Advances in Purple Tea Research: Chemical Compositions, Anthocyanin Synthesis and Regulation, Processing, and Health Benefits
Abstract
1. Introduction
2. The Chemical Compositions of Purple Tea
2.1. Anthocyanins in Purple Tea
2.2. Flavonoids in Purple Tea
2.3. Other Compounds in Purple Tea
3. Biosynthesis and Regulation of Anthocyanins in Purple Tea
3.1. Biosynthetic Pathway of Anthocyanin in Purple Tea
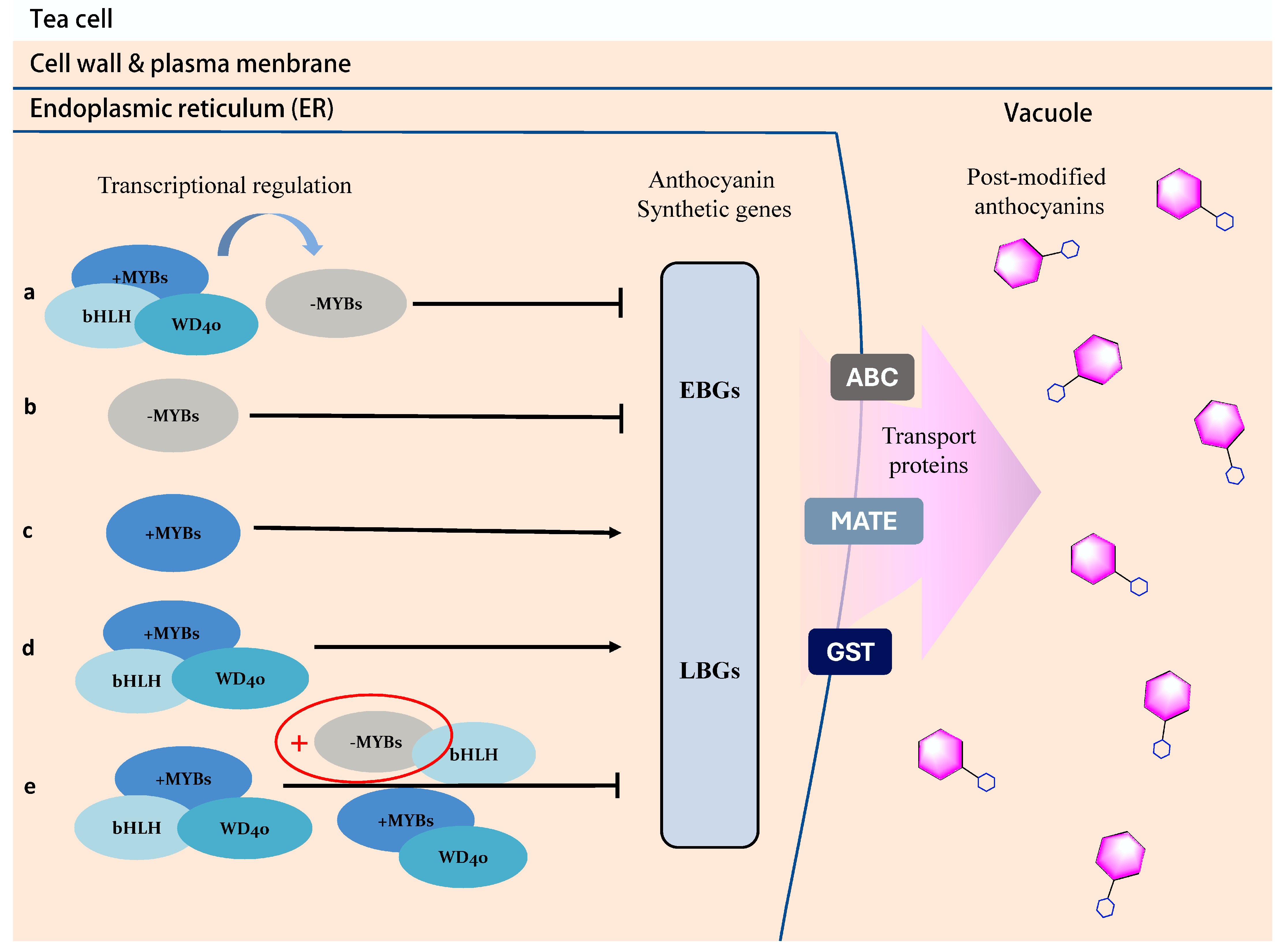
3.2. The Transcriptional Regulation of Anthocyanin in Purple Tea
| Function | Gene | Type | Impact Factors | Accession | References |
|---|---|---|---|---|---|
| positive | CsAN1/ CsMYB75 | R2R3-MYB | CsGSTF1 | CSS0010687 | [49,54,55,56] |
| positive | CsMYB5a | R2R3-MYB | / | AT2G16720 | [57] |
| positive | CsMYB5e | R2R3-MYB | / | AT3G13540 | [57] |
| positive | CsMYB5b | R2R3-MYB | CsLAR, CsANR | AT1G22640 | [58] |
| positive | CsMYB6a | R2R3-MYB | CsCHS, Cs3GT | AT1G56650 | [56] |
| positive | CsMYB113 | R2R3-MYB | CsF3H, CsCHI, CsUF3G and CsDFR | AT1G66370 | [59] |
| negative | CsMYB4a | R2R3-MYB | CsC4H, Cs4CL, CsCHS, CsLAR, and CsANR | MN894521 | [60] |
| negative | CsMYBL2a | R2R3-MYB | CsDFR, CsANR, | MW837257 | [61] |
| negative | CsMYBL2b | R2R3-MYB | CsDFR, CsANR | MW837258 | [61] |
| CsTTG1 | WD40 | interacts with MYB TFs and bHLH TFs | XM_028248215 | [62] | |
| CsGL3 | bHLH | interacts with MYB TFs and WD40 TFs | AT5G41315 | [61] | |
| CsEGL3 | bHLH | interacts with MYB TFs and WD40 TFs | AT1G63650 | [61] | |
| CsTT8 | bHLH | interacts with MYB TFs and WD40 TFs | AT4G09820 | [61] |
3.3. Other Influencing Factors
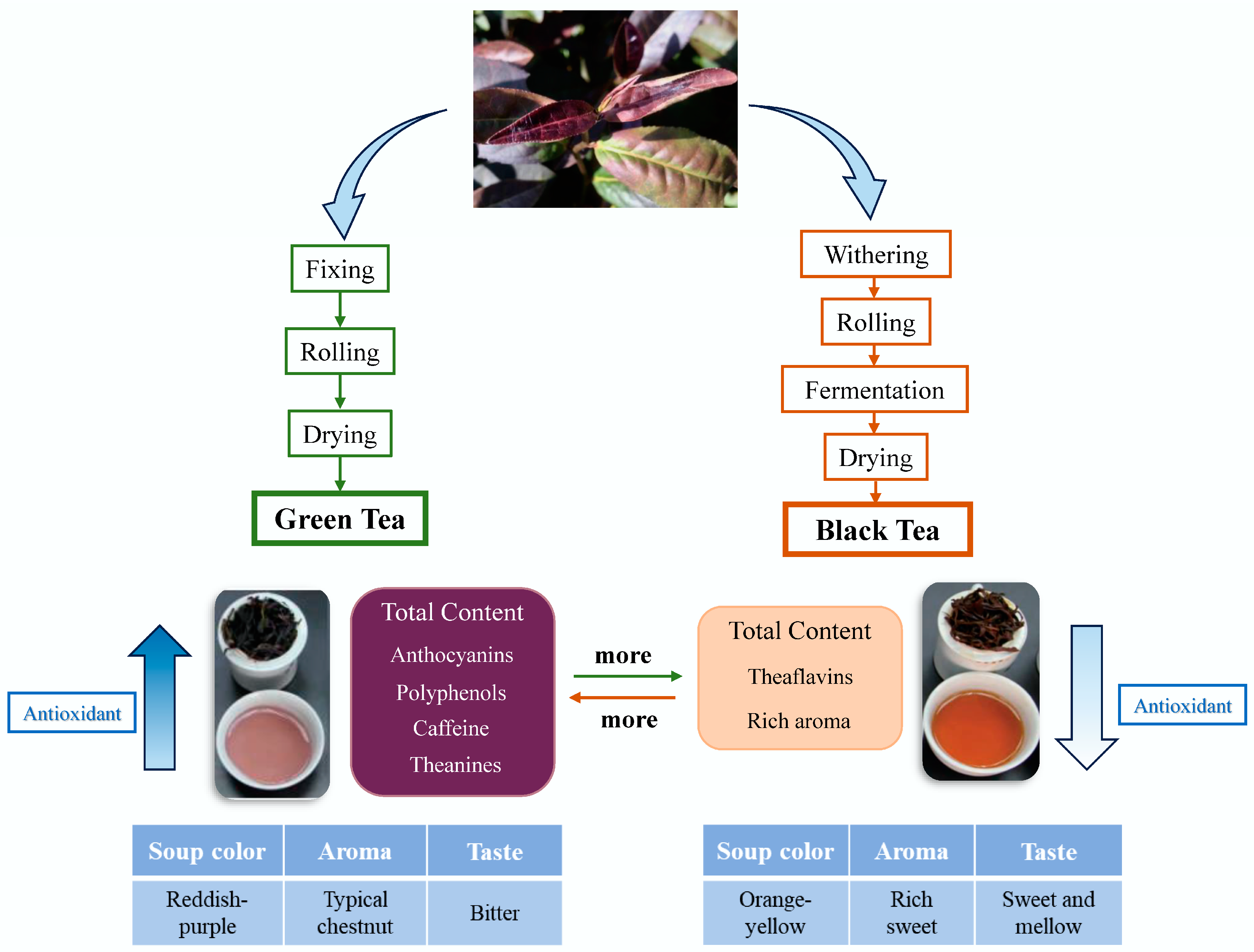
4. The Processing of Purple Tea
5. Health Benefits of Purple Tea
5.1. Antioxidant and Anti-Inflammatory
5.2. Metabolic Syndrome Regulation
5.3. Anti-Cancer
6. Conclusions and Prospect
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hazra, A.; Dasgupta, N.; Sengupta, C.; Das, S. Next generation crop improvement program: Progress and prospect in tea (Camellia sinensis (L.) O. Kuntze). Ann. Agrar. Sci. 2018, 16, 128–135. [Google Scholar] [CrossRef]
- Li, M.; Liu, Y.; Yang, Y.; An, S.; Guo, X.; Xu, T.; Hao, Q. Research advance in chemical constituents and pharmacological activities of Zijuan tea. J. Food Saf. Qual. 2019, 10, 2293–2299. [Google Scholar]
- Zhang, K.; Su, H.; Lin, Y.; Zhang, L. Research progress on the mechanism of color generation and anthocyanin accumulation in anthocyanin-rich tea. J. Food Saf. Qual. 2022, 13, 3585–3592. [Google Scholar]
- Wang, X.; Liu, B.-Y.; Zhao, Q.; Sun, X.; Li, Y.; Duan, Z.; Miao, X.; Luo, S.; Li, J. Genomic variance and transcriptional comparisons reveal the mechanisms of leaf color affecting palatability and stressed defense in tea plant. Genes 2019, 10, 929. [Google Scholar] [CrossRef] [PubMed]
- Kerio, L.C.; Wachira, F.N.; Wanyoko, J.K.; Rotich, M.K. Characterization of anthocyanins in Kenyan teas: Extraction and identification. Food Chem. 2012, 131, 31–38. [Google Scholar] [CrossRef]
- Saito, T.; Honma, D.; Tagashira, M.; Kanda, T.; Nesumi, A.; Maeda-Yamamoto, M. Anthocyanins from new red leaf tea ‘Sunrouge’. J. Agric. Food Chem. 2011, 59, 4779–4782. [Google Scholar] [CrossRef]
- Tian, Y.; Yin, Z.; Tang, Q. Water extraction process of anthocyanins from "Ziyan" tea and the antitumor activity of its extracts. J. Anhui Agric. Univ. 2019, 46, 1–7. [Google Scholar]
- Lin, Y.; Yu, W.; Li, X.; Zheng, Y.; Ye, N. Analysis on the protection status and characteristics of geographical indication products of oolong tea in Fujian province. Tea Fujian 2018, 40, 1–2. [Google Scholar]
- Wei, M.; Lu, M.; Chen, X.; Xu, B.; Lin, C.; Chen, X.; Wang, P.; Ye, N. Biological traits of three new tea tree lines including JinMingzhao survey. Tea Fujian 2021, 43, 30–32. [Google Scholar]
- Tang, X.; Sun, W.; Hong, Y.; Xie, F.; Chen, J.; Chen, Z. Differential expression of genes associated with anthocyanin synthesis in Camellia sinensis varieties with different leaf colors. J. Fujian Agric. For. Univ. Nat. Sci. Ed. 2019, 48, 742–745. [Google Scholar]
- Cai, J.; Lv, L.; Zeng, X.; Zhang, F.; Chen, Y.; Tian, W.; Li, J.; Li, X.; Li, Y. Integrative analysis of metabolomics and transcriptomics reveals molecular mechanisms of anthocyanin metabolism in the Zikui tea plant (Camellia sinensis cv. Zikui). Int. J. Mol. Sci. 2022, 23, 4780. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Deng, X.; Huang, Z.; Zhao, Z.; Li, C.; Song, Q.; He, Y.; Niu, S. Integrated transcriptome and metabolome provide insights into flavonoid biosynthesis in ‘P113’, a new purple tea of Camellia tachangensis. Beverage Plant Res. 2023, 3, 1–11. [Google Scholar] [CrossRef]
- Wu, H.; Qiao, X.; Li, J.; Chen, D.; He, Y.; Huang, H.; Liu, J.; Guan, M. Biological characters of new tea germplasms with reddishviolet shoots. Chin. J. Trop. Crops 2011, 32, 1009–1015. [Google Scholar]
- Shen, J.; Zou, Z.; Zhang, X.; Zhou, L.; Wang, Y.; Fang, W.; Zhu, X. Metabolic analyses reveal different mechanisms of leaf color change in two purple-leaf tea plant (Camellia sinensis L.) cultivars. Hortic. Res. 2018, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Terahara, N.; Takeda, Y.; Nesumi, A.; Honda, T. Anthocyanins from red flower tea (Benibana-cha), Camellia sinensis. Phytochemistry 2001, 56, 359–361. [Google Scholar] [CrossRef] [PubMed]
- Rashid, K.; Wachira, F.N.; Nyabuga, J.N.; Wanyonyi, B.; Murilla, G.; Isaac, A.O. Kenyan purple tea anthocyanins ability to cross the blood brain barrier and reinforce brain antioxidant capacity in mice. Nutr. Neurosci. 2014, 17, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, Z.; Zhu, W.; Wang, Y.; Liang, Y.; Wang, K.; Ye, J.; Lu, J.; Zheng, X. Anthocyanin metabolism and its differential regulation in purple tea (Camellia sinensis). Plant Physiol. Biochem. 2023, 201, 107875. [Google Scholar] [CrossRef]
- Wei, K.; Zhang, Y.; Wu, L.; Li, H.; Ruan, L.; Bai, P.; Zhang, C.; Zhang, F.; Xu, L.; Wang, L. Gene expression analysis of bud and leaf color in tea. Plant Physiol. Biochem. 2016, 107, 310–318. [Google Scholar] [CrossRef]
- Jiang, L.; Shen, X.; Shoji, T.; Kanda, T.; Zhou, J.; Zhao, L. Characterization and Activity of Anthocyanins in Zijuan Tea (Camellia sinensis var. kitamura). J. Agric. Food Chem. 2013, 61, 3306–3310. [Google Scholar] [CrossRef]
- Lai, Y.; Li, S.; Tang, Q.; Li, H.; Chen, S.; Li, P.; Xu, J.; Xu, Y.; Guo, X. The dark-purple tea cultivar ‘Ziyan’ accumulates a large amount of delphinidin-related anthocyanins. J. Agric. Food Chem. 2016, 64, 2719–2726. [Google Scholar] [CrossRef]
- Abdel-Aal, E.-S.M.; Rabalski, I.; Mats, L.; Rai, I. Identification and quantification of anthocyanin and catechin compounds in purple tea leaves and flakes. Molecules 2022, 27, 6676. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Xie, H.; Liu, S.; Zhu, J.; Zhao, S.; Wei, C. Metabolites and transcriptional profiling analysis reveal the molecular mechanisms of the anthocyanin metabolism in the “Zijuan” tea plant (Camellia sinensis var. assamica). J. Agric. Food Chem. 2021, 69, 414–427. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yi, B.; Li, Y.; Jiang, H.; Duan, Z.; Shang, W.; Yang, Y.; Yang, S.; Liu, B. Analysis on differences of major biochemical components of purple-bud tea tree germplasm resources. Shandong Agric. Sci. 2015, 47, 14–19. [Google Scholar]
- Tan, X.; Li, W.; Wang, C.; Huang, J.; Yang, Y.; Tang, Q. Seasonal changes of anthocyanins and main biochemical components in ‘Ziyan’ and ‘Zijuan’ tea plants. Chin. J. Trop. Crops 2021, 42, 168–174. [Google Scholar]
- Lv, H.; Dai, W.; Tan, J.; Guo, L.; Zhu, Y.; Lin, Z. Identification of the anthocyanins from the purple leaf coloured tea cultivar Zijuan (Camellia sinensis var. assamica) and characterization of their antioxidant activities. J. Funct. Foods. 2015, 17, 449–458. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, J.; Meng, Q.; Tong, H. Non-volatile metabolites profiling analysis reveals the tea flavor of “Zijuan” in different tea plantations. Food Chem. 2023, 412, 135534. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Zhang, P.; Cui, D.; Yang, X.; Zhang, D.; Yang, Y.; Chen, W.; Tang, D.; Tang, Q.; Li, P. Multi-omics analysis revealed anthocyanin accumulation differences in purple tea plants ‘Ziyan’, ‘Zijuan’ and their dark-purple hybrid. Sci. Hortic. 2023, 321, 112275. [Google Scholar] [CrossRef]
- Shi, J.; Simal-Gandara, J.; Mei, J.; Ma, W.; Peng, Q.; Shi, Y.; Xu, Q.; Lin, Z.; Lv, H. Insight into the pigmented anthocyanins and the major potential co-pigmented flavonoids in purple-coloured leaf teas. Food Chem. 2021, 363, 130278. [Google Scholar] [CrossRef]
- Zhu, M.; Zhou, F.; Ran, L.; Li, Y.; Tan, B.; Wang, K.; Huang, J.; Liu, Z. Metabolic profiling and gene expression analyses of purple-leaf formation in tea cultivars (Camellia sinensis var. sinensis and var. assamica). Front. Plant Sci. 2021, 12, 606962. [Google Scholar] [CrossRef]
- Chen, X.; Wang, P.; Zheng, Y.; Gu, M.; Lin, X.; Wang, S.; Jin, S.; Ye, N. Comparison of metabolome and transcriptome of flavonoid biosynthesis pathway in a purple-leaf tea germplasm Jinmingzao and a green-leaf tea germplasm Huangdan reveals their relationship with genetic mechanisms of color formation. Int. J. Mol. Sci. 2020, 21, 4167. [Google Scholar] [CrossRef]
- Liu, Z.; Shi, X.; Duan, S.; Nian, B.; Chen, L.; Zhang, G.; Lv, C.; Ma, Y.; Zhao, M. Multiomics analysis of the mechanisms behind flavonoid differences between purple and green tender shoots of Camellia sinensis var. assamica. G3 2022, 13, jkac297. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Hu, J.; Liu, M.; Shi, Y.; De Vos, R.C.H.; Ruan, J. Stimulated biosynthesis of delphinidin-related anthocyanins in tea shoots reducing the quality of green tea in summer. J. Sci. Food Agric. 2020, 100, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Zhang, M.; Liu, J.; Cai, J. Metabolomic and transcriptomic analyses reveal the characteristics of tea flavonoids and caffeine accumulation and regulation between Chinese varieties (Camellia sinensis var. sinensis) and Assam varieties (C. sinensis var. assamica). Genes 2022, 13, 1994. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Xia, E.; Fu, J.; Xu, Y.; Zhao, X.; Tong, W.; Tang, Q.; Tadege, M.; Fernie, A.R.; Zhao, J. Diverse roles of MYB transcription factors in regulating secondary metabolite biosynthesis, shoot development, and stress responses in tea plants (Camellia sinensis). Plant J. 2022, 110, 1144–1165. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, J.; Guan, M.; Zhao, C.; Geng, P.; Zhao, Q. Arabidopsis MYB4 plays dual roles in flavonoid biosynthesis. Plant J. 2020, 101, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Schäffner, A.R. Flavonoid biosynthesis and Arabidopsis genetics: More good music. J. Exp. Bot. 2016, 67, 1203–1204. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schwinn, K.; Miosic, S.; Davies, K.; Thill, J.; Gotame, T.P.; Stich, K.; Halbwirth, H. The B-ring hydroxylation pattern of anthocyanins can be determined through activity of the flavonoid 3′-hydroxylase on leucoanthocyanidins. Planta 2014, 240, 1003–1010. [Google Scholar] [CrossRef]
- Krol, A.R.V.D.; Mur, L.A.; Lange, P.D.; Mol, J.N.M.; Stuitje, A.R. Inhibition of flower pigmentation by antisense CHS genes: Promoter and minimal sequence requirements for the antisense effect. Plant Mol. Biol. 1990, 14, 457–466. [Google Scholar] [CrossRef]
- Kobayashi, S.; Ishimaru, M.; Hiraoka, K.; Honda, C. Myb-related genes of the Kyoho grape (Vitis labruscana) regulate anthocyanin biosynthesis. Planta 2002, 215, 924–933. [Google Scholar] [CrossRef]
- Sun, L.; Huo, J.; Liu, J.; Yu, J.; Zhou, J.; Sun, C.; Wang, Y.; Leng, F. Anthocyanins distribution, transcriptional regulation, epigenetic and post-translational modification in fruits. Food Chem. 2023, 411, 135540. [Google Scholar] [CrossRef]
- Zhang, Y.; Butelli, E.; Martin, C. Engineering anthocyanin biosynthesis in plants. Curr. Opin. Plant Biol. 2014, 19, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Provenzano, S.; Spelt, C.; Hosokawa, S.; Nakamura, N.; Brugliera, F.; Demelis, L.; Geerke, D.P.; Schubert, A.; Tanaka, Y.; Quattrocchio, F.; et al. Genetic control and evolution of anthocyanin methylation. Plant Physiol. 2014, 165, 962–977. [Google Scholar] [CrossRef] [PubMed]
- Broun, P. Transcriptional control of flavonoid biosynthesis: A complex network of conserved regulators involved in multiple aspects of differentiation in Arabidopsis. Curr. Opin. Plant Biol. 2005, 8, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Alfenito, M.R.; Souer, E.; Goodman, C.D.; Buell, R.; Mol, J.; Koes, R.; Walbot, V. Functional complementation of anthocyanin sequestration in the vacuole by widely divergent glutathione S-transferases. Plant Cell 1998, 10, 1135–1149. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xiao, Y.; Zhou, X.; Liao, Y.; Wu, S.; Chen, J.; Qian, J.; Yan, Y.; Tang, J.; Zeng, L. Characterizing the cultivar-specific mechanisms underlying the accumulation of quality-related metabolites in specific Chinese tea (Camellia sinensis) germplasms to diversify tea products. Food Res. Int. 2022, 161, 111824. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Sun, W.; Lai, Z. Differential expression of genes in purple-shoot tea tender leaves and mature leaves during leaf growth. J. Sci. Food Agric. 2016, 96, 1982–1989. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Huang, R.; Liu, L.; Li, Y.; Wang, W.; Xu, Q.; Yu, Y.; Zhou, T. CsUGT78A15 catalyzes the anthocyanidin 3-O-galactoside biosynthesis in tea plants. Plant Physiol. Biochem. 2021, 166, 738–749. [Google Scholar] [CrossRef]
- Maritim, T.K.; Masand, M.; Seth, R.; Sharma, R.K. Transcriptional analysis reveals key insights into seasonal induced anthocyanin degradation and leaf color transition in purple tea (Camellia sinensis (L.) O. Kuntze). Sci. Rep. 2021, 11, 1244. [Google Scholar] [CrossRef]
- Wei, K.; Wang, L.; Zhang, Y.; Ruan, L.; Li, H.; Wu, L.; Xu, L.; Zhang, C.; Zhou, X.; Cheng, H.; et al. A coupled role for CsMYB75 and CsGSTF1 in anthocyanin hyperaccumulation in purple tea. Plant J. 2019, 97, 825–840. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, H.; Zhao, Y.; Li, X.; Xia, T. Three Camellia sinensis glutathione S-transferases are involved in the storage of anthocyanins, flavonols, and proanthocyanidins. Planta 2019, 250, 1163–1175. [Google Scholar] [CrossRef]
- Stracke, R.; Ishihara, H.; Huep, G.; Barsch, A.; Weisshaar, B. Differential regulation of closely related R2R3-MYB transcription factors controls flavonol accumulation in different parts of the Arabidopsis thaliana seedling. Plant J. 2010, 50, 660–677. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Hu, B.; Qin, Y.; Hu, G.; Zhao, J. Advance of the negative regulation of anthocyanin biosynthesis by MYB transcription factors. Plant Physiol. Biochem. 2019, 136, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Chagné, D.; Wang, K.; Espley, R.V.; Volz, R.K.; How, N.M.; Rouse, S.; Brendolise, C.; Carlisle, C.M.; Kumar, S.; De Silva, N.; et al. An ancient duplication of apple MYB transcription factors is responsible for novel red fruit-flesh phenotypes. Plant Physiol. 2012, 161, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Duan, J.; Lei, Y.; Kang, Y.; Luo, Y.; Chen, Y.; Ding, D.; Li, S. Metabolomic and transcriptomic analyses reveal a MYB gene, CsAN1, involved in anthocyanins accumulation separation in F1 between ‘Zijuan’ (Camellia sinensis var. assamica) and ‘Fudingdabaicha’ (C. sinensis var. sinensis) tea plants. Front. Plant Sci. 2022, 13, 1008588. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Zhu, Z.; Cao, P.; Chen, H.; Chen, C.; Zhou, X.; Mao, Y.; Lei, J.; Jiang, Y.; Meng, W.; et al. Purple foliage coloration in tea (Camellia sinensis L.) arises from activation of the R2R3-MYB transcription factor CsAN1. Sci. Rep. 2016, 6, 32534. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhao, X.; Gao, L.; Shi, X.; Dai, X.; Liu, Y.; Xia, T.; Wang, Y. Isolation and characterization of key genes that promote flavonoid accumulation in purple-leaf tea (Camellia sinensis L.). Sci. Rep. 2018, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Huang, K.; Zheng, G.; Hou, H.; Wang, P.; Jiang, H.; Zhao, X.; Li, M.; Zhang, S.; Liu, Y. CsMYB5a and CsMYB5e from Camellia sinensis differentially regulate anthocyanin and proanthocyanidin biosynthesis. Plant Sci. 2018, 270, 209–220. [Google Scholar] [CrossRef]
- Wang, P.; Ma, G.; Zhang, L.; Li, Y.; Fu, Z.; Kan, X.; Han, Y.; Wang, H.; Jiang, X.; Liu, Y. A sucrose-induced MYB (SIMYB) transcription factor promoting proanthocyanidin accumulation in the tea plant (Camellia sinensis). J. Agric. Food Chem. 2019, 67, 1418–1428. [Google Scholar] [CrossRef]
- Shui, L.; Li, W.; Yan, M.; Li, H.; Guo, F. Characterization of the R2R3-MYB transcription factor CsMYB113 regulates anthocyanin biosynthesis in tea plants (Camellia sinensis). Plant Mol. Biol. Rep. 2022, 41, 46–58. [Google Scholar] [CrossRef]
- Li, M.; Li, Y.; Guo, L.; Gong, N.; Pang, Y.; Jiang, W.; Liu, Y.; Jiang, X.; Zhao, L.; Wang, Y.; et al. Functional characterization of tea (Camellia sinensis) MYB4a transcription factor using an integrative approach. Front. Plant Sci. 2017, 8, 00943. [Google Scholar] [CrossRef]
- Zhao, X.; Li, P.; Zuo, H.; Peng, A.; Lin, J.; Li, P.; Wang, K.; Tang, Q.; Tadege, M.; Liu, Z.; et al. CsMYBL2 homologs modulate the light and temperature stress-regulated anthocyanin and catechins biosynthesis in tea plants (Camellia sinensis). Plant J. 2023, 115, 1051–1070. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hou, H.; Jiang, X.; Wang, P.; Dai, X.; Chen, W.; Gao, L.; Xia, T. A WD40 repeat protein from Camellia sinensis regulates anthocyanin and proanthocyanidin accumulation through the formation of MYB–bHLH–WD40 ternary complexes. Int. J. Mol. Sci. 2018, 19, 1686. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Jeong, C.Y.; Kwon, J.; Van Kien, V.; Lee, D.; Hong, S.-W.; Lee, H. Drastic anthocyanin increase in response to PAP1 overexpression in fls1 knockout mutant confers enhanced osmotic stress tolerance in Arabidopsis thaliana. Plant Cell Rep. 2016, 35, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, X.; Zhao, Y.; Yang, J.; He, Y.; Li, G.; Ma, W.; Huang, X.; Su, J. Transcription factor PyHY5 binds to the promoters of PyWD40 and PyMYB10 and regulates its expression in red pear ‘Yunhongli No. 1’. Plant Physiol. Biochem. 2020, 154, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ahammed, G.J.; Li, Z.; Zhang, L.; Wei, J.; Shen, C.; Yan, P.; Zhang, L.; Han, W. Brassinosteroids improve quality of summer tea (Camellia sinensis L.) by balancing biosynthesis of polyphenols and amino acids. Front. Plant Sci. 2016, 7, 01304. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, L.; Li, Y.; Chen, Q.; Ye, Y.; Zhang, Y.; Luo, Y.; Sun, B.; Wang, X.; Tang, H. Effect of red and blue light on anthocyanin accumulation and differential gene expression in strawberry (Fragaria × ananassa). Molecules 2018, 23, 820. [Google Scholar] [CrossRef]
- Wang, L.; Pan, D.; Liang, M.; Abubakar, Y.S.; Li, J.; Lin, J.; Chen, S.; Chen, W. Regulation of Anthocyanin Biosynthesis in Purple Leaves of Zijuan Tea (Camellia sinensis var. kitamura). Int. J. Mol. Sci. 2017, 18, 833. [Google Scholar] [CrossRef]
- Lin, J.; Liu, F.; Zhou, X.; Tu, Z.; Chen, L.; Wang, Y.; Yang, Y.; Wu, X.; Lv, H.; Zhu, H.; et al. Effect of red light on the composition of metabolites in tea leaves during the withering process using untargeted metabolomics. J. Sci. Food Agric. 2022, 102, 1628–1639. [Google Scholar] [CrossRef]
- Shirin, A.; Zhang, Y.; Mao, P.; Lei, Y.; Bai, P.; Wang, Y.; Ruan, L.; Xun, H.; Wu, L.; Cheng, H.; et al. Responses of secondary metabolites and transcriptomes in the tea cultivar ‘Zhong Ming 6’ (Camellia sinensis) to blue light and red light. Plant Growth Regul. 2022, 98, 343–358. [Google Scholar] [CrossRef]
- Zheng, T.; Tan, W.; Yang, H.; Zhang, L.; Li, T.; Liu, B.; Zhang, D.; Lin, H. Regulation of anthocyanin accumulation via MYB75/HAT1/TPL-mediated transcriptional repression. PLoS Genet. 2019, 15, e1007993. [Google Scholar] [CrossRef]
- Yu, S.; Li, P.; Zhao, X.; Tan, M.; Ahmad, M.Z.; Xu, Y.; Tadege, M.; Zhao, J. CsTCPs regulate shoot tip development and catechin biosynthesis in tea plant (Camellia sinensis). Hortic. Res. 2021, 8, 104. [Google Scholar] [CrossRef] [PubMed]
- Ke, S.-W.; Chen, G.; Chen, C.; Tzen, J.T.C.; Yang, C. Ethylene signaling modulates contents of catechin and ability of antioxidant in Camellia sinensis. Bot. Stud. 2018, 59, 11. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Zhang, S.; Xiong, L.; Zhou, J.; Huang, J.; Zhao, A.; Liu, Z.; Liu, A. A comprehensive review of polyphenol oxidase in tea (Camellia sinensis): Physiological characteristics, oxidation manufacturing, and biosynthesis of functional constituents. Compr. Rev. Food Sci. Food Saf. 2023, 22, 2267–2291. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, L.; Sheng, Y.; Wang, K.; Shi, Y.; Liang, Y.; Zheng, X. Screening tea hybrid with abundant anthocyanins and investigating the effect of tea processing on foliar anthocyanins in tea. Folia Hortic. 2020, 32, 279–290. [Google Scholar] [CrossRef]
- Joshi, R.; Rana, A.; Gulati, A. Studies on quality of orthodox teas made from anthocyanin-rich tea clones growing in Kangra valley, India. Food Chem. 2015, 176, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Kerio, L.C.; Wachira, F.N.; Wanyoko, J.K.; Rotich, M.K. Total polyphenols, catechin profiles and antioxidant activity of tea products from purple leaf coloured tea cultivars. Food Chem. 2013, 136, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Cao, B.; Wang, Q.; Qin, D.; Pan, C.; Li, B.; Li, H.; Fang, K.; Ni, E.; Jiang, X.; et al. Research on the suitability of teas made from fresh tea leaves of purple tea. Sci. Technol. Food Ind. 2022, 43, 279–288. [Google Scholar]
- Wei, L.; Lai, Z.; Deng, H.; Luo, X.; Qiu, Y.; Zhuge, T.; Huang, J. Study on the suitability of different tea types of the tea tree species Zijuan. Agric. Technol. 2023, 43, 4–6. [Google Scholar]
- Zhou, Q.; Chen, Z.; Lee, J.; Li, X.; Sun, W. Proteomic analysis of tea plants (Camellia sinensis) with purple young shoots during leaf development. PLoS ONE 2017, 12, 0177816. [Google Scholar] [CrossRef]
- Wang, X.; Zeng, X.; Qin, C.; Yan, X.; Chen, X.; Zhang, L.; Zhou, Y. Herbaspirillum sp. ZXN111 colonization characters to different tea cultivars and the effects on tea metabolites profiling on Zijuan (Camellia sinensis var. assamica). J. Agric. Food Chem. 2023, 71, 5283–5292. [Google Scholar] [CrossRef]
- Mohammed, H.A.; Khan, R.A. Anthocyanins: Traditional uses, structural and functional variations, approaches to increase yields and products’ quality, hepatoprotection, liver longevity, and commercial products. Int. J. Mol. Sci. 2022, 23, 2149. [Google Scholar] [CrossRef] [PubMed]
- Hou, D.; Yanagita, T.; Uto, T.; Masuzaki, S.; Fujii, M. Anthocyanidins inhibit cyclooxygenase-2 expression in LPS-evoked macrophages: Structure–activity relationship and molecular mechanisms involved. Biochem. Pharmacol. 2005, 70, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Horio, F.; Osawa, T. Cyanidin 3-O-β-D-glucoside Suppresses Nitric Oxide Production during a Zymosan Treatment in Rats. J. Nutr. Sci. Vitaminol. 2002, 48, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Rana, A.; Kumar, V.; Kumar, D.; Padwad, Y.S.; Yadav, S.K.; Gulati, A. Anthocyanins enriched purple tea exhibits antioxidant, immunostimulatory and anticancer activities. J. Food Sci. Technol. 2017, 54, 1953–1963. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Lin, H.; Chang, H.; Chuang, L.; Hsieh, C.; Lu, S.; Hung, C.; Chang, J. Prophylactic effects of purple shoot green tea on cytokine immunomodulation through scavenging free radicals and NO in LPS-stimulated macrophages. Curr. Issues Mol. Biol. 2022, 44, 3980–4000. [Google Scholar] [CrossRef] [PubMed]
- de Moura, C.; Kabbas Junior, T.; Pedreira, F.R.d.O.; Azevedo, L.; Furtado, M.M.; Sant’Ana, A.S.; Franchin, M.; Gonzaga, V.R.; Cui, Y.; Wen, M.; et al. Purple tea (Camellia sinensis var. assamica) leaves as a potential functional ingredient: From extraction of phenolic compounds to cell-based antioxidant/biological activities. Food Chem. Toxicol. 2022, 159, 112668. [Google Scholar] [CrossRef]
- van Zwieten, P.A.; Mancia, G. Background and treatment of metabolic syndrome: A therapeutic challenge. Semin. Cardiothorac. Vasc. Anesth. 2006, 10, 206–214. [Google Scholar] [CrossRef]
- Shimoda, H.; Hitoe, H.; Nakamura, S.; Matsuda, H. Purple tea and its extract suppress diet-induced fat accumulation in mice and human subjects by inhibiting fat absorption and enhancing hepatic carnitine palmitoyltransferase expression. Int. J. Biomed. Sci. 2015, 11, 67–75. [Google Scholar] [CrossRef]
- da Silva, T.B.V.; Castilho, P.A.; Sá-Nakanishi, A.B.d.; Seixas, F.A.V.; Dias, M.I.; Barros, L.; Ferreira, I.C.F.R.; Bracht, A.; Peralta, R.M. The inhibitory action of purple tea on in vivo starch digestion compared to other Camellia sinensis teas. Food Res. Int. 2021, 150, 110781. [Google Scholar] [CrossRef]
- da Silva, T.B.V.; Dias, M.I.; Pereira, C.; Mandim, F.; Ivanov, M.; Soković, M.; Ferreira, I.; Barros, L.; Seixas, F.A.V.; Bracht, A.; et al. Purple tea: Chemical characterization and evaluation as inhibitor of pancreatic lipase and fat digestion in mice. Food Funct. 2023, 14, 1761–1772. [Google Scholar] [CrossRef]
- Lin, Y.; Lu, H.; Chen, J.; Huang, H.; Chen, Y.; Su, Y.; Tung, C.; Huang, C. Purple-leaf tea (Camellia sinensis L.) ameliorates high-fat diet induced obesity and metabolic disorder through the modulation of the gut microbiota in mice. BMC Complement. Med. Ther. 2020, 20, 376. [Google Scholar] [CrossRef] [PubMed]
- Mbuthia, K.S.; Mireji, P.O.; Ngure, R.M.; Stomeo, F.; Kyallo, M.; Muoki, C.; Wachira, F.N. Tea (Camellia sinensis) infusions ameliorate cancer in 4TI metastatic breast cancer model. BMC Complement. Altern. Med. 2017, 17, 202. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.; Shih, Y.; Lin, B.; Chiu, C.; Lin, C. Inhibitory Effect and Mechanisms of an Anthocyanins- and Anthocyanidins-Rich Extract from Purple-Shoot Tea on Colorectal Carcinoma Cell Proliferation. J. Agric. Food Chem. 2012, 60, 3686–3692. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Hsu, C.; Chen, C.; Liao, T.; Chiu, C.; Lien, L.P.J.; Shih, Y. Anti-proliferation and radiation-sensitizing effect of an anthocyanidin-rich extract from purple-shoot tea on colon cancer cells. J. Food Drug Anal. 2012, 20, 328–331. [Google Scholar] [CrossRef]
- de Moura, C.; Kabbas, T.; Mendanha Cruz, T.; Boscacci Marques, M.; Araújo Vieira do Carmo, M.; Turnes Pasini Deolindo, C.; Daguer, H.; Azevedo, L.; Xu, Y.-Q.; Granato, D. Sustainable and effective approach to recover antioxidant compounds from purple tea (Camellia sinensis var. assamica cv. Zijuan) leaves. Food Res. Int. 2023, 164, 112402. [Google Scholar] [CrossRef]
- Bashir, A.; Khan, F.; Al Mughairbi, F. Purple tea composition and inhibitory effect of anthocyanin-rich extract on cancer cell proliferation. Front Hum. Neurosci. 2019, 229, 00028. [Google Scholar] [CrossRef]
- Maeda-Yamamoto, M.; Nishimura, M.; Kitaichi, N.; Nesumi, A.; Monobe, M.; Nomura, S.; Horie, Y.; Tachibana, H.; Nishihira, J. A randomized, placebo-controlled study on the safety and efficacy of daily ingestion of green tea (Camellia sinensis L.) cv. “Yabukita” and “Sunrouge” on eyestrain and blood pressure in healthy adults. Nutrients 2018, 10, 569. [Google Scholar] [CrossRef]



| Samples | Detection Method | Anthocyanin Compounds | References |
|---|---|---|---|
| Zijuan from Yunnan Province | HPLC, LC-ESI-MS | Delphinidin-3-O-β-D-galactoside, Cyanidin-3-O-β-D-galactoside, Delphinidin-3-O-β-D-(6-(E)-p-coumaroyl) galactopyranoside, Cyanidin-3-O-β-D-(6-(E)-p-coumaroyl) galactopyranoside | [19] |
| Zijuan from Yunnan Province | HPLC and LC-MS | Pelargonidin-3,5-diglucoside, Cyanidin-3-O-galactoside, Cyanidin-3-O-glucoside, Delphinidin, Cyanidin, Pelargonidin, Peonidin, Malvidin | [25] |
| Zijuan from different provinces of China(Yunnan, Qijiang, and Ersheng) | UPLC-ESI-MS/MS | Delphinidin-3-O-sambubioside-5-O-glucoside, Delphinidlin-3-O-(6-O-malonyl-beta-D-glucoside),Delphinidin-3-O-(6-O-p-coumaroy)-glucaside, Delphinidin-3-O-galactoside, Delphinidin-3-O-glucoside, Delphinidin-3-O-rutinoside, Delphinidin-3,5-O-diglucoside, Delphinidin-3-O-rutinoside-5-O-glucoside, Delphinidin-3-O-sophoroside, Delphinidin, Cyanidin-3-(6-O-p-caffeoyl)-glucoside, Cyanidin-3-O-xyloside, Cyanidin-3-O-rutinoside, Cyanidin-3-O-(6-O-p-coumaroyl-glucoside, Cyanidin-3-O-galactoside, Cyanidin-3-O-arabinoside, Cyanidin-3-O-glucoside, Pelargonidin-3-O-(6-O-p-coumaroyl)-glucoside, Pelargonidin-3-O-arabinoside, Pelargonidin-3,5-O-diglucoside, Pelargonidin-3-sophoroside-5-glucoside, Pelargonidin-3-O-galactoside, Pelargonidin-3-O-rutinoside, Pelargonidin-3-O-sambubioside-5-O-glucoside, Peonidin-3-O-(6-O-p-coumaroyl)-glucoside, Peonidin-3-O-rutinoside, Peonidin-3-O-arabinoside, Peonidin-3,5-O-diglucoside, Petunidin-3-O-sambubioside-5-O-glucoside, Petunidin-3-O-arabinoside, Petunidin-3-O-glucoside, Petunidin-3-O-sambubioside, Petunidin-3-O-galactoside, Malvidin-3-O-galactoside | [26] |
| Ziyan from Sichuan Province | HPLC | Delphinidin, Cyanidin, Pelargonidin | [20] |
| Chuanzi 3 (ZZ), Zijuan, Ziyan from Sichuan Province | UPLC-ESI-MS/MS | Cyanidin-3-(6-O-p-caffeoyl)-glucoside, Cyanidin-3,5-O-diglucoside, Cyanidin-3-O-arabinoside, Cyanidin-3-O-galactoside, Cyanidin-3-O-glucoside, Cyanidin-3-O-rutinoside, Delphinidin, Delphinidin-3,5-O-diglucoside, Delphinidin-3-O-(6-O-p-coumaroyl)-galactoside, Delphinidin-3-O-5-O-(6-O-coumaroyl)-diglucoside, Delphinidin-3-O-galactoside, Delphinidin-3-O-glucoside, Delphinidin-3-O-sophoroside, Pelargonidin-3-O-(6-O-p-coumaroyl)-galactoside, Pelargonidin-3-O-arabinoside, Pelargonidin-3-O-galactoside, Pelargonidin-3-O-glucoside, Pelargonidin-3-O-rutinoside, Peonidin-3-O-(6-O-p-coumaroyl)-glucoside, Peonidin-3-O-galactoside, Petunidin-3-O-arabinoside, Petunidin-3-O-glucoside | [27] |
| Zikui from Guizhou | UPLC MS/MS ESI-QTRAP-MS/MS | Petunidin 3-O-glucoside, Cyanidin 3-O-galactoside, Cyanidin 3-O-glucosid | [11] |
| Purple-leaf tea cultivars from Jiangsu Province | UPLC-ESI-MS/MS | Cyanidin 3-O-(6-O-malonyl-beta-D-glucoside), Cyanidin 3-O-arabinoside, Cyanidin 3-O-galactoside, Cyanidin 3-O-glucoside, Delphinidin 3,5-O-diglucoside, Delphinidin 3-O-(6’’-O-malonyl)-beta-D-glucoside, Delphinidin 3-O-arabinoside, Delphinidin 3-O-galactoside, Delphinidin 3-O-glucoside, Malvidin 3-O-arabinoside, Malvidin 3-O-galactoside, Malvidin 3-O-glucoside, Pelargonidin 3-O-(6-O-malonyl-beta-D-glucoside), Pelargonidin 3-O-arabinoside, Pelargonidin 3-O-galactoside, Pelargonidin 3-O-glucoside, Pelargonidin 3-O-rutinoside, Peonidin 3-O-glucoside, Peonidin 3-O-arabinoside, Peonidin 3-O-galactoside, Petunidin 3,5-diglucoside, Petunidin 3-O-arabinoside, Petunidin 3-O-galactoside, Petunidin 3-O-glucoside | [28] |
| Hongyecha, Zijuan, 9803, Hongyafoshou from Hunan Province | (UPLC-DAD-QTOF-MS | Delphinidin 3-O-galactoside, Cyanidin 3-O-galactoside, Pelargonidin-O-hexose, Pelargonidin-O-dihexose, Delphinidin-(Z)-p-coumaroylgalactoside, Delphinidin-(E)-p-coumaroylgalactoside, Cyanidin-(Z)-p-coumaroylgalactoside | [29] |
| Jinmingzao from Fujian Province | LC-ESI-MS/MS | Cyanidin 3-O-glucoside, Delphinidin 3-O-glucoside, Peonidin 3-O-glucoside chloride, Cyanidin 3-rutinoside, Cyanidin 3-O-galactoside | [30] |
| Purple tea leaves from Asilia Inc. | LC-MS/MS | Delphinidin-3-galactoside, Delphinidin-3-glucoside, Cyanidin-3-galactoside, Cyanidin-3-glucoside, Delphinidin-coumaroyl-hexoside isomers 1, 2, and 3, Cyanidin-coumaroyl-hexoside isomers 1 and 2, Petunidin-coumaroyl-hexoside | [21] |
| Zijuan from Yunnan, Puer | LC-ESI-MS/MS | Cyanidin 3-O-galactoside, Cyanidin 3-O-glucoside, Delphinidin 3-O-glucoside, Petunidin 3-O-glucoside, Pelargonidin 3-O-glucoside | [31] |
| TRFK 306 from Kenya | HPLC | Cyanidin-3-O-galactoside, Cyanidin-3-O-glucoside, Delphinidin, Cyanidin, Pelargonidin, Peonidin, Malvidin | [16] |
| Purple-colored tea cultivars from Kenya | HPLC-UV–Visible | Delphinidin, Cyanidin, Pelargonidin, Peonidin, Malvidin, Cyanidin-3-O-galactoside, Cyanidin-3-O-glucoside | [5] |
| Sunrouge from Japan | HPLC, LC/MS/MS, and NMR | Delphinidin-3-O-β-D-(6-(E)-p-coumaroyl)galactopyranoside, Delphinidin-3-O-β-D-(6-(E)-p-coumaroyl)glucopyranoside, Cyanidin-3-O-β-D-(6-(E)-p-coumaroyl)galactopyranoside, Cyanidin-3-O-β-D-(6-(E)-p-coumaroyl)glucopyranoside, Delphinidin-(Z)-p-coumaroylgalactopyranoside, Petunidin-(E)-p-coumaroylgalactopyranoside | [6] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, M.; Huang, X.; Xie, N.; Zhao, T.; Zhu, M.; Li, J.; Wang, K. Advances in Purple Tea Research: Chemical Compositions, Anthocyanin Synthesis and Regulation, Processing, and Health Benefits. Horticulturae 2024, 10, 50. https://doi.org/10.3390/horticulturae10010050
Yan M, Huang X, Xie N, Zhao T, Zhu M, Li J, Wang K. Advances in Purple Tea Research: Chemical Compositions, Anthocyanin Synthesis and Regulation, Processing, and Health Benefits. Horticulturae. 2024; 10(1):50. https://doi.org/10.3390/horticulturae10010050
Chicago/Turabian StyleYan, Meihong, Xiangxiang Huang, Nianci Xie, Tiyue Zhao, Mingzhi Zhu, Juan Li, and Kunbo Wang. 2024. "Advances in Purple Tea Research: Chemical Compositions, Anthocyanin Synthesis and Regulation, Processing, and Health Benefits" Horticulturae 10, no. 1: 50. https://doi.org/10.3390/horticulturae10010050
APA StyleYan, M., Huang, X., Xie, N., Zhao, T., Zhu, M., Li, J., & Wang, K. (2024). Advances in Purple Tea Research: Chemical Compositions, Anthocyanin Synthesis and Regulation, Processing, and Health Benefits. Horticulturae, 10(1), 50. https://doi.org/10.3390/horticulturae10010050







