The Production of Useful Phenol Compounds with Antioxidant Potential in Gametophytes and Sporophytes from In Vitro Cultures in Four Ornamental Ferns Species
Abstract
1. Introduction
2. Materials and Methods
2.1. In Vitro Sporophyte and Gametophyte Cultures
Biomass Production Evaluation
2.2. Biochemical Analyses
2.2.1. Determination of Total Phenolic Content (PC)
2.2.2. Determination of Total Flavonoid Content (FC)
2.2.3. Determination of the Total Content of Triterpenoids (TT)
2.2.4. Determination of Total Tannin Content (TC)
2.2.5. Assessment of Antioxidant Activity through the DPPH, FRAP, CUPRAC, and TEAC Assays
2.2.6. Quantitative Determinations Using the HPLC-DAD Analysis
2.2.7. Chemicals
2.2.8. Statistical Analysis
3. Results
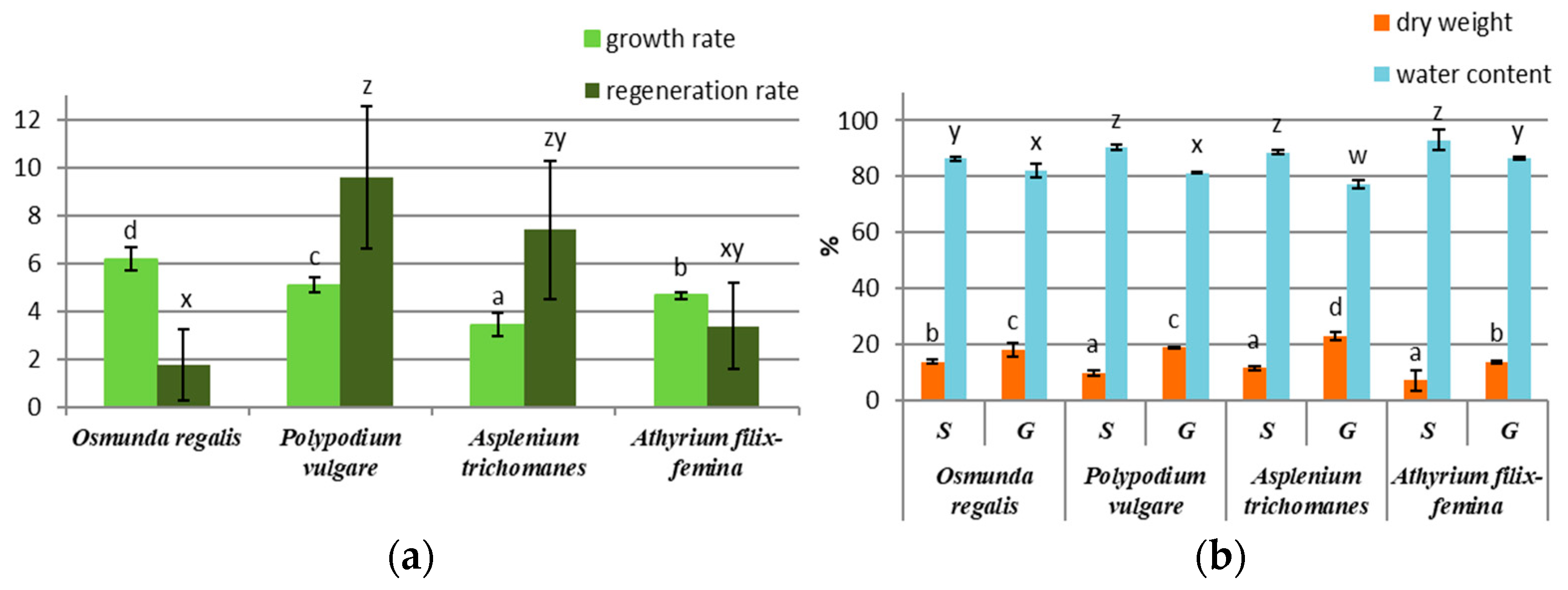
3.1. Gametophyte and Sporophyte Biomass Production
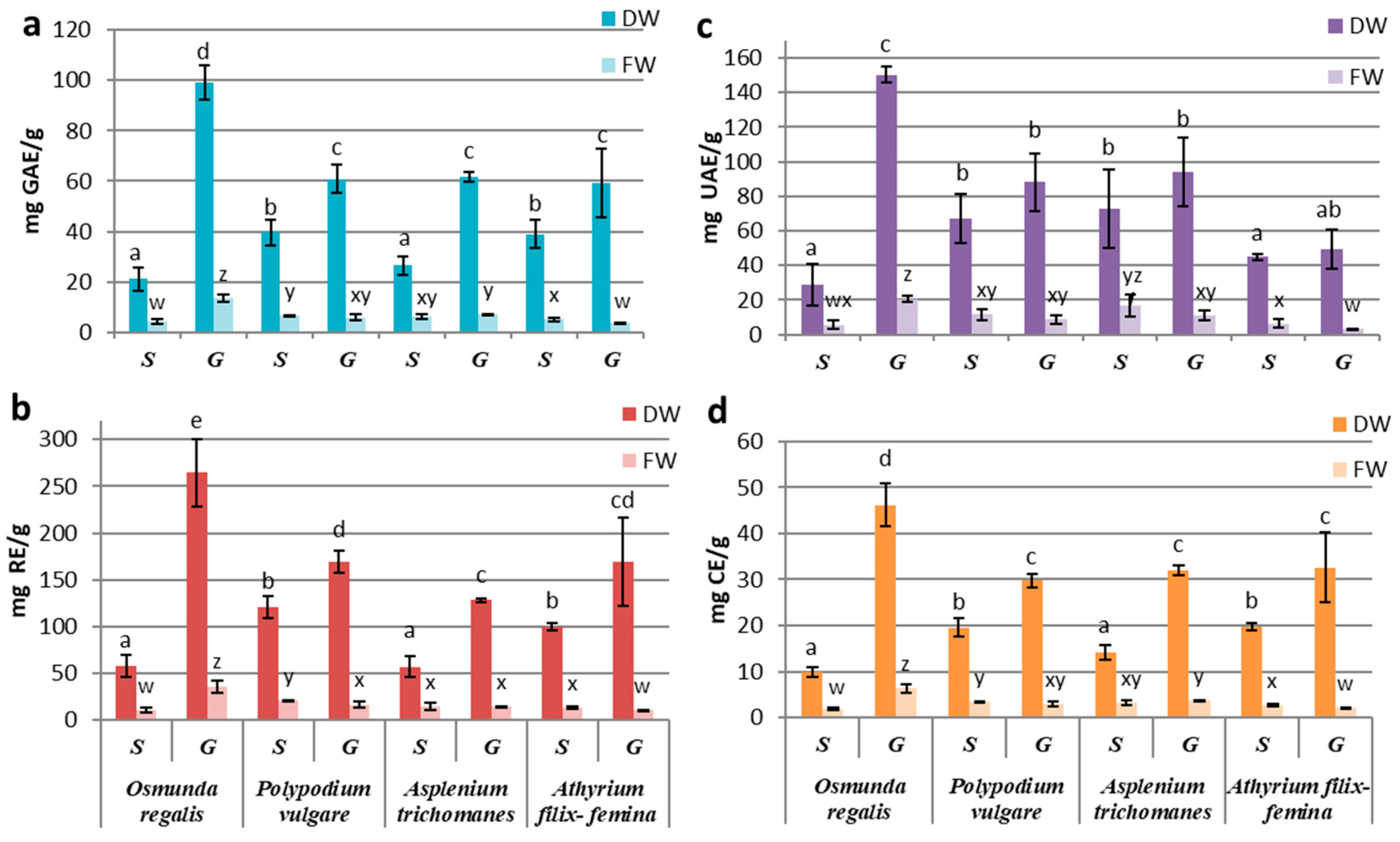
3.2. Determination of Polyphenols (PC), Flavonoids (FC), Tannins (TC), and Triterpenoids (TT) Contents
3.3. Antioxidant Activities Evaluation by DPPH, FRAP, CUPRAC, and TEAC Methods and Correlations with the Contents of Metabolites
3.4. HP-TLC Analyses
3.5. Quantitative HPLC–UV–VIS–DAD Determinations of Some Known Phytoterapeutic Chemicals
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fernández, H.; Revilla, M.A. In vitro culture of ornamental ferns. Plant Cell Tissue Org. Cult. 2003, 73, 1–13. [Google Scholar] [CrossRef]
- Ojosnegros, S.; Alvarez, J.M.; Grossmann, J.; Gagliardini, V.; Quintanilla, L.G.; Grossniklaus, U.; Fernández, H. Proteome and Interactome Linked to Metabolism, Genetic Information Processing, and Abiotic Stress in Gametophytes of Two Woodferns. Int. J. Mol. Sci. 2023, 24, 12429. [Google Scholar] [CrossRef]
- Langhansová, L.; Pumprová, K.; Haisel, D.; Ekrt, L.; Pavicic, A.; Zajíčková, M.; Vaněk, T.; Dvořáková, M. European ferns as rich sources of antioxidants in the human diet. Food Chem. 2021, 356, 129637. [Google Scholar] [CrossRef] [PubMed]
- Dvorakova, M.; Pumprova, K.; Antoninova, Z.; Rezek, J.; Haisel, D.; Ekrt, L.; Vanek, T.; Langhansova, L. Nutritional and Antioxidant Potential of Fiddleheads from European Ferns. Foods 2021, 10, 460. [Google Scholar] [CrossRef] [PubMed]
- Farràs, A.; Mitjans, M.; Maggi, F.; Caprioli, G.; Vinardell, M.P.; López, V. Polypodium vulgare L. (Polypodiaceae) as a Source of Bioactive Compounds: Polyphenolic Profile, Cytotoxicity and Cytoprotective Properties in Different Cell Lines. Front. Pharmacol. 2021, 12, 727528. [Google Scholar] [CrossRef] [PubMed]
- Farràs, A.; Cásedas, G.; Les, F.; Terrado, E.M.; Mitjans, M.; López, V. Evaluation of Anti-Tyrosinase and Antioxidant Properties of Four Fern Species for Potential Cosmetic Applications. Forests 2019, 10, 179. [Google Scholar] [CrossRef]
- Farràs, A.; Mitjans, M.; Maggi, F.; Caprioli, G.; Vinardell, M.P.; López, V. Exploring wild Aspleniaceae ferns as safety sources of polyphenols: The case of Asplenium trichomanes L. and Ceterach officinarum Willd. Front. Nutr. 2022, 12, 994215. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency (EMA). Assessment Report on Polypodium vulgare L.; Rizoma European Medicines Agency: London, UK, 2008; p. 22. [Google Scholar]
- Gleńsk, M.; Dudek, M.K.; Ciach, M.; Wlodarczyk, M. Isolation and structural determination of flavan-3-ol derivatives from the Polypodium vulgare L. rhizomes water extract. Nat. Prod. Res. 2019, in press. [Google Scholar] [CrossRef]
- Leporatti, M.L.; Corradi, L. Ethnopharmacobotanical remarks on the province of Chieti town (Abruzzo, central Italy). J. Ethnopharmacol. 2001, 74, 17–40. [Google Scholar] [CrossRef] [PubMed]
- Bonet, M.A.; Vallès, J. Ethnobotany of Montseny Biosphere reserve (Catalonia, Iberian Peninsula): Plants Used in Veterinary Medicine. J. Ethnopharmacol. 2007, 110, 30–47. [Google Scholar] [CrossRef]
- Jizba, J.; Dolejš, L.; Herout, V.; Šorm, F. The structure of osladin—The sweet principle of the rhizomes of Polypodium vulgare L. Tetrahedron Lett. 1971, 12, 1329–1332. [Google Scholar] [CrossRef]
- Prakash, C.V.S.; Prakash, I. Isolation and structural characterization of lupine triterpenes from Polypodium Vulgare. Res. J. Pharm. Sci. 2012, 1, 23–27. Available online: http://www.isca.in/IJPS/Archive/v1i1/5.ISCA-RJPcS-2012-009.pdf (accessed on 15 June 2024).
- Jizba, J.; Herout, V. Plant substances. XXVI. Isolation of constituents of common polypody rhizomes (Polypodium vulgare L.). Collect. Czech. Chem. Commun. 1967, 32, 2867–2874. [Google Scholar] [CrossRef]
- Arai, Y.; Yamaide, M.; Yamazaki, S.; Ageta, H. Fern constituents–Triterpenoids isolated from Polypodium-vulgare, Polypodium-fauriei and Polypodium-virginianum. Phytochemistry 1991, 30, 3369–3377. [Google Scholar] [CrossRef]
- Messeguer, J.; Mele, E.; Reixach, N.; Irurre-Santilari, J.; Casas, J. Polypodium vulgare L.(Wood Fern): In vitro cultures and the production of phytoecdysteroids. In Biotechnology in Agriculture and Forestry, Medicinal and Aromatic Plants X; Bajaj, Y.P.S., Ed.; Springer: Berlin, Germany, 1998; Volume 41, pp. 333–348. [Google Scholar]
- Simon, A.; Vanyolos, A.; Beni, Z.; Dekany, M.; Toth, G.; Bathori, M. Ecdysteroids from Polypodium vulgare L. Steroids 2011, 76, 1419–1424. [Google Scholar] [CrossRef] [PubMed]
- Bahadori, M.B.; Kordi, F.M.; Ahmadi, A.A.; Bahadori, S.; Valizadeh, H. Antibacterial evaluation and preliminary phytochemical screening of selected ferns from Iran. Res. J. Pharmacogn. 2015, 2, 53–59. [Google Scholar]
- Sofiane, G.; Wafa, N.; Ouarda, D. Antioxidant, antimicrobial and anti-inflammatory activities of flavonoids and tannins extracted from Polypodium vulgare L. Asian. J. Biochem. Pharm. Res. 2015, 5, 114–122. [Google Scholar]
- Batur, S.; Ayla, S.; Sakul, A.A.; Okur, M.E.; Karadag, A.E.; Daylan, B.; Ozdemir, E.M.; Kepil, N.; Gunal, M.Y. An Alternative Approach Wound Healing Field with Polypodium Vulgare. Medeni Med. J. 2020, 35, 315–323. [Google Scholar] [CrossRef]
- Saeedi, M.; Babaie, K.; Karimpour-Razkenari, E.; Vazirian, M.; Akbarzadeh, T.; Khanavi, M.; Hajimahmoodi, M.; Shams Ardekani, M.R. In Vitro Cholinesterase Inhibitory Activity of Some Plants Used in Iranian Traditional Medicine. Nat. Prod. Res. 2017, 31, 2690–2694. [Google Scholar] [CrossRef]
- Rigat, M.; Vallès, J.; Iglésias, J.; Garnatje, T. Traditional and alternative natural therapeutic products used in the treatment of respiratory tract infectious diseases in the eastern Catalan Pyrenees (Iberian Peninsula). J. Ethnopharmacol. 2013, 148, 411–422. [Google Scholar] [CrossRef]
- Bonet, M.À.; Agelet, A.; Vallès, J.; Villar Pérez, L. Contribution à la connaissance ethnobotanique des ptéridophytes dans les Pyrénées. Bocconea 2001, 13, 605–612. Available online: http://www.herbmedit.org/bocconea/13-605.pdf (accessed on 15 June 2024).
- Fiorin, E.; Sáez, L.; Malgosa, A. Ferns as healing plants in medieval Mallorca, Spain? Evidence from human dental calculus. Int. J. Osteoarchaeol. 2019, 29, 82–90. [Google Scholar] [CrossRef]
- Imperato, F. Two novel flavonol glycosides from the fern Cheilanthes fragrans. Tetrahedron 1989, 45, 215–218. [Google Scholar] [CrossRef]
- Durdevic, L.; Mitrovic, M.; Pavlovic, P.; Bojovic, S.; Jaric, S.; Oberan, L.; Gajic, G.; Kostic, O. Total phenolics and phenolic acids content in leaves, rhizomes and rhizosphere soil under Ceterach officinarum D.C., Asplenium trichomanes L., and A. adiantum-nigrum L. in The Gorge of Sicevo (Serbia). Ekológia 2007, 26, 164–173. [Google Scholar]
- Froissard, D.; Fons, F.; Bessière, J.M.; Buatois, B.; Rapior, S. Volatiles of French ferns and “fougère” scent in perfumery. Nat. Prod. Commun. 2011, 6, 1723–1726. [Google Scholar] [CrossRef] [PubMed]
- Practical Plants. Available online: https://practicalplants.org/wiki/athyrium_filix-femina (accessed on 15 June 2024).
- Native American Ethnobotany Database. Available online: http://naeb.brit.org/uses/search/?string=Athyrium+filix-femina (accessed on 15 June 2024).
- Fons, F.; Froissard, D.; Bessière, J.-M.; Buatois, B.; Rapior, S. Biodiversity of Volatile Organic Compounds from Five French Ferns. Nat. Prod. Commun. 2010, 5, 1934578X1000501028. [Google Scholar] [CrossRef]
- Soare, L.C.; Ferdes, M.; Stefanov, S.; Denkova, Z.; Nicolova, R.; Denev, P.; Bejan, C.; Paunescu, A. Antioxidant activity, polyphenols content and antimicrobial activity of several native pteridophytes of Romania. Not. Bot. Hort. Agrobot. Cluj Napoca 2012, 40, 53–57. Available online: https://www.notulaebotanicae.ro/index.php/nbha/article/view/6648/6816 (accessed on 15 June 2024). [CrossRef]
- Valizadeh, H.; Sonboli, A.; Kordi, F.M.; Dehghan, H.; Bahadori, M.B. Cytotoxicity, antioxidant activity and phenolic content of eight fern species, from north of Iran. Pharm. Sci. 2015, 21, 18–24. [Google Scholar] [CrossRef]
- IUCN. The IUCN Red List of Threatened Species 2014: e.T164368A63306495. Available online: https://doi.org/10.2305/IUCN.UK.2014-2.RLTS.T164368A63306495.en (accessed on 19 June 2024).
- Makowski, D.; Tomiczak, K.; Rybczyński, J.J.; Mikuła, A. Integration of tissue culture and cryopreservation methods for propagation and conservation of the fern Osmunda regalis L. Acta Physiol. Plant. 2016, 38, 19. [Google Scholar] [CrossRef]
- Boșcaiu, N.; Coldea, G.; Horeanu, C. The Red list of extinct, endangered, vulnerable and rare vascular plants from Romania’s flora. Ocrot. Nat. Med. Înconj. 1994, 1, 45–56. [Google Scholar]
- Dihoru, G.; Dihoru, A. Rare, endangered and endemic plants from Romania’s flora—Red List. Acta Bot. Horti Bucharest 1993–1994, 23, 173–197. Available online: https://ahbb.unibuc.ro/wp-content/uploads/2020/03/ABHB-1993-1994-Page-177-201.pdf (accessed on 15 June 2024).
- Molina, M.; Reyes-García, V.; Pardo-de-Santayana, M. Local Knowledge and Management of the Royal Fern (Osmunda regalis L.) in Northern Spain: Implications for Biodiversity Conservation. Am. Fern J. 2009, 99, 45–55. [Google Scholar] [CrossRef]
- Hartwell, J.L. Plants Used against Cancer; Quartermain Publishing: Lawrence, MA, USA, 1982; p. 694. [Google Scholar]
- Schmidt, M.Q.; David, J.; Yoshida, E.J.; Scher, K.; Mita, A.; Shiao, S.L.; Ho, A.S.; Zumsteg, Z.S. Predictors of survival in head and neck mucosal melanoma. Oral Oncol. 2017, 73, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Chen, K.; Li, B.; Tian, Y.; Liu, S.; Dong, J. Chemical constituents, cytotoxic and antioxidant activities of extract from the rhizomes of Osmunda japonica Thunb. Nat. Prod. Res. 2018, 34, 847–850. [Google Scholar] [CrossRef]
- Zhu, X.-X.; Li, Y.-J.; Yang, L.; Zhang, N.; Chen, Y.; Kmoníčková, E.; Weng, X.-G.; Yang, Q.; Zídek, Z. Divergent immunomodulatory effects of extracts and phenolic compounds from the fern Osmunda japonica Thunb. Chin. J. Integr. Med. 2013, 19, 761–770. [Google Scholar] [CrossRef]
- Numata, A.; Takahashi, C.; Fujiki, R.; Kitano, E.; Kitajima, A.; Takemura, T. Plant Constituents Biologically Active to Insects. VI. Antifeedants for Larvae of the Yellow Butterfly, Eurema Hecabe Mandarina, in Osmunda Japonica. (2). Chem. Pharm. Bull. 1990, 38, 2862–2865. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Gamborg, O.; Miller, R.; Ojimi, K. Nutrient Requirements of Suspension Cultures of Soybean Root Cells. Exp. Cell Res. 1968, 50, 151–158. [Google Scholar] [CrossRef]
- Stankovic, M.S.; Niciforovic, N.; Mihailovic, V.; Topuzovic, M.; Solujic, S. Antioxidant activity, total phenolic content and flavonoid concentrations of different plant parts of Teucrium polium L. subsp. polium. Acta Soc. Bot. Pol. 2012, 81, 117–122. Available online: https://pbsociety.org.pl/journals/index.php/asbp/article/view/asbp.2012.010 (accessed on 15 June 2024). [CrossRef]
- Cai, W.; Gu, X.; Tang, J. Extraction, purification, and characterisation of the flavonoids from Opuntia milpa alta skin. Czech J. Food Sci. 2010, 28, 108–116. Available online: https://cjfs.agriculturejournals.cz/artkey/cjf-201002-0003_extraction-purification-and-characterisation-of-the-flavonoids-from-opuntia-milpa-alta-skin.php (accessed on 15 June 2024). [CrossRef]
- Ke, Z.C.; Zhu, Z.P.; Xu, Z.Y.; Fang, C.; Hu, S.Q. Response surface optimized extraction of total triterpene acids from Eriobotrya japonica (Thunb) Lindl (Loquat) leaf and evaluation of their in vitro antioxidant activities. Trop. J. Pharm. Res. 2014, 13, 787–792. Available online: https://www.ajol.info/index.php/tjpr/article/view/107603 (accessed on 15 June 2024). [CrossRef]
- Makkar, H.P.; Blümmel, M.; Borowy, N.K.; Becker, K. Gravimetric determination of tannins and their correlations with chemical and protein precipitation methods. J. Sci. Food Agric. 1993, 61, 161–165. Available online: https://scijournals.onlinelibrary.wiley.com/doi/10.1002/jsfa.2740610205 (accessed on 15 June 2024). [CrossRef]
- Marxen, K.; Vanselow, K.H.; Lippemeier, S.; Hintze, R.; Ruser, A.; Hansen, U.-P. Determination of DPPH Radical Oxidation Caused by Methanolic Extracts of Some Microalgal Species by Linear Regression Analysis of Spectrophotometric Measurements. Sensors 2007, 7, 2080–2095. [Google Scholar] [CrossRef] [PubMed]
- Simirgiotis, M.J.; Bórquez, J.; Schmeda-Hirschmann, G. Antioxidant Capacity, Polyphenolic Content and Tandem HPLC–DAD–ESI/MS Profiling of Phenolic Compounds from the South American Berries Luma Apiculata and L. Chequén. Food Chem. 2013, 139, 289–299. [Google Scholar] [CrossRef]
- Chamorro, M.F.; Reiner, G.; Theoduloz, C.; Ladio, A.; Schmeda-Hirschmann, G.; Gómez-Alonso, S.; Jiménez-Aspee, F. Polyphenol Composition and (Bio)Activity of Berberis Species and Wild Strawberry from the Argentinean Patagonia. Molecules 2019, 24, 3331. [Google Scholar] [CrossRef] [PubMed]
- Jesionek, W.; Majer-Dziedzic, B.; Choma, I.M. Separation, Identification, and Investigation of Antioxidant Ability of Plant Extract Components Using TLC, LC-MS, and TLC-DPPH. J. Liq. Chromatogr. Relat. Technol. 2015, 38, 1147–1153. [Google Scholar] [CrossRef]
- Frum, A.; Dobrea, C.M.; Rus, L.L.; Virchea, L.I.; Morgovan, C.; Chis, A.A.; Arseniu, A.M.; Butuca, A.; Gligor, F.G.; Vicas, L.G.; et al. Valorization of Grape Pomace and Berries as a New and Sustainable Dietary Supplement: Development, Characterization, and Antioxidant Activity Testing. Nutrients 2022, 14, 3065. [Google Scholar] [CrossRef] [PubMed]
- Popescu, D.I.; Frum, A.; Dobrea, C.M.; Cristea, R.; Gligor, F.G.; Vicas, L.G.; Ionete, R.E.; Sutan, N.A.; Georgescu, C. Comparative Antioxidant and Antimicrobial Activities of Several Conifer Needles and Bark Extracts. Pharmaceutics 2024, 16, 52. [Google Scholar] [CrossRef]
- Aldea, F.; Banciu, C.; Brezeanu, A.; Helepciuc, F.E.; Soare, L.C. In vitro micropropagation of fern species (Pteridophyta) of biotechnological interest, for ex situ conservation. Olten. Stud. Comunicări Ştiinţele Nat. 2016, 32, 27–35. [Google Scholar]
- Roginsky, V.; Lissi, E. Review of methods to determine chain-breaking antioxidant activity in food. Food Chem. 2005, 92, 235–254. [Google Scholar] [CrossRef]
- Graßmann, J. Terpenoids as Plant Antioxidants. Vitam. Horm. 2005, 72, 505–535. [Google Scholar] [CrossRef] [PubMed]
- Tomou, E.M.; Skaltsa, H. Phytochemical investigation of the fern Asplenium ceterach (Aspleniaceae). Nat. Prod. Commun. 2018, 13, 849–850. [Google Scholar] [CrossRef]


| Phytochemical Compounds | Osmunda regalis | Polypodium vulgare | Asplenium trichomanes | |||
|---|---|---|---|---|---|---|
| G | S | G | S | G | S | |
| Gallic acid | 46.78 ± 0.59 | 110.14 ± 0.14 | 48.95 ± 0.38 * | 83.96 ± 0.60 | 126.95 ± 0.51 * | 44.61 ± 0.52 |
| Catechin | 535.81 ± 0.36 | 673.11 ± 0.99 | 2019.56 ± 1.07 | 3504.51 ± 0.97 | - | 763.02 ± 0.20 |
| Syringic acid | - | - | - | - | 19.49 ± 0.55 | 14.53 ± 0.34 |
| Cinnamic acid | 37.59 ± 0.57 | 27.43 ± 0.51 | 48.26 ± 0.77 * | 51.00 ± 0.97 | 43.71 ± 0.74 | - |
| Resveratrol | - | - | 20.82 ± 0.62 | 58.34 ± 0.68 | 34.13 ± 0.82 | - |
| Caffeic acid | 79.80 ± 0.70 | 103.08 ± 0.95 | 26.33 ± 0.67 | - | - | - |
| Ferulic acid | 39.60 ± 0.27 | 32.26 ± 0.89 | 112.87 ± 0.82 | 152.97 ± 0.25 | 7.82 ± 0.32 | 12.52 ± 0.73 * |
| Chlorogenic acid | - | - | - | 18.40 ± 0.89 | 11.07 ± 0.52 | 7.17 ± 0.12 |
| Rutin | 18.50 ± 0.55 | 21.05 ± 0.47 | 35.94 ± 0.82 | 646.60 ± 0.93 | 247.23 ± 0.40 | 47.62 ± 0.39 |
| Quercetin | - | - | 29.30 ± 0.51 | 21.38 ± 0.89 | 128.03 ± 0.55 * | 12.88 ± 0.90 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitoi, E.M.; Aldea, F.; Helepciuc, F.E.; Ciocan, A.-G.; Frum, A.; Popescu, D.I.; Luțu, O.A.; Șuțan, N.A.; Soare, L.C. The Production of Useful Phenol Compounds with Antioxidant Potential in Gametophytes and Sporophytes from In Vitro Cultures in Four Ornamental Ferns Species. Horticulturae 2024, 10, 799. https://doi.org/10.3390/horticulturae10080799
Mitoi EM, Aldea F, Helepciuc FE, Ciocan A-G, Frum A, Popescu DI, Luțu OA, Șuțan NA, Soare LC. The Production of Useful Phenol Compounds with Antioxidant Potential in Gametophytes and Sporophytes from In Vitro Cultures in Four Ornamental Ferns Species. Horticulturae. 2024; 10(8):799. https://doi.org/10.3390/horticulturae10080799
Chicago/Turabian StyleMitoi, Elena Monica, Florentina Aldea, Florența Elena Helepciuc, Alexandra-Gabriela Ciocan, Adina Frum, Diana Ionela Popescu, Oana Alexandra Luțu, Nicoleta Anca Șuțan, and Liliana Cristina Soare. 2024. "The Production of Useful Phenol Compounds with Antioxidant Potential in Gametophytes and Sporophytes from In Vitro Cultures in Four Ornamental Ferns Species" Horticulturae 10, no. 8: 799. https://doi.org/10.3390/horticulturae10080799
APA StyleMitoi, E. M., Aldea, F., Helepciuc, F. E., Ciocan, A.-G., Frum, A., Popescu, D. I., Luțu, O. A., Șuțan, N. A., & Soare, L. C. (2024). The Production of Useful Phenol Compounds with Antioxidant Potential in Gametophytes and Sporophytes from In Vitro Cultures in Four Ornamental Ferns Species. Horticulturae, 10(8), 799. https://doi.org/10.3390/horticulturae10080799







