Photofermentative Hydrogen Production from Real Dark Fermentation Effluents: A Sequential Valorization of Orange Peel Waste
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Culture Media
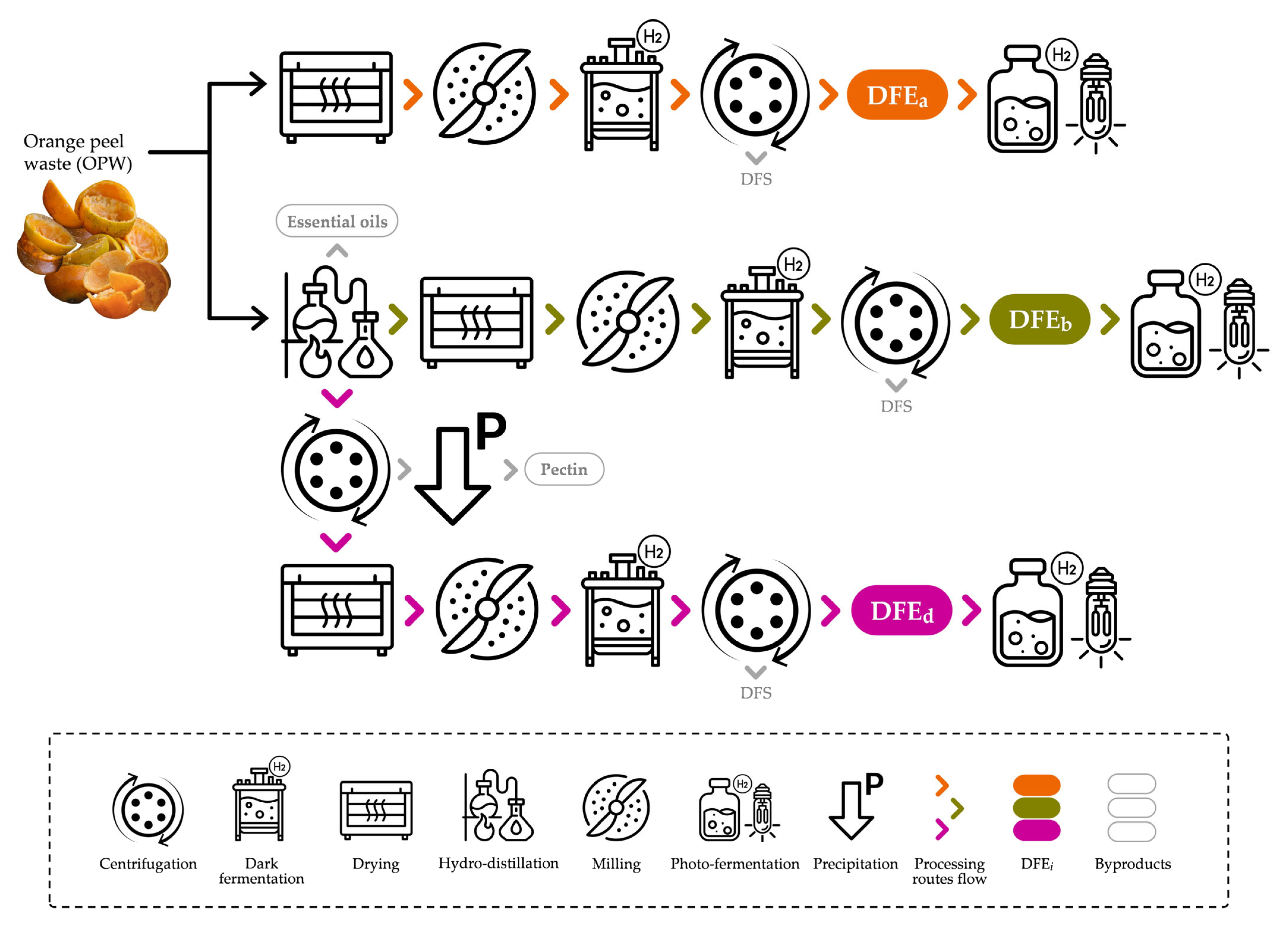
2.2. Substrate Preparation: Production of Dark Fermentation Effluents
2.3. Photofermentative Hydrogen Production Evaluation from Dark Fermentation Effluents
2.4. Analytical Methods
2.5. Kinetic Models Fitting
2.6. Statistical Analysis
3. Results and Discussion
Photofermentative Hydrogen Production
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CDW | Cell Dry Weight |
| DFE | Dark fermentation effluents |
| DFEa | Dark fermentation effluents from OPW without byproduct extraction |
| DFEb | Dark fermentation effluents from OPW after essential oils extraction |
| DFEd | Dark fermentation effluents from OPW after essential oils and pectin extraction |
| DFS | Dark fermentation solids |
| dx | Slope fit parameter |
| GHG | Greenhouse gas |
| H0 | Initial hydrogen production |
| Hmax | Maximum cumulative hydrogen production |
| hmax | Maximum cumulative specific hydrogen production |
| OPW | Orange peel waste |
| PNSB | Purple non-sulfur bacteria |
| Rmax | Maximum hydrogen production rate |
| rmax | Maximum specific hydrogen production rate |
| RMSEP | Root mean square error of prediction |
| Ti | Time at the inflection point |
| t50 | Time to reach half of Hmax and hmax |
| VFA | Volatile fatty acids |
| λ | Lag phase |
References
- Meher Kotay, S.; Das, D. Biohydrogen as a Renewable Energy Resource—Prospects and Potentials. Int. J. Hydrogen Energy 2008, 33, 258–263. [Google Scholar] [CrossRef]
- Marzouk, O.A. 2030 Ambitions for Hydrogen, Clean Hydrogen, and Green Hydrogen. Eng. Proc. 2023, 56, 14. [Google Scholar]
- Koleva, M.; Guerra, O.J.; Eichman, J.; Hodge, B.-M.; Kurtz, J. Optimal Design of Solar-Driven Electrolytic Hydrogen Production Systems within Electricity Markets. J. Power Sources 2021, 483, 229183. [Google Scholar] [CrossRef]
- SCLCI Swiss Centre for Life Cycle Inventories. Ecoinvent Data, v3.3; The Ecoinvent Association: Zurich, Switzerland, 2010. [Google Scholar]
- Madigan, M.T.; Jung, D.O. Overview of Purple Bacteria: Systematics, Physiology, and Habitats. In The Purple Phototrophic Bacteria; Hunter, C.N., Daldal, F., Thurnauer, M.C., Beatty, J.T., Eds.; Springer: Dordrecht, The Netherlands, 2009; Volume 28, pp. 1–15. ISBN 978-1-4020-8814-8. [Google Scholar]
- Harwood, C.S. Degradation of Aromatic Compounds by Purple Nonsulfur Bacteria. In The Purple Phototrophic Bacteria. Advances in Photosynthesis and Respiration; Hunter, C.N., Daldal, F., Thurnauer, M.C., Beatty, J.T., Eds.; Springer: Dordrecht, The Netherlands, 2009; Volume 28, pp. 577–594. ISBN 978-1-4020-8814-8. [Google Scholar]
- Hallenbeck, P.C. Microbial Paths to Renewable Hydrogen Production. Biofuels 2011, 2, 285–302. [Google Scholar] [CrossRef]
- Keskin, T.; Abo-Hashesh, M.; Hallenbeck, P.C. Photofermentative Hydrogen Production from Wastes. Bioresour. Technol. 2011, 102, 8557–8568. [Google Scholar] [CrossRef]
- Rai, P.K.; Singh, S.P. Integrated Dark- and Photo-Fermentation: Recent Advances and Provisions for Improvement. Int. J. Hydrogen Energy 2016, 41, 19957–19971. [Google Scholar] [CrossRef]
- Velenturf, A.P.M.; Purnell, P. Principles for a Sustainable Circular Economy. Sustain. Prod. Consum. 2021, 27, 1437–1457. [Google Scholar] [CrossRef]
- Rodríguez-Valderrama, S.; Escamilla-Alvarado, C.; Magnin, J.-P.; Rivas-García, P.; Amézquita-García, H.J.; Cano-Gómez, J.J. Photo-Fermentative Hydrogen Production from Organic Acids Mixtures Evaluated through Predictive Models for Rhodobacter Capsulatus Strains. Rev. Int. Contam. Ambient. 2022, 38, 93–110. [Google Scholar] [CrossRef]
- López-Hernández, B.N.; Escamilla-Alvarado, C.; Guadalupe Paredes, M.; Albalate-Ramírez, A.; Rodríguez-Valderrama, S.; Magnin, J.-P.; Rivas-García, P. Environmental Impact Assessment of Biohydrogen Production from Orange Peel Waste by Lab-Scale Dark and Photofermentation Processes. Rev. Int. Contam. Ambient. 2024, 40, 27–40. [Google Scholar] [CrossRef]
- He, D.; Bultel, Y.; Magnin, J.-P.; Willison, J.C. Kinetic Analysis of Photosynthetic Growth and Photohydrogen Production of Two Strains of Rhodobacter Capsulatus. Enzym. Microb. Technol. 2006, 38, 253–259. [Google Scholar] [CrossRef]
- Zhang, Q.; Jiao, Y.; He, C.; Ruan, R.; Hu, J.; Ren, J.; Toniolo, S.; Jiang, D.; Lu, C.; Li, Y.; et al. Biological Fermentation Pilot-Scale Systems and Evaluation for Commercial Viability towards Sustainable Biohydrogen Production. Nat. Commun. 2024, 15, 4539. [Google Scholar] [CrossRef]
- Sagnak, R.; Kargi, F. Photo-Fermentative Hydrogen Gas Production from Dark Fermentation Effluent of Acid Hydrolyzed Wheat Starch with Periodic Feeding. Int. J. Hydrogen Energy 2011, 36, 4348–4353. [Google Scholar] [CrossRef]
- Hitit, Z.Y.; Zampol Lazaro, C.; Hallenbeck, P.C. Increased Hydrogen Yield and COD Removal from Starch/Glucose Based Medium by Sequential Dark and Photo-Fermentation Using Clostridium Butyricum and Rhodopseudomonas Palustris. Int. J. Hydrogen Energy 2017, 42, 18832–18843. [Google Scholar] [CrossRef]
- Yokoi, H.; Maki, R.; Hirose, J.; Hayashi, S. Microbial Production of Hydrogen from Starch-Manufacturing Wastes. Biomass Bioenergy 2002, 22, 389–395. [Google Scholar] [CrossRef]
- Cabecas Segura, P.; De Meur, Q.; Alloul, A.; Tanghe, A.; Onderwater, R.; Vlaeminck, S.E.; Wouwer, A.V.; Wattiez, R.; Dewasme, L.; Leroy, B. Preferential Photoassimilation of Volatile Fatty Acids by Purple Non-Sulfur Bacteria: Experimental Kinetics and Dynamic Modelling. Biochem. Eng. J. 2022, 186, 108547. [Google Scholar] [CrossRef]
- Albuquerque, M.M.; Sartor, G.d.B.; Martinez-Burgos, W.J.; Scapini, T.; Edwiges, T.; Soccol, C.R.; Medeiros, A.B.P. Biohydrogen Produced via Dark Fermentation: A Review. Methane 2024, 3, 500–532. [Google Scholar] [CrossRef]
- López-Hernández, B.N.; Escamilla-Alvarado, C.; Albalate-Ramírez, A.; Rodríguez-Valderrama, S.; Rivas-García, P.; Paredes, M.G. Transforming Orange Peel Waste into Hydrogen: The Effect of Biocompound Extraction and Inoculum-to-Substrate Ratio on Dark Fermentation. Int. J. Hydrogen Energy 2025, 141, 1261–1270. [Google Scholar] [CrossRef]
- Martín, M.A.; Siles, J.A.; Chica, A.F.; Martín, A. Biomethanization of Orange Peel Waste. Bioresour. Technol. 2010, 101, 8993–8999. [Google Scholar] [CrossRef] [PubMed]
- Santiago, B.; Moreira, M.T.; Feijoo, G.; González-García, S. Identification of Environmental Aspects of Citrus Waste Valorization into D-Limonene from a Biorefinery Approach. Biomass Bioenergy 2020, 143, 105844. [Google Scholar] [CrossRef]
- Albalate-Ramírez, A.; Rueda-Avellaneda, J.F.; López-Hernández, B.N.; Alcalá-Rodríguez, M.M.; García-Balandrán, E.E.; Evrard, D.; Rivas-García, P. Geographic Life Cycle Assessment of Food Loss and Waste Management in Mexico: The Reality of Distribution and Retail Centers. Sustain. Prod. Consum. 2024, 48, 289–300. [Google Scholar] [CrossRef]
- Rodríguez-Valderrama, S.; Escamilla-Alvarado, C.; Magnin, J.-P.; Rivas-García, P.; Valdez-Vazquez, I.; Ríos-Leal, E. Batch Biohydrogen Production from Dilute Acid Hydrolyzates of Fruits-and-Vegetables Wastes and Corn Stover as Co-Substrates. Biomass Bioenergy 2020, 140, 105666. [Google Scholar] [CrossRef]
- Zinatizadeh, A.A.; Mirghorayshi, M.; Birgani, P.M.; Mohammadi, P.; Ibrahim, S. Influence of Thermal and Chemical Pretreatment on Structural Stability of Granular Sludge for High-Rate Hydrogen Production in an UASB Bioreactor. Int. J. Hydrogen Energy 2017, 42, 20512–20519. [Google Scholar] [CrossRef]
- Valdez-Vazquez, I.; Poggi-Varaldo, H.M. Hydrogen Production by Fermentative Consortia. Renew. Sustain. Energy Rev. 2009, 13, 1000–1013. [Google Scholar] [CrossRef]
- Carrillo-Verástegui, K.A.; Escamilla-Alvarado, C.; Escárcega-González, C.E.; Cano-Gómez, J.J.; Paniagua-Vega, D.; Nava-Bravo, I.; Ríos-Leal, E. Biohydrogen Potential Assessment of Opuntia Spp.: Effect of Inoculum-to-Substrate Ratio and Residual Biomass Evaluation. Int. J. Hydrogen Energy 2022, 47, 30085–30096. [Google Scholar] [CrossRef]
- Secretaría de Comercio y Fomento Industrial Norma Mexicana NMX-AA-25-1984. In Environmental Protection—Soil Contamination—Solid Residues—pH Determination—Potentiometric Method; Mexico City, Mexico, 1984; pp. 1–5.
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; Van’t Riet, K. Modeling of the Bacterial Growth Curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [CrossRef]
- Tjørve, K.M.C.; Tjørve, E. The Use of Gompertz Models in Growth Analyses, and New Gompertz-Model Approach: An Addition to the Unified-Richards Family. PLoS ONE 2017, 12, e0178691. [Google Scholar] [CrossRef]
- Magnin, J.-P.; Deseure, J. Hydrogen Generation in a Pressurized Photobioreactor: Unexpected Enhancement of Biohydrogen Production by the Phototrophic Bacterium Rhodobacter Capsulatus. Appl. Energy 2019, 239, 635–643. [Google Scholar] [CrossRef]
- Argun, H.; Kargi, F.; Kapdan, I.K. Light Fermentation of Dark Fermentation Effluent for Bio-Hydrogen Production by Different Rhodobacter Species at Different Initial Volatile Fatty Acid (VFA) Concentrations. Int. J. Hydrogen Energy 2008, 33, 7405–7412. [Google Scholar] [CrossRef]
- Cahyari, K.; Syamsiah, S.; Hidayat, M.; Sarto, S. Inhibitory Kinetics Study of Limonene and Eugenol towards Mixed Culture of Dark Fermentative Biohydrogen Production. In Proceedings of the 2nd International Conference on Chemistry, Chemical Process and Engineering (IC3PE), Yogyakarta, Indonesia, 14 August 2018; Volume 2026, p. 020038. [Google Scholar]
- Camargo, F.P.; Sarti, A.; Alécio, A.C.; Sabatini, C.A.; Adorno, M.A.T.; Duarte, I.C.S.; Varesche, M.B.A. Limonene Quantification by Gas Chromatography with Mass Spectrometry (GC-MS) and Its Effects on Hydrogen and Volatile Fatty Acids Production in Anaerobic Reactors. Quim. Nova 2020, 43, 844–850. [Google Scholar] [CrossRef]
- de Groot, A. Limonene Hydroperoxides. Dermatitis 2019, 30, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Kern, S.; Granier, T.; Dkhil, H.; Haupt, T.; Ellis, G.; Natsch, A. Stability of Limonene and Monitoring of a Hydroperoxide in Fragranced Products. Flavour Fragr. J. 2014, 29, 277–286. [Google Scholar] [CrossRef]
- Chubukov, V.; Mingardon, F.; Schackwitz, W.; Baidoo, E.E.K.; Alonso-Gutierrez, J.; Hu, Q.; Lee, T.S.; Keasling, J.D.; Mukhopadhyay, A. Acute Limonene Toxicity in Escherichia Coli Is Caused by Limonene Hydroperoxide and Alleviated by a Point Mutation in Alkyl Hydroperoxidase AhpC. Appl. Environ. Microbiol. 2015, 81, 4690–4696. [Google Scholar] [CrossRef]
- Dari, D.N.; Freitas, I.S.; Aires, F.I.d.S.; Melo, R.L.F.; dos Santos, K.M.; da Silva Sousa, P.; Gonçalves de Sousa Junior, P.; Luthierre Gama Cavalcante, A.; Neto, F.S.; da Silva, J.L.; et al. An Updated Review of Recent Applications and Perspectives of Hydrogen Production from Biomass by Fermentation: A Comprehensive Analysis. Biomass 2024, 4, 132–163. [Google Scholar] [CrossRef]
- Smith, S.C.; Toledo-Alarcón, J.; Schiappacasse, M.C.; Tapia-Venegas, E. Enrichment of a Mixed Culture of Purple Non-Sulfur Bacteria for Hydrogen Production from Organic Acids. Sustainability 2023, 15, 16607. [Google Scholar] [CrossRef]
- Bayon-Vicente, G.; Toubeau, L.; Gilson, M.; Gégo, G.; Landgey, N.; Krings, S.; Leroy, B. Metabolic Pathways to Sustainability: Review of Purple Non-Sulfur Bacteria Potential in Agri-Food Waste Valorization. Front. Bioeng. Biotechnol. 2025, 13, 1529032. [Google Scholar] [CrossRef] [PubMed]
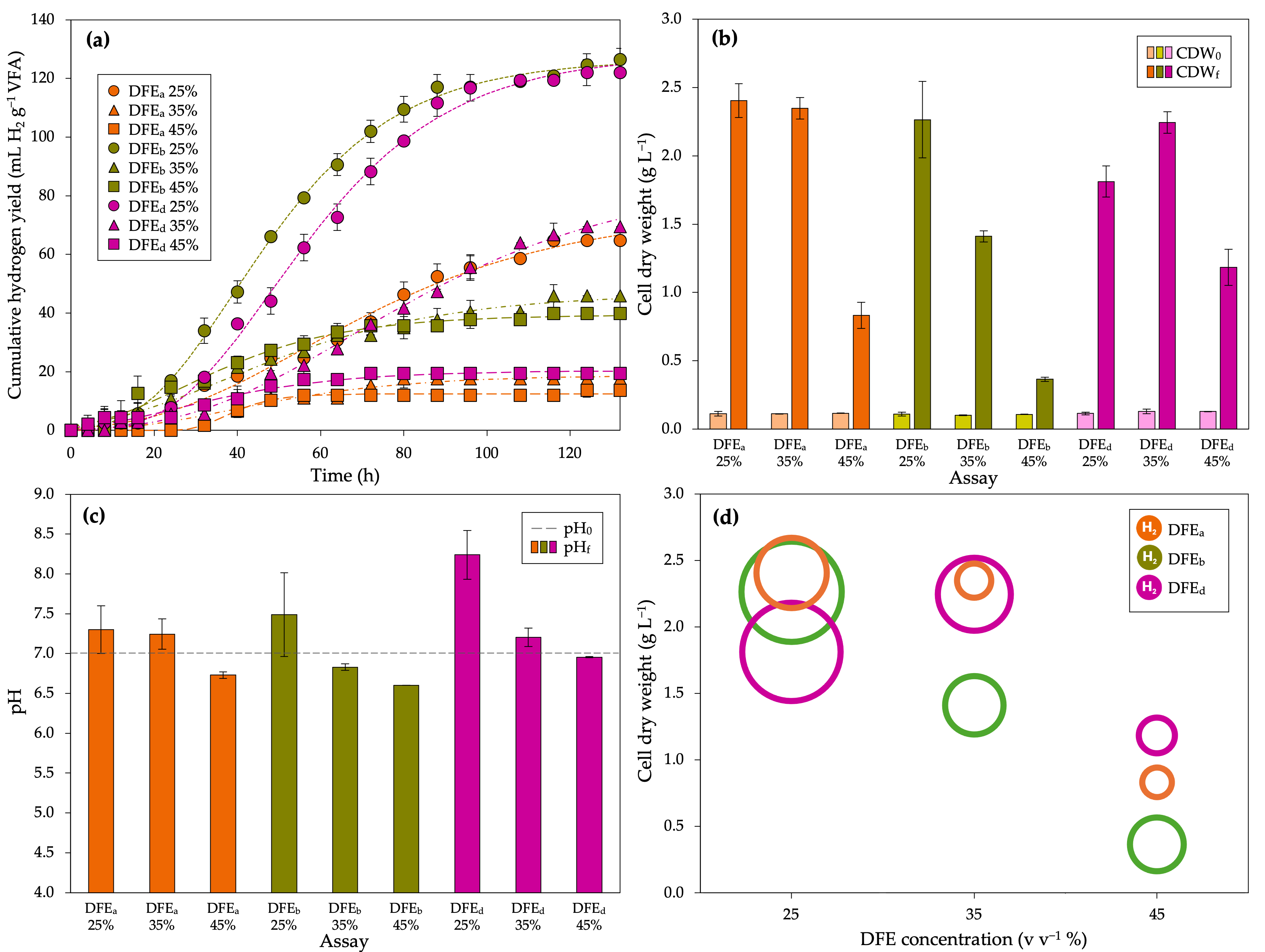
| Parameter | DFEa | DFEb | DFEd | ||||||
|---|---|---|---|---|---|---|---|---|---|
| DFE concentration (v v−1 %) | 25 | 35 | 45 | 25 | 35 | 45 | 25 | 35 | 45 |
| Production Hmax,e (mL H2) | 21.00 ± 4.24 | 8.00 ± 2.83 | 8.00 ± 2.83 | 33.50 ± 6.61 | 17.00 ± 4.24 | 19.00 ± 4.24 | 31.33 ± 5.03 | 25.00 ± 1.41 | 9.00 ± 4.24 |
| Modified Gompertz model | |||||||||
| Hmax (mL H2) | 24.22 | 8.53 | 7.25 | 33.51 | 17.35 | 18.79 | 32.99 | 31.12 | 9.42 |
| Rmax (mL H2/h) | 0.25 | 0.13 | 0.40 | 0.57 | 0.21 | 0.32 | 0.51 | 0.30 | 0.16 |
| λ (h) | 20.14 | 14.25 | 29.81 | 17.24 | 6.99 | 5.08 | 24.23 | 28.59 | 3.60 |
| RMSEP | 0.77 | 0.48 | 0.28 | 0.39 | 0.57 | 0.83 | 0.63 | 0.51 | 0.51 |
| Ti-Gompertz model | |||||||||
| Hmax (mL H2) | 24.22 | 8.53 | 7.25 | 33.51 | 17.35 | 18.79 | 32.99 | 31.12 | 9.42 |
| Rmax (mL H2/h) | 0.25 | 0.13 | 0.40 | 0.57 | 0.21 | 0.32 | 0.51 | 0.30 | 0.16 |
| Ti (h) | 55.46 | 38.38 | 36.56 | 39.01 | 36.84 | 26.55 | 48.05 | 66.84 | 25.59 |
| RMSEP | 0.77 | 0.48 | 0.28 | 0.39 | 0.57 | 0.83 | 0.63 | 0.51 | 0.51 |
| Boltzmann’s sigmoidal model 1 | |||||||||
| Hmax (mL H2) | 21.80 | 8.21 | 7.21 | 32.34 | 16.47 | 18.33 | 31.49 | 26.66 | 9.17 |
| Rmax (mL H2/h) | 0.28 | 0.14 | 0.40 | 0.59 | 0.22 | 0.32 | 0.54 | 0.34 | 0.16 |
| t50 (h) | 64.94 | 47.75 | 39.57 | 47.52 | 47.39 | 35.44 | 56.91 | 73.98 | 34.70 |
| dx (h) | 19.54 | 15.07 | 4.52 | 13.62 | 18.88 | 14.19 | 14.61 | 19.76 | 13.91 |
| RMSEP | 0.91 | 0.42 | 0.30 | 0.83 | 0.84 | 1.03 | 0.72 | 0.54 | 0.37 |
| Parameter | DFEa | DFEb | DFEd | ||||||
|---|---|---|---|---|---|---|---|---|---|
| DFE concentration (v v−1 %) | 25 | 35 | 45 | 25 | 35 | 45 | 25 | 35 | 45 |
| Yield, hmax,e (mL H2 g−1 VFA) | 64.77 ± 13.09 | 17.63 ± 6.23 | 13.71 ± 4.85 | 126.50 ± 24.95 | 45.85 ± 11.44 | 39.86 ± 8.90 | 122.01 ± 19.60 | 69.54 ± 3.93 | 19.47 ± 9.18 |
| Modified Gompertz model | |||||||||
| hmax (mL H2 g−1 VFA) | 74.71 | 18.78 | 12.43 | 126.55 | 46.79 | 39.42 | 128.45 | 86.55 | 20.37 |
| rmax (mL H2 g−1 VFA h) | 0.78 | 0.29 | 0.68 | 2.14 | 0.58 | 0.68 | 1.98 | 0.83 | 0.34 |
| λ (h) | 20.14 | 14.25 | 29.81 | 17.24 | 6.99 | 5.08 | 24.23 | 28.59 | 3.60 |
| RMSEP | 2.37 | 1.05 | 0.48 | 1.47 | 1.55 | 1.74 | 2.46 | 1.41 | 1.10 |
| Ti-Gompertz model | |||||||||
| hmax (mL H2 g−1 VFA) | 74.71 | 18.78 | 12.43 | 126.55 | 46.79 | 39.42 | 128.45 | 86.55 | 20.37 |
| rmax (mL H2 g−1 VFA h) | 0.78 | 0.29 | 0.68 | 2.14 | 0.58 | 0.68 | 1.98 | 0.83 | 0.34 |
| Ti (h) | 55.46 | 38.38 | 36.56 | 39.01 | 36.84 | 26.55 | 48.05 | 66.84 | 25.59 |
| RMSEP | 2.37 | 1.05 | 0.48 | 1.47 | 1.55 | 1.74 | 2.46 | 1.41 | 1.10 |
| Boltzmann’s sigmoidal model 1 | |||||||||
| hmax (mL H2 g−1 VFA) | 67.25 | 18.09 | 12.36 | 122.11 | 44.43 | 38.46 | 122.64 | 74.16 | 19.84 |
| rmax (mL H2 g−1 VFA h) | 0.86 | 0.30 | 0.68 | 2.24 | 0.59 | 0.68 | 2.10 | 0.94 | 0.36 |
| t50 (h) | 64.94 | 47.75 | 39.57 | 47.52 | 47.39 | 35.44 | 56.91 | 73.98 | 34.70 |
| dx (h) | 19.54 | 15.07 | 4.52 | 13.62 | 18.88 | 14.19 | 14.61 | 19.76 | 13.91 |
| RMSEP | 2.80 | 0.93 | 0.52 | 3.12 | 2.27 | 2.16 | 2.80 | 1.51 | 0.81 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Hernández, B.N.; Escamilla-Alvarado, C.; Albalate-Ramírez, A.; Rivas-García, P.; Amézquita-García, H.J.; Rodríguez-Valderrama, S.; Paredes, M.G. Photofermentative Hydrogen Production from Real Dark Fermentation Effluents: A Sequential Valorization of Orange Peel Waste. Fermentation 2025, 11, 504. https://doi.org/10.3390/fermentation11090504
López-Hernández BN, Escamilla-Alvarado C, Albalate-Ramírez A, Rivas-García P, Amézquita-García HJ, Rodríguez-Valderrama S, Paredes MG. Photofermentative Hydrogen Production from Real Dark Fermentation Effluents: A Sequential Valorization of Orange Peel Waste. Fermentation. 2025; 11(9):504. https://doi.org/10.3390/fermentation11090504
Chicago/Turabian StyleLópez-Hernández, Brenda Nelly, Carlos Escamilla-Alvarado, Alonso Albalate-Ramírez, Pasiano Rivas-García, Héctor Javier Amézquita-García, Santiago Rodríguez-Valderrama, and María Guadalupe Paredes. 2025. "Photofermentative Hydrogen Production from Real Dark Fermentation Effluents: A Sequential Valorization of Orange Peel Waste" Fermentation 11, no. 9: 504. https://doi.org/10.3390/fermentation11090504
APA StyleLópez-Hernández, B. N., Escamilla-Alvarado, C., Albalate-Ramírez, A., Rivas-García, P., Amézquita-García, H. J., Rodríguez-Valderrama, S., & Paredes, M. G. (2025). Photofermentative Hydrogen Production from Real Dark Fermentation Effluents: A Sequential Valorization of Orange Peel Waste. Fermentation, 11(9), 504. https://doi.org/10.3390/fermentation11090504






