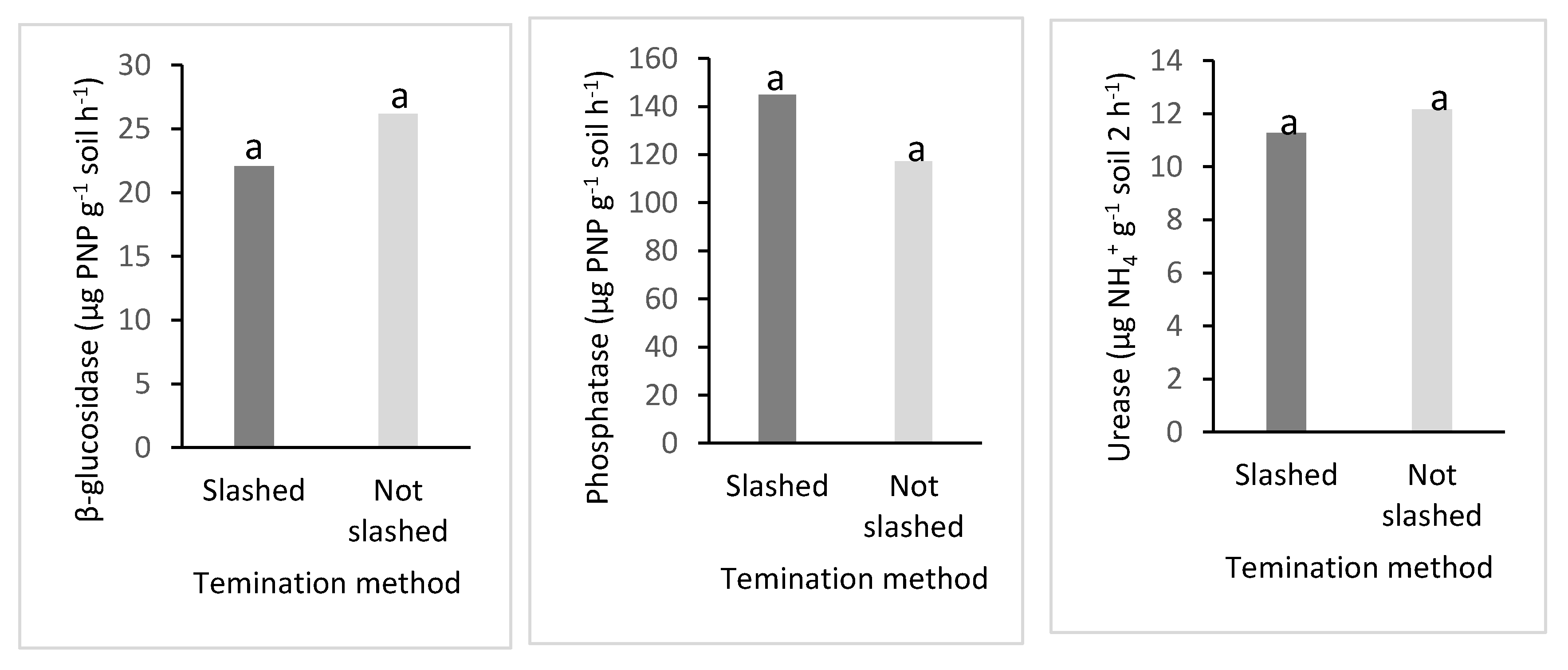
3.5. Soil Enzyme Activity and Termination Methods (Slashed and Non-Slashed Methods)
3.6. Correlation Between Soil Carbon, Soil Moisture and Soil Enzyme Activities Using Pearson Method
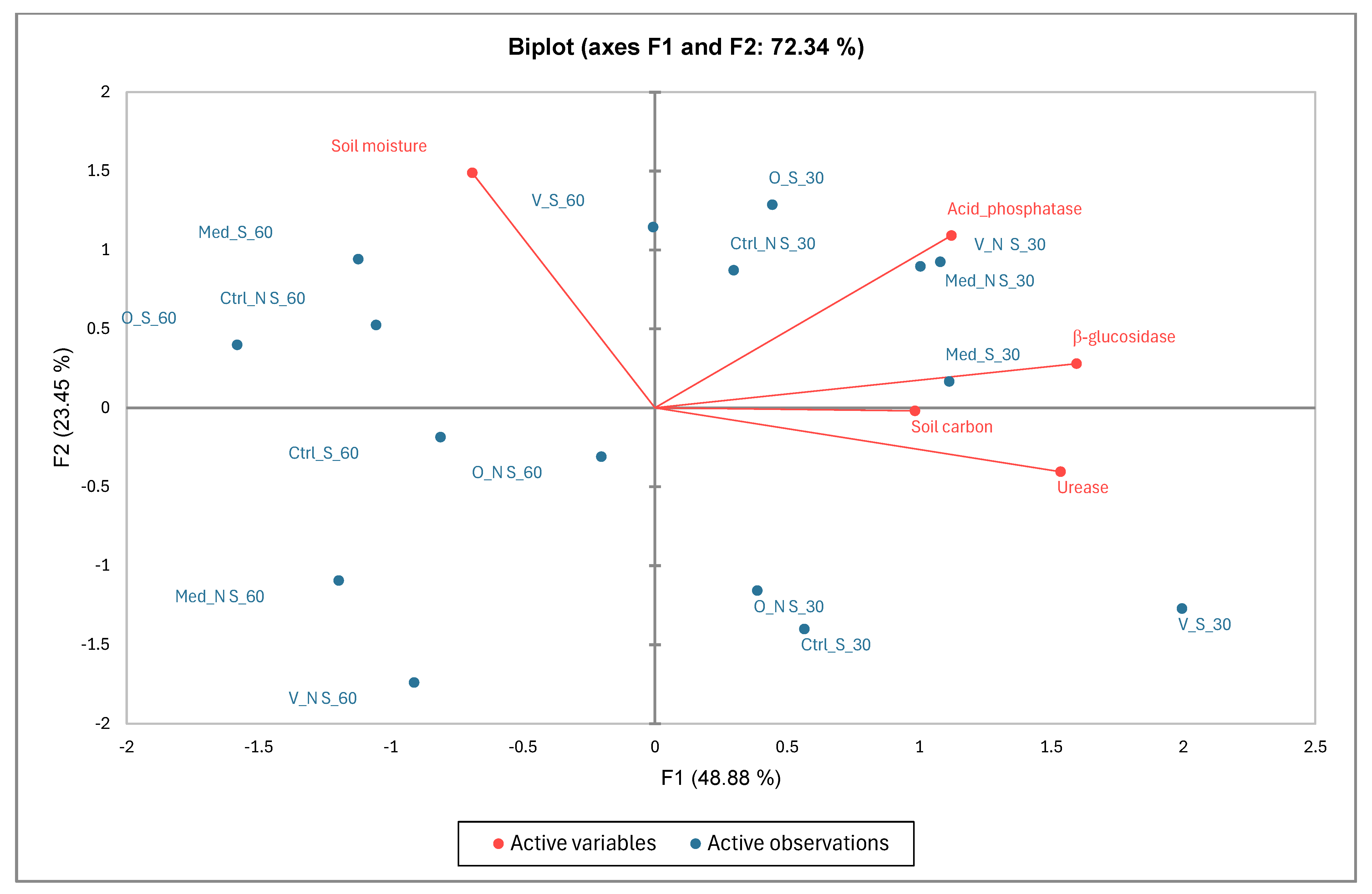
3.7. Contribution of Variables and Factors (Soil Carbon, Soil Moisture and Soil Enzyme Activities) Using Principal Component Analysis
3.8. Correlation Between Soil Carbon, Soil Moisture, Soil Activities, Cover Crop and Weed Dry Weights Using Pearson Method
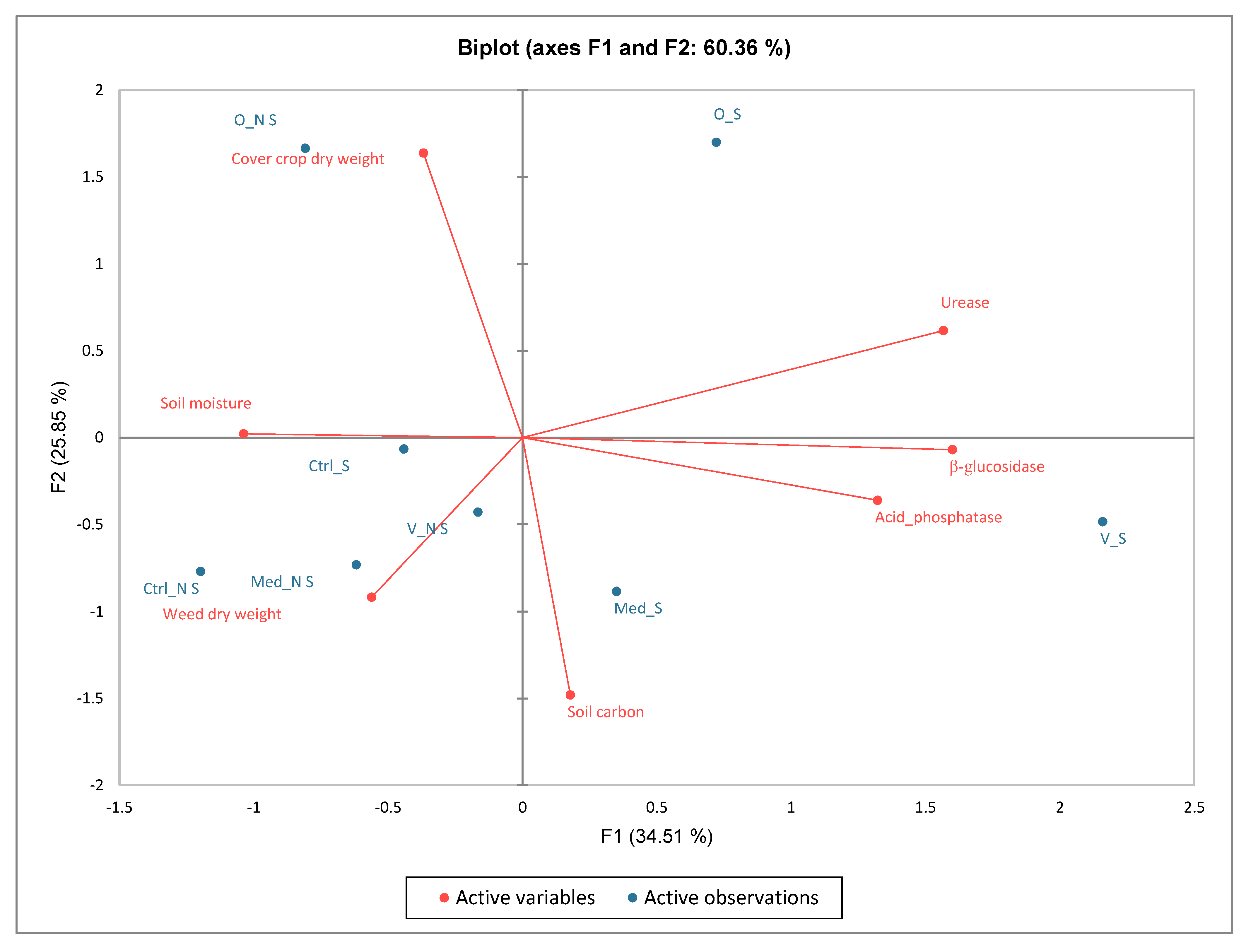
3.9. Contribution of Variables and Factors (Soil Carbon, Soil Moisture, Soil Activities, Cover Crop and Weed Dry Weights) Using Principal Component Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Bruin, A.M. An Evaluation of the Risks in South African Citrus Farming. Ph.D. Thesis, North-West University, Potchefstroom, South Africa, 2023. [Google Scholar]
- Mukhametzyanov, R.R.; Brusenko, S.V.; Khezhev, A.M.; Kelemetov, E.M.; Kirillova, S.S. Changing the Global Production and Trade of Citrus Fruits. In Sustainable Development of the Agrarian Economy Based on Digital Technologies and Smart Innovations; Springer Nature: Cham, Switzerland, 2024; pp. 19–24. [Google Scholar]
- El-Otmani, M.; Ait-Oubahou, A.; Zacarías, L. Citrus spp.: Orange, mandarin, tangerine, clementine, grapefruit, pomelo, lemon and lime. In Postharvest Biology and Technology of Tropical and Subtropical Fruits; Woodhead Publishing: Cambridge, UK, 2011; pp. 437–516. [Google Scholar]
- Bindraban, P.S.; van der Velde, M.; Ye, L.; Van den Berg, M.; Materechera, S.; Kiba, D.I.; Tamene, L.; Ragnarsdóttir, K.V.; Jongschaap, R.; Hoogmoed, M.; et al. Assessing the impact of soil degradation on food production. Curr. Opin. Environ. Sustain. 2012, 4, 478–488. [Google Scholar] [CrossRef]
- Mulinge, J.; Saha, H.; Mounde, L.; Wasilwa, L. Effects of legume cover crops on orange (Citrus sinensis) fruit weight and brix. Int. J. Plant Soil Sci. 2018, 21, 1–9. [Google Scholar] [CrossRef]
- Karaca, A.; Cetin, S.C.; Turgay, O.C.; Kizilkaya, R. Soil enzymes as indication of soil quality. In Soil Enzymology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 119–148. [Google Scholar]
- Adetunji, A.T.; Lewu, F.B.; Mulidzi, R.; Ncube, B. The biological activities of β-glucosidase, phosphatase and urease as soil quality indicators: A review. J. Soil Sci. Plant Nutr. 2017, 17, 794–807. [Google Scholar] [CrossRef]
- Knowler, D.; Bradshaw, B. Farmers’ adoption of conservation agriculture: A review and synthesis of recent research. Food Policy 2007, 32, 25–48. [Google Scholar] [CrossRef]
- Storr, T. The Effect of Cover Crops on Soil Quality Indicators in a Cereal and Salad Rotation. Ph.D. Thesis, Cranfield University, Cambridgeshire, UK, 2019. [Google Scholar]
- Thilakarathna, M.S.; Serran, S.; Lauzon, J.; Janovicek, K.; Deen, B. Management of manure nitrogen using cover crops. Agron. J. 2015, 107, 1595–1607. [Google Scholar] [CrossRef]
- Sievers, T.; Cook, R.L. Aboveground and root decomposition of cereal rye and hairy vetch cover crops. Soil Sci. Soc. Am. J. 2018, 82, 147–155. [Google Scholar] [CrossRef]
- Das, S.K.; Varma, A. Role of enzymes in maintaining soil health. In Soil Enzymology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 25–42. [Google Scholar]
- Li, H.; Song, Y.; Li, Q.; He, J.; Song, Y. Effective microbial calcite precipitation by a new mutant and precipitating regulation of extracellular urease. Bioresour. Technol. 2014, 167, 269–275. [Google Scholar] [CrossRef]
- Gil-Sotres, F.; Trasar-Cepeda, C.; Leirós, M.C.; Seoane, S. Different approaches to evaluating soil quality using biochemical properties. Soil Biol. Biochem. 2005, 37, 877–887. [Google Scholar] [CrossRef]
- Adetunji, A.T. Impact of Cover Crop Species, Termination Stage and Termination Method on Soil Nitrogen, Organic Carbon and Enzyme Activities. Ph.D. Thesis, Cape Peninsula University of Technology, Cape Town, South Africa, 2019. [Google Scholar]
- Martinez, C.E.; Tabatabai, M.A. Decomposition of Biotechnology By-Products in Soils; American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America: Madison, WI, USA, 1997; Volume 26, pp. 625–632. [Google Scholar]
- Ghosh, A.; Paul, R.; Sarkar, A.; Manna, M.C.; Bhattacharjya, S.; Alam, K.; Choudhury, S.; Mondal, P. Carbon-, Nitrogen-, Phosphorus-, and Sulfur-Cycling Enzymes and Functional Diversity in Agricultural Systems. In Agricultural Biocatalysis; Jenny Stanford Publishing: Uttar Pradesh, India, 2022; pp. 335–363. [Google Scholar]
- Merino, C.; Godoy, R.; Matus, F. Soil enzymes and biological activity at different levels of organic matter stability. J. Soil Sci. Plant Nutr. 2016, 16, 14–30. [Google Scholar]
- Makoi, J.H.; Ndakidemi, P.A. Selected soil enzymes: Examples of their potential roles in the ecosystem. Afr. J. Biotechnol. 2008, 7, 181–191. [Google Scholar]
- Gabasawa, A.I. Phosphorus cycle enzymes to remedy soil phosphorus deficiency. In Sustainable Agriculture Reviews 58: Phosphorus Use Efficiency for Sustainable Agriculture; Springer: Berlin/Heidelberg, Germany, 2022; pp. 177–205. [Google Scholar]
- Condron, L.M.; Turner, B.L.; Cade-Menun, B.J. Chemistry and dynamics of soil organic phosphorus. In Phosphorus: Agriculture and the Environment; Wiley Online Library: Hoboken, NJ, USA, 2005; Volume 46, pp. 87–121. [Google Scholar]
- Soil Classification Working Group (SCWG). Soil Classification: A Taxonomic System for South Africa; Department of Agricultural Development: Pretoria, South Africa, 1991; Volume 15.
- Jannoyer, M.L.; Le Bellec, F.; Lavigne, C.; Achard, R.; Malézieux, E. Choosing cover crops to enhance ecological services in orchards: A multiple criteria and systemic approach applied to tropical areas. Procedia Environ. Sci. 2011, 9, 104–112. [Google Scholar] [CrossRef]
- Silwana, S.; Mulidzi, A.R.; Jovanovic, N. Evaluating the effects and benefits of cover crops in citrus orchards: A review. S. Afr. J. Plant Soil 2023, 40, 117–126. [Google Scholar] [CrossRef]
- Oliveira, F.É.R.D.; Oliveira, J.D.M.; Xavier, F.A.D.S. Changes in soil organic carbon fractions in response to cover crops in an orange orchard. Rev. Bras. Cienc. Solo 2016, 40, e0150105. [Google Scholar] [CrossRef]
- Adetunji, A.T.; Ncube, B.; Meyer, A.H.; Olatunji, O.S.; Mulidzi, R.; Lewu, F.B. Soil pH, nitrogen, phosphatase and urease activities in response to cover crop species, termination stage and termination method. Heliyon 2021, 7, e05980. [Google Scholar] [CrossRef]
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Eivazi, F.; Tabatabai, M.A. Factors affecting glucosidase and galactosidase activities in soils. Soil Biol. Biochem. 1990, 22, 891–897. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Mishra, P.; Pandey, C.M.; Singh, U.; Gupta, A.; Sahu, C.; Keshri, A. Descriptive statistics and normality tests for statistical data. Ann. Card. Anaesth. 2019, 22, 67–72. [Google Scholar] [CrossRef]
- Ott, R.L.; Longnecker, M. An Introduction to Statistical Methods and Data Analysis; Cengage Learning: Boston, MA, USA, 2016. [Google Scholar]
- Srivastava, A.K.; Singh, S. Citrus decline: Soil fertility and plant nutrition. J. Plant Nutr. 2009, 32, 197–245. [Google Scholar] [CrossRef]
- Silva, M.B.D.; Kliemann, H.J.; Silveira, P.M.D.; Lanna, A.C. Biological attributes of the soil under the influence of vegetation cover and management system. Pesqui. Agropecu. Bras. 2007, 42, 1755–1761. [Google Scholar] [CrossRef]
- Vanzolini, J.I.; Galantini, J.A.; Martínez, J.M.; Suñer, L. Changes in soil pH and phosphorus availability during decomposition of cover crop residues. Arch. Agron. Soil Sci. 2017, 63, 1864–1874. [Google Scholar] [CrossRef]
- Dick, W.A.; Cheng, L.; Wang, P. Soil acid and alkaline phosphatase activity as pH adjustment indicators. Soil Biol. Biochem. 2000, 32, 1915–1919. [Google Scholar] [CrossRef]
- Maltais-Landry, G. Legumes have a greater effect on rhizosphere properties (pH, organic acids and enzyme activity) but a smaller impact on soil P compared to other cover crops. Plant Soil 2015, 394, 139–154. [Google Scholar] [CrossRef]
- Spera, S.T.; Escosteguy, P.A.V.; dos Santos, H.P.; Klein, V.A. The soil solution of a dystrophic red latosol subjected to three types of soil and crop management. Nativa. Sinop. 2014, 2, 58–64. [Google Scholar] [CrossRef]
- Medeiros, J.C.; Dalla Rosa, J.; Mafra, A.L.; de Sousa Mendes, W. Chemical attributes of agricultural soil after the cultivation of cover crops. Aust. J. Crop Sci. 2017, 11, 1497–1503. [Google Scholar]
- Hontoria, C.; García-González, I.; Quemada, M.; Roldán, A.; Alguacil, M.M. The cover crop determines the AMF community composition in soil and in roots of maize after a ten-year continuous crop rotation. Sci. Total Environ. 2019, 660, 913–922. [Google Scholar] [CrossRef] [PubMed]
- Ajwa, H.A.; Tabatabai, M.A. Decomposition of different organic materials in soils. Biol. Fertil. Soils 1994, 18, 175–182. [Google Scholar] [CrossRef]
- Singh, J.; Kumar, S. Seasonal changes of soil carbon fractions and enzyme activities in response to winter cover crops under long-term rotation and tillage systems. Eur. J. Soil Sci. 2021, 72, 886–899. [Google Scholar] [CrossRef]
- Turner, S.; Schippers, A.; Meyer-Stüve, S.; Guggenberger, G.; Gentsch, N.; Dohrmann, R.; Condron, L.M.; Eger, A.; Almond, P.C.; Peltzer, D.A.; et al. Mineralogical impact on long-term patterns of soil nitrogen and phosphorus enzyme activities. Soil Biol. Biochem. 2014, 68, 31–43. [Google Scholar] [CrossRef]
- Deng, J.; Ni, H.; Zhang, Z.; Usman, S.; Yang, X.; Shen, Y.; Li, Y. Designing productive, energy-efficient, and environmentally friendly production systems by replacing fallow period with annual forage cultivation on the Loess Plateau of China. J. Clean. Prod. 2021, 320, 128660. [Google Scholar] [CrossRef]
- Agustí, M.; Mesejo, C.; Reig, C.; Martínez-Fuentes, A. Citrus production. In Horticulture: Plants for People and Places, Volume 1: Production Horticulture; Springer: Dordrecht, The Netherlands, 2014; pp. 159–195. [Google Scholar]
- Zekri, M.; Obreza, T.A. Plant Nutrients for Citrus Trees; University of Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, EDIS: Gainesville, FL, USA, 2003; pp. 1–2. [Google Scholar]
- Schroth, A.W.; Bostick, B.C.; Graham, M.; Kaste, J.M.; Mitchell, M.J.; Friedland, A.J. Sulfur species behavior in soil organic matter during decomposition. J. Geophys. Res. Biogeosci. 2007, 112. [Google Scholar] [CrossRef]
- Qian, X.; Gu, J.; Pan, H.J.; Zhang, K.Y.; Sun, W.; Wang, X.J.; Gao, H. Effects of living mulches on the soil nutrient contents, enzyme activities, and bacterial community diversities of apple orchard soils. Eur. J. Soil Biol. 2015, 70, 23–30. [Google Scholar] [CrossRef]
- Maseko, S.T.; Dakora, F.D. Rhizosphere acid and alkaline phosphatase activity as a marker of P nutrition in nodulated Cyclopia and Aspalathus species in the Cape fynbos of South Africa. S. Afr. J. Bot. 2013, 89, 289–295. [Google Scholar] [CrossRef]
- Parr, M.; Grossman, J.M.; Reberg-Horton, S.C.; Brinton, C.; Crozier, C. Nitrogen delivery from legume cover crops in no-till organic corn production. Agron. J. 2011, 103, 1578–1590. [Google Scholar] [CrossRef]
- Jani, A.D.; Grossman, J.; Smyth, T.J.; Hu, S. Winter legume cover-crop root decomposition and N release dynamics under disking and roller-crimping termination approaches. Renew. Agric. Food Syst. 2016, 31, 214–229. [Google Scholar] [CrossRef]
- García-Orenes, F.; Guerrero, C.; Roldán, A.; Mataix-Solera, J.; Cerdà, A.; Campoy, M.; Zornoza, R.; Bárcenas, G.; Caravaca, F. Soil microbial biomass and activity under different agricultural management systems in a semiarid Mediterranean agroecosystem. Soil Tillage Res. 2010, 109, 110–115. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef]
- Yadav, S.S.; Guzman, J.G.; Meena, R.S.; Lal, R.; Yadav, G.S. Long term crop management effects on soil organic carbon, structure, and water retention in a cropland soil in central Ohio, USA. J. Plant Nutr. Soil Sci. 2020, 183, 200–207. [Google Scholar] [CrossRef]
- Lajtha, K.; Bowden, R.D.; Nadelhoffer, K. Litter and root manipulations provide insights into soil organic matter dynamics and stability. Soil Sci. Soc. Am. J. 2014, 78, S261–S269. [Google Scholar] [CrossRef]
- Zhang, Y.; Ding, S.; Fan, J.; Wang, L. Depth-dependent soil microbial and enzymatic responses to land use changes in agroecosystems. Appl. Soil Ecol. 2020, 155, 103648. [Google Scholar]
- Pervaiz, Z.H.; Iqbal, J.; Zhang, Q.; Chen, D.; Wei, H.; Saleem, M. Continuous cropping alters multiple biotic and abiotic indicators of soil health. Soil Syst. 2020, 4, 59. [Google Scholar] [CrossRef]

| Year | April | May | June | July | August | Total (mm) |
|---|---|---|---|---|---|---|
| 2022 | 21.3 | 79.8 | 169.2 | 101.3 | 111.3 | 482.9 |
| 2023 | 107 | 160 | 390.1 | 123.9 | 2.1 | 783.1 |
| Soil Characteristics | 2022 | 2023 |
|---|---|---|
| Soil texture | Sand | Sand |
| Carbon% (Walkley back) | 0.46% | 0.59% |
| PH (KCl) | 5.42 | 6.02 |
| P (Ambic 1) | 24.5 mg kg−1 | 28.4 mg kg−1 |
| K (ammonium acetate extraction) | 64 mg kg−1 | 54 mg kg−1 |
| NO3-N (KCl) | - | 3.95 mg kg−1 |
| NH4-N (KCl | - | 11.88 mg kg−1 |
| Soil Depth | Sampling Time | Season | β-Glucosidase (µg PNP g−1 Soil h−1) | Phosphatase (µg PNP g−1 Soil h−1) | Urease (μg NH4+ g−1 Soil 2 h−1) |
|---|---|---|---|---|---|
| 0–30 cm | Before planting a cover crop. | 2022 | 59.66 (±18.34) a | 98.57 (±42.85) a | 16.53 (±4.42) a |
| A year after planting cover crops. | 2023 | 41.12 (±17.56) a | 154.52 (±83.01) a | 21.71 (±3.83) a | |
| 30–60 cm | Before planting a cover crop. | 2022 | 35.76 (±7.61) a | 82.28 (±20.10) a | 8.22 (±3.95) a |
| A year after planting cover crops. | 2023 | 24.12 (±16.50) a | 131.01 (±59.46) a | 11.72 (±5.20) a |
| Soil Depth | Sampling Time | Cover Crops | β-Glucosidase (µg PNP g−1 Soil h−1) | Acid Phosphatase (µg PNP g−1 Soil h−1) | Urease (μg NH4+ g−1 Soil 2 h−1) |
|---|---|---|---|---|---|
| 0–30 cm | After one year | Medics | 41.60 (±22.59) a | 173.29 (±134.50) a | 20.29 (±8.85) a |
| Vetch | 47.78 (±29.55) a | 156.24 (±90.129) a | 25.77 (±14.56) a | ||
| Oats | 36.06 (±26.81) a | 154.14 (±105.35) a | 21.97 (±10.78) a | ||
| Control | 39.02 (±32.522) a | 134.40 (± 120.61) a | 18.83 (±11.15) a | ||
| 30–60 cm | After one year | Medics | 19.75 (±14.04) a | 132.76 (±116.71) a | 7.30 (±4.86) b |
| Vetch | 24.62 (±17.74) a | 132.36 (±98.01)) a | 13.96 (±7.49) a | ||
| Oats | 28.67 (±34.63) a | 127.45 (±90.76) a | 13.17 (±8.93) a | ||
| Control | 23.50 (±23.89) a | 131.46 (±131.46) a | 12.46 (±7.65) a |
| Variables | Soil Carbon % | Soil Moisture % | β-Glucosidase (µg PNP g−1 Soil h−1) | Acid Phosphatase (µg PNP g−1 Soil h−1) | Urease (μg NH4+ g−1 Soil 2 h−1) |
|---|---|---|---|---|---|
| Soil carbon | 1 | −0.176 | 0.348 | 0.258 | 0.296 |
| Soil moisture | −0.176 | 1 | −0.175 | 0.114 | −0.439 |
| β-glucosidase | 0.348 | −0.175 | 1 | 0.578 | 0.777 |
| Acid phosphatase | 0.258 | 0.114 | 0.578 | 1 | 0.341 |
| Urease | 0.296 | −0.439 | 0.777 | 0.341 | 1 |
| Variables | Soil Carbon % | Soil Moisture % | β-Glucosidase (µg PNP g−1 Soil h−1) | Acid Phosphatase (µg PNP g−1 Soil h−1) | Urease (μg NH4+ g−1 Soil 2 h−1) | Cover Crop Dry Weight (g m−2) | Weed Dry Weight (g m−2) |
|---|---|---|---|---|---|---|---|
| Soil carbon | 1 | −0.053 | 0.226 | −0.009 | −0.139 | −0.538 | 0.237 |
| Soil moisture | −0.053 | 1 | −0.177 | −0.255 | −0.422 | 0.172 | 0.203 |
| β-glucosidase | 0.226 | −0.177 | 1 | 0.487 | 0.861 | −0.068 | −0.043 |
| Acid phosphatase | −0.009 | −0.255 | 0.487 | 1 | 0.313 | −0.452 | −0.307 |
| Urease | −0.139 | −0.422 | 0.861 | 0.313 | 1 | 0.202 | −0.156 |
| Cover crop dry weight | −0.538 | 0.172 | −0.068 | −0.452 | 0.202 | 1 | −0.253 |
| Weed dry weight | 0.237 | 0.203 | −0.043 | −0.307 | −0.156 | −0.253 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silwana, S.; Mulidzi, A.R.; Jovanovic, N. Short-Term Effects of Cover Crop Species and Termination Methods on Soil pH and Key Enzymatic Activities (β-Glucosidase, Phosphatase and Urease Activities) in a Citrus Orchard (Eureka Lemons). Horticulturae 2025, 11, 1014. https://doi.org/10.3390/horticulturae11091014
Silwana S, Mulidzi AR, Jovanovic N. Short-Term Effects of Cover Crop Species and Termination Methods on Soil pH and Key Enzymatic Activities (β-Glucosidase, Phosphatase and Urease Activities) in a Citrus Orchard (Eureka Lemons). Horticulturae. 2025; 11(9):1014. https://doi.org/10.3390/horticulturae11091014
Chicago/Turabian StyleSilwana, Sibongiseni, Azwimbavhi Reckson Mulidzi, and Nebo Jovanovic. 2025. "Short-Term Effects of Cover Crop Species and Termination Methods on Soil pH and Key Enzymatic Activities (β-Glucosidase, Phosphatase and Urease Activities) in a Citrus Orchard (Eureka Lemons)" Horticulturae 11, no. 9: 1014. https://doi.org/10.3390/horticulturae11091014
APA StyleSilwana, S., Mulidzi, A. R., & Jovanovic, N. (2025). Short-Term Effects of Cover Crop Species and Termination Methods on Soil pH and Key Enzymatic Activities (β-Glucosidase, Phosphatase and Urease Activities) in a Citrus Orchard (Eureka Lemons). Horticulturae, 11(9), 1014. https://doi.org/10.3390/horticulturae11091014







