1. Introduction
Microbial fermentation and its metabolites have attracted increasing attention for their roles in health promotion and disease prevention. In particular, fermentation using grain substrates has been extensively studied because it produces various bioactive compounds such as proteins, peptides, lipids, and polyphenols, which exhibit antioxidant, anti-inflammatory, and even anticancer effects [
1,
2,
3].
Recently, among the microbial strains used for fermentation,
B. amyloliquefaciens has emerged as a useful candidate strain for improving the functional quality of fermented foods [
4,
5].
B. amyloliquefaciens-generated metabolites are known to have significant biological activities. They have also been reported to reduce oxidative stress and inflammation, which are major causes of several chronic diseases, by scavenging reactive oxygen species (ROS), such as superoxide anion and hydroxyl radicals [
6]. These fermented products have been proposed as supplementary therapies for the purpose of disease treatment or as functional food ingredients aimed at immune regulation and overall health promotion [
7,
8].
A novel
B. amyloliquefaciens strain, designated NPKE6, was isolated from traditional kimchi. Genomic analysis, including 16S rRNA gene sequencing and whole-genome comparison, confirmed that NPKE6 belongs to the same species as
B. amyloliquefaciens with genomic traits not observed in existing strains, which may contribute to distinct functional profiles (see
Supplementary Figure S2). This strain, we hypothesized that its fermentation metabolites could exhibit potent antioxidant and anti-inflammatory properties similar to those of the strain.
However, in order to use this as a supplement drug or for therapeutic and preventive purposes in vivo, it is important not only for the in vitro activity effect but also for the physiological relevance and actual function in vivo, especially for strain-specific products such as those derived from NPKE6. To better translate these in vitro activities in vivo, we investigated the biological functions of NPKE6-derived fermented metabolites using in vivo disease models. This study specifically targets two diseases commonly encountered in modern clinical practice: ulcerative colitis (UC) and acetaminophen-induced liver injury.
Ulcerative colitis is a chronic inflammatory bowel disease of unknown etiology that primarily affects the rectum and colon. UC is often associated with dysbiosis and is characterized by infiltration of immune cells such as neutrophils and macrophages, which generate high levels of reactive oxygen species (ROS), which exacerbate mucosal damage and chronic inflammation [
9,
10,
11]. Currently, drug therapy including 5-aminosalicylic acid, corticosteroids, and immunosuppressants is the standard, but it causes significant side effects, especially with long-term use, highlighting the need for safer adjuvant treatment strategies [
12,
13]. Also, long-term, high-dose acetaminophen-induced liver damage is primarily caused by oxidative stress resulting from hepatic glutathione (GSH) depletion and accumulation of its toxic metabolite, NAPQI. This activates Kupffer cells and secretes proinflammatory cytokines such as TNF-α, IL-6, and IL-1β, thereby amplifying liver damage [
14,
15,
16].
Given the central role of oxidative stress in the pathogenesis of both diseases, it is plausible that phenolic compounds, flavonoids, and organic acids—likely enriched in NPKE6-derived fermented metabolites—may contribute to disease mitigation by enhancing endogenous antioxidant defense mechanisms such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) [
17,
18], inhibiting lipid peroxidation [
19,
20], and downregulating pro-inflammatory signaling pathways including NF-κB and MAPK [
20,
21]. While the antioxidant and anti-inflammatory properties are the primary focus of this study, digestive enzymes present in the fermented products may also alleviate digestive burden, suppress the production of harmful metabolites, and restore gut microbial homeostasis. Collectively, these effects could reduce systemic inflammation and limit the translocation of endotoxins to the liver [
22,
23].
Therefore, this study aimed to evaluate the therapeutic efficacy of NPKE6-derived fermented metabolites in DSS-induced colitis and acetaminophen-induced liver injury models. Specifically, this study aimed to verify whether the observed in vitro bioactivities of NPKE6-derived metabolites are preserved in vivo after oral administration, and to assess their feasibility as functional food ingredients targeting inflammation-associated diseases.
2. Materials and Methods
2.1. Reagents and Solution Preparation
Dextran sulfate sodium (DSS; Cat#160110, MP Biomedicals, Irvine, CA, USA) was dissolved in 490 mL of distilled water (DW) to prepare a 1% (w/v) solution, filtered, and adjusted to a final volume of 1 L with additional DW. The solution was stored at 4 °C and refreshed every 2–3 days during the administration period. Acetaminophen (Cat#103-90-2, Sigma-Aldrich, Yongin, Republic of Korea) was dissolved in DW and administered intraperitoneally at 300 mg/kg three times per week (Monday, Wednesday, and Friday).
2.2. Preparation of Bacillus amyloliquefaciens NPKE6
The NPKE6 strain, isolated from Korean water kimchi, was cultured in BM medium containing D-glucose monohydrate (13.5 g/L), hydrolyzed soy protein (3 g/L), yeast extract (3 g/L), sodium carbonate (0.3 g/L), and MgSO4·7H2O (0.2 g/L). Cultures were incubated at 30 °C with shaking at 150 rpm for 24 h, followed by a 1% (v/v) subculture into fresh medium under identical conditions.
2.3. Production of NPKE6-FM
The puffed mixture of white soybean and white rice was obtained from Cheongwon Life (Cheongju, Republic of Korea). The puffed mixture was soaked in sterile DW at 60 °C for 1 min and drained. Grains were inoculated with NPKE6 at 5% (w/w) and fermented at 30 °C and >80% relative humidity for 24 h. The fermented mixture was then air-dried at 70 °C for 24 h, pulverized into powder, and stored at room temperature.
The NPKE6-FM used in this study was produced under Good Manufacturing Practice (GMP)–certified conditions by NPK Inc. (Damyang, Jeonnam, 57309, Republic of Korea). All experiments were conducted using the product with manufacturer part number 20100515088588.
2.4. Digestive Enzyme Activity Assays
For sample preparation, 1 g of the sample was extracted with 19 mL of solvent (distilled water for α-amylase analysis and 0.1 M sodium acetate buffer, pH 6.0, for protease analysis) by stirring for 1 h at room temperature using a magnetic stirrer (MS33, Misung Scientific Co., Ltd., Seoul, Republic of Korea). The extracts were centrifuged at 4000 rpm for 10 min using a refrigerated centrifuge (Supra R22, Hanil Scientific Inc., Gimpo, Republic of Korea), and the supernatants were filtered through Whatman No. 1 filter paper prior to analysis.
α-Amylase activity was determined using the dinitrosalicylic acid (DNS) method. The sample extract was reacted with the substrate solution at 37 °C in a temperature-controlled water bath (WB-07G, DAEYANG ETS, Incheon, Republic of Korea). After incubation, color development was achieved by adding DNS reagent and heating in a boiling water bath. Absorbance was measured at 540 nm using a UV–Vis spectrophotometer (SpectraMax iD3, Molecular Devices, San Jose, CA, USA). Enzyme activity was calculated based on a glucose calibration curve prepared using D-glucose (Sigma-Aldrich, St. Louis, MO, USA) and expressed as units per gram of sample (U/g), where one unit was defined as the amount of enzyme producing 1 μg of glucose per minute under the assay conditions.
Protease activity was measured using the casein–Folin method. The sample extract was reacted with casein substrate at 37 °C in a temperature-controlled water bath (WB-07G, DAEYANG ETS, Incheon, Republic of Korea). The reaction was terminated by adding trichloroacetic acid (TCA), followed by filtration. The filtrate was reacted with Folin–Ciocalteu reagent for color development, and absorbance was measured at 660 nm using the same UV–Vis spectrophotometer (SpectraMax iD3, Molecular Devices, San Jose, CA, USA). Enzyme activity was calculated using an L-tyrosine calibration curve prepared with L-tyrosine (Sigma-Aldrich, St. Louis, MO, USA). One unit of protease activity was defined as the amount of enzyme releasing 1 μg of L-tyrosine per minute under the assay conditions and expressed as U/g.
2.5. Antioxidant Enzyme Activity Assays
NPKE6-FM (1 g) was homogenized in 10 mL of DW, centrifuged at 10,000 rpm for 10 min, and the supernatant was used for all assays. SOD activity was measured using an EZ-SOD Kit (DoGen Bio, Seoul, Republic of Korea; Cat. No. DG-SOD400) by mixing 20 μL of the sample with WST solution and xanthine oxidase, incubating at 37 °C for 20 min, and reading absorbance at 450 nm. CAT activity was determined using the EZ-Catalase Kit (DoGen Bio, Seoul, Republic of Korea; Cat. No. DG-CAT400) by reacting 25 μL of sample with 40 μM H2O2, followed by Oxi-Probe/HRP, and reading at 560 nm. GPx activity (Abcam, Cambridge, UK; Cat. No. ab102530) was measured by mixing 50 μL of supernatant with colorimetric reagents and cumene hydroperoxide, with absorbance decline recorded at 340 nm. One unit of GPx activity was defined as the oxidation of 1 μmol NADPH per minute at 25 °C. All absorbance values were measured using a multi-mode microplate reader (SpectraMax iD3, Molecular Devices, San Jose, CA, USA).
2.6. Animals and Housing Conditions
Male C57BL/6 mice (6 weeks old; Koatech, Pyeongtaek, Republic of Korea) were housed in AAALAC- and MFDS-accredited facilities (K-MEDI Hub Preclinical Research Center, Daegu-Gyeongbuk Medical Innovation Foundation; IACUC Approval No. KMEDI-24082301-01, Date Approval. 2 September 2024) under controlled conditions (22 ± 1 °C, 50 ± 10% RH, 150–300 lux, 10–20 air changes/h). Mice were monitored daily, with cage and bedding replacements conducted weekly. During animal experiments, euthanasia was established in cases where necrosis, infection, ulceration interfered with food or water intake, rapid weight loss occurred, the animal was unable to walk or spent a significant amount of time in a lateral position, ulcers and infections, persistent self-trauma, severe dehydration, hypothermia, terminal illness, or morbidity occurred. Furthermore, euthanasia was performed using CO2 gas for exhalation or cervical dislocation. The carcasses were disposed of in a dedicated carcass disposal container in the corpse room of the Preclinical Research Animal Center by a designated carcass disposal company.
2.7. DSS-Induced Colitis Model
Chronic colitis was induced by providing 1% DSS (36–50 kDa) in drinking water. Following 1 week of acclimatization, mice were randomly assigned to four groups (n = 10): Normal control, Negative control (DSS + saline), and DSS + NPKE6-FM at 10 or 50 mg/kg. The entire experimental protocol, including model induction and treatment, was conducted over a total period of 28 days. DSS was administered in two exposure cycles it was first provided via free drinking for 5 days, followed by a recovery period, and then re-administered for 3 days via free drinking starting on day 19. Body weight and clinical signs were monitored regularly throughout the study, and tissues were collected at the study endpoint. During animal experiments, euthanasia was established in cases where necrosis, infection, ulceration interfered with food or water intake, rapid weight loss occurred, the animal was unable to walk or spent a significant amount of time in a lateral position, ulcers and infections, persistent self-trauma, severe dehydration, hypothermia, terminal illness, or morbidity occurred. Furthermore, euthanasia was performed using CO2 gas for exhalation or cervical dislocation. The carcasses were disposed of in a dedicated carcass disposal container in the corpse room of the Preclinical Research Animal Center by a designated carcass disposal company.
2.8. Acetaminophen-Induced Liver Injury Model
Liver injury was induced by intraperitoneal injection of acetaminophen (300 mg/kg in 0.1 mL), three times per week for 51 days. Mice were allocated into four groups (n = 10): Normal control, Negative control (APAP + saline), and APAP + NPKE6-FM at 10 or 50 mg/kg. Oral treatments (0.2 mL) were administered daily. Liver function was assessed via weekly blood collection and biochemical analyses.
2.9. Necropsy and Sample Collection
At the experimental endpoint, body weight was measured for each animal. Mice were anesthetized by inhalation of isoflurane (1–2.5%), and blood was collected from the abdominal vena cava, followed by exsanguination. Collected blood samples were centrifuged to obtain serum, which was subsequently used for biochemical analyses. The colon and major organs were excised and weighed. Colon tissues were processed for histopathological examination.
2.10. Biochemical Analyses
Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP) levels were measured using an automated biochemical analyzer (TBA-120FR, Toshiba, Tokyo, Japan). Levels of malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) were determined using commercially available ELISA kits according to the manufacturers’ protocols. Absorbance was measured using a microplate reader, and concentrations were calculated from standard curves.
2.11. Tissue and Cytokine Analyses
Body weights were recorded three times per week, and clinical signs were monitored daily. Blood samples were collected at baseline, week 2, and week 4 for biochemical and antioxidant measurements. Colon tissues were harvested at necropsy for histological evaluation (hematoxylin and eosin staining), cytokine quantification, and gene expression analysis. Tissue sections were examined using a Pannoramic 250 FlashIII digital scanner (3D Histech, Budaepst, Hungary). Whole –slide images were acquired and analyzed using Pannoramic Digital Scanner Software (version 3.0.7, Histech).
Levels of tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) in colon tissue homogenates were quantified using Mouse TNF-α Uncoated ELISA Kit (Invitrogen, Carlsbad, CA, USA, 88-7324-88) and Mouse IL-6 Uncoated ELISA Kit (Invitrogen, 88-7064-88), according to the manufacturers’ instructions.
Quantitative real-time PCR was performed using a Roche Light Cycler 96 system (Roche, Basel, Switzerland) under the following cycling conditions an initial denaturation step at 95 °C for 30 s, followed by 40 cycles of denaturation at 95 °C for 5 s and annealing/extension at 60 °C for 30 s.
Total RNA was extracted from colon tissues using TRIzol reagent (Invitrogen, Catalog Nos. 15596026 and 15596018). Complementary DNA (cDNA) was synthesized from 1 μg of total RNA using an M-MLV cDNA Synthesis Kit (Invitrogen, Burlington, MA, USA) following the manufacturer’s protocol. Quantitative real-time PCR was performed using an Applied Biosystems StepOne Real-Time PCR System (Desktop, Invitrogen, Burlington, MA, USA, Catalog No. 4376374).
Gene expression levels of inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), AKT1, and monocyte chemoattractant protein-1 (MCP-1) were analyzed using the following primer pairs (
Table 1).
2.12. Statistical Analysis
We employed GraphPad Prism version 9 for the statistical analysis and used the unpaired Student’s test to analyze statistical significance. The data from each group are expressed as the mean ± standard error of the mean, and statistical significance was set at * p < 0.05, ** p < 0.01, *** p < 0.001, and **** p < 0.0001.
3. Results
3.1. In Vitro Antioxidant and Digestive Enzyme Activities of NPKE6-FM
Fermentation of puffed grains with Bacillus amyloliquefaciens NPKE6 resulted in the production of a bioactive fermented extract with enhanced antioxidant and digestive enzyme activities, hereafter referred to as NPKE6-FM. Enzymatic activities of the fermented product were quantitatively compared with those of the unfermented raw material without NPKE6 inoculation.
At a concentration of 100 mg/mL, NPKE6-FM exhibited markedly increased antioxidant enzyme activities compared with the unfermented raw material (
Figure 1). In particular, superoxide dismutase (SOD) activity was observed to be 123,716 mU/mL after fermentation. SOD activity was defined as one unit corresponding to the concentration of SOD required to inhibit 50% of the WST-1 reduction reaction (IC
50). In contrast, the unfermented raw material did not reach 50% inhibition under the experimental conditions; therefore, SOD activity could not be calculated in units for the unfermented sample. Catalase (CAT) activity was substantially elevated to 3854 mU/mL, representing an approximately 124-fold increase compared with the unfermented sample. In addition, glutathione peroxidase (GPx) activity increased to 39.85 mU/mL, corresponding to an approximately 9.0-fold increase following fermentation.
Digestive enzyme activities were also markedly enhanced by fermentation. α-Amylase activity increased to 177,885 U/g, representing an approximately 304-fold increase relative to the unfermented raw material. Similarly, protease activity increased to 5661 U/g, corresponding to an approximately 83-fold increase after fermentation.
Collectively, these results demonstrate that fermentation with B. amyloliquefaciens NPKE6 significantly amplifies both antioxidant and digestive enzyme activities. The pronounced increases in SOD, CAT, and GPx activities indicate enhanced antioxidative potential, while the dramatic elevation of α-amylase and protease activities supports a substantial improvement in digestive functionality. These findings highlight the functional value of NPKE6 fermentation for the production of enzyme-enriched bioactive materials.
3.2. In Vivo Evaluation in a DSS-Induced Ulcerative Colitis Mouse Model
To evaluate whether the antioxidant and anti-inflammatory effects observed in vitro could be replicated in vivo, a dextran sulfate sodium (DSS)-induced ulcerative colitis (UC) mouse model was employed. UC was induced through two cycles of 1% DSS administration followed by a recovery phase. All DSS-treated groups showed significant reductions in body weight, ranging from approximately 30% to 35% (
Supplementary Figure S1).
In the DSS- Negative control group, serum biochemical analyses revealed substantial increases in hepatic injury markers (ALT, AST, ALP), the oxidative stress marker (MDA), pro-inflammatory cytokines (IL-6, TNF-α), and inflammatory mediators (iNOS, COX-2), alongside a marked decrease in the levels of key antioxidant enzymes (SOD, CAT, GPx).
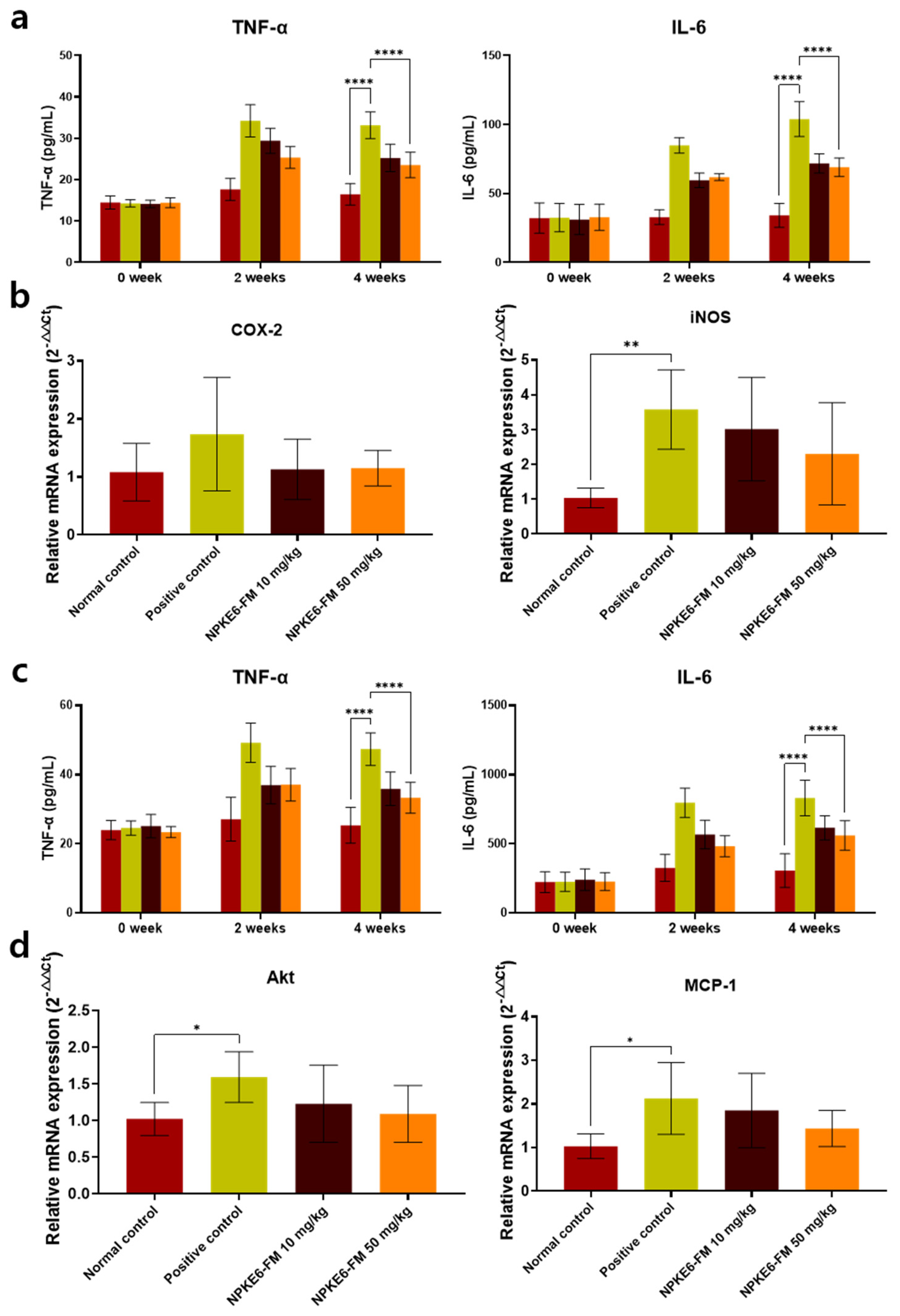
NPKE6-FM was administered orally at doses of 10 mg/kg and 50 mg/kg daily for four weeks. A dose-dependent improvement was observed in both serum biochemical parameters and antioxidant enzyme activities (
Figure 2a and
Figure 3a). At the higher dose of 50 mg/kg, the levels of ALT, AST, ALP, IL-6, TNF-α, iNOS, COX-2, and MDA were reduced to approximately 70.0%, 75.5%, 89.0%, 66.4%, 71.0%, 64.4%, 66.1%, and 70.2%, respectively, compared to the DSS-only group (
Table 2).
Meanwhile, antioxidant enzyme activities that were suppressed by DSS treatment—down to 69.2% for SOD, 71.4% for CAT, and 63.0% for GPx—recovered to 90.4%, 80.0%, and 86.4%, respectively, after four weeks of high-dose NPKE6-FM treatment.
Ex vivo analysis of colon tissues revealed that DSS treatment reduced colon length and weight by approximately 20% and 60%, respectively, compared to the normal control group. These parameters were significantly restored in the NPKE6-FM-treated groups (
Figure 4a,b). Histological analysis using hematoxylin and eosin (H&E) staining further confirmed the protective effects of NPKE6-FM, with lesion scores significantly decreased from 4.22 ± 1.30 in the DSS group to 2.88 ± 1.89 and 2.56 ± 1.51 in the NPKE6-FM 10 mg/kg and 50 mg/kg groups, respectively (
Figure 4c and
Figure 4d and
Figure 4e,
Table 2).
3.3. In Vivo Evaluation in an Acetaminophen-Induced Liver Injury Model
To further assess the hepatoprotective effects of NPKE6-FM, a liver injury model was established via intraperitoneal administration of acetaminophen (300 mg/kg, three times per week). Body weights remained stable across all groups, including the acetaminophen-treated control group, indicating that the treatments did not cause systemic toxicity (
Supplementary Figure S1).
Acetaminophen exposure significantly elevated serum levels of liver damage markers—ALT, AST, and ALP—as well as hepatic levels of IL-6, TNF-α, Akt, MCP-1, and MDA, with increases ranging from 1.35- to 9.2-fold compared to the negative control group. Concurrently, antioxidant enzyme levels were notably suppressed: SOD, CAT, and GPx activities were reduced by factors of 1.3, 3.39, and 1.38, respectively.
Oral administration of NPKE6-FM led to a dose-dependent improvement in both inflammatory and oxidative stress markers. At the higher dose of 50 mg/kg, levels of ALT, AST, ALP, IL-6, TNF-α, Akt, MCP-1, and MDA were decreased to 55.4%, 64.2%, 78.2%, 67.5%, 70.0%, 68.6%, 67.6%, and 66.4%, respectively, relative to the untreated acetaminophen group (
Figure 2b,
Figure 3b and
Figure 5b). Antioxidant enzyme activities were also significantly restored: SOD increased from 77.3% to 89.2%, CAT from 29.5% to 65.4%, and GPx from 72.6% to 90.0%.
Histological analysis of liver tissue showed no marked structural abnormalities between groups. This may be attributed to partial tissue recovery by the time of sample collection. Representative hematoxylin and eosin (H&E) staining images are shown in
Figure 4e.
4. Discussion
The health-promoting potential of
Bacillus amyloliquefaciens has been widely studied, particularly due to its ability to generate bioactive fermentation metabolites and enzymatic byproducts [
24,
25,
26,
27]. Kimchi, a traditional Korean fermented food enriched with functional microorganisms, is a natural source of
B. amyloliquefaciens strains. Among these, the NPKE6 strain—isolated from Korean water kimchi—was selected in this study to ferment puffed grains.
To determine whether NPKE6 represents merely another isolate or a genomically distinctive strain, whole-genome sequencing (WGS) was performed. ANI analysis demonstrated 99.58% identity with B. amyloliquefaciens, confirming its taxonomic classification at the species level. However, the complete genome (4.28 Mb, 45.1% G + C content) comprising 4412 predicted coding sequences revealed strain-level genomic characteristics. Notably, multiple oxidative stress–response genes, including superoxide dismutase (SOD1, SOD2), catalase (katE/katA family), and glutathione peroxidase (gpx), were identified. These genes may enhance cellular stability under fermentation and processing conditions and provide a genomic basis for the elevated antioxidant and enzymatic activities observed in NPKE6-derived preparations. Furthermore, genome-wide safety assessment confirmed the absence of major antibiotic resistance and toxin-related genes, supporting its suitability for food and industrial applications. Therefore, NPKE6 should be regarded as a genomically characterized and functionally distinctive strain rather than merely an uncharacterized variant.
NPKE6 was selected for its favorable safety profile and demonstrated ability to produce a diverse range of bioactive compounds. Preliminary in-house screening showed significant anti-inflammatory potential, consistent with reported antioxidant, immunomodulatory, and inflammation-modulating effects of B. amyloliquefaciens, including regulation of NF-Κβ and MAPK pathways. The strain exhibited stable growth and reliable fermentation performance, enabling reproducible production of enzyme-rich preparations. NPKE6-derived materials significantly reduced inflammatory responses in cell-based assays, supporting further evaluation in an in vivo gastritis model. Unlike strains studied for exopolysaccharides or postbiotic fractions, NPKE6’s enzyme-focused preparation suggests distinct bioactive mechanisms. These results support NPKE6 as a biologically relevant and technically robust candidate for functional food development. We subsequently evaluated the biological activities of the resulting fermentation product, NPKE6-FM, through both in vitro and in vivo analyses.
Previous studies have focused on the health benefits of individual bioactive compounds produced during grain fermentation. Notably, microbial fermentation is known to facilitate the bioconversion of isoflavone glycosides into more bioactive aglycones via β-glucosidase activity, enhancing antioxidant potential. These processes often coincide with elevated levels of flavonoids, phenolic compounds, and peptides [
28,
29]. In this context, the observed increase in antioxidant and digestive enzyme activities in NPKE6-FM is consistent with such biochemical transformations.
However, the precise identity and composition of the active metabolites responsible for these effects remain unclear. While quantitative and qualitative analyses of individual compounds are important, the inherent complexity of fermented matrices complicates the direct attribution of bioactivity to a single molecule. It is more plausible that a synergistic interplay of multiple components—such as lipopeptides, enzymes, flavonoids, and other microbial metabolites—underlies the observed functional properties of NPKE6-FM [
30,
31].
Among the enzyme-related components generated during NPKE6-mediated fermentation, superoxide dismutase (SOD) is a key antioxidant metalloenzyme that catalyzes the dismutation of superoxide radicals into hydrogen peroxide and molecular oxygen. As an enzymatic protein, SOD is best characterized by functional parameters such as enzymatic activity and redox-regulatory capacity rather than by mass-based compositional ratios. Functioning as the first line of defense in a sequential antioxidant enzyme system, SOD acts in concert with catalase and glutathione peroxidase to regulate cellular redox balance. Accordingly, the antioxidant properties of NPKE6-derived fermented metabolites were evaluated based on the integrated activities of these enzymes, providing a mechanistic basis for the modulation of redox-sensitive inflammatory signaling pathways discussed below.
Mechanistically, the observed suppression of iNOS, COX-2, Akt, and MCP-1 suggests the involvement of upstream inflammatory pathways. Canonical NF-Κβ signaling is a central transcriptional regulator of iNOS and COX-2 and controls the expression of several pro-inflammatory cytokines and chemokines, including MCP-1. Therefore, the inhibition of NF-kB activity represents a plausible mechanism underlying the downregulation of these mediators. In parallel, the Nrf2 pathway orchestrates antioxidant and cytoprotective responses and can attenuate NF-Κβ activity through HO-1 and other downstream effectors. Activation of Nrf2 has been shown to reduce oxidative stress and suppress iNOS and COX-2 expression, raising the possibility that NPKE6 exerts anti-inflammatory effects through coordinated modulation of both NF-Κβ and Nrf2 pathways. Although these pathways were not directly measured in this study, the combined reduction in inflammatory mediators strongly supports their potential involvement and warrants targeted mechanistic investigation.
The growing interest in fermented products as functional food ingredients stems from their potential to mitigate chronic conditions linked to oxidative stress and inflammation. In our in vitro assays, NPKE6-FM significantly improved radical-scavenging activities (e.g., DPPH, ORAC), reducing power, and the activities of digestive enzymes such as α-amylase and glucosidase [
32]. However, in vitro results alone are insufficient for confirming physiological efficacy. To validate its potential as a dietary adjunct for disease prevention or management, in vivo assessment is essential, given the complex interplay of bioavailability, metabolic fate, and tissue-specific delivery.
Accordingly, we evaluated NPKE6-FM in two well-established animal models: DSS-induced ulcerative colitis and acetaminophen-induced liver injury. Although these models differ in their etiology, they share oxidative stress and inflammation as central pathogenic mechanisms. Based on dose ranges reported in previous studies and investigations using similar
Bacillus-derived probiotics, an exploratory dose range of 5–100 mg/kg is commonly applied in preclinical evaluations. Given the favorable safety profile of this strain, we selected doses that avoid unnecessarily high exposure while still enabling the detection of meaningful efficacy-related changes. A dose of 10 mg/kg was designated as a low dose to assess the minimal effective level, whereas 50 mg/kg was chosen as a representative mid-range dose to evaluate potential dose dependency and to explore the upper range of efficacy. A five-fold separation between the two doses was implemented to facilitate clear biological and statistical discrimination. Furthermore, existing data indicate that
Bacillus amyloliquefaciens strains exhibit low toxicity, with doses up to 50 mg/kg showing no adverse effects on body weight, food intake, or organ morphology. These safety findings supported the selection of the dose range used in the present study. Consistent with this, NPKE6-FM administration partially restored antioxidant enzyme activity and reduced pro-inflammatory cytokine levels in both models. In the colitis model, therapeutic effects were evident both physiologically and histologically, supporting its potential as a therapeutic candidate [
33,
34].
In contrast, in the liver injury model, NPKE6-FM significantly improved biochemical markers such as ALT, AST, ALP and MDA; however, histological changes were less pronounced. This discrepancy may reflect the early-stage nature of acetaminophen-induced liver injury—dominated by subcellular dysfunctions such as mitochondrial stress and ROS accumulation—that are not easily detectable by conventional H&E staining. Additionally, the liver’s strong regenerative capacity may have masked more distinct morphological changes [
35].
The present study also has limitations. The relatively short treatment duration allowed evaluation of only acute effects, limiting conclusions about long-term efficacy or chronic inflammation. Also, comprehensive compositional profiling of NPKE6-FM was not performed, and identifying specific active components will be essential for understanding the mechanism and ensuring batch consistency. Additionally, long-term toxicity and safety assessments were not included and will be required before human application. These limitations underscore the need for additional mechanistic, compositional, and safety investigations.
Taken together, these findings support the functional potential of NPKE6-FM as a bioactive agent with antioxidant and anti-inflammatory properties. NPKE6-FM consistently demonstrated therapeutic and preventive effects across both models. These results highlight its promise as a functional food ingredient or an adjunctive therapeutic option for managing inflammation-related diseases.