1. Introduction
Due to the adverse impacts of chemical control, such as environmental pollution and the disruption of the ecological balance, many developed countries and regions worldwide have restricted the use of some toxic chemical reagents in plant disease control [
1]. With the global focus on ecological environmental protection, biological control has emerged as the preferred alternative to chemical agents in plant disease control because of its environmentally friendly, sustainable, and long-lasting effects [
2]. Numerous studies have demonstrated that
Bacillus pumilus is a widely used microorganism in the biological control of plant diseases [
3,
4], such as chickpea root rot [
5], rice blast [
6], pine shoot blight [
7], pine seedling damping-off disease [
8], black rot in strawberries [
9], and cucumber wilt [
10], etc.
In the realm of microbial preparation research and production, cost reduction is often achieved by employing inexpensive raw materials and enhancing the effectiveness of the fermentation liquid. The improvement of a fermentation broth’s effectiveness necessitates fermentation process technology, which requires a lot of human and material resources to be invested in exploration. Laboratory-based fermentation process optimization typically utilizes one-way tests combined with orthogonal tests to swiftly identify the optimal fermentation medium ratio. Through fermentation optimization, Pan [
11] increased the antifungal rate of
B. pumilus LYMC-3 against the pathogen of pine shoot blight by 15.84% compared to before optimization. Dai [
12] optimized the fermentation conditions of
Bacillus pumilus HR10 to achieve an 87.04% inhibition of
Sphaeropsis sapinea. Yao [
13] optimized the fermentation conditions of
Bacillus licheniformis to achieve a final fibrinolytic enzyme activity (FEA) of 2434.45 ± 28.49 IU/mL. Shi [
14] significantly enhanced the number of
Bacillus velezensis YH-18 fermentation broth spores and enhanced the antifungal ability of the fermentation broth’s de-bacterialized filtrate through optimized design. After optimizing the design, Yu [
15] increased the antifungal activity and antagonistic substance production of a
Bacillus pumilus BSH-4 fermentation broth by more than three times. In order to enhance the antifungal activity of the
Bacillus pumilus LYMC-3 strain against
P. macrospora and
P. vesicolor and expand its production application, the optimization of its fermentation conditions is very significant.
As an important class of microbial resources, Bacillus has garnered considerable attention. However, scholars from various countries hold different opinions regarding the inhibition mechanism of Bacillus. The physicochemical properties of the LYMC-3 strain are useful for a further understanding of the mechanisms of action and the composition of inhibitory substances. Therefore, this study conducted preliminary research on the physicochemical properties of the LYMC-3 strain.
Preliminary experiments have revealed that the
B. pumilus LYMC-3, a strain with a potent antagonistic effect, was isolated from
Pinus massoniana stems. It exhibited a broad spectrum of antifungal inhibition and a strong inhibitory effect on the poplar canker pathogen (
P. macrospora) and the pine shoot blight pathogen (
S. sapinea) [
11,
16]. It also has strong nematicidal activity against
Bursaphelenchus xylophilu, which is the causal agent of pine wilt disease. In order to elucidate the effect of a fermentation broth of
B. pumilus LYMC-3 on the control of poplar canker and tea gray blight, this study conducted an indoor biocontrol test of the LYMC-3 stain. The aim was to provide realistic and feasible methods and a theoretical basis for developing the application technology of this strain for the control of
P. macrospora and
P. vesicolor. Moreover, the study explored the optimal fermentation conditions for producing
P. macrospora and
P. vesicolor inhibitory substances using a one-way experimental design combined with an orthogonal design to search for a more efficient and cost-effective fermentation condition. This would provide a theoretical basis for the research and development of biofungicides. Subsequently, the physicochemical properties, such as polarity, thermal stability, and acid-base stability, of the inhibitory substances of the
B. pumilus LYMC-3 strain were initially explored. This exploration paved the way for the purification and identification of the inhibitory substances at a later stage.
2. Materials and Methods
2.1. Test Strains
A poplar canker pathogen (Phomopsis macrospora) and tea gray blight pathogen (Pestalotiopsis vesicolor) were provided by the Pathology Laboratory of the College of Forestry and Grassland of Nanjing Forestry University.
The Bacillus pumilus LYMC-3 strain was isolated from the stems of P. massoniana at Sui Tang Botanical Gardens in Luoyang, China.
2.2. Culture Media
Bacterial culture medium (NA): Beef paste, 5 g; peptone, 10 g; sodium chloride, 5 g; agar, 15–20 g; pH 7.2; distilled water, 1 L;
Bacterial fermentation medium (NB): Beef paste, 5 g; peptone, 10 g; glucose, 10 g; sodium chloride, 5 g; pH 8; distilled water, 1 L;
Phytopathogenic fungus medium (PDA): Potato, 200 g; glucose, 20 g; agar, 20 g; distilled water, 1 L;
Seed medium: Peptone, 10 g; glucose, 10 g; NaCl, 10 g; deionized water, 1000 mL; pH 7.0; autoclaved at 121 °C for 20 min.
2.3. Cultivation Method for the Test Strains
2.3.1. LYMC-3 Strain Culture
- (1)
Seed solution culture
The
B. pumilus LYMC-3 strain was inoculated on the NA plate using the plate scribing method [
17], and a single colony was selected for purification. The purified single colony was placed into 50 mL of seed medium in 100 mL conical flasks and cultured for 16–24 h with shaking on a constant-temperature shaking incubator at 28 °C and 200 r/min.
- (2)
Shake flask fermentation culture
A 100 mL conical flask containing 50 mL of the NB medium with 2% inoculum was incubated on a constant-temperature shaking incubator at 28 °C and 200 r/min with shaking for 48 h. Each single-factor and orthogonal test was conducted separately to optimize the fermentation conditions.
- (3)
Fermentation broth acquisition
The LYMC-3 strain was activated via streaking on an NA plate, and single colonies were selected to be transferred to the NB culture medium. The shake flasks were incubated on a shaker at a constant temperature for 24 h until the logarithmic growth stage was reached to obtain the seed solution (28 °C, 200 r/min). The seed solution was transferred to the bacterial fermentation medium (NB) with an inoculum of 2% by volume and incubated in a shaker for 48 h (28 °C, 200 r/min) for an indoor control effect test.
2.3.2. Culture of Pathogenic Fungi and Preparation of Spore Solution
P. macrospora and P. vesicolor were inoculated in the center of PDA plates. When P. macrospora and P. vesicolor had grown all over the plates, they were placed under a UV light (100 µw/cm2) in the dark to promote spore production The pathogen spores were collected after 7 d. The spores were prepared into a spore solution containing a concentration of approximately 1 × 106 cfu with sterile water.
2.4. Indoor Control Effect Test of Fermentation Broth on Poplar Canker and Tea Gray Blight Disease
2.4.1. Inoculation of Pathogenic Fungi
- (1)
Inoculation of P. macrospora
Poplar branches and trunks were inoculated using the puncture method [
18]. A sterilized inoculation needle was used to create perforations at equal points on the seedling trunks, reaching deep into the xylem. Subsequently, spore liquid was injected into the wounds, and sterile, water-soaked cotton wool was placed to keep the wounds moisturized. Finally, the wounds were wrapped using a sealing film (BKMANLAB).
- (2)
Inoculation of P. vesicolor
Tea tree seedlings were inoculated using the spore suspension needling method [
19]. The tea tree leaves’ surfaces were sterilized with 75% alcohol. Subsequently, the leaves were gently touched with a red-hot inoculation needle to create punctate scalded spots, and the diameter of the initial wound was measured. After the scald treatment, the spore solution was sprayed onto the leaf surface, and the inoculated area was moisturized and treated for 24 h post-inoculation.
2.4.2. Indoor Control Effect Test of Fermentation Broth on Poplar Canker Disease
- (1)
Experiment on the prevention of poplar canker disease
The fermentation broth was incubated for 48 h and then diluted 2 times for subsequent use. The specific treatments were conducted as follows:
Irrigation treatment: One week prior to inoculation with the pathogenic fungus, each poplar seedling was irrigated with 100 mL of diluted fermentation broth;
Coating treatment: One week before the inoculation with the pathogenic fungus, each poplar seedling was coated with 100 mL of diluted fermentation broth. After drying, the coating was repeated three times;
Root irrigation and coating treatment: One week before the inoculation with the pathogenic fungus, each poplar sapling was inoculated with 50 mL of diluted fermentation broth via root irrigation and was then coated with 50 mL of diluted fermentation broth. After drying, the coating was repeated 3 times.
Each treatment was placed in a greenhouse and inoculated with sterile water as a control with 10 replicates for both the treatment and control groups. The size of the greenhouse is 125 × 85 × 150 cm, and it is covered with polyvinyl chloride. A constant temperature of 28 °C and approximately 60% humidity were maintained within the greenhouse.
- (2)
Experiment on the treatment of poplar canker disease
After a 2-month period of inoculation with the pathogenic fungus, the fermentation broth incubated for 48 h was utilized and diluted 2 times for subsequent use. The specific treatments remained consistent with those described in
Section 2.4.2 (1). Each treatment was then placed within the greenhouse and subjected to inoculation with sterile water as the control. There were 10 replicates for each treatment and control group.
2.4.3. Indoor Control Effect Test of Fermentation Broth on Tea Gray Blight Disease
- (1)
Preventive test of tea gray blight disease
The fermentation broth, cultured for 48 h and diluted 2 times, was used for the preventive test. The specific treatments were as follows:
Rooting treatment: The same as that described in
Section 2.4.2 (1);
Spray treatment: Ten tea leaves were selected from each tea sapling one week before inoculation. These tea leaves were sprayed with 50 mL of diluted fermentation broth until they started dripping. After drying, the spraying process was repeated three times;
Root irrigation and spraying treatment: One week before inoculation, each tea sapling was subjected to root irrigation with 50 mL of diluted fermentation broth. Additionally, ten tea leaves were selected for a spraying treatment, which was repeated three times after drying.
Each treatment was performed in the greenhouse. Sterile water was used as a negative control, and pathogenic fungus alone was used as a positive control. Five replicates were performed for each treatment and control.
- (2)
Treatment test of tea gray blight disease
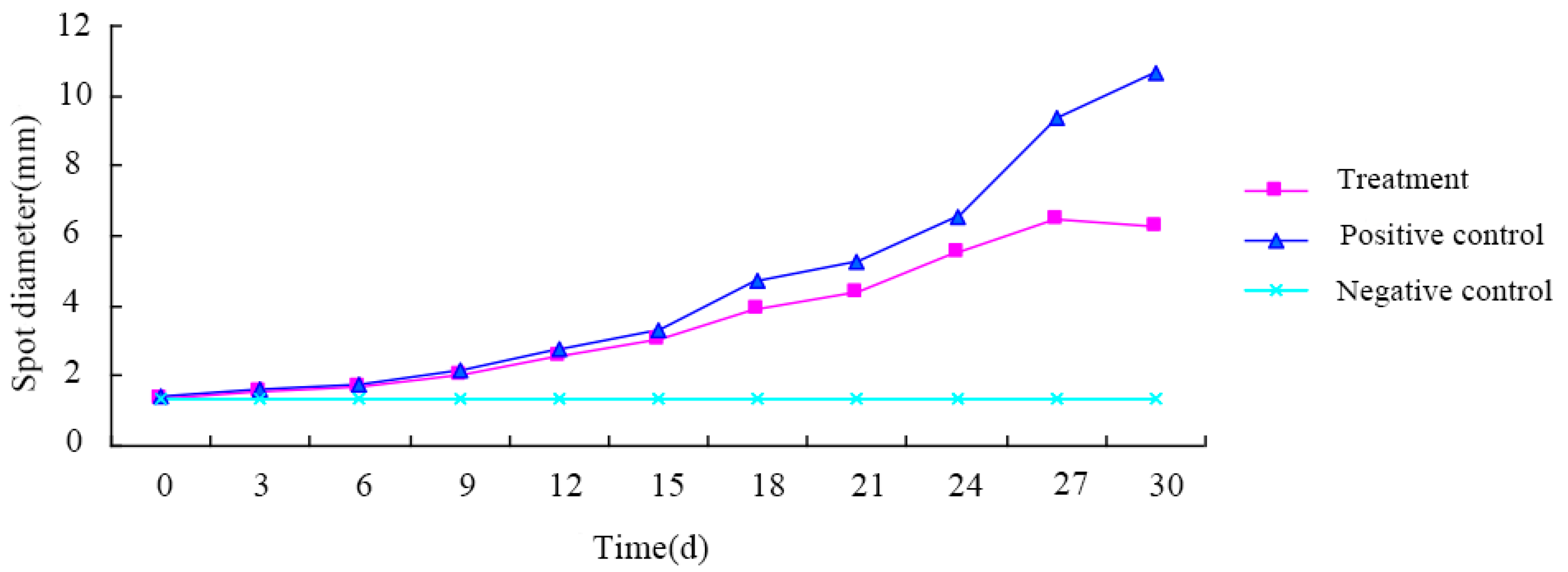
One week after inoculation with the pathogenic fungus, the tea tree seedlings were uniformly treated with the fermentation broth cultured for 48 h. Each tea tree seedling was sprayed and left to dry, with the spraying process repeated three times. Inoculation with sterile water was used as the negative control, and only the pathogen was used as the positive control. Five replications were conducted for each treatment and control. Each treatment was performed in the greenhouse. Leaf spot expansion was observed every 3 days, and the size of the spots’ diameter was recorded using a vernier caliper (Guilin Guanglu Measuring Instrument Co., Ltd., Guilin, China).
2.4.4. Investigation of Disease Development
The morbidity following pathogen inoculation was investigated, and the disease index and control effect were calculated. The disease grading standards of poplar canker and tea gray blight disease can be found in
Table 1 and
Table 2, respectively. The formulas for disease index and control effect were as follows:
Disease index formula: Disease index = 100 × ∑ (number of diseased plants (leaves) at each grade × representative value of each grade)/(total number of plants (leaves) surveyed × representative value of the highest grade);
Control effect formula: Control effect (%) = 100 × (disease index of control − disease index after treatment)/disease index of control).
2.5. Determination of the Bacterial Biomass and Antifungal Activity of the LYMC-3 Strain
2.5.1. Measurement of Bacterial Biomass
A specific volume of LYMC-3 fermentation broth was measured, and the optical density (OD) value was determined at a wavelength of 600 nm using a spectrophotometer, with an NB medium serving as the control. The biomass of the LYMC-3 strain was quantified using the OD value, for which a high OD value indicated a large amount of bacterial biomass, and vice versa.
2.5.2. Antifungal Activity Assay
Toxic plate method [
20]: PDA medium and bacterial metabolism liquid from decontaminated organisms were thoroughly mixed in a ratio of 4:1 to create a poisoned plate. After the plate solidified and dried, a pathogenic fungus cake (d = 5 mm) was placed at the center of the plate, and the diameter of the pathogenic fungus colony was measured after 4–5 d.
Inhibition rate (%) [
11] = (diameter of control colony − diameter of treated colony) × 100/(diameter of control colony − 5).
2.6. Optimization of the Composition of the Fermentation Medium for Inhibitory Substances
2.6.1. Effects of Different Carbon Sources on the Inhibitory Activity of Antifungal Substances
The carbon sources in the basal medium (NB) were replaced with 5 g/L of maltose, glucose, sucrose, lactose, and mannitol, respectively, to formulate five kinds of media. The medium without a carbon source was used as the control. A 2% seed solution by total volume was added to each of the six media, and after incubation at 28 °C and 200 rpm for 48 h, the metabolite containing antifungal substances was filtered through a 0.22 µm bacterial filter, following centrifugation at 4 °C and 10,000 rpm for 10 min using a refrigerated high-speed centrifuge. The activity of the inhibitory substances in the different carbon source media was detected using the toxic plate method, with the medium without a metabolic solution serving as the control. Three replications were performed.
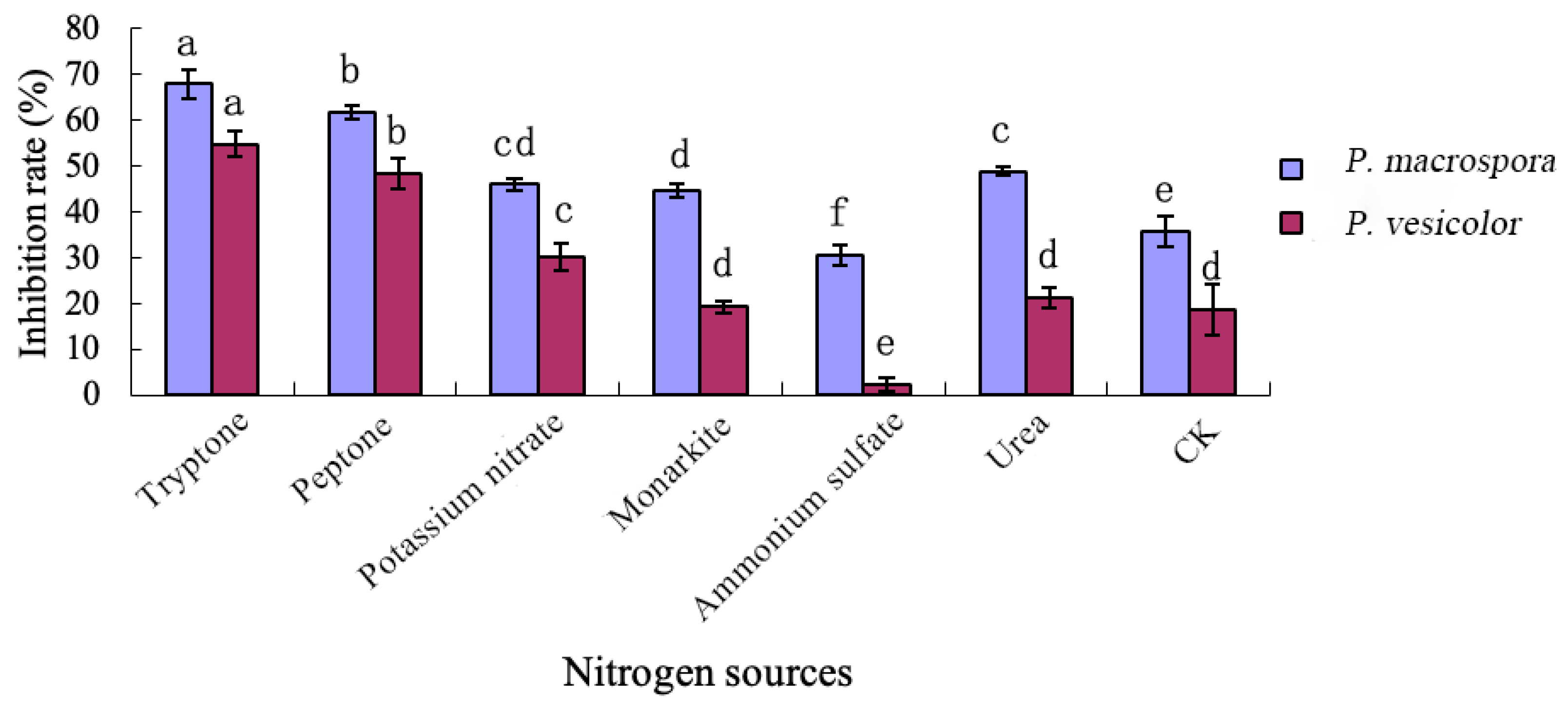
2.6.2. Effects of Different Nitrogen Sources on the Inhibitory Activity of Antifungal Substances
The nitrogen sources in the basal medium were replaced with tryptone, peptone, potassium nitrate, monarkite, ammonium sulfate, and urea, respectively, to formulate six different nitrogen sources, and the medium without nitrogen sources was used as the control. A 2% seed solution by total volume was added to each of the seven media, and the inhibition rates of strain LYMC-3 against the two pathogenic fungi were determined according to the method described in
Section 2.6.1.
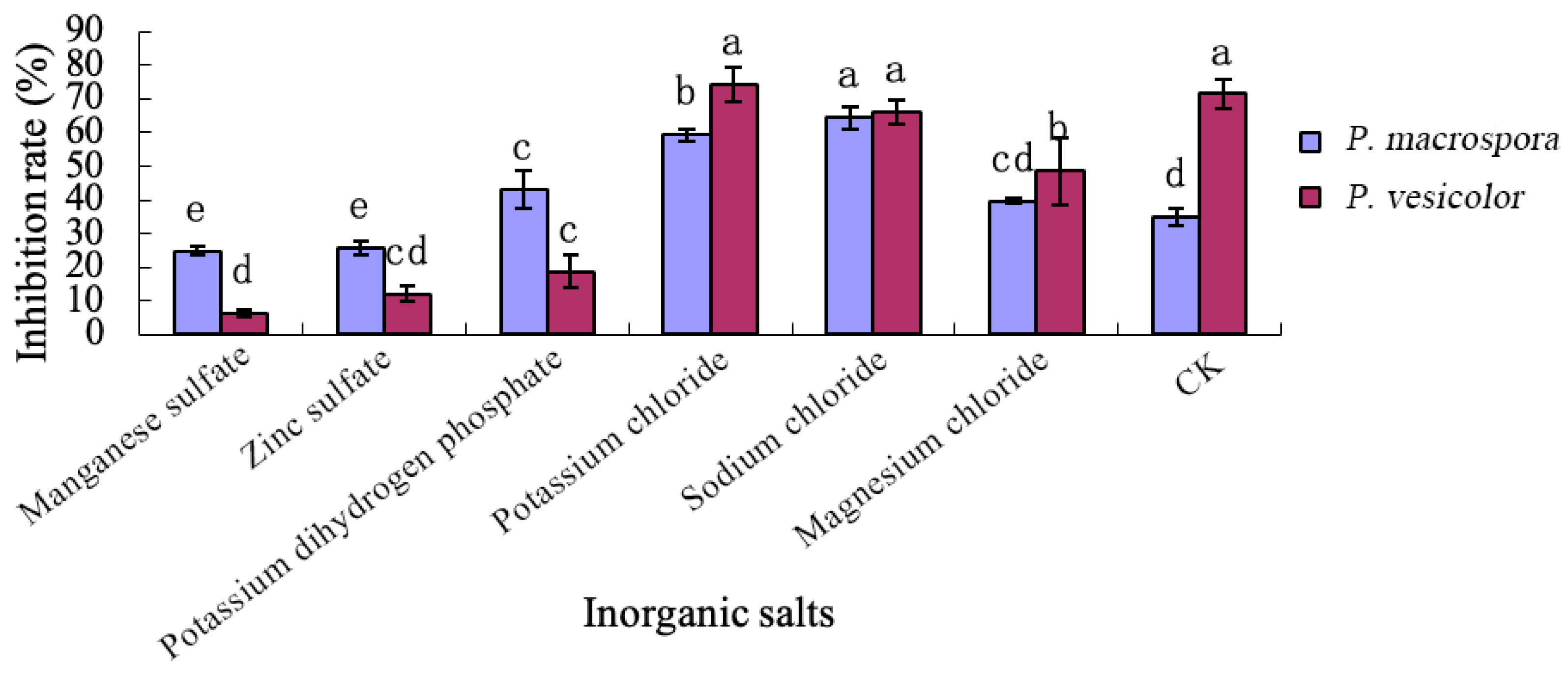
2.6.3. Effects of Different Inorganic Salts on the Inhibitory Activity of Antifungal Substances
The inorganic salts in the basal medium were replaced with manganese sulfate, zinc sulfate, potassium dihydrogen phosphate, potassium chloride, sodium chloride, and magnesium chloride, respectively, to formulate six kinds of media containing different inorganic salts, and the medium without inorganic salts was used as the control. A 2% seed solution by total volume was added to each of the seven media, and the inhibition rates of strain LYMC-3 against the two pathogenic fungi were determined according to the method described in
Section 2.6.1.
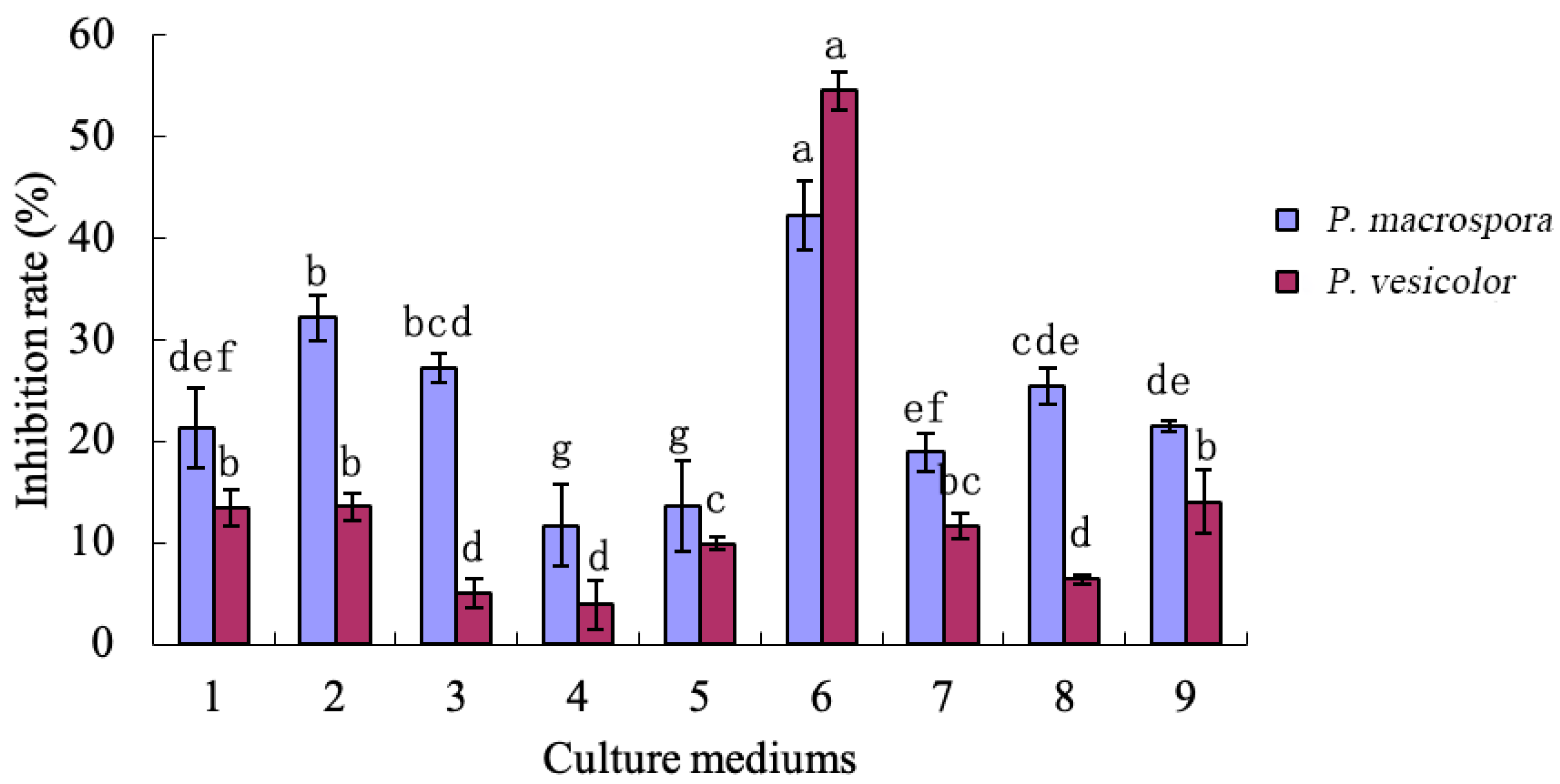
2.6.4. Orthogonal Tests with Different Components of the Culture Medium
Based on the optimal screening results obtained via the aforementioned three experiments and the optimal dosage ranges of each nutrient ingredient derived from the tests, nine sets of orthogonal tests were designed based on
Table 3. These tests comprised varying concentrations of the screened optimal ingredients to identify the optimal medium ingredients and their ratios [
21].
2.7. Optimization of Culture Conditions for the Fermentation of the Inhibitory Substances
2.7.1. Effects of Different Fermentation Times on the Inhibitory Activity of Antifungal Substances
A 2% LYMC-3 strain seed solution was introduced into 50 mL of the modified medium and incubated under constant conditions at 28 °C and 200 rpm with shaking. The biomass of the bacteria was measured at regular intervals after 12, 24, 36, 48, 60, 72, 84, and 96 h of fermentation incubation. The inhibition rates on
P. macrospora and
P. vesicolor were determined following the method described in
Section 2.6.1.
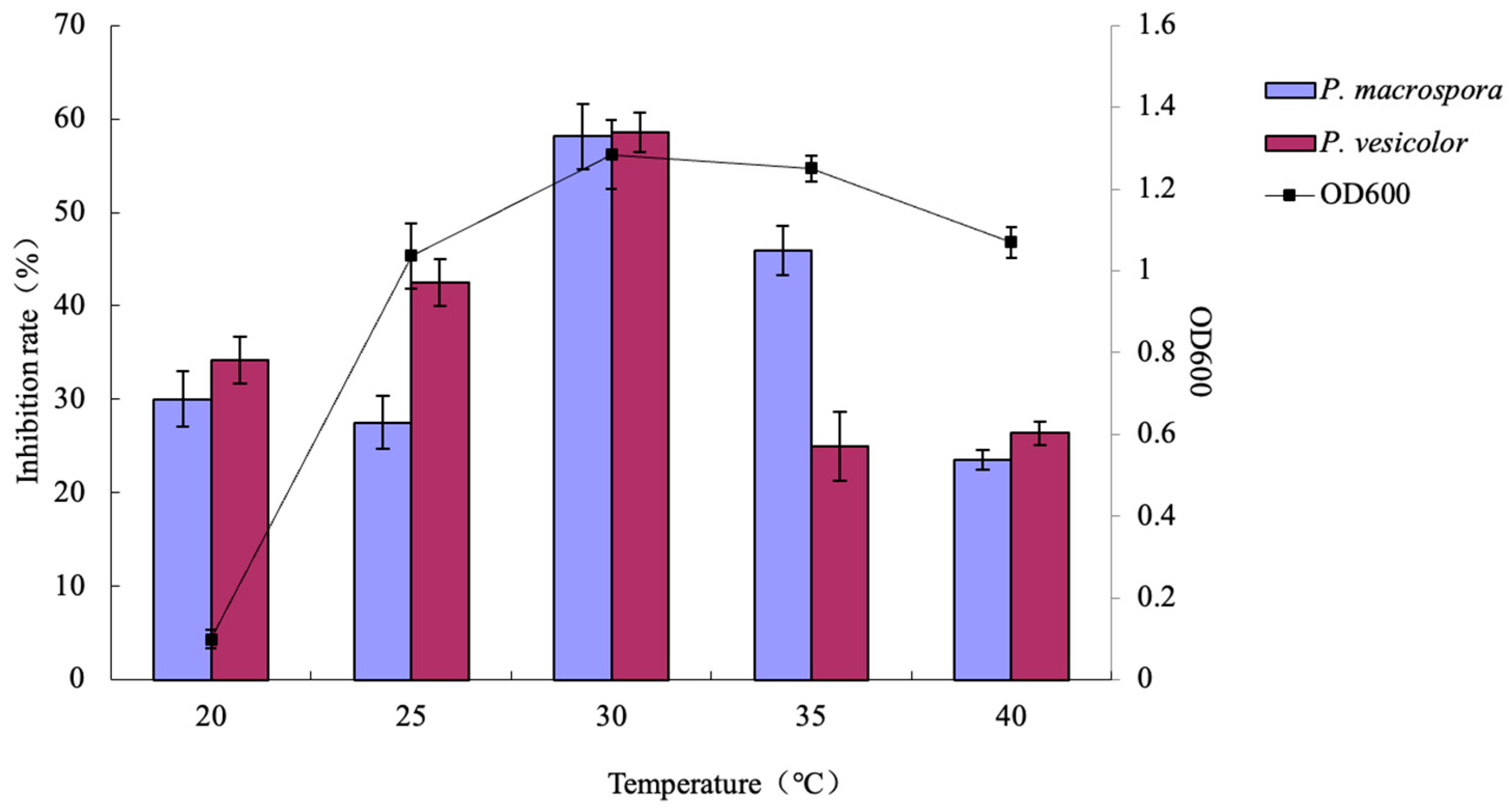
2.7.2. Effects of Different Temperatures on the Inhibitory Activity of Antifungal Substances
A 2% LYMC-3 strain seed solution was introduced into 50 mL of the modified medium while the other conditions were kept constant. Five temperature gradients (20 °C, 25 °C, 30 °C, 35 °C, and 40 °C) were set using a horizontal shaker operating at a constant speed of 200 rpm. After 48 h of incubation, the OD600 was determined for each temperature gradient. The inhibition rates were assessed following the method described in
Section 2.6.1.
2.7.3. Effects of Different Initial pH Levels on the Inhibitory Activity of Antifungal Substances
A 2% LYMC-3 strain seed solution was introduced into 50 mL of the modified medium, and the pH was adjusted to 5.0, 6.0, 7.0, 8.0, 9.0, 10.0, and 11.0 using sodium hydroxide and hydrochloric acid solutions. The mixture was then incubated for 48 h at 28 °C and 200 rpm, after which OD600 was measured. The inhibition rates were calculated following the method described in
Section 2.6.1.
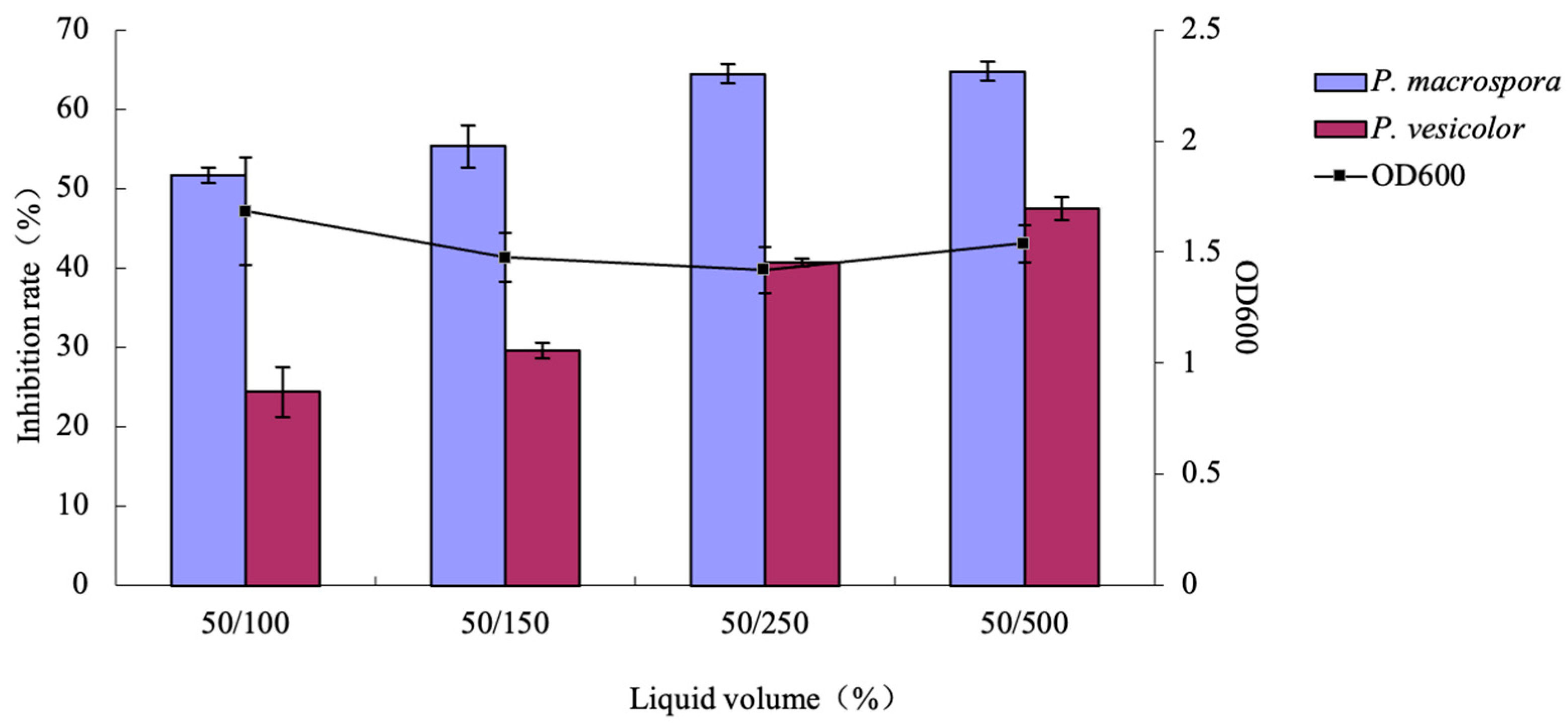
2.7.4. Effects of Different Liquid Volumes on the Inhibitory Activity of Antifungal Substances
Four conical flasks of varying sizes (100 mL, 150 mL, 250 mL, and 500 mL) were utilized to hold 50 mL of the modified medium. A 2% LYMC-3 strain seed liquid was added to each flask, followed by incubation at 28 °C and 200 rpm for 48 h. The OD600 was subsequently measured, and the inhibition rates were calculated following the method described in
Section 2.6.1.
2.7.5. Effects of Different Inoculum Amounts on the Inhibitory Activity of Antifungal Substances
Different concentrations of the seed solution (1%, 2%, 4%, 6%, 8%, and 10%) were introduced to 50 mL of the modified medium. The mixture was incubated in a constant-temperature horizontal shaker at 28 °C and 200 rpm for 48 h. Subsequently, the OD600 was measured using a spectrophotometer. The inhibition rates were calculated following the method described in
Section 2.6.1.
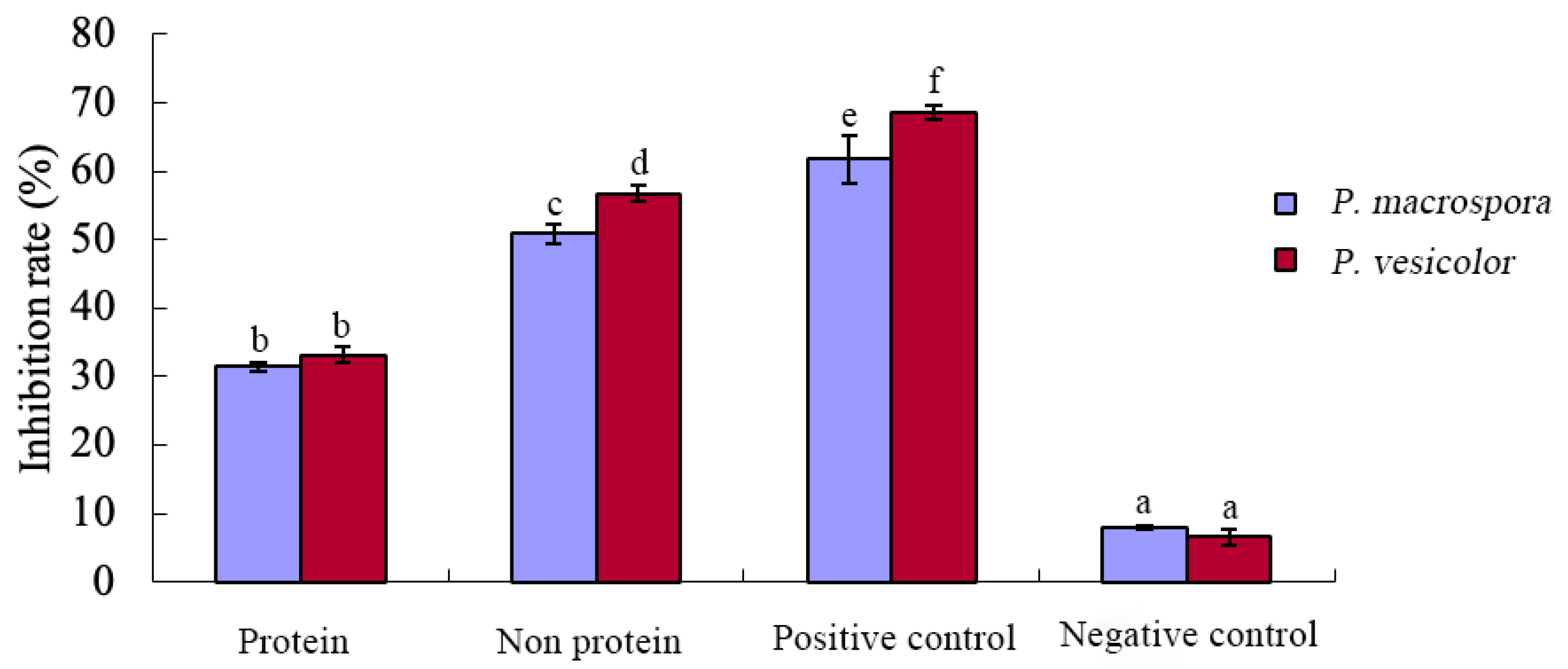
2.8. Determination of Protein and Non-Protein Inhibitory Activities in the Fermentation Filtrate
The anhydrous ethanol precipitation method [
22] was employed to separate proteins from non-proteins in the fermentation filtrate. Specifically, 2–3 times the volume of anhydrous ethanol was added to a certain volume of sterilized fermentation filtrate (performed in an ice bath) and stirred slowly for a period of time to allow for the complete precipitation of macromolecular proteins, polysaccharides, and nucleic acids. The mixture was then frozen and centrifuged for 15 min at 2 °C and 5000 r/ min to collect the protein fraction and the non-protein fraction, respectively. The protein fraction was dissolved in deionized water and restored to the original volume. The non-protein fraction was rotary-evaporated at 40 °C for 120 r/min until the ethanol was completely removed, and then deionized water was added, and the original volume was restored. The activity of the inhibitory substances of each fraction was detected using the toxic plate method, with the fermentation filtrate serving as the positive control and the culture medium serving as a negative control. Three replications were performed.
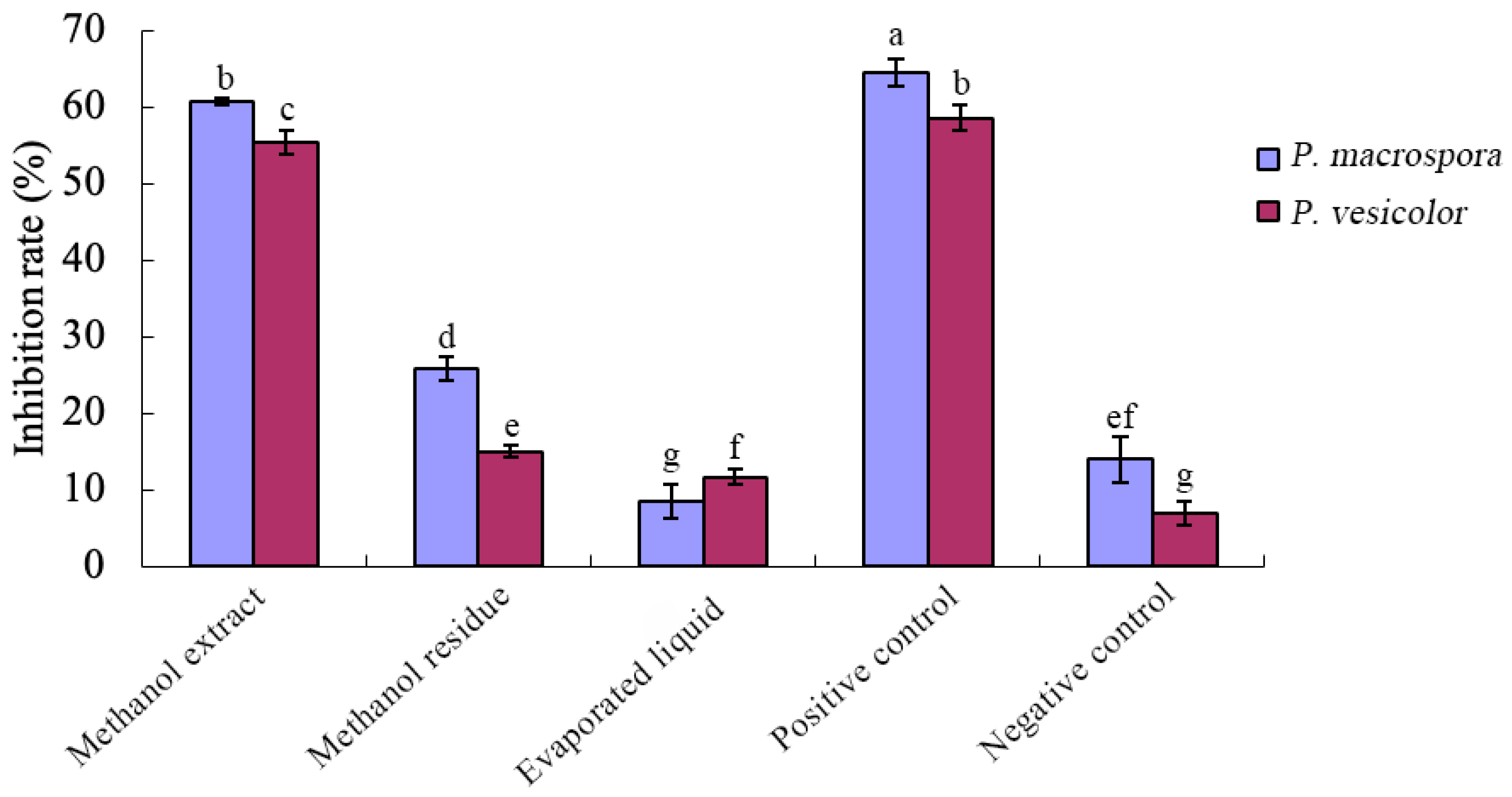
2.9. Preparation of Antifungal Crude Extracts
A certain volume of fermentation filtrate was measured and rotary-evaporated at 50 °C to dryness, and then the evaporated liquid was collected. The dry material was then mixed thoroughly with an appropriate amount of methanol through three slow oscillations. The mixture was centrifuged, and the supernatant fluid was retained. The supernatant fluid was rotary-evaporated to dryness at 40 °C and dissolved in distilled water to the original volume to obtain the antifungal crude extract (methanol extract). The residue after centrifugation (mainly proteins, nucleic acids, polysaccharides, and other macromolecules) was redissolved in distilled water to its original volume. The inhibitory activity of the crude extract (methanol extract), the residue after centrifugation (methanol residue), and the evaporated liquid was determined using the toxic plate method. The fermentation filtrate was used as the positive control, and the NB medium was used as the negative control. Three replications were performed.
2.10. Determination of Antifungal Crude Extract Activity at Different Temperatures
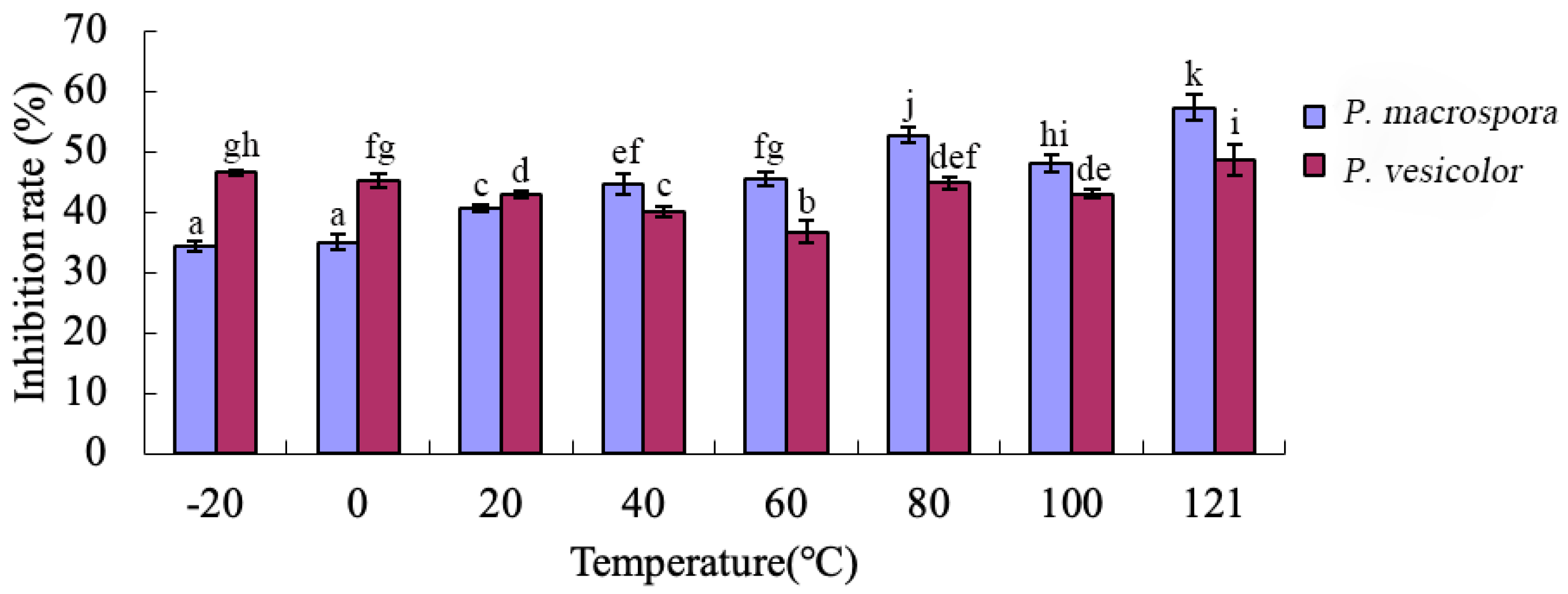
The antifungal crude extracts were subjected to treatments at various temperatures (−20 °C, 0 °C, 20 °C, 40 °C, 60 °C, 80 °C, 100 °C, and 120 °C) for a duration of 1 h. Bacteria present in the filtrate were eliminated using a 0.22 µm bacterial filter. The inhibitory activity of the antifungal crude extracts treated at different temperatures was evaluated using the toxic plate method. The experiment was repeated three times.
2.11. Determination of Antifungal Crude Extract Activity at Different pH Values
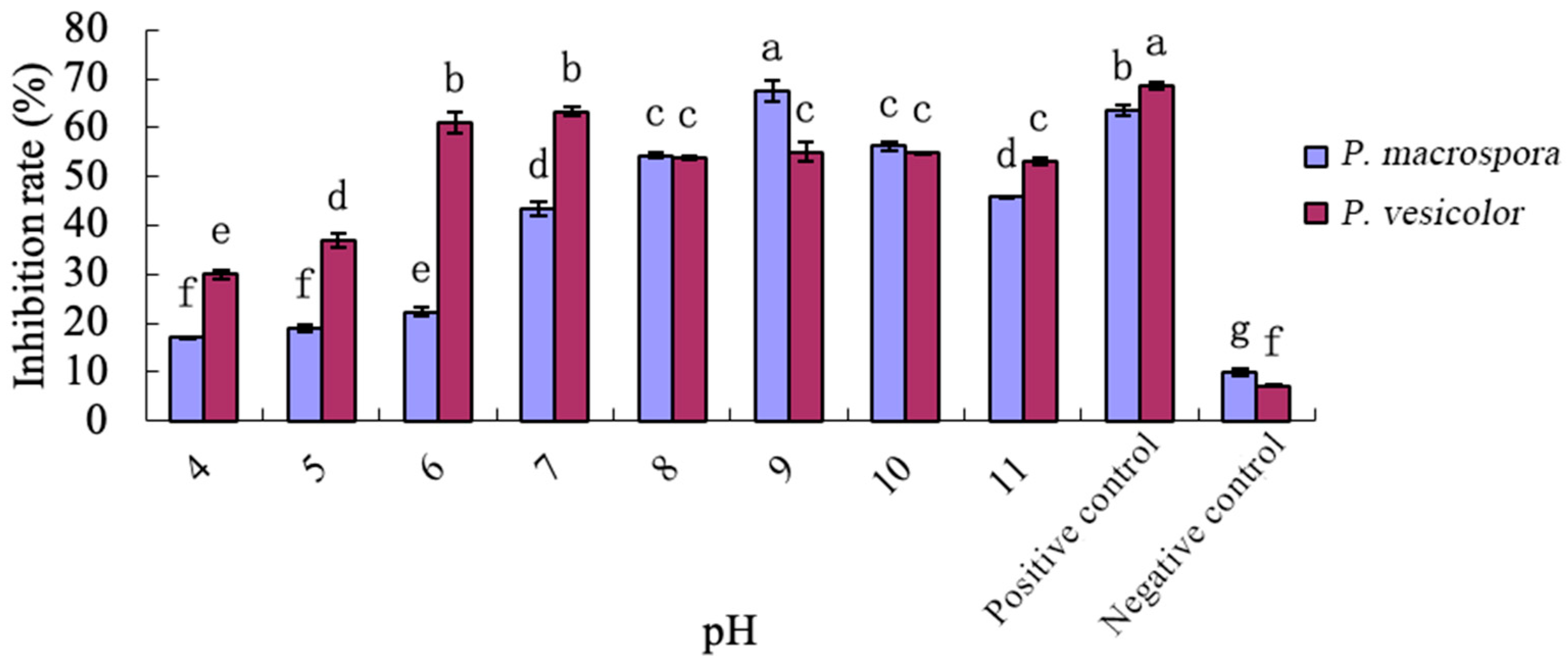
The pH value of the inhibition crude extract was adjusted to 4.0, 5.0, 6.0, 7.0, 8.0, 9.0, 10.0, and 11.0 using 1 mol/ L HCl and NaOH solutions, respectively. The inhibitory activity was assessed by treating the inhibition crude extract with P. macrospora and P. vesicolor as the target strains. The experiment was repeated three times with untreated inhibitory substances serving as the positive control and the NB medium as the negative control.
2.12. Solubility of Antifungal Substances in Organic Solvents of Different Polarities
A volume of 20 mL of fermentation filtrate was measured and combined with 40 mL of organic solvents with varying polarities, including ethyl acetate, chloroform, dichloromethane, benzene, n-butanol, and carbon tetrachloride, for extraction. The mixture was vigorously shaken and allowed to stand in a refrigerator at 2 °C for 24 h. Subsequently, the organic and aqueous phases were separated in a separating funnel under a fume hood. The organic solvent was then removed from the organic phase using a rotary evaporator, and the original volume of distilled water was added to obtain an aqueous solution of the organic phase extract. Antifungal activity tests were conducted on the aqueous solutions extracted from the various organic phases with the fermentation filtrate serving as the positive control and the NB culture medium as the negative control. The experiment was repeated three times.
4. Discussion
In recent years, biological control has gained increasing popularity due to its environmentally friendly nature. Among the various biocontrol microorganisms that have been researched and developed, biocontrol bacteria to control plant diseases has become a focal point for scholars in the field of biocontrol. In this study, we found that the endophytic bacterium B. pumilus LYMC-3 has certain preventive and therapeutic effects on poplar canker and tea gray blight disease through indoor control effect experiments. For poplar canker, the highest indoor preventive effect reached 44.12%, while the therapeutic effect reached only 28.83%. For tea gray blight, the highest indoor preventive effect was 48.59%, which was slightly higher than the therapeutic effect. These findings suggest that the indoor preventive effect of the B. pumilus strain LYMC-3 is superior to its therapeutic effect, highlighting the importance of emphasizing prevention when applying this strain for the control of poplar canker and tea gray blight. However, it is important to note that this trial had a limited scope, and further improvements in application methods are necessary. Additionally, the potential of this biocontrol bacteria against these two diseases requires further exploration.
The optimization of the fermentation process plays a crucial role in the industrialization of microbial fermentation products, as it ensures their effectiveness as biopesticides. Various experimental techniques and design methods have been employed for fermentation process optimization [
23]. The single-factor method is the most traditional approach to optimization. However, due to the complexity of medium composition and fermentation conditions in most fermentation processes, relying solely on the single-factor method may not achieve the desired results. It can increase the workload, prolong the experimental period, and potentially lead to unreliable or incorrect conclusions due to variations in experimental conditions and batches [
24]. Moreover, conclusions cannot be drawn about the presence of interactions between factors based solely on the result of a single-factor test. Hence, it is commonly incorporated into an orthogonal test [
25]. Therefore, single-factor tests are frequently utilized in conjunction with orthogonal tests or response surface methodology.
The approach of arranging multifactorial experiments with orthogonal tables is referred to as the orthogonal experimental design method. Orthogonal tests have found extensive application in fermentation process optimization. Chen [
26] employed the orthogonal test method to optimize the fermentation medium of
Streptomyces aureus, resulting in a 16% increase in the demeclocycline yield. Madjirebaye [
27] determined that a combination of one-way and orthogonal tests optimized the conditions for fermented soybean milk, leading to an improved flavor and enhanced storage stability. Wu [
28] developed a low-cost medium and significantly enhanced the production capacity of fibrinolytic enzymes in
Bacillus subtilis WR350 through the utilization of one-way and orthogonal tests, with optimized fibrinolytic activity reaching 5865 U/mL. While the results of the orthogonal test may have aligned with those obtained using the single-factor method, the orthogonal test offers advantages, such as rationality in factor and level section, uniform distribution, the estimation of errors without the need for replicated experiments, and high accuracy [
29]. In our study, the fermentation conditions of the LYMC-3 strain were initially investigated via a combination of the single-factor and orthogonal tests. The bacterium was found to be adaptable to a broad temperature range (25–40 °C) but unable to thrive under strong acidic or alkaline environments, with its optimal pH range being 7–10. The optimal fermentation conditions for antifungal substances’ production can be summarized as follows: a culture medium consisting of 10 g/L of glucose, 15 g/L of tryptone, and 5 g/L of sodium chloride; an initial pH of 8; a 10% liquid volume; a 4% inoculum amount; and a shaking culture at 30 °C for 84 h.
Through the crude isolation of inhibitory substances in the fermentation broth and the preliminary study of their properties, it was determined that these substances belonged to a class of small, strongly polar non-protein molecules. They exhibited good thermal stability but were sensitive to acid and alkali, with their inhibitory activity significantly reduced under strong acidic and alkaline conditions. Various organic solvents were employed for the extraction of these non-protein antifungal substances. Methanol extraction yielded crude antifungal substances that displayed a notable inhibitory effect on
P. macrospora, while n-butanol extraction resulted in crude substances with a strong inhibitory effect on t
P. vesicolor. Lin [
30] discovered that the anti-
Listeria substance produced via
Bacillus amyloliquefaciens maintains stable activity across a wide pH and temperature range. Lei [
31] compared the inhibition substance of
Lactobacillus plantarum zrx03′s inhibition substance extracted using different organic solvents (such as n-butanol, n-hexane, dichloromethane, and trichloromethane) and ultimately determined that ethyl acetate was the optimal solution for crude extraction.
Pan [
32] extracted and purified the inhibition substance from
Bacillus megaterium L2 and identified it as a non-protein phenylacetic acid (PAA) that antagonized
Agrobacterium tumefaciens. Furthermore, PAA exerted its antifungal activity by disrupting cell membrane integrity and cell ultrastructure. Tian [
33] demonstrated that
Talaromyces purpurogenus Q2 effectively reduced the relative tolerance of
Fusarium oxysporum by secreting cell wall degrading enzymes, thereby achieving excellent biological control effects on
Fusarium wilt. It has been hypothesized that the main disease prevention mechanism of LYMC-3 involves the following: (1) the production of antagonistic substances (non-protein antimicrobial substances), which may play a significant role in disease prevention; (2) the competition for space and nutrition with pathogenic fungus. The LYMC-3 strain exhibits a vigorous growth ability in comparison to other microorganisms that inhabit the same environment. This characteristic enables the strain to rapidly acquire the resources needed for growth and reproduction, consequently impeding the proliferation of pathogenic fungi. However, it is important to note that disease prevention may be influenced by various factors, such as induced host resistance. In conclusion, the control mechanism employed by the LYMC-3 strain to combat poplar canker and tea gray blight encompasses multiple factors, necessitating further investigations into its intricate nature. Additionally, the specific substances inhibiting fungal growth also warrant further exploration.
5. Conclusions
In this study, we conducted an indoor biocontrol test on the B. pumilus LYMC-3 strain and found that its fermentation broth exhibited the highest indoor preventive efficacy of 44.12% against poplar canker and 48.59% against tea gray blight. Subsequently, we optimized the growth conditions of the LYMC-3 strain and the fermentation conditions of inhibitory substances using a single-way test combined with an orthogonal design. Our results indicated that a medium consisting of 10 g/L of glucose, 15 g/L of tryptone, and 5 g/L of sodium chloride was conducive to the production of inhibitory substances via strain LYMC-3. Moreover, a culture medium with a shaking cultivation temperature of 30 °C, a cultivation time of 84 h, an initial pH of 8.0, a 10% liquid volume, and a 4% inoculum amount was found to be suitable for the production of inhibitory substances using strain LYMC-3. Finally, we crudely extracted the inhibitory substances from the fermentation broth and preliminarily investigated their properties. Our findings indicate that the non-protein extracts exhibited a notable inhibitory effect on P. macrospora and P. vesicolor. Additionally, the inhibitory substances displayed strong polarity and good thermal stability, and they were more sensitive to strong acidic or strong alkaline environments.