Abstract
Koji, which is used for manufacturing Japanese traditional fermented foods, has long been safely used as a cosmetic product. Although its cosmetic effect has been empirically established, the underlying mechanism has not been reported. We and other groups have previously elucidated that koji contains glycosylceramides, including N-2′-hydroxyoctadecanoyl-1-O-β-d-glucosyl-9-methyl-4,8-sphingadienine and N-2′-hydroxyoctadecanoyl-1-O-β-d-galactosyl-9-methyl-4,8-sphingadienine. This led us to hypothesise that koji exerts its cosmetic effect by acting on the keratinocytes through glycosylceramides on the gene level. Therefore, in this study, we investigated the effects of glycosylceramides from various sources on gene expression in normal human epidermal keratinocytes. The results revealed that glycosylceramides purified from white koji and the white koji-producing non-pathogenic fungus Aspergillus luchuensis and A. oryzae increased the expression of occludin (OCLN, an epidermal tight junction protein) and ATP-binding cassette sub-family A member 12 (ABCA12, a cellular membrane transporter), albeit the effect was modest relative to that of ceramides. Indeed, ceramide was increased in the keratinocytes upon koji lipid extract addition. These results indicate that glycosylceramides, which are the major sphingolipids of most natural materials, have an effect of increasing ABCA12 and OCLN expression, and suggest that koji exerts its cosmetic effect by increasing ceramide and tight junctions via glycosylceramides.
1. Introduction
Skin health is of significant concern, especially given the recent increase in cases of atopic dermatitis in developed countries [1]. The epidermis provides protective functions against skin dryness and microbial infection. In particular, the differentiated stratum corneum retards transcutaneous evaporative water loss. The stratum corneum is a multi-layered tissue composed of flattened, geometrical, and anucleated corneocytes, and it is surrounded by multiple stacks of broad, planar lamellar layers [2]. Ceramides, which are the degradation products of glucosylceramide and sphingomyelin by phospholipases and β-glucocerebrosidase, are provided to the extracellular spaces of the stratum corneum by the secretion of the two substrates from the outermost stratum granulosum cells via ATP-binding cassette sub-family A member 12 (ABCA12) [3]. Therefore, the lamellae in the stratum corneum are enriched in ceramides (50%), cholesterol, and free fatty acids and contain small amounts of cholesterol sulphates and cholesterol esters. In patients with atopic dermatitis, a decreased level of ceramides is observed [4]. Ceramides in the stratum corneum are divided into seven species (Ceramide I to VII), depending on their structures [5].
Since atopic dermatitis exhibits an aspect of inflammatory dermatoses, many drugs and substances intended for repressing inflammatory responses have been developed to treat this skin condition [6]. By considering atopic dermatitis as a skin barrier dysfunction, skin barrier repair therapies to normalise the epidermal barrier function have been attempted by reducing transepidermal water loss and improving stratum corneum hydration [7]. The lipids in the lamellae of patients with atopic dermatitis contain less ceramides and more cholesterol than those of unaffected individuals [8]. Furthermore, sphingomyelin deacylase, which degrades sphingomyelin and produces sphingosylphosphocholine instead of ceramide, might be up-regulated in patients with atopic diseases [9]. Therefore, the topical application of ceramide is reported to be beneficial for patients with atopic dermatitis [10]. Indeed, an equimolar ratio (1:1:1) of ceramide, cholesterol, and free fatty acids has been found to be efficient for barrier recovery in acute injury models [11]. Although non-physiological agents (e.g., petrolatum, lanolin mineral oil, and silicone) are effective in the short term, they might impede the biochemical process of skin barrier formation in the long term [12]. Therefore, natural and empirical substances that can improve skin barrier function are desired.
The ceramide-mediated activation of genes involved in skin barrier function in keratinocytes has been reported. For example, ceramide up-regulates the ABCA12 gene, which is involved in the transport of lipids in the keratinocytes [13]. Moreover, sphingoid bases such as 4,8-sphingadienine and 4-hydroxy-8-sphingenine activate ceramide synthesis genes [5]. However, ceramides or sphingoid bases constitute a minor proportion in most natural materials, whereas glucosylceramides, glycosylceramides, and sphingomyelins are the major sphingolipids.
Japanese traditional fermented foods and drinks manufactured using koji (steamed rice fermented with the non-pathogenic fungus Aspergillus oryzae or A. luchuensis) has long been effectively and safely used as cosmetics in Japan, but the mechanism behind its cosmetic effect remains unknown. Japanese traditional foods and drinks have been shown to contain various glycosylceramides, including N-2′-hydroxyoctadecanoyl-1-O-β-d-glucosyl-9-methyl-4,8-sphingadienine (d19:2/C18:0h) and N-2′-hydroxyoctadecanoyl-1-O-β-d-galactosyl-9-methyl-4,8-sphingadienine (d19:2/C18:0h) [14,15,16,17]. However, the effects of glycosylceramides contained in natural substances, including koji, on gene expression in normal human epidermal cells are not known. In order to explore the availability of natural substances to cure skin disorders, the effects of the glycosylceramides contained in koji on gene expression in normal human epidermal cells were investigated in this study.
2. Materials and Methods
2.1. Materials
Conidia of Aspergillus luchuensis and A. oryzae were purchased from Higuchi Matsunosukeshoten Co. Ltd. (Osaka, Japan). The yellow koji and white koji were purchased from Tokushima Seiko (Tokushima, Japan). Nine kinds of plant-derived glucosylceramides were purchased from Nagara Science (Gifu, Japan). The normal human epidermal keratinocyte (NHEK)-c adult (lot number: 3111203.6, taken from skin/breast, 36/female/Caucasian) cell line was obtained from PromoCell (Heidelberg, Germany). Ceramide II (NS) was purchased from Takasago International Corporation (Tokyo, Japan) and Olbracht Serdary Research Laboratories (Toronto, Canada). Ceramides III (NP) and VI (AP) were purchased from Evonik Nutrition & Care GmbH, (Essen, Germany). These chemically synthesised ceramides have the same structure as those in the human body.
2.2. Extraction and Purification of Glycosylceramides from A. luchuensis, A. Oryzae, White Koji, and Yellow Koji
Conidia of A. luchuensis and A. oryzae were inoculated with 200 mL of 24 g/L potato dextrose broth (Difco, Beckton Dickinson, Sparks, USA). The incubated culture was centrifuged and washed with sterile distilled water, and the pellet was lyophilised. Total lipids were extracted from 0.2 g of lyophilised A. luchuensis pellets using a chloroform–methanol solution (1:1, v/v), fractionated with the Bligh and Dyer method followed by alkaline treatment. The extracted lipids were dried in an evaporator and dissolved in 4 mL of chloroform.
Silica gel chromatography was applied to further purify the lipid fraction. Firstly, the lipid sample was eluted with 600 mL of chloroform and the chloroform-eluted fraction was discarded. Then, the remaining lipid sample was eluted with 400 mL of an ethyl acetate–methanol solution (9:1, v/v) and the back 350 mL was collected. The collected lipid sample was dried in an evaporator and dissolved in 1 mL of a chloroform–methanol solution (2:1, v/v).
Total lipids were extracted from 100 g of white koji or 100 g of yellow koji, which were crushed with a mixer. The koji was dissolved with 180 mL of a chloroform–methanol solution (1:1, v/v) for 5 min with ultrasonic treatment. Then, it was subjected to ultrasonication for 10 min after the further addition of 90 mL of chloroform. The collected lipid samples were dried in an evaporator and dissolved in 4 mL of hexane.
The lipid sample was eluted from silica gel with 500 mL of an ethyl acetate–methanol solution (9:1, v/v). The first 100 mL and the last 270 mL of eluents were discarded, whereas the middle 130 mL was collected. The collected lipid sample was dried in an evaporator and dissolved in 5 mL of a chloroform–methanol solution (2:1, v/v).
These lipid solutions containing glycosylceramides were spotted onto a TLC plate. Cerebroside derived from bovine brain was spotted as a control. A chloroform–methanol–acetic acid–water solution (20:3.5:2.3:0.7, v/v) was used as the solvent to develop the TLC plate. A part of the TLC plate was heated at 100 °C for 40 min. The rest of the TLC plate was scratched and collected according to the location of the detected glycosylceramides. The silica gel was eluted with 6 mL of a chloroform–methanol solution (2:1, v/v), and the collected sample was dried in an evaporator and dissolved in 1 mL of chloroform.
The glycosylceramide fractions purified from A. luchuensis, A. oryzae, white koji, and yellow koji were further purified using HPLC with the following gradient program (using the same buffer solutions as described above): 00.00 min of A 100%/B 0%, 30.00 min of A 92.5%/B 7.5%, 60.00 min of A 91%/B 9%, 80.00 min of A 10%/B 90%, 80.01–90.00 min of A 10%/B 90%, 90.01–100.00 min of A 100%/B 0%, and 100.01 min STOP. The collected fractions were subjected to TLC analysis for verification of their identity.
The glycosylceramide contents were quantitated on the basis of the intensities of the spots on TLC plates. Glucosylceramide derived from Grifola frondosa (Nagara Science, Gifu, Japan) was used as the standard sample. By applying a known quantity of standard glucosylceramide on the same TLC plate, the quantity of glycosylceramide was calculated from the following expression: y = k × √x (where x represents the intensity multiplied by the area, y represents the predicted quantity of glycosylceramide, and k represents a constant), as previously described [18].
2.3. NHEK Cell Assay
NHEK cells were cultured with keratinocyte basal medium 2 (KBM) at 37 °C under 5% CO2 in an incubator. Cells detached by 0.25% trypsin-EDTA were inoculated in a 24-well plate at a concentration of 5 × 104 cells/well and cultured for 24 h. The glycosylceramides and glucosylceramides were dissolved in KBM using 0.5% (v/v) dimethyl sulfoxide (DMSO). The ceramides were dissolved in KBM using an ethanol–n-dodecane solution (98:2, v/v). KBM (in 0.5% DMSO) with the test substances included were added to the cultured cells at concentrations of 5 or 20 μg/mL and the cells were cultured for 48 h.
2.4. Analysis of the Expression Levels of Genes Involved in Skin Barrier Function
RNA extraction from NHEK cells cultured with the test substances was conducted with the RNeasy Mini Kit (QIAGEN) according to the manufacturer’s protocol, and the extracted total RNA samples were stored at −80 °C. The expression levels of genes involved in skin barrier function were measured using the quantitative real-time polymerase chain reaction (qPCR) (Bio-Rad, Hercules, CA, USA). The following kit and reagents were used: One-Step SYBR PrimeScript PLUS RT-PCR Kit (Perfect Real Time, TaKaRa Bio Inc., Otsu, Shiga, Japan) with 5 μL of 2× One Step RT-PCR Buffer 4, 0.6 μL of TaKaRa Ex Taq HS Mix, 0.2 μL of Prime Script PLUS RTase Mix, 0.8 μL (or 1 μL) of primer, 1 μL of total RNA, and 2.4 μL (or 2.2 μL) of RNase-free dH2O for a total 10 μL (or total 20 μL) reaction volume. The reaction temperatures were as follows: reverse transcription at 42 °C for 5 min, a hold at 95 °C for 10 s, and PCR at 95 °C for 5 s and 60 °C for 30 s. (We applied 55 °C for 30 s, when using only the serine palmitoyltransferase (SPTLC) and involucrin (IVL) primers.) The ΔΔCq method was used for quantitation of the gene expression levels, and GAPDH was used as the reference gene.
2.5. Analysis of Ceramide in NHEK Cells
Lipids extracted from white koji were dissolved in DMSO at 20 mg/mL and then diluted 200-fold with KBM (with a final of 0.5% (v/v) DMSO and 100 μg/mL white koji extract). The solution was then serially diluted to 10, 20, and 50 μg/mL glycosylceramide. The cells were collected by treating with trypsin for 5 to 10 min and then inoculated into 24-well plates at 5 × 104 cells/well. The medium was removed and the white koji lipid extracts at 10, 20, and 50 μg/mL were added to their respective wells with NHEK cells and the plates were then incubated in KBM in 75-cm2 flasks at 37 °C under 5% CO2 for 48 h. After incubation, the medium was removed, the cells were washed three times with 1 mL of phosphate-buffered saline (PBS), and 500 μL of Cell Counting Kit-8 reagent diluted in serum-free Dulbecco’s modified Eagle’s medium was added. To each well, 200 μL of the samples was transferred, incubated for 1 to 4 hr and the optical density at 450 nm was measured. The cell viability was calculated according to the following equation:
% of control = (Data sample – Data blank)/(Data control – Data blank) × 100
2.6. Quantitation of the Ceramide Content of Cultured Cells
After removal of the medium from the cell culture, the cells were washed twice with 500 µL of PBS/well. To detach the cells, 500 µL of trypsin (diluted 3-fold with PBS) was added and the culture was incubated for 5–10 min. The detached cells (corresponding to 3 wells) were collected into a 5-mL plastic tube and centrifuged at 400× g for 3 min. The pellet was then washed twice with PBS. Thereafter, 500 µL of PBS was added to the pellet and the suspension was sonicated. Then, 1250 µL of methanol and 625 µL of chloroform were added and the suspension was incubated at 42 °C at 125 rpm for 20 min and then centrifuged at 1500× g for 5 min. The supernatant was collected and 625 µL of chloroform was added. After vortexing the mixture, 625 µL of PBS was added and the mixture was centrifuged at 1500× g for 15 min until the white substance in the upper layer reached the boundary of the separated layers. The lower layer was collected and dried under N2 gas, following which the sample was dissolved in 100 µL of a chloroform–methanol solution (2:1, v/v).
A 10 µL volume of the lipid sample was spotted onto an HPTLC plate at 1.5 cm from the bottom. Ceramide II (NS), ceramide III (NP), and ceramide VI (AP) were dissolved in a chloroform–methanol solution (2:1, v/v) at concentrations of 4, 20, 100, and 500 µg/mL, and 5 µL of the solutions were spotted onto the HPTLC plate, which was then developed in a pre-saturated chamber using a chloroform–methanol–acetic acid solvent (190:9:1, v/v). Spots were visualised by spraying with a 10% CuSO4–8% H3PO4 solvent and heating at 180 °C for 10 min. The spots were quantitated using ChemiDoc XRS and Image Lab software (Bio-Rad, Hercules, CA, USA).
3. Statistical Analyses
Statistical differences among mean values of multiple data groups were judged by Dunnett’s test or the unpaired one-tailed Student’s t-test followed by Bonferroni correction.
4. Results
4.1. Separation and Purification of Glycosylceramides from Total Lipids Extracted from Koji
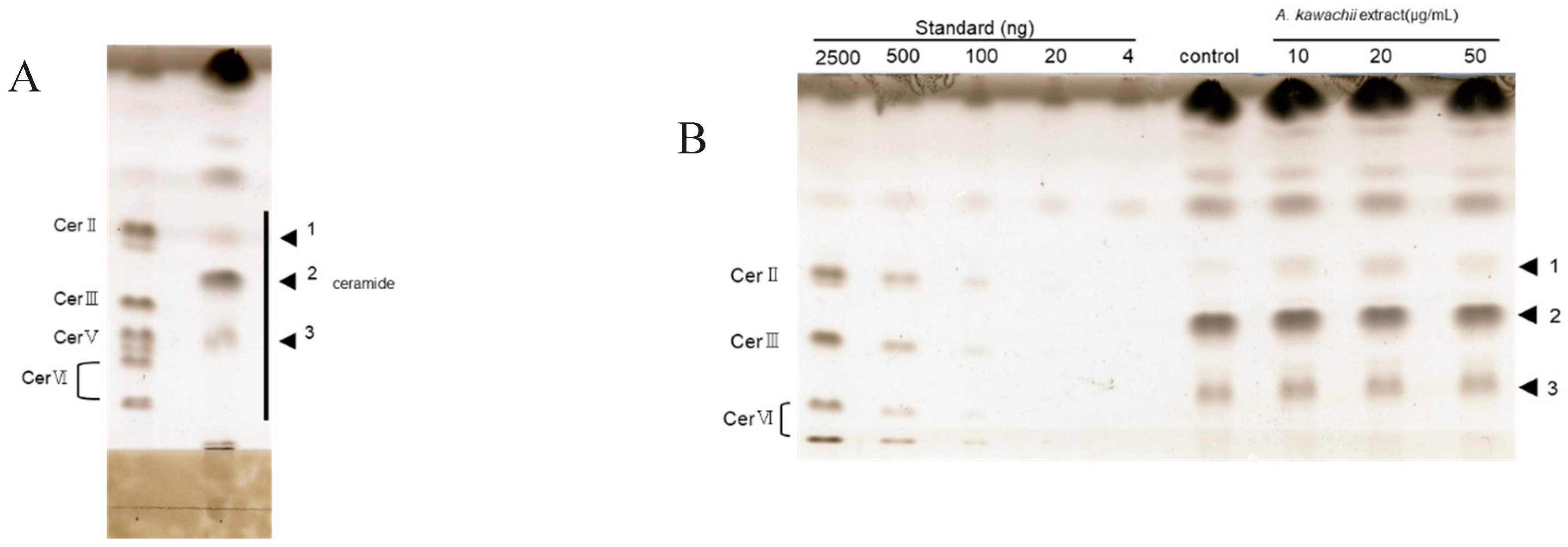
Firstly, glycosylceramides were purified from the mycelia of A. oryzae and A. luchuensis. To achieve this, total lipids were first extracted from the mycelia of the fungi and the glycosylceramides were then purified from the total lipids using HPLC (Figure 1A) to apparent homogeneity (Figure 1B).
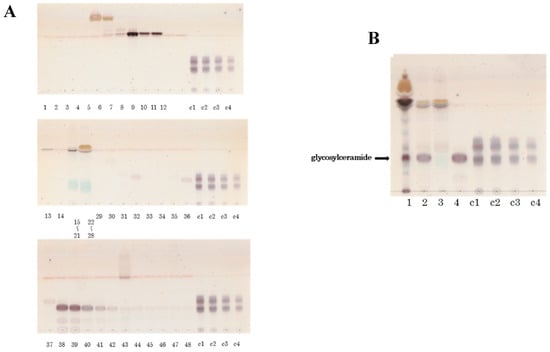
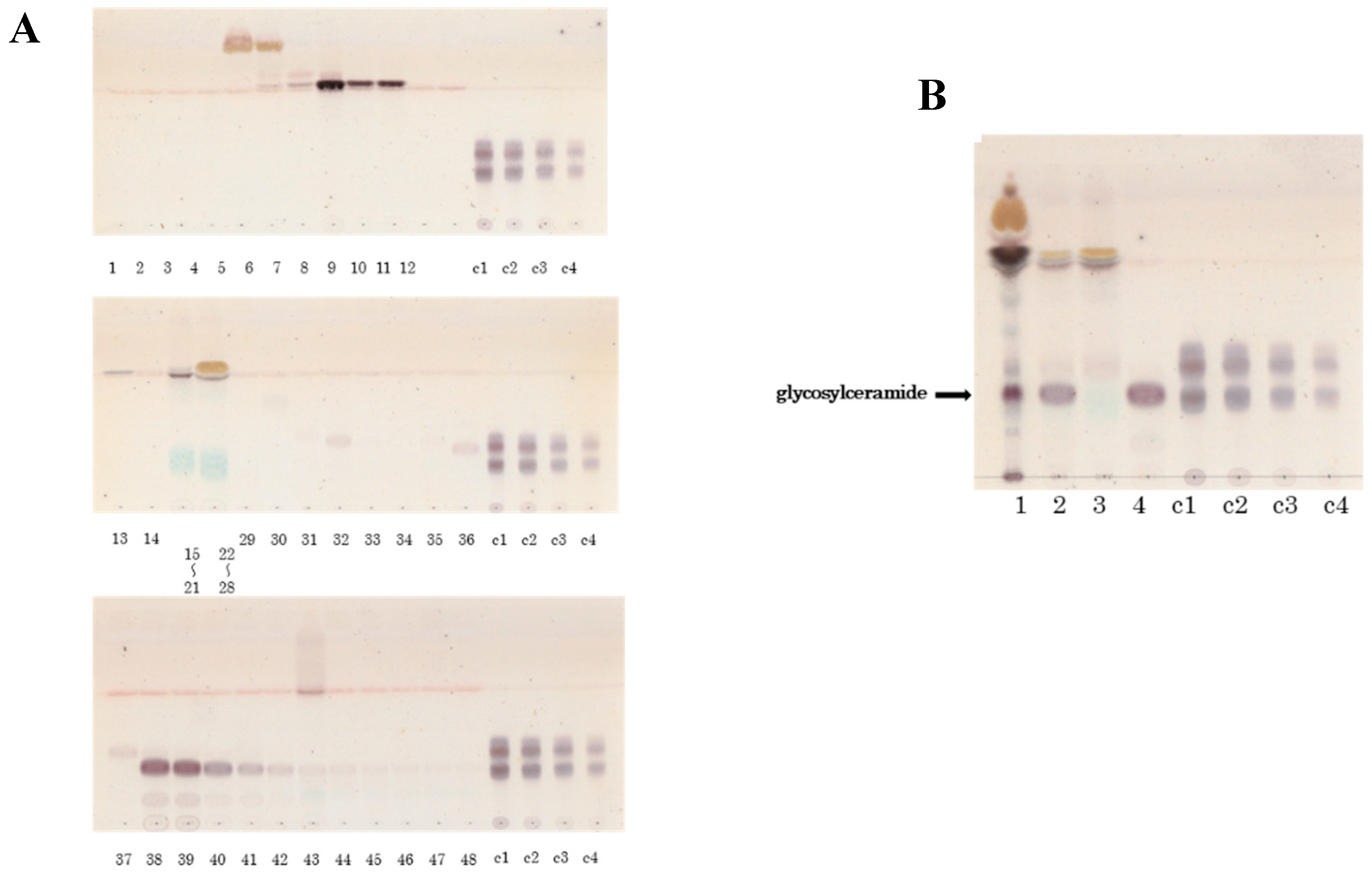
Figure 1.
Glycosylceramide from mycelia of Aspergillus luchuensis using HPLC. (A) Purification of glycosylceramide from koji using HPLC. Lanes 1–48: Elution fractions collected during HPLC. Lanes c1, c2, c3, c4: Standard cerebroside (20 μg, 10 μg, 5 μg, and 2.5 μg, respectively). Fractions 38–42 were identified as the glycosylceramide-containing fractions. (B) Purified glycosylceramides. Lane 1: Total lipid fraction extracted from 0.01 g mycelia of A. luchuensis. Lane 2: Eluted fractions 11–42. Lane 3: Eluted fractions 11–37. Lane 4: Eluted fractions 38–42 (glycosylceramide fraction). Lanes c1, c2, c3, c4: Standard cerebroside (20 μg, 10 μg, 5 μg, and 2.5 μg, respectively).
To compare the glycosylceramides from different sources, glycosylceramides were also purified from total lipids of lyophilised white koji and yellow koji using column chromatography, TLC silica gel fractionation, and HPLC (Figure 2).
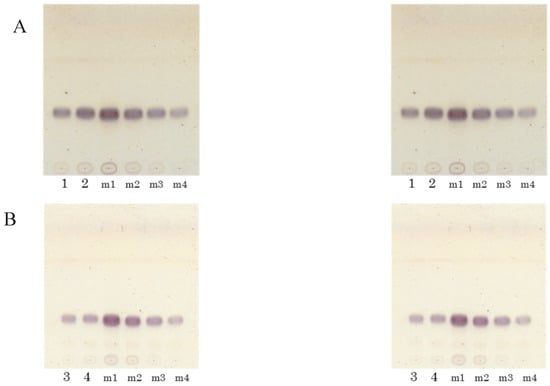
Figure 2.
Glycosylceramides purified from Aspergillus luchuensis, Aspergillus oryzae, white koji, and yellow koji by HPLC for quantitation. (A) Purified glycosylceramides from mycelia of fungi. Lane 1: Purified glycosylceramides from A. luchuensis. Lane 2: Purified glycosylceramides from A. oryzae. (B) Purification of glycosylceramides from koji sanples. Lane 3: Purified glycosylceramides from white koji.Lane 4: Purified glycosylceramides from yellow koji. Lanes m1, m2, m3, m4: Glucosylceramides derived from Grifola frondosa (30 μg, 15 μg, 7.5 μg, and 3.75 μg, respectively).
4.2. Effects of Glycosylceramides Purified from A. Luchuensis Mycelia on Gene Expression in NHEK Cells
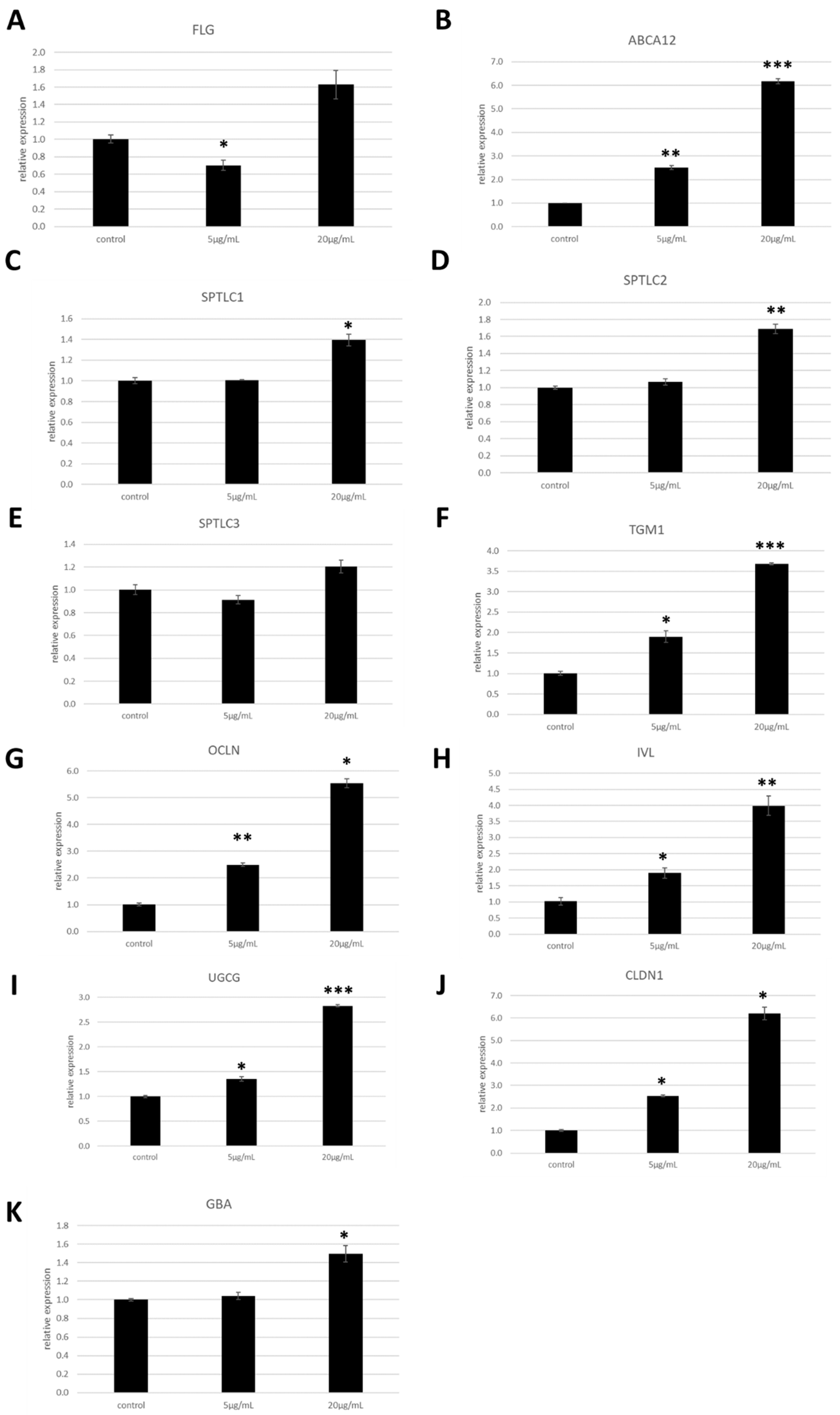
In order to analyse the effects of these glycosylceramides on primary human epidermal keratinocytes, NHEK cells were cultured for 48 h with 5 or 20 μg/mL of the glycosylceramide purified from A. luchuensis. The expression levels of genes involved in skin barrier function were measured using qPCR. The glycosylceramide significantly increased the expression of the genes encoding occludin (OCLN, an epidermal tight junction protein), ABCA12 (a cellular membrane transporter), serine palmitoyltransferase long chain base subunits 1 and 2 (SPTLC1 and SPTLC2), transglutaminase 1 (TGM1), ceramide glucosyltransferase (UGCG), claudin 1 (CLDN1), and glucosylceramidase beta (GBA) in the NHEK cells (n = 3, p < 0.05) (Figure 3). However, it did not significantly increase the expression of the genes encoding filaggrin (FLG, a natural moisturising factor) or serine palmitoyltransferase long chain base subunit 3 (SPTLC3, which is involved in the synthesis of the sphingoid bases) (n = 3, p > 0.05) (Figure 3).
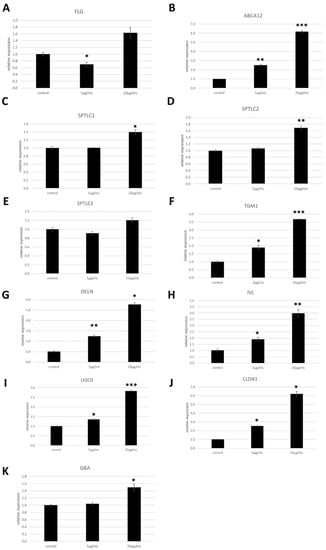
Figure 3.
Expression of genes involved in skin barrier function in NHEK cells upon addition of the glycosylceramide purified from Aspergillus luchuensis. NHEK cells were incubated with the glycosylceramide purified from A. luchuensis, and the mRNA expression levels of the indicated genes were measured using qPCR. The results indicated are the mean values ± standard errors. The values on the x-axis indicate the concentration of glycosylceramides (μg/mL) added. Statistical significance of differences was determined by the unpaired one-tailed Student’s t-test followed by Bonferroni correction (n = 3, *, p < 0.05; **, p < 0.01; ***, p < 0.001). (A) Filaggrin (FLG); (B) ATP-binding cassette sub-family A member 12 (ABCA12); (C) serine palmitoyltransferase long chain base subunit 1 (SPTLC1); (D) SPTLC2; (E) SPTLC3, (F) transglutaminase 1 (TGM1); (G) occludin (OCLN); (H) involucrin (IVL); (I) ceramide glucosyltransferase (UGCG); (J) claudin 1 (CLDN1); (K) glucosylceramidase beta (GBA).
4.3. Effects of Glycosylceramides and Ceramide from Other Sources on OCLN and ABCA12 Expression in NHEK Cells
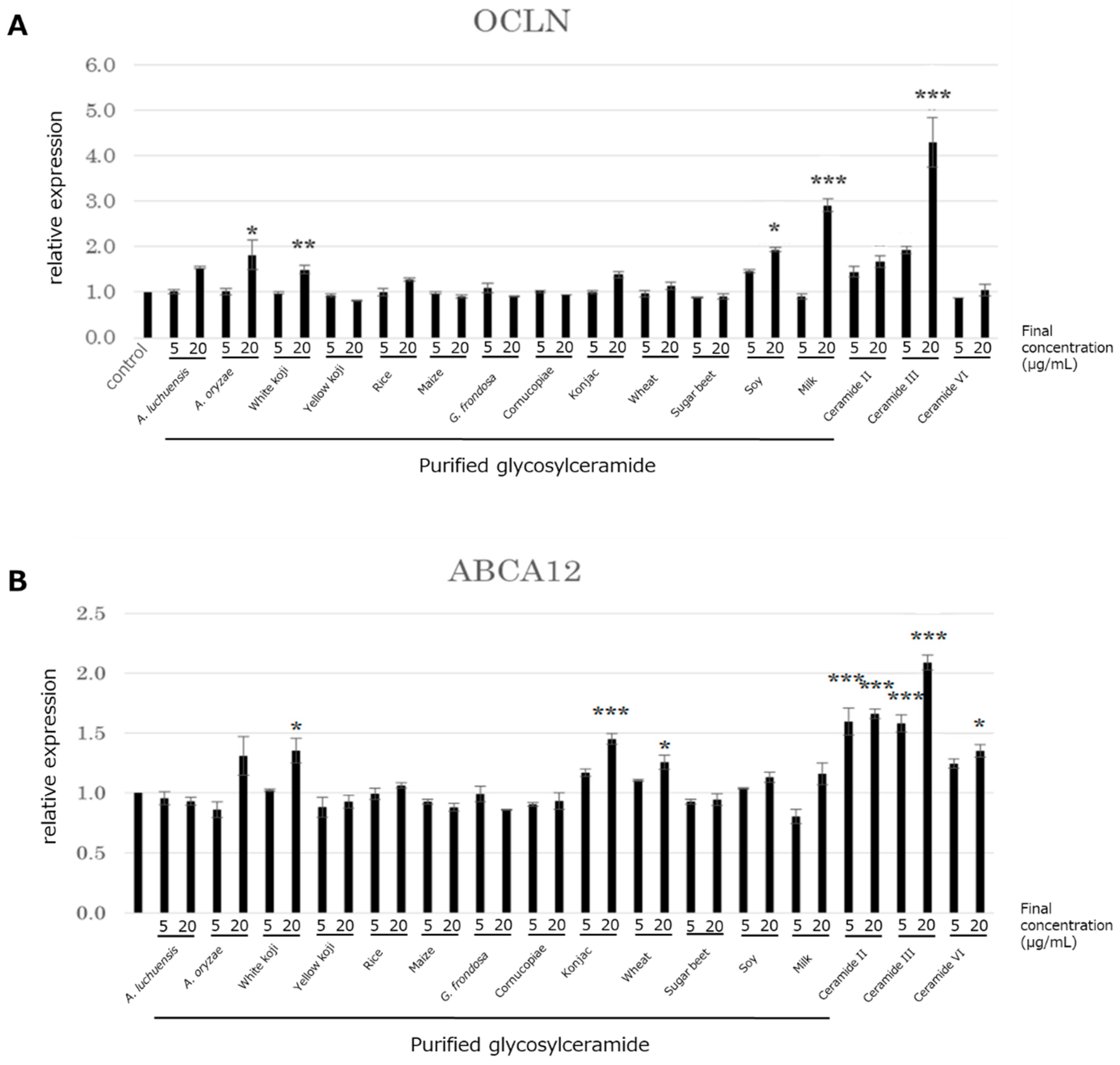
The above results indicated that the glycosylceramide purified from mycelia of A. luchuensis affected the expression of genes involved in skin barrier function in NHEK cells. In order to investigate if these effects are specific to the A. luchuensis glycosylceramide, the effects of glycosylceramides from other sources on the expression of ABCA12 and OCLN in NHEK cells was investigated; namely, glycosylceramide from koji, glucosylceramides from nine kinds of plants, and ceramide. The glycosylceramides purified from A. oryzae, white koji, soy, milk, and ceramide III (NP) significantly increased the expression of OCLN, similarly to that purified from A. luchuensis (n = 3, Dunnett’s test, p < 0.05) (Figure 4A). The glycosylceramides purified from white koji, konjac, wheat, and ceramides II (NS), III (NP), and VI (AP) significantly increased the expression of ABCA12 (n = 3, Dunnett’s test, p < 0.05) (Figure 4B). The results indicated that glycosylceramides and ceramides similarly up-regulated the OCLN and ABCA12 genes, although the effects of the glycosylceramides were generally modest as compared with those of the ceramides.
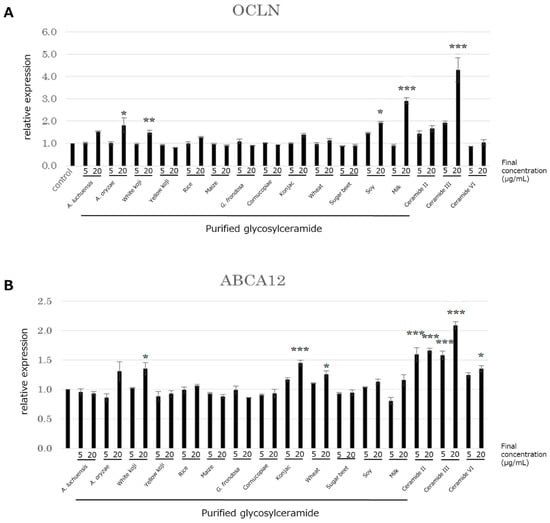
Figure 4.
Expression of the OCLN and ABCA12 genes in NHEK cells upon addition of various glycosylceramides and ceramides. Glycosylceramides and ceramides were incubated with NHEK cells, whereupon the total RNA was extracted and the expression of (A) OCLN and (B) ABCA12 were analysed by real-time PCR. The values on the x-axis indicate the final concentrations of sphingolipids (μg/mL) added (n = 3, Dunnett’s test, *, p < 0.05; **, p < 0.01; ***, p < 0.001).
4.4. Effect of Koji Lipid Extract on the Ceramide Level in Normal Human Epidermal Keratinocytes
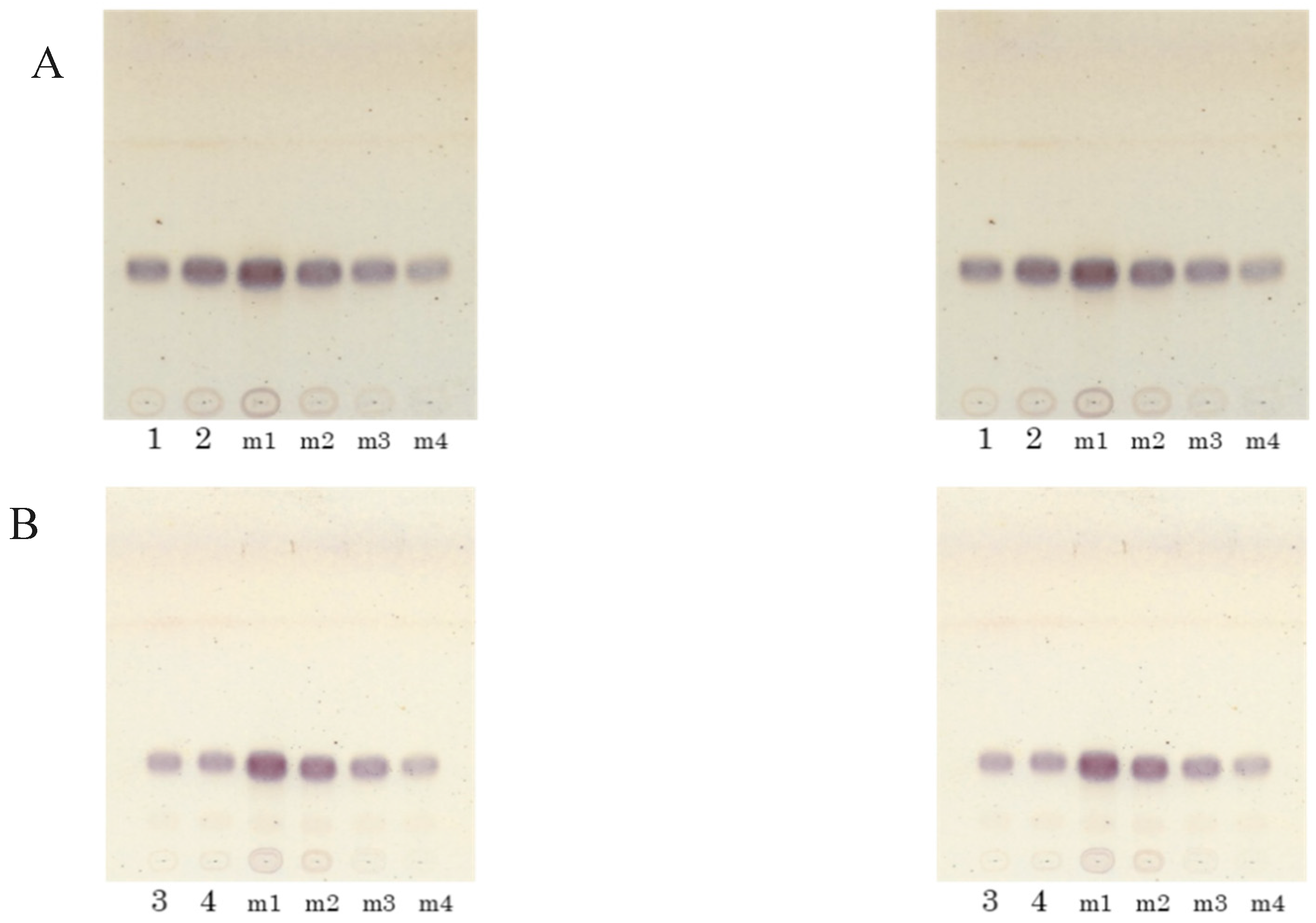
The results described above indicated that koji-derived glycosylceramides up-regulated genes involved in sphingolipid synthesis. Since koji contains glycosylceramides, it can be hypothesised that the lipid extract of koji similarly should increase the ceramide content of the cells. In order to verify this hypothesis, the ceramide content of NHEK cells to which the koji lipid extract had been added was investigated. The Rf value of the ceramides in the keratinocytes was confirmed by HPTLC (Figure 5A). Consistent with the hypothesis, the addition of lipid extract at 10 and 20 μg/mL significantly increased the ceramide II (NS) content of the NHEK cells (n = 3, p < 0.05) (Figure 5B). This result indicated that the koji-derived glycosylceramides functioned to increase the ceramide content in these cells.
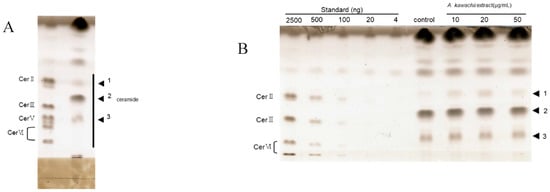
Figure 5.
Increase of ceramide in NHEK cells upon the addition of koji lipid extract. (A) Total lipids were extracted from NHEK cells which had been incubated for 24 h with 10, 20, or 50 μg/mL of lipids (dissolved in 0.5% DMSO) extracted from white koji. (B) The lipid constituents were analysed with HPTLC, developed using a chloroform–methanol–acetic acid solvent (190:9:1, v/v). Band 1 corresponds to ceramide II (NS), and band 3 corresponds to ceramide V (AS).
5. Discussion
Until this study, the mechanism behind the cosmetic effect of koji or Japanese traditional foods (e.g., koji and sake lees) was not known. Furthermore, the effect of glycosylceramides on the gene expression levels in keratinocytes was also not known. In this study, we first elucidated that glycosylceramides or glucosylceramides increased the expression of genes involved in skin barrier function. Indeed, the addition of the lipid extract from koji increased the content of ceramide II in the keratinocytes. These novel results provide a mechanism for the empirical skin barrier-improving effects of glycosylceramide-containing substances, including koji and related traditional Japanese fermented products.
The glycosylceramide purified from white koji increased the expression of OCLN and ABCA12. OCLN encodes occludin, one of the proteins of epidermal tight junctions. Since the expression of OCLN restores the function of tight junctions in disturbed epithelial cells [19], it can be speculated that the increase of OCLN expression will restore skin barrier function. ABCA12, as described earlier, encodes a transporter of lipids that delivers glucosylceramide to epidermal lamellar bodies in keratinocytes, and its dysfunction causes skin-related diseases, such as harlequin-type ichthyosis [20]. Therefore, it can be speculated that the increase of ABCA12 expression would restore the delivery of complex sphingolipids to the lamellar bodies in keratinocytes and accelerate maturation of the skin’s permeability barrier function.
Sphingolipids derived from other sources exhibited effects on the gene expression of OCLN and ABCA12. Other than the glycosylceramide from white koji, konjac glucosylceramide and wheat glucosylceramide had a significant effect on ABCA12 expression. Although the glycosylceramides purified from A. luchuensis, A. oryzae, yellow koji, maize, G. frondosa, tamogitake, sugar beet, soy, and milk also increased the expression of these genes, the increase was not statistically significant (Figure 4A,B). The difference in effects might be attributed to the chemical structure of the glycosylceramides or the relatively small difference in the effect on gene expression, which is not known from the results obtained in this study.
Consistent with a previous report [13], ceramide had a greater effect on ABCA12 expression than glycosylceramide did (Figure 4B). This is likely because ceramide is more hydrophobic than glycosylceramide, resulting in its ability to exert a stronger effect. Alternatively, a small portion of ceramide generated from glycosylceramide might have exerted its effect, since rice koji extract increases the β-glucocerebrosidase levels in human epidermal keratinocytes [21]. Furthermore, ceramide is a signalling molecule, with crucial roles in processes, such as protein kinase C and protein phosphatase 2A activation, in addition to structural components [22]. Moreover, since sphingoid bases (the degraded form of ceramide) activate peroxisome proliferator-activated receptor gamma (PPARγ) [23], which in turn activates ABCA12 [13], the glycosylceramide might have been degraded to sphingoid bases and exerted its effect in this way. These hypotheses await further study.
In conclusion, the effect of the glycosylceramides purified from koji or A. luchuensis on the expression levels of ABCA12 and OCLN in keratinocytes was elucidated in this study. Although the degraded form of glycosylceramide—namely, ceramide—had previously been shown to increase the expression of these genes, it was not known until this study that glycosylceramide exerted an effect on the gene expression of keratinocytes. Indeed, the ceramide content was increased in keratinocytes to which glycosylceramide had been added. The novel information obtained in this study provides an underlying mechanism for the cosmetic effect of koji, which has long been used safely for cosmetic purposes.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, H.K., S.K. and T.O.; Methodology, S.K. and T.O.; Formal Analysis, M.M. and A.F; Investigation, M.M., A.F., M.N and K.K.; Data Curation, M.M.; Writing-Original Draft Preparation, M.M. and A.F.; Writing-Review & Editing, H.K.; Visualization, H.K.; Supervision, H.K.; Project Administration, H.K.; Funding Acquisition, H.K.
Funding
This research was funded by Toyo Shinyaku Co., Ltd.
Acknowledgments
In this section you can acknowledge any support given which is not covered by the author contribution or funding sections. This may include administrative and technical support, or donations in kind (e.g., materials used for experiments).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Available online: https://atopicdermatitis.net/eczema-statistics/ (accessed on 21 May 2019).
- Feingold, K.R.; Elias, P.M. Role of Lipids in the Formation and Maintenance of the Cutaneous Permeability Barrier. Biochim. Biophys. Acta 2014, 1841, 280–294. [Google Scholar] [CrossRef] [PubMed]
- Feingold, K.R. Thematic Review Series: Skin Lipids. The Role of Epidermal Lipids in Cutaneous Permeability Barrier Homeostasis. J. Lipid Res. 2007, 48, 2531–2546. [Google Scholar] [CrossRef] [PubMed]
- Imokawa, G.; Abe, A.; Jin, K.; Higaki, Y.; Kawashima, M.; Hidano, A. Decreased Level of Ceramides in Stratum Corneum of Atopic Dermatitis: An Etiologic Factor in Atopic Dry Skin? J. Investig. Dermatol. 1991, 96, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Shirakura, Y.; Kikuchi, K.; Matsumura, K.; Mukai, K.; Mitsutake, S.; Igarashi, Y. 4,8-Sphingadienine and 4-Hydroxy-8-Sphingenine Activate Ceramide Production in the Skin. Lipids Health Dis. 2012, 11, 108. [Google Scholar] [CrossRef] [PubMed]
- Meurer, M.; Fölster-Holst, R.; Wozel, G.; Weidinger, G.; Jünger, M.; Bräutigam, M.; CASM-DE-01 Study Group. Pimecrolimus Cream in the Long-Term Management of Atopic Dermatitis in Adults: A Six-Month Study. Dermatology 2002, 205, 271–277. [Google Scholar] [CrossRef]
- Del Rosso, J.Q.; Harper, J.; Kircik, L.; Albon, G.; Berson, D.; Hebert, A.; Day, D. Consensus Recommendations on Adjunctive Topical Management of Atopic Dermatitis. J. Drugs Dermatol. 2018, 17, 1070–1076. [Google Scholar] [PubMed]
- Macheleidt, O.; Kaiser, H.W.; Sandhoff, K. Deficiency of Epidermal Protein-Bound Omega-Hydroxyceramides in Atopic Dermatitis. J. Investig. Dermatol. 2002, 119, 166–173. [Google Scholar] [CrossRef]
- Hara, J.; Higuchi, K.; Okamoto, R.; Kawashima, M.; Imokawa, G. High-Expression of Sphingomyelin Deacylase is an Important Determinant of Ceramide Deficiency Leading to Barrier Disruption in Atopic Dermatitis. J. Investig. Dermatol. 2000, 115, 406–413. [Google Scholar] [CrossRef]
- Linter, K.; Mondon, P.; Girard, F.; Gibaud, C. The Effect of a Synthetic Ceramide-2 on Transepidermal Water Loss After Stripping or Sodium Lauryl Sulfate Treatment: An in Vivo Study. Int. J. Cosmet. Sci. 1997, 19, 15–25. [Google Scholar]
- Man, M.Q.; Feingold, K.R.; Thornfeldt, C.R.; Elias, P.M. Optimization of Physiological Lipid Mixtures for Barrier Repair. J. Investig. Dermatol. 1996, 106, 1096–1101. [Google Scholar]
- Shimada, E.; Aida, K.; Sugawara, T.; Hirata, T. Inhibitory Effect of Topical Maize Glucosylceramide on Skin Photoaging in UVA-Irradiated Hairless Mice. J. Oleo Sci. 2011, 60, 321–325. [Google Scholar] [CrossRef][Green Version]
- Jiang, Y.-J.; Uchida, Y.; Lu, B.; Kim, P.; Mao, C.; Akiyama, M.; Elias, P.M.; Holleran, W.M.; Grunfeld, C.; Feingold, K.R. Ceramide Stimulates ABCA12 Expression Via Peroxisome Proliferator-Activated Receptor δ in Human Keratinocytes. J. Biol. Chem. 2009, 284, 18942–18952. [Google Scholar] [CrossRef] [PubMed]
- Hirata, M.; Tsuge, K.; Jayakody, L.N.; Urano, Y.; Sawada, K.; Inaba, S.; Nagao, K.; Kitagaki, H. Structural Determination of Glucosylceramides in the Distillation Remnants of Shochu, the Japanese Traditional Liquor, and Its Production by Aspergillus luchuensis. J. Agric. Food Chem. 2012, 60, 11473–11482. [Google Scholar] [CrossRef]
- Hamajima, H.; Fujikawa, A.; Yamashiro, M.; Ogami, T.; Kitamura, S.; Tsubata, M.; Tan, S.; Matsunaga, H.; Sawada, K.; Kumagai, S.; et al. Chemical Analysis of the Sugar Moiety of Monohexosylceramide Contained in Koji, Japanese Traditional Rice Fermented with Aspergillus. Fermentation 2016, 2, 2. [Google Scholar] [CrossRef]
- Fujino, Y.; Ohnishi, M. Structure of Cerebroside in Aspergillus Oryzae. Biochim. Biophys. Acta 1976, 486, 161–171. [Google Scholar]
- Tani, Y.; Amaishi, Y.; Funatsu, T.; Ito, M.; Itonori, S.; Hata, Y.; Ashida, H.; Yamamoto, K. Structural Analysis of Cerebrosides from Aspergillus Fungi: The Existence of Galactosylceramide in A. Oryzae. Biotechnol. Lett. 2014, 36, 2507–2513. [Google Scholar] [CrossRef]
- Takahashi, K.; Izumi, K.; Nakahata, E.; Hirata, M.; Sawada, K.; Tsuge, K.; Nagao, K.; Kitagaki, H. Quantitation and Structural Determination of Glucosylceramides Contained in Sake Lees. J. Oleo Sci. 2014, 63, 15–23. [Google Scholar] [CrossRef]
- Li, D.; Mrsny, R.J. Oncogenic Raf-1 Disrupts Epithelial Tight Junctions Via Downregulation of Occludin. J. Cell Biol. 2000, 148, 791–800. [Google Scholar] [CrossRef]
- Akiyama, M.; Sugiyama-Nakagiri, Y.; Sakai, K.; McMillan, J.R.; Goto, M.; Arita, K.; Tsuji-Abe, Y.; Tabata, N.; Matsuoka, K.; Sasaki, R.; et al. Mutations in Lipid Transporter ABCA12 in Harlequin Ichthyosis and Functional Recovery by Corrective Gene Transfer. J. Clin. Investig. 2005, 115, 1777–1784. [Google Scholar] [CrossRef]
- Maeda, K.; Ogino, Y.; Nakamura, A.; Nakata, K.; Kitagawa, M.; Ito, S. Identification of Rice Koji Extract Components that Increase β-Glucocerebrosidase Levels in Human Epidermal Keratinocytes. Foods 2018, 7, 94. [Google Scholar] [CrossRef]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and Their Metabolism in Physiology and Disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Esaki, S.; Nagasawa, T.; Tanaka, H.; Tominaga, A.; Mikami, D.; Usuki, S.; Hamajima, H.; Hanamatsu, H.; Sakai, S.; Hama, Y.; et al. The Fungal 9-Methyl-Sphingadiene is a Novel Ligand for Both PPARγ and GPR120. J. Food Biochem. 2018, 42, e12624. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).