Phytohormones and Effects on Growth and Metabolites of Microalgae: A Review
Abstract
1. Introduction
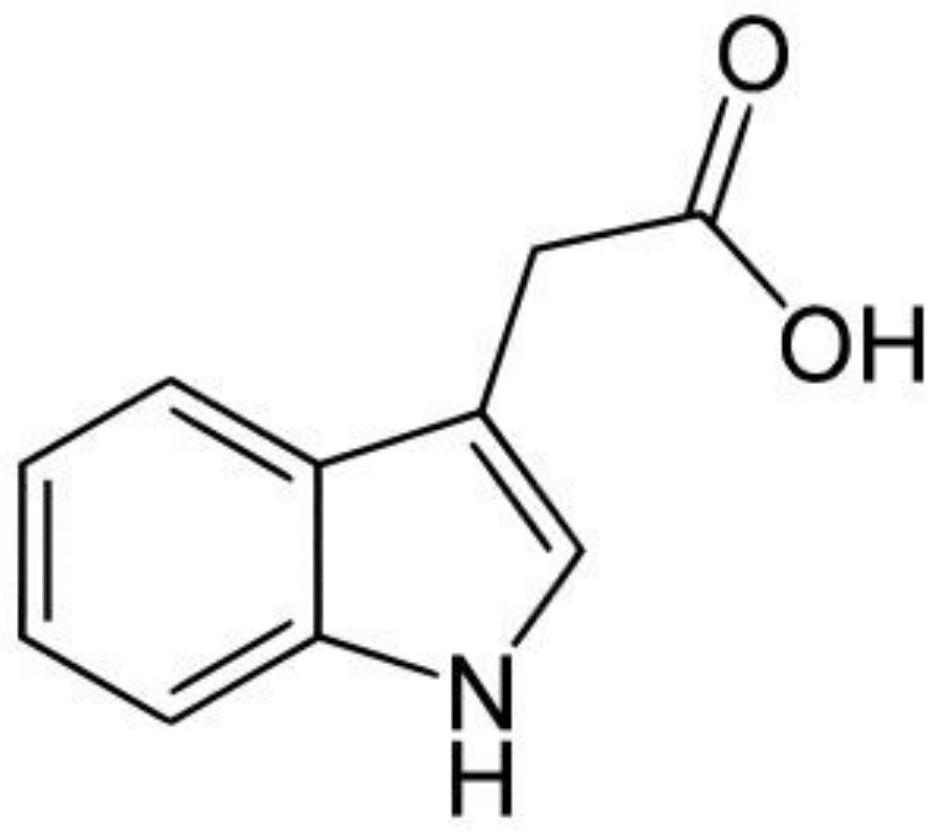
2. Auxins
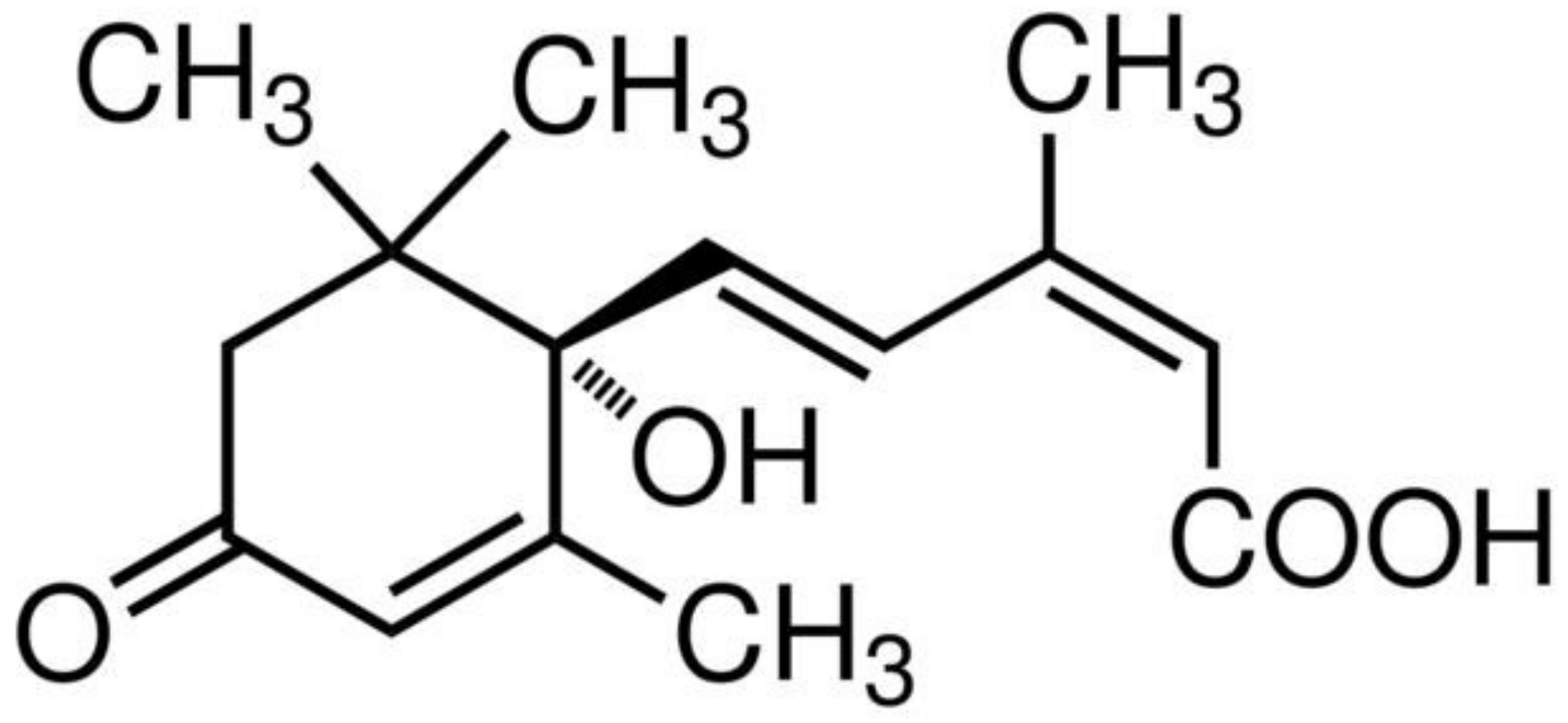
3. Abscisic Acid
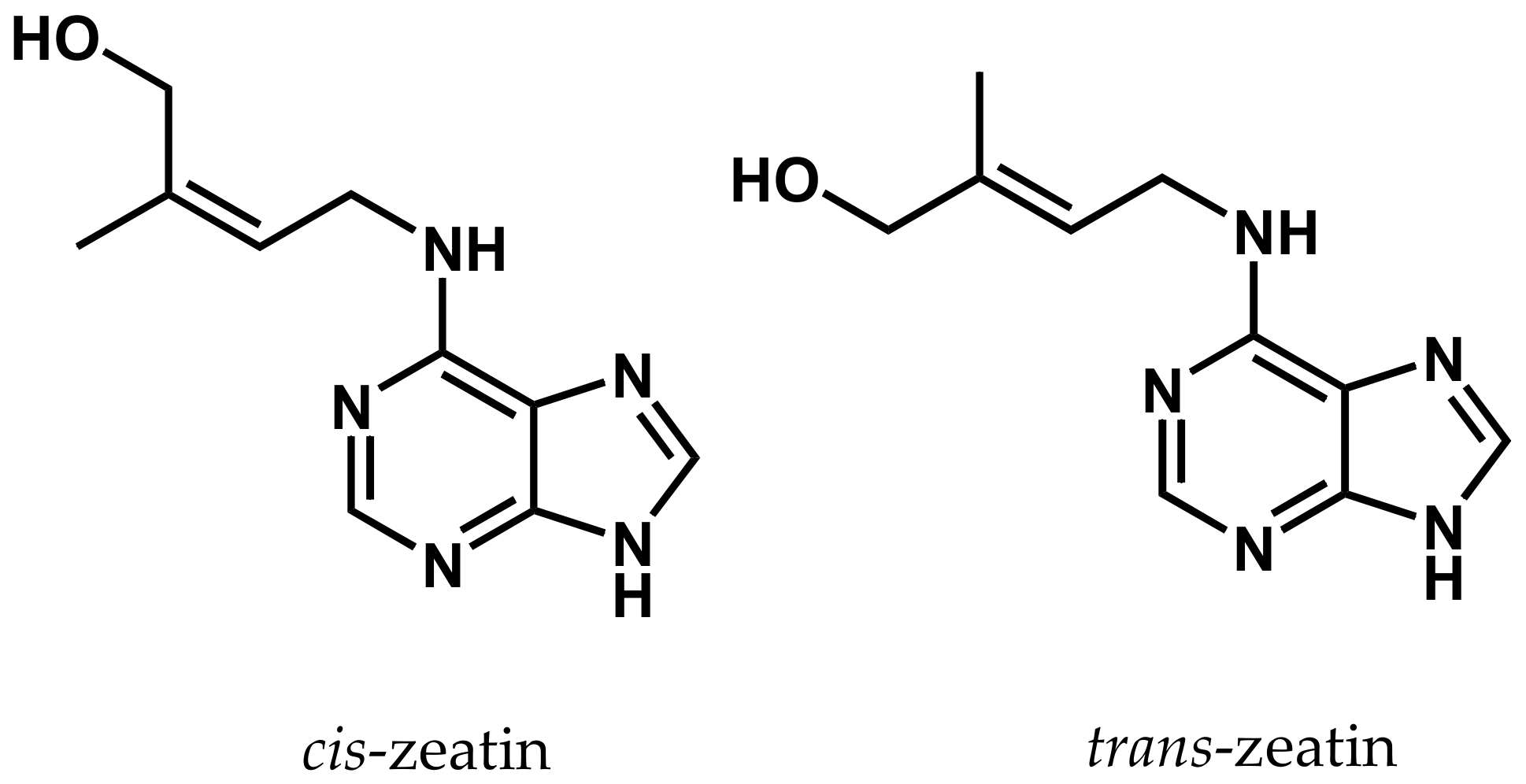
4. Cytokinins
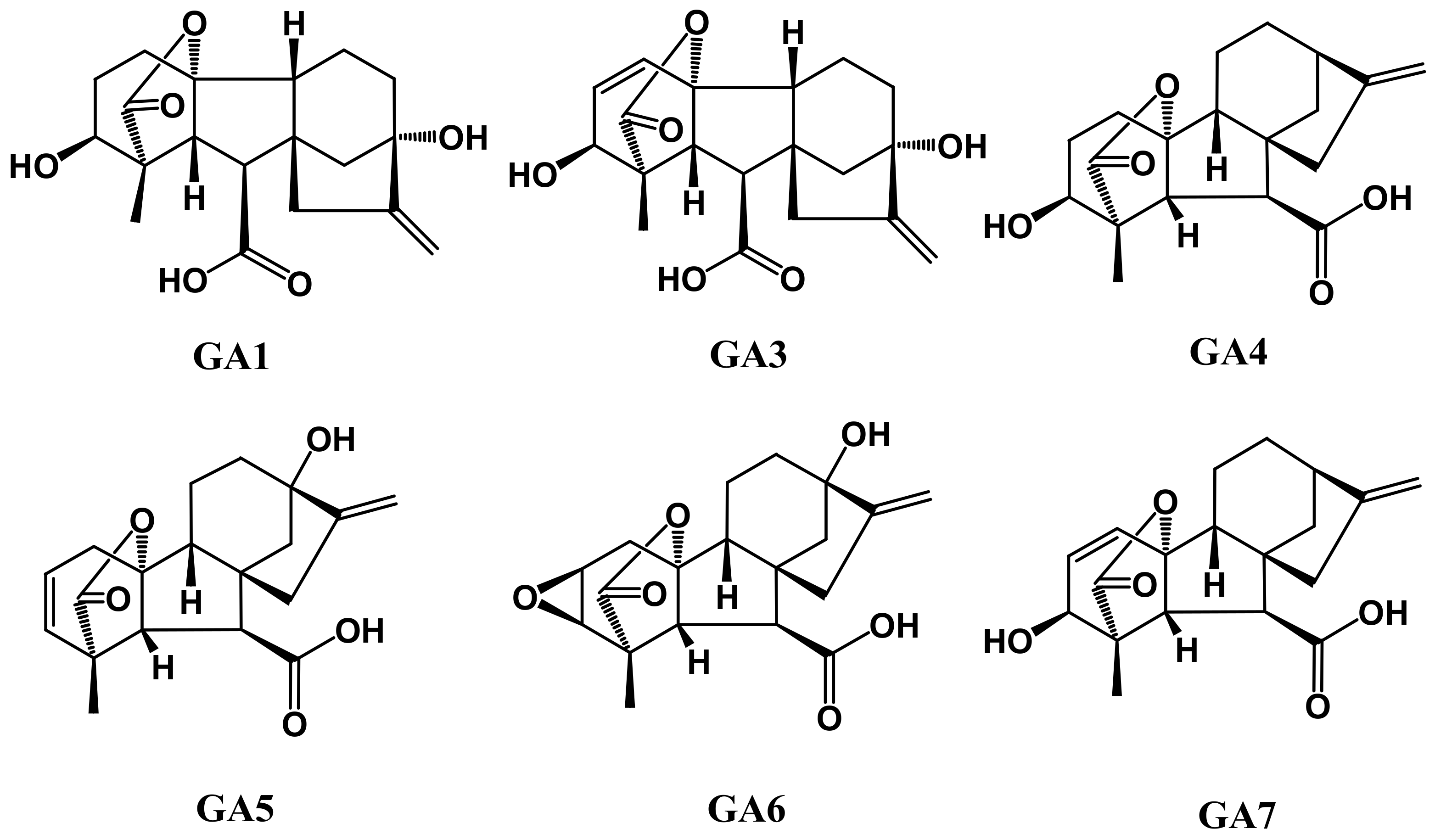
5. Gibberellins
6. Ethylene
7. The Crosstalk for Different Phytohormones
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Darwin, C.; Sir Darwin, F. The Power of Movement in Plants; D. Appleton and Company: New York, NY, USA, 1881; p. 592. [Google Scholar]
- Cardozo, K.H.; Guaratini, T.; Barros, M.P.; Falcão, V.R.; Tonon, A.P.; Lopes, N.P.; Campos, S.; Torres, M.A.; Souza, A.O.; Colepicolo, P.; et al. Metabolites from algae with economical impact. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2007, 146, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshani, I.; Rath, B. Commercial and industrial applications of micro algae—A review. J. Algal Biomass Utln. 2012, 3, 89–100. [Google Scholar]
- Greenwell, H.C.; Laurens, L.M.L.; Shields, R.J.; Lovitt, R.W.; Flynn, K.J. Placing microalgae on the biofuels priority list: A review of the technological challenges. J. R. Soc. Interface 2010, 7, 703–726. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.J. Regulatory factors in hormone action: Level, location and signal transduction. In Plant Hormones; Springer: Dordrecht, The Netherlands, 2010; pp. 16–35. [Google Scholar]
- George, E.F.; Hall, M.A.; De Klerk, G.J. Plant growth regulators I: Introduction; auxins, their analogues and inhibitors. In Plant Propagation by Tissue Culture; Springer: Dordrecht, The Netherlands, 2008; pp. 175–204. [Google Scholar] [CrossRef]
- Del Pozo, J.C.; Lopez-Matas, M.; Ramirez-Parra, E.; Gutierrez, C. Hormonal control of the plant cell cycle. Physiol. Plant. 2005, 123, 173–183. [Google Scholar] [CrossRef]
- Lau, S.; Shao, N.; Bock, R.; Jürgens, G.; De Smet, I. Auxin signaling in algal lineages: Fact or myth? Trends Plant Sci. 2009, 14, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Romanenko, E.A.; Kosakovskaya, I.V.; Romanenko, P.A. Phytohormones of Microalgae: Biological Role and Involvement in the Regulation of Physiological Processes. Pt I. Auxins, Abscisic Acid, Ethylene. Int. J. Algae 2015, 17, 275–289. [Google Scholar] [CrossRef]
- Went, F.W. Phytohormones; The Macmillan Company: New York, NY, USA, 1937. [Google Scholar]
- Rotino, G.L.; Perri, E.; Zottini, M.; Sommer, H.; Spena, A. Genetic engineering of parthenocarpic plants. Nat. Biotechnol. 1997, 15, 1398–1401. [Google Scholar] [CrossRef] [PubMed]
- Yokoya, N.S.; Handro, W. Effects of auxins and cytokinins on tissue culture of Grateloupia dichotoma (Gigartinales, Rhodophyta). Hydrobiologia 1996, 326, 393–400. [Google Scholar] [CrossRef]
- Stirk, W.A.; Van Staden, J. Comparison of cytokinin-and auxin-like activity in some commercially used seaweed extracts. J. Appl. Phycol. 1996, 8, 503–508. [Google Scholar] [CrossRef]
- Le Bail, A.; Billoud, B.; Kowalczyk, N.; Kowalczyk, M.; Gicquel, M.; Le Panse, S.; Stewart, S.; Scornet, D.; Cock, J.M.; Ljung, K.; et al. Auxin metabolism and function in the multicellular brown alga Ectocarpus siliculosus. Plant Physiol. 2010, 153, 128–144. [Google Scholar] [CrossRef] [PubMed]
- Kozlova, T.A.; Hardy, B.P.; Krishna, P.; Levin, D.B. Effect of phytohormones on growth and accumulation of pigments and fatty acids in the microalgae Scenedesmus quadricauda. Algal Res. 2017, 27, 325–334. [Google Scholar] [CrossRef]
- Pratt, R. Influence of auxins on the growth of Chlorella vulgaris. Am. J. Bot. 1938, 25, 498–501. [Google Scholar] [CrossRef]
- Salama, E.S.; Kabra, A.N.; Ji, M.K.; Kim, J.R.; Min, B.; Jeon, B.H. Enhancement of microalgae growth and fatty acid content under the influence of phytohormones. Bioresour. Technol. 2014, 172, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Kokkiligadda, S.; Pandey, B.; Ronda, S.R. Effect of plant growth regulators on production of alpha-linolenic acid from microalgae Chlorella pyrenoidosa. Sādhanā 2017, 42, 1821–1824. [Google Scholar] [CrossRef]
- Dao, G.H.; Wu, G.X.; Wang, X.X.; Zhuang, L.L.; Zhang, T.Y.; Hu, H.Y. Enhanced growth and fatty acid accumulation of microalgae Scenedesmus sp. LX1 by two types of auxin. Bioresour. Technol. 2018, 247, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska-Niczyporuk, A.; Bajguz, A. The effect of natural and synthetic auxins on the growth, metabolite content and antioxidant response of green alga Chlorella vulgaris (Trebouxiophyceae). Plant Growth Regul. 2014, 73, 57–66. [Google Scholar] [CrossRef]
- Liu, J.; Qiu, W.; Song, Y. Stimulatory effect of auxins on the growth and lipid productivity of Chlorella pyrenoidosa and Scenedesmus quadricauda. Algal Res. 2016, 18, 273–280. [Google Scholar] [CrossRef]
- Parsaeimehr, A.; Mancera-Andrade, E.I.; Robledo-Padilla, F.; Iqbal, H.M.; Parra-Saldivar, R. A chemical approach to manipulate the algal growth, lipid content and high-value alpha-linolenic acid for biodiesel production. Algal Res. 2017, 26, 312–322. [Google Scholar] [CrossRef]
- Jusoh, M.; Loh, S.H.; Chuah, T.S.; Aziz, A.; San Cha, T. Indole-3-acetic acid (IAA) induced changes in oil content, fatty acid profiles and expression of four fatty acid biosynthetic genes in Chlorella vulgaris at early stationary growth phase. Phytochemistry 2015, 111, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Czerpak, R.; Bajguz, A.; Białecka, B.; Wierzchołowska, L.E.; Wolańska, M.M. Effect of auxin precursors and chemical analogues on the growth and chemical composition in Chlorella pyrenoidosa Chick. Acta Soc. Bot. Pol. 1994, 63, 279–286. [Google Scholar] [CrossRef]
- Piotrowska-Niczyporuk, A.; Bajguz, A.; Zambrzycka, E.; Godlewska-Żyłkiewicz, B. Phytohormones as regulators of heavy metal biosorption and toxicity in green alga Chlorella vulgaris (Chlorophyceae). Plant Physiol. Biochem. 2012, 52, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Qiu, W.; Song, Y.; Peng, H.; Zhao, Y. The growth and lipid productivity of Chlorella pyrenoidosa enhanced by plant hormones under ammonium stress. Environ. Prog. Sustain. Energy 2017, 36, 1187–1193. [Google Scholar] [CrossRef]
- Park, W.K.; Yoo, G.; Moon, M.; Kim, C.W.; Choi, Y.E.; Yang, J.W. Phytohormone supplementation significantly increases growth of Chlamydomonas reinhardtii cultivated for biodiesel production. Appl. Biochem. Biotechnol. 2013, 171, 1128–1142. [Google Scholar] [CrossRef] [PubMed]
- El Arroussi, H.; Benhima, R.; Bennis, I.; El Mernissi, N.; Wahby, I. Improvement of the potential of Dunaliella tertiolecta as a source of biodiesel by auxin treatment coupled to salt stress. Renew. Energy 2015, 77, 15–19. [Google Scholar] [CrossRef]
- Trinh, C.T.; Tran, T.H.; Bui, T.V. Effects of plant growth regulators on the growth and lipid accumulation of Nannochloropsis oculata (droop) Hibberd. In AIP Conference Proceedings; No. 1, 020017; AIP Publishing: Melville, NY, USA, 2017; Volume 1878. [Google Scholar] [CrossRef]
- Udayan, A.; Arumugam, M. Selective enrichment of Eicosapentaenoic acid (20:5n-3) in N. oceanica CASA CC201 by natural auxin supplementation. Bioresour. Technol. 2017, 242, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, P.; Morowvat, M.H.; Montazeri-Najafabady, N.; Abolhassanzadeh, Z.; Mohagheghzadeh, A.; Hamidi, M.; Niazi, A.; Ghasemi, Y. Investigating the effects of phytohormones on growth and β-carotene production in a naturally isolates stain of Dunaliella salina. J. Appl. Pharm. Sci. 2016, 6, 164–171. [Google Scholar] [CrossRef]
- Wu, G.; Gao, Z.; Du, H.; Lin, B.; Yan, Y.; Li, G.; Guo, Y.; Fu, S.; Wei, G.; Wang, M.; et al. The effects of abscisic acid, salicylic acid and jasmonic acid on lipid accumulation in two freshwater Chlorella strains. J. Gen. Appl. Microbiol. 2018, 64, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Pool, P.Y.; Peraza-Echeverria, S.; Ku-González, Á.F.; Herrera-Valencia, V.A. The phytohormone abscisic acid increases triacylglycerol content in the green microalga Chlorella saccharophila (Chlorophyta). Algae 2016, 31, 267–276. [Google Scholar] [CrossRef]
- Kobayashi, M.; Hirai, N.; Kurimura, Y.; Ohigashi, H.; Tsuji, Y. Abscisic acid-dependent algal morphogenesis in the unicellular green alga Haematococcus pluvialis. Plant Growth Regul. 1997, 22, 79–85. [Google Scholar] [CrossRef]
- Cowan, A.K.; Rose, P.D. Abscisic acid metabolism in salt-stressed cells of Dunaliella salina: Possible interrelationship with β-carotene accumulation. Plant Physiol. 1991, 97, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Renuka, N.; Guldhe, A.; Singh, P.; Ansari, F.A.; Rawat, I.; Bux, F. Evaluating the potential of cytokinins for biomass and lipid enhancement in microalga Acutodesmus obliquus under nitrogen stress. Energy Convers. Manag. 2017, 140, 14–23. [Google Scholar] [CrossRef]
- Piotrowska, A.; Czerpak, R. Cellular response of light/dark-grown green alga Chlorella vulgaris Beijerinck (Chlorophyceae) to exogenous adenine-and phenylurea-type cytokinins. Acta Physiol. Plant. 2009, 31, 573–585. [Google Scholar] [CrossRef]
- Souza, J.M.; Yokoya, N.S. Effects of cytokinins on physiological and biochemical responses of the agar-producing red alga Gracilaria caudata (Gracilariales, Rhodophyta). J. Appl. Phycol. 2016, 28, 3491–3499. [Google Scholar] [CrossRef]
- Du, K.; Tao, H.; Wen, X.; Geng, Y.; Li, Y. Enhanced growth and lipid production of Chlorella pyrenoidosa by plant growth regulator GA3. Fresenius Environ. Bull. 2015, 24, 3414–3419. [Google Scholar]
- González-Garcinuño, Á.; Sánchez-Álvarez, J.M.; Galán, M.A.; Martin del Valle, E.M. Understanding and optimizing the addition of phytohormones in the culture of microalgae for lipid production. Biotechnol. Prog. 2016, 32, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Falkowska, M.; Pietryczuk, A.; Piotrowska, A.; Bajguz, A.; Grygoruk, A.; Czerpak, R. The effect of gibberellic acid (GA3) on growth, metal biosorption and metabolism of the green algae Chlorella vulgaris (Chlorophyceae) Beijerinck exposed to cadmium and lead stress. Pol. J. Environ. Stud. 2011, 20, 53–59. [Google Scholar]
- Pan, X.; Chang, F.; Kang, L.; Liu, Y.; Li, G.; Li, D. Effects of gibberellin A3 on growth and microcystin production in Microcystis aeruginosa (cyanophyta). J. Plant Physiol. 2008, 165, 1691–1697. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Lim, S.R.; Hong, S.J.; Cho, B.K.; Lee, H.; Lee, C.G.; Choi, H.K. Effect of ethephon as an ethylene-releasing compound on the metabolic profile of Chlorella vulgaris. J. Agric. Food Chem. 2016, 64, 4807–4816. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.Q.; Meng, C.X. Impact of extraneous ethylene concentrations to astaxanthin accumulation of Haematoccus pluvialis. Food Sci. 2007, 10, 093. [Google Scholar]
- Vo, T.T.; Lee, C.; Han, S.I.; Kim, J.Y.; Kim, S.; Choi, Y.E. Effect of the ethylene precursor, 1-aminocyclopropane-1-carboxylic acid on different growth stages of Haematococcus pluvialis. Bioresour. Technol. 2016, 220, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Hunt, R.W.; Chinnasamy, S.; Bhatnagar, A.; Das, K.C. Effect of biochemical stimulants on biomass productivity and metabolite content of the microalga, Chlorella sorokiniana. Appl. Biochem. Biotechnol. 2010, 162, 2400–2414. [Google Scholar] [CrossRef] [PubMed]
- Bajguz, A.; Piotrowska-Niczyporuk, A. Synergistic effect of auxins and brassinosteroids on the growth and regulation of metabolite content in the green alga Chlorella vulgaris (Trebouxiophyceae). Plant Physiol. Biochem. 2013, 71, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Bajguz, A.; Piotrowska-Niczyporuk, A. Interactive effect of brassinosteroids and cytokinins on growth, chlorophyll, monosaccharide and protein content in the green alga Chlorella vulgaris (Trebouxiophyceae). Plant Physiol. Biochem. 2014, 80, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Pei, H.; Jiang, L.; Hou, Q.; Nie, C.; Zhang, L. Phytohormone addition coupled with nitrogen depletion almost tripled the lipid productivities in two algae. Bioresour. Technol. 2018, 247, 904–914. [Google Scholar] [CrossRef]
- Salama, E.S.; Jeon, B.H.; Chang, S.W.; Lee, S.H.; Roh, H.S.; Yang, I.S.; Kurade, M.B.; El-Dalatony, M.M.; Kim, D.H.; Kim, K.H.; et al. Interactive effect of indole-3-acetic acid and diethyl aminoethyl hexanoate on the growth and fatty acid content of some microalgae for biodiesel production. J. Clean. Prod. 2017, 168, 1017–1024. [Google Scholar] [CrossRef]
- Tatkowska, E.; Buczek, J. Effect of phytohormones on the growth of Scenedesmus quadricauda (Turp.) Bréh. Acta Soc. Bot. Pol. 2014, 49, 211–220. [Google Scholar] [CrossRef][Green Version]
- Liu, W.C.; Carnsdagger, H.R. Isolation of abscisin, an abscission accelerating substance. Science 1961, 134, 384–385. [Google Scholar] [CrossRef] [PubMed]
- Wareing, P.F.; Eagles, C.F.; Robinson, P.M. Natural inhibitors as dormancy agents. Coll. Int. Centr. Natl. Rech. Sci. 1964, 123, 377–378. [Google Scholar]
- Addicott, F.T.; Lyon, J.L.; Ohkuma, K.; Thiessen, W.E.; Carns, H.R.; Smith, O.E.; Cornforth, J.W.; Milborrow, B.V.; Ryback, G.; Wareing, P.F. Abscisic acid: A new name for abscisin II (dormin). Science 1968, 159, 1493. [Google Scholar] [CrossRef] [PubMed]
- Tietz, A.; Ruttkowski, U.; Kohler, R.; Kasprik, W. Further investigations on the occurrence and the effects of abscisic acid in algae. Biochem. Physiol. Pflanz. 1989, 184, 259–266. [Google Scholar] [CrossRef]
- Xu, Z.Y.; Yoo, Y.J.; Hwang, I. ABA Conjugates and Their Physiological Roles in Plant Cells. In Abscisic Acid: Metabolism, Transport and Signaling; Springer: Dordrecht, The Netherlands, 2014; pp. 77–87. [Google Scholar] [CrossRef]
- Frey, A.; Audran, C.; Marin, E.; Sotta, B.; Marion-Poll, A. Engineering seed dormancy by the modification of zeaxanthin epoxidase gene expression. Plant Mol. Biol. 1999, 39, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Zakharychev, V.V. Fitogormony, Ikh Analogi i Antagonisty v Kachestve Gerbitsidov i Regulyatorov Rosta Rastenii (Phytohormones, Their Analogues and Antagonists as Herbicides and Plant Growth Regulators); RKhTU Im. D.I. Mendeleeva: Moscow, Russia, 1999. [Google Scholar]
- Kentzer, T.; Mazur, H. Abscisic acid as endogenous inhibitor of the marine diatom Coscinodiscus granii. Acta Physiol. Plant. 1991, 13, 153–157. [Google Scholar]
- Lu, Y.; Tarkowská, D.; Turečková, V.; Luo, T.; Xin, Y.; Li, J.; Wang, Q.; Jiao, N.; Strnad, M.; Xu, J. Antagonistic roles of abscisic acid and cytokinin during response to nitrogen depletion in oleaginous microalga Nannochloropsis oceanica expand the evolutionary breadth of phytohormone function. Plant J. 2014, 80, 52–68. [Google Scholar] [CrossRef] [PubMed]
- Khasin, M.; Cahoon, R.E.; Alvarez, S.; Beckeris, R.G.; Eyun, S.I.; Jia, Q.; Riethoven, J.J.; Nickerson, K.W.; Riekhof, W.R. Synthesis, secretion, and perception of abscisic acid regulates stress responses in Chlorella sorokiniana. bioRxiv 2017, 180547. [Google Scholar] [CrossRef]
- Beck, E.H.; Fettig, S.; Knake, C.; Hartig, K.; Bhattarai, T. Specific and unspecific responses of plants to cold and drought stress. J. Biosci. 2007, 32, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Zeevaart, J.A. Overexpression of a 9-cis-epoxycarotenoid dioxygenase gene in Nicotiana plumbaginifolia increases abscisic acid and phaseic acid levels and enhances drought tolerance. Plant Physiol. 2002, 128, 544–551. [Google Scholar] [CrossRef] [PubMed]
- George, E.F.; Hall, M.A.; De Klerk, G.J. Plant Growth Regulators III: Gibberellins, Ethylene, Abscisic Acid, their Analogues and Inhibitors; Miscellaneous Compounds. In Plant Propagation by Tissue Culture; Springer: Dordrecht, The Netherlands, 2008; pp. 227–281. [Google Scholar] [CrossRef]
- Maršálek, B.; Zahradníčková, H.; Hronková, M. Extracellular abscisic acid produced by cyanobacteria under salt stress. J. Plant Physiol. 1992, 139, 506–508. [Google Scholar] [CrossRef]
- Yoshida, K.; Igarashi, E.; Wakatsuki, E.; Miyamoto, K.; Hirata, K. Mitigation of osmotic and salt stresses by abscisic acid through reduction of stress-derived oxidative damage in Chlamydomonas reinhardtii. Plant Sci. 2004, 167, 1335–1341. [Google Scholar] [CrossRef]
- Sulochana, S.B.; Arumugam, M. Influence of abscisic acid on growth, biomass and lipid yield of Scenedesmus quadricauda under nitrogen starved condition. Bioresour. Technol. 2016, 213, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.O.; Skoog, F.; Von Saltza, M.H.; Strong, F.M. Kinetin, a cell division factor from deoxyribonucleic acid. J. Am. Chem. Soc. 1955, 77, 1392–1392. [Google Scholar] [CrossRef]
- Hirose, N.; Takei, K.; Kuroha, T.; Kamada-Nobusada, T.; Hayashi, H.; Sakakibara, H. Regulation of cytokinin biosynthesis, compartmentalization and translocation. J. Exp. Bot. 2007, 59, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Werner, T.; Schmülling, T. Cytokinin action in plant development. Curr. Opin. Plant Biol. 2009, 12, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Yamane, H.; Takahashi, N.; Chapman, D.J.; Phinney, B.O. Identification of a cytokinin in the green alga Chara globularis. Phytochemistry 1989, 28, 337–338. [Google Scholar] [CrossRef]
- Romanenko, K.O.; Kosakovskaya, I.V.; Romanenko, P.O. Phytohormones of Microalgae: Biological Role and Involvement in the Regulation of Physiological Processes. Pt II. Cytokinins and Gibberellins. Int. J. Algae 2016, 18, 179–201. [Google Scholar] [CrossRef]
- Stirk, W.A.; Ördög, V.; Novák, O.; Rolčík, J.; Strnad, M.; Bálint, P.; Staden, J. Auxin and cytokinin relationships in 24 microalgal strains1. J. Phycol. 2013, 49, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Mok, D.W.; Mok, M.C. Cytokinin metabolism and action. Annu. Rev. Plant Biol. 2001, 52, 89–118. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.D.; Karas, B.J.; Sato, S.; Tabata, S.; Amyot, L.; Szczyglowski, K. A cytokinin perception mutant colonized by Rhizobium in the absence of nodule organogenesis. Science 2007, 315, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.; Vankova, R.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Cytokinins: Metabolism and function in plant adaptation to environmental stresses. Trends Plant Sci. 2012, 17, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Zalabák, D.; Pospíšilová, H.; Šmehilová, M.; Mrízová, K.; Frébort, I.; Galuszka, P. Genetic engineering of cytokinin metabolism: Prospective way to improve agricultural traits of crop plants. Biotechnol. Adv. 2013, 31, 97–117. [Google Scholar] [CrossRef] [PubMed]
- Stirk, W.A.; Bálint, P.; Tarkowská, D.; Novák, O.; Maróti, G.; Ljung, K.; Turečková, V.; Strnad, M.; Ördög, V.; Van Staden, J. Effect of light on growth and endogenous hormones in Chlorella minutissima (Trebouxiophyceae). Plant Physiol. Biochem. 2014, 79, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Stirk, W.A.; van Staden, J.; Novák, O.; Doležal, K.; Strnad, M.; Dobrev, P.I.; Sipos, G.; Ördög, V.; Balint, P. Changes in endogenous cytokinin concentrations in Chlorella (Chlorophyceae) in relation to light and the cell cycle. J. Phycol. 2011, 47, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.J.; Wang, Y.; Zhu, Y.R.; Lü, X.Y.; Huang, K.; Shao, N.; Beck, C.F. Synthesis of the photorespiratory key enzyme serine: Glyoxylate aminotransferase in C. reinhardtii is modulated by the light regime and cytokinin. Physiol. Plant. 2006, 127, 571–582. [Google Scholar] [CrossRef]
- Kurosawa, E. Experimental studies on the nature of the substance secreted by the “bakanae” fungus. Nat. Hist. Soc. Formosa 1926, 16, 213–227. [Google Scholar]
- Yabuta, T. On the crystal of gibberellin, a substance to promote plant growth. J. Agric. Chem. Soc. Jpn. 1938, 14, 1526–1938. [Google Scholar]
- Mowat, J.A. A survey of results on the occurrence of auxins and gibberellins in algae. Bot. Mar. 1965, 8, 149–155. [Google Scholar] [CrossRef]
- Sponsel, V.M.; Hedden, P. Gibberellin biosynthesis and inactivation. In Plant Hormones; Springer: Dordrecht, The Netherlands, 2010; pp. 63–94. [Google Scholar] [CrossRef]
- Huang, S.; Raman, A.S.; Ream, J.E.; Fujiwara, H.; Cerny, R.E.; Brown, S.M. Overexpression of 20-oxidase confers a gibberellin-overproduction phenotype in Arabidopsis. Plant Physiol. 1998, 118, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Croker, S.J.; García-Lepe, R.; Lewis, M.J.; Hedden, P. Modification of gibberellin production and plant development in Arabidopsis by sense and antisense expression of gibberellin 20-oxidase genes. Plant J. 1999, 17, 547–556. [Google Scholar] [CrossRef]
- Stirk, W.A.; Bálint, P.; Tarkowská, D.; Novák, O.; Strnad, M.; Ördög, V.; Van Staden, J. Hormone profiles in microalgae: Gibberellins and brassinosteroids. Plant Physiol. Biochem. 2013, 70, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.J.; Sun, J.; Sun, Y.Q.; Zheng, J.Y.; Wang, Z. Metabolomics analysis of phytohormone gibberellin improving lipid and DHA accumulation in Aurantiochytrium sp. Biochem. Eng. J. 2016, 112, 258–268. [Google Scholar] [CrossRef]
- Pierik, R.; Tholen, D.; Poorter, H.; Visser, E.J.; Voesenek, L.A. The Janus face of ethylene: Growth inhibition and stimulation. Trends Plant Sci. 2006, 11, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.L.C.; Li, H.; Ecker, J.R. Ethylene biosynthesis and signaling networks. Plant Cell 2002, 14 (Suppl. 1), S131–S151. [Google Scholar] [CrossRef]
- Neljubow, D. Uber die horizontale Nutation der Stengel von Pisum Sativum und einiger anderen Planzen. Bot. Centralbl. Beih. 1901, 10, 128–139. [Google Scholar]
- Denny, F.E. Effect of Ethylene upon Respiration of Lemons. Bot. Gaz. 1924, 77, 322–329. [Google Scholar] [CrossRef]
- Abeles, F.B. Fruit ripening, abscission, and postharvest disorders. In Ethylene in Plant Biology; Academic Press: New York, NY, USA, 1992; pp. 182–221. [Google Scholar] [CrossRef]
- Ju, C.; Van de Poel, B.; Cooper, E.D.; Thierer, J.H.; Gibbons, T.R.; Delwiche, C.F.; Chang, C. Conservation of ethylene as a plant hormone over 450 million years of evolution. Nat. Plants 2015, 1, 14004. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.C.; Chow, T.J. Ethylene production by blue-green algae. Bot. Bull. Acad. Sin. 1984, 25, 81–86. [Google Scholar]
- Maillard, P.; Thepenier, C.; Gudin, C. Determination of an ethylene biosynthesis pathway in the unicellular green alga, Haematococcus pluvialis. Relationship between growth and ethylene production. J. Appl. Phycol. 1993, 5, 93–98. [Google Scholar] [CrossRef]
- Plettner, I.N.A.; Steinke, M.; Malin, G. Ethene (ethylene) production in the marine macroalga Ulva (Enteromorpha) intestinalis L. (Chlorophyta, Ulvophyceae): Effect of light-stress and co-production with dimethyl sulphide. Plant Cell Environ. 2005, 28, 1136–1145. [Google Scholar] [CrossRef]
- Achard, P.; Vriezen, W.H.; Van Der Straeten, D.; Harberd, N.P. Ethylene Regulates Arabidopsis Development via the Modulation of DELLA Protein Growth Repressor Function. Plant Cell 2003, 15, 2816–2825. [Google Scholar] [CrossRef] [PubMed]
- Bajguz, A. Brassinosteroid enhanced the level of abscisic acid in Chlorella vulgaris subjected to short-term heat stress. J. Plant Physiol. 2009, 166, 882–886. [Google Scholar] [CrossRef] [PubMed]
- De Jesus Raposo, M.F. Influence of the growth regulators kinetin and 2, 4-D on the growth of two chlorophyte microalgae, Haematococcus pluvialis and Dunaliella salina. J. Basic Appl. Sci. 2013, 9, 302–308. [Google Scholar] [CrossRef]
- Teng, Y.; Wang, X.; Li, X. Effects of Gibberellin and Naphthyleneacetic Acid on Growth of Skeletonema costatum. Chin. Agric. Sci. Bull. 2006, 5, 113. [Google Scholar]
- Yu, Z.; Song, M.; Pei, H.; Jiang, L.; Hou, Q.; Nie, C.; Zhang, L. The effects of combined agricultural phytohormones on the growth, carbon partitioning and cell morphology of two screened algae. Bioresour. Technol. 2017, 239, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, M.J.; Thorne, J.L.; Wikström, N.; Bremer, K. Molecular evidence on plant divergence times. Am. J. Bot. 2004, 91, 1656–1665. [Google Scholar] [CrossRef] [PubMed]
| Specific | Species | Targets Promoted | Reference | |
|---|---|---|---|---|
| Auxins | Auxins | Scenedesmus quadricauda | Cell size, growth, biomass, chlorophyll-a, carotenoids, fatty acids | [15] |
| IAA | Nannochloropsis oculata | Cell density, division, chlorophyll-a | [29] | |
| IAA | Nannochloropsis oceanica | Growth, lipid, PUFA, EPA | [30] | |
| IAA | Scenedesmus obliquus | Growth, fatty acid, protein, carbohydrate | [17] | |
| Auxins | Chlorella vulgaris | Pigments, soluble proteins, monosaccharides | [20] | |
| IAA | Dunaliella salina | Growth, β-carotene | [31] | |
| ABA | ABA | Dunaliella salina | Growth, β-carotene | [31] |
| ABA | Chlorella pyrenoidosa | Lipid | [26] | |
| ABA | Chlorella sp. | Lipid | [32] | |
| ABA | Chlorella saccharophila | Lipid, TAG | [33] | |
| ABA | Haematococcus pluvialis | Carotenoids | [34] | |
| ABA | Dunaliella salina | β-carotene | [35] | |
| CKs | Kinetin, Zeatin | Acutodesmus obliquus | Biomass, lipid, carbohydrate | [36] |
| CK | Chlorella protothecoides | Biomass, lipid, ALA | [22] | |
| Kinetin | Dunaliella salina | Growth, β-carotene | [31] | |
| CKs | Chlorella vulgaris | Cell divisions, pigments | [37] | |
| CK | Gracilaria caudata | Pigments, proteins | [38] | |
| GAs | GA3 | Chlorella pyrenoldosa | Growth, lipid, UFAs | [39] |
| GA | Nannochloropsis oculata | Cell diameter, lipid | [29] | |
| GA | Chlorella ellipsoidea | Growth, lipid | [40] | |
| GA3 | Chlorella vulgaris | Cell number, pigment, protein, monosaccharide | [41] | |
| GA3 | Microcystis aeruginosa | Dry weight, cell number, chlorophyll-a, phycocyanin, protein | [42] | |
| GA3 | Chlamydomonas reinhardtii | Cell number and size, dry weight, chlorophylls, biodiesel, proteins | [27] | |
| Ethylene and its precusors | Ethephon | Chlorella vulgaris | SFAs, a-tocopherol, c-aminobutyric acid, asparagine, proline | [43] |
| Ethylene | Haematococcus pluvialis | Astaxanthin | [44] | |
| ACC | Haematococcus pluvialis | Astaxanthin | [45] | |
| Combinations | NAA + Zeatin | Chlorella sorokiniana | Biomas, chlorophyll-a, lipid | [46] |
| Auxins + BL | Chlorella vulgaris | Chlorophylls, proteins, monosaccharides | [47] | |
| CKs + BR | Chlorella vulgaris | Cell number, chlorophylls, proteins, monosaccharides | [48] | |
| IBA + NAA | Scenedesmus sp., Chlorella sorokiniana | Lipid | [49] | |
| IAA + DAH | Scenedesmu obliquusi, Ourococcus multisporus, Chlorella vulgaris | Growth, PUFAs | [50] | |
| GA + Kinetin | Scenedesmus quadricauda | Chlorophyl, protein | [51] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, X.; Zeng, H.; Bartocci, P.; Fantozzi, F.; Yan, Y. Phytohormones and Effects on Growth and Metabolites of Microalgae: A Review. Fermentation 2018, 4, 25. https://doi.org/10.3390/fermentation4020025
Han X, Zeng H, Bartocci P, Fantozzi F, Yan Y. Phytohormones and Effects on Growth and Metabolites of Microalgae: A Review. Fermentation. 2018; 4(2):25. https://doi.org/10.3390/fermentation4020025
Chicago/Turabian StyleHan, Xingfeng, Huiru Zeng, Pietro Bartocci, Francesco Fantozzi, and Yunjun Yan. 2018. "Phytohormones and Effects on Growth and Metabolites of Microalgae: A Review" Fermentation 4, no. 2: 25. https://doi.org/10.3390/fermentation4020025
APA StyleHan, X., Zeng, H., Bartocci, P., Fantozzi, F., & Yan, Y. (2018). Phytohormones and Effects on Growth and Metabolites of Microalgae: A Review. Fermentation, 4(2), 25. https://doi.org/10.3390/fermentation4020025








